Abstract
Resistance to anti-PD-1 therapy remains a significant challenge in gastric cancer (GC) treatment. Here, we revealed that the USP14-IMP2-CXCL2 axis in tumor-associated macrophages (TAMs) drove resistance by recruiting myeloid-derived suppressor cells (MDSCs). Endoscopic biopsy samples were obtained from patients with inoperable GC who were candidates for anti-PD-1 therapy. Single-cell RNA sequencing (scRNA-seq) analysis showed a higher prevalence of USP14+ TAMs in therapy-resistant cases, where USP14 was linked to the immunosuppressive phenotype of TAMs. Clinically, GC samples with elevated USP14+ TAM infiltration exhibited decreased CD8+ T cell presence and increased MDSC infiltration. In vivo experiments further confirmed that USP14+ TAMs facilitated resistance to anti-PD-1 therapy in GC, reduced the infiltration of CD8+ T cells, and significantly increased the infiltration of MDSCs. In particular, USP14+ TAMs markedly enhanced the recruitment of MDSCs into the GC microenvironment through the secretion of CXCL2. Mechanistically, USP14 stabilized the m6A reader IMP2 through deubiquitination, thus enhancing CXCL2 expression and secretion. Conversely, the E3 ligase RNF40 facilitated IMP2 degradation via increasing its ubiquitination, with USP14 and RNF40 dynamically balancing IMP2’s protein abundance. Furthermore, animal experiments demonstrated that targeted intervention of USP14 markedly enhanced the sensitivity of GC to anti-PD-1 therapy. This study provided a comprehensive exploration of USP14’s oncogenic roles in TAMs, suggesting a novel strategy to enhance the efficacy of anti-PD-1 therapy by inhibiting the USP14/IMP2/CXCL2 signaling axis in GC.

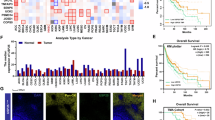
The sc-RNA analysis revealed that infiltration of USP14+ TAM was significantly higher in anti-PD-1 resistant GC compared to anti-PD-1 sensitive GC (top). USP14 stabilized IMP2 protein in tumor-associated macrophages (middle-left), leading to recruitment of MDSCs into GC microenvironment through secretion of CXCL2 (middle-right), thus reducing the infiltration of CD8+ T cells and facilitating the resistance to anti-PD-1 therapy in GC (bottom). Comment: Dear editors, in formally published articles, figure legends could be placed beneath their corresponding figures. In this version, is this arrangement adopted for the sake of convenience during the proofreading process? If so, we understand and accept this layout.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The single-cell RNA sequencing data (FUSCC Cohort 1) have been deposited in National Omics Data Encyclopedia (http://www.biosino.org/node): OEZ00017591. The readers can also obtained these data through contacting the corresponding author, Prof. Yu Zhang (zyu722320@163.com). Other materials in current study is available from the corresponding author upon reasonable requests.
Code availability
Our code used in this study can be available from the corresponding authors upon reasonable requests.
References
Wang Z, Han W, Xue F, Zhao Y, Wu P, Chen Y, et al. Nationwide gastric cancer prevention in China, 2021-2035: a decision analysis on effect, affordability and cost-effectiveness optimisation. Gut. 2022;71:2391–2400.
You L, Dou Y, Zhang Y, Xiao H, Lv H, Wei GH, et al. SDC2 Stabilization by USP14 Promotes Gastric Cancer Progression through Co-option of PDK1. Int J Biol Sci. 2023;19:3483–98.
Xue W, Xu C, Zhang K, Cui L, Huang X, Nan Y, et al. Enhancing antitumor efficacy of CLDN18.2-directed antibody-drug conjugates through autophagy inhibition in gastric cancer. Cell Death Discov. 2024;10:393.
Janjigian YY, Shitara K, Moehler M, Garrido M, Salman P, Shen L, et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet. 2021;398:27–40.
Rha SY, Oh DY, Yanez P, Bai Y, Ryu MH, Lee J, et al. Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for HER2-negative advanced gastric cancer (KEYNOTE-859): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol. 2023;24:1181–95.
Xu J, Jiang H, Pan Y, Gu K, Cang S, Han L, et al. Sintilimab Plus Chemotherapy for Unresectable Gastric or Gastroesophageal Junction Cancer: The ORIENT-16 Randomized Clinical Trial. JAMA. 2023;330:2064–74.
Yukami H, Kawazoe A, Lin YT, Koyama S, Fukuoka S, Hara H, et al. Updated Efficacy Outcomes of Anti-PD-1 Antibodies plus Multikinase Inhibitors for Patients with Advanced Gastric Cancer with or without Liver Metastases in Clinical Trials. Clin Cancer Res. 2022;28:3480–8.
Kwon M, An M, Klempner SJ, Lee H, Kim KM, Sa JK, et al. Determinants of Response and Intrinsic Resistance to PD-1 Blockade in Microsatellite Instability-High Gastric Cancer. Cancer Discov. 2021;11:2168–85.
Bain CC, Mowat AM. Macrophages in intestinal homeostasis and inflammation. Immunol Rev. 2014;260:102–17.
Li J, Sun J, Zeng Z, Liu Z, Ma M, Zheng Z, et al. Tumour-associated macrophages in gastric cancer: From function and mechanism to application. Clin Transl Med. 2023;13:e1386.
Lv K, Sun M, Fang H, Wang J, Lin C, Liu H et al. Targeting myeloid checkpoint Siglec-10 reactivates antitumor immunity and improves anti-programmed cell death 1 efficacy in gastric cancer. J Immunother Cancer. 2023;11:e007669.
Eissmann MF, Dijkstra C, Jarnicki A, Phesse T, Brunnberg J, Poh AR, et al. IL-33-mediated mast cell activation promotes gastric cancer through macrophage mobilization. Nat Commun. 2019;10:2735.
Wang YC, Wang X, Yu J, Ma F, Li Z, Zhou Y, et al. Targeting monoamine oxidase A-regulated tumor-associated macrophage polarization for cancer immunotherapy. Nat Commun. 2021;12:3530.
Dong X, Fan J, Xie W, Wu X, Wei J, He Z, et al. Efficacy evaluation of chimeric antigen receptor-modified human peritoneal macrophages in the treatment of gastric cancer. Br J Cancer. 2023;129:551–62.
Candido JB, Morton JP, Bailey P, Campbell AD, Karim SA, Jamieson T, et al. CSF1R(+) Macrophages Sustain Pancreatic Tumor Growth through T Cell Suppression and Maintenance of Key Gene Programs that Define the Squamous Subtype. Cell Rep. 2018;23:1448–60.
Veglia F, Sanseviero E, Gabrilovich DI. Myeloid-derived suppressor cells in the era of increasing myeloid cell diversity. Nat Rev Immunol. 2021;21:485–98.
Liu X, Tang R, Xu J, Tan Z, Liang C, Meng Q, et al. CRIP1 fosters MDSC trafficking and resets tumour microenvironment via facilitating NF-kappaB/p65 nuclear translocation in pancreatic ductal adenocarcinoma. Gut. 2023;72:2329–43.
Bao Y, Zhai J, Chen H, Wong CC, Liang C, Ding Y, et al. Targeting m(6)A reader YTHDF1 augments antitumour immunity and boosts anti-PD-1 efficacy in colorectal cancer. Gut. 2023;72:1497–509.
Liao W, Overman MJ, Boutin AT, Shang X, Zhao D, Dey P, et al. KRAS-IRF2 Axis Drives Immune Suppression and Immune Therapy Resistance in Colorectal Cancer. Cancer Cell. 2019;35:559–572.e557.
Huang B, Pan PY, Li Q, Sato AI, Levy DE, Bromberg J, et al. Gr-1+CD115+ immature myeloid suppressor cells mediate the development of tumor-induced T regulatory cells and T-cell anergy in tumor-bearing host. Cancer Res. 2006;66:1123–31.
Li N, Kang Y, Wang L, Huff S, Tang R, Hui H, et al. ALKBH5 regulates anti-PD-1 therapy response by modulating lactate and suppressive immune cell accumulation in tumor microenvironment. Proc Natl Acad Sci USA. 2020;117:20159–70.
Nagarsheth N, Wicha MS, Zou W. Chemokines in the cancer microenvironment and their relevance in cancer immunotherapy. Nat Rev Immunol. 2017;17:559–72.
Schiwon M, Weisheit C, Franken L, Gutweiler S, Dixit A, Meyer-Schwesinger C, et al. Crosstalk between sentinel and helper macrophages permits neutrophil migration into infected uroepithelium. Cell. 2014;156:456–68.
Wang X, Tokheim C, Gu SS, Wang B, Tang Q, Li Y, et al. In vivo CRISPR screens identify the E3 ligase Cop1 as a modulator of macrophage infiltration and cancer immunotherapy target. Cell. 2021;184:5357–5374.e5322.
Shi D, Wu X, Jian Y, Wang J, Huang C, Mo S, et al. USP14 promotes tryptophan metabolism and immune suppression by stabilizing IDO1 in colorectal cancer. Nat Commun. 2022;13:5644.
Zhao C, Gong J, Bai Y, Yin T, Zhou M, Pan S, et al. A self-amplifying USP14-TAZ loop drives the progression and liver metastasis of pancreatic ductal adenocarcinoma. Cell Death Differ. 2023;30:1–15.
Lange SM, Armstrong LA, Kulathu Y. Deubiquitinases: From mechanisms to their inhibition by small molecules. Mol Cell. 2022;82:15–29.
Zhang S, Zou S, Yin D, Zhao L, Finley D, Wu Z, et al. USP14-regulated allostery of the human proteasome by time-resolved cryo-EM. Nature. 2022;605:567–74.
Yang J, Xu P, Chen Z, Zhang X, Xia Y, Fang L, et al. N6-methyadenosine modified SUV39H2 regulates homologous recombination through epigenetic repression of DUSP6 in gastric cancer. Cancer Lett. 2023;558:216092.
Weng H, Huang F, Yu Z, Chen Z, Prince E, Kang Y, et al. The m(6)A reader IGF2BP2 regulates glutamine metabolism and represents a therapeutic target in acute myeloid leukemia. Cancer Cell. 2022;40:1566–82 e1510.
Huang H, Weng H, Sun W, Qin X, Shi H, Wu H, et al. Recognition of RNA N(6)-methyladenosine by IGF2BP proteins enhances mRNA stability and translation. Nat Cell Biol. 2018;20:285–95.
Kessler SM, Pokorny J, Zimmer V, Laggai S, Lammert F, Bohle RM, et al. IGF2 mRNA binding protein p62/IMP2-2 in hepatocellular carcinoma: antiapoptotic action is independent of IGF2/PI3K signaling. Am J Physiol Gastrointest Liver Physiol. 2013;304:G328–336.
Allen WL, Jithesh PV, Oliver GR, Proutski I, Longley DB, Lenz HJ, et al. The colorectal cancer disease-specific transcriptome may facilitate the discovery of more biologically and clinically relevant information. BMC Cancer. 2010;10:687.
Simon Y, Kessler SM, Bohle RM, Haybaeck J, Kiemer AK. The insulin-like growth factor 2 (IGF2) mRNA-binding protein p62/IGF2BP2-2 as a promoter of NAFLD and HCC? Gut. 2014;63:861–3.
Kessler SM, Laggai S, Barghash A, Schultheiss CS, Lederer E, Artl M, et al. IMP2/p62 induces genomic instability and an aggressive hepatocellular carcinoma phenotype. Cell Death Dis. 2015;6:e1894.
Valverde R, Edwards L, Regan L. Structure and function of KH domains. FEBS J. 2008;275:2712–26.
Cao T, Zhang W, Wang Q, Wang C, Ma W, Zhang C et al. Cancer SLC6A6-mediated taurine uptake transactivates immune checkpoint genes and induces exhaustion in CD8(+) T cells. Cell. 2024;187:2288–304.e27.
Yao Y, Liu Q, Adrianto I, Wu X, Glassbrook J, Khalasawi N, et al. Histone deacetylase 3 controls lung alveolar macrophage development and homeostasis. Nat Commun. 2020;11:3822.
Chen J, Yao Y, Gong C, Yu F, Su S, Chen J, et al. CCL18 from tumor-associated macrophages promotes breast cancer metastasis via PITPNM3. Cancer Cell. 2011;19:541–55.
Tsvetkov P, Detappe A, Cai K, Keys HR, Brune Z, Ying W, et al. Mitochondrial metabolism promotes adaptation to proteotoxic stress. Nat Chem Biol. 2019;15:681–9.
Willenborg S, Sanin DE, Jais A, Ding X, Ulas T, Nuchel J, et al. Mitochondrial metabolism coordinates stage-specific repair processes in macrophages during wound healing. Cell Metab. 2021;33:2398–414 e2399.
Chaib S, Lopez-Dominguez JA, Lalinde-Gutierrez M, Prats N, Marin I, Boix O, et al. The efficacy of chemotherapy is limited by intratumoral senescent cells expressing PD-L2. Nat Cancer. 2024;5:448–62.
Xie M, Lin Z, Ji X, Luo X, Zhang Z, Sun M, et al. FGF19/FGFR4-mediated elevation of ETV4 facilitates hepatocellular carcinoma metastasis by upregulating PD-L1 and CCL2. J Hepatol. 2023;79:109–25.
Brina D, Ponzoni A, Troiani M, Cali B, Pasquini E, Attanasio G, et al. The Akt/mTOR and MNK/eIF4E pathways rewire the prostate cancer translatome to secrete HGF, SPP1 and BGN and recruit suppressive myeloid cells. Nat Cancer. 2023;4:1102–21.
Kumar V, Ramnarayanan K, Sundar R, Padmanabhan N, Srivastava S, Koiwa M, et al. Single-Cell Atlas of Lineage States, Tumor Microenvironment, and Subtype-Specific Expression Programs in Gastric Cancer. Cancer Discov. 2022;12:670–91.
Qiu MZ, Oh DY, Kato K, Arkenau T, Tabernero J, Correa MC, et al. Tislelizumab plus chemotherapy versus placebo plus chemotherapy as first line treatment for advanced gastric or gastro-oesophageal junction adenocarcinoma: RATIONALE-305 randomised, double blind, phase 3 trial. BMJ. 2024;385:e078876.
Chen Y, Jia K, Sun Y, Zhang C, Li Y, Zhang L, et al. Predicting response to immunotherapy in gastric cancer via multi-dimensional analyses of the tumour immune microenvironment. Nat Commun. 2022;13:4851.
Bell HN, Zou W. Beyond the Barrier: Unraveling the Mechanisms of Immunotherapy Resistance. Annu Rev Immunol. 2024;42:521–50.
Kono K, Nakajima S, Mimura K. Biomarker-oriented chemo-immunotherapy for advanced gastric cancer. Int J Clin Oncol. 2024;29:865–72.
Chong X, Madeti Y, Cai J, Li W, Cong L, Lu J, et al. Recent developments in immunotherapy for gastrointestinal tract cancers. J Hematol Oncol. 2024;17:65.
Xiang J, Zhang N, Du A, Li J, Luo M, Wang Y, et al. A Ubiquitin-Dependent Switch on MEF2D Senses Pro-Metastatic Niche Signals to Facilitate Intrahepatic Metastasis of Liver Cancer. Adv Sci. 2023;10:e2305550.
Liu D, Li M, Zhao Z, Zhou L, Zhi F, Guo Z et al. Targeting the TRIM14/USP14 Axis Enhances Immunotherapy Efficacy by Inducing Autophagic Degradation of PD-L1. Cancer Res. 2024;84:2806–19.
Rowinsky EK, Paner A, Berdeja JG, Paba-Prada C, Venugopal P, Porkka K, et al. Phase 1 study of the protein deubiquitinase inhibitor VLX1570 in patients with relapsed and/or refractory multiple myeloma. Invest N Drugs. 2020;38:1448–53.
Liu B, Chen J, Zhang S. Emerging role of ubiquitin-specific protease 14 in oncogenesis and development of tumor: Therapeutic implication. Life Sci. 2019;239:116875.
Wang J, Chen L, Qiang P. The role of IGF2BP2, an m6A reader gene, in human metabolic diseases and cancers. Cancer Cell Int. 2021;21:99.
O’Reilly ME, Ho S, Coronel J, Zhu L, Liu W, Xue C, et al. linc-ADAIN, a human adipose lincRNA, regulates adipogenesis by modulating KLF5 and IL-8 mRNA stability. Cell Rep. 2024;43:114240.
Arshad J, Rao A, Repp ML, Rao R, Wu C, Merchant JL. Myeloid-Derived Suppressor Cells: Therapeutic Target for Gastrointestinal Cancers. Int J Mol Sci. 2024;25:2985.
Zhou X, Fang D, Liu H, Ou X, Zhang C, Zhao Z, et al. PMN-MDSCs accumulation induced by CXCL1 promotes CD8(+) T cells exhaustion in gastric cancer. Cancer Lett. 2022;532:215598.
Shitara K, Di Bartolomeo M, Mandala M, Ryu MH, Caglevic C, Olesinski T et al. Association between gene expression signatures and clinical outcomes of pembrolizumab versus paclitaxel in advanced gastric cancer: exploratory analysis from the randomized, controlled, phase III KEYNOTE-061 trial. J Immunother Cancer. 2023;11:e006920.
Acknowledgements
We express our gratitude to Prof. Gong-Hong Wei, chair of the Department of Molecular Biology and Biochemistry, Fudan University, Shanghai, and Prof. Dazhi Xu, chair of the Department of Gastric Surgery, Fudan University Shanghai Cancer Center, for thier very detailed and useful suggestions on this study.
Funding
This study was supported by research funding of FUSCC (SCIP-2023146) and by the National Natural Science Foundation of China (No. 32200745).
Author information
Authors and Affiliations
Contributions
Li You, Xuyao Zhang, and Yu Zhang designed the research. Li You, Qian Wang, Hongwei Xiao and Mengjiao Lv performed the experiments. Hong Lv assessed the immunohistochemical scores. Li You, Tianxue Zhang, Li Deng and Yu Zhang analyzed data and wrote the manuscript. Xuyao Zhang and Yu Zhang supervised the process of this study and revised manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics
Informed consent forms were signed by all GC patients enrolled in this study, and the study was approved by the Ethics Committee of Fudan University Shanghai Cancer Center (Approval No. 2012228-14). The animal experiments were also approved by the Ethics Committee of Fudan University Shanghai Cancer Center (Approval No. IACUC-2023301). Additionally, the care and handling of the animals were conducted in accordance with applicable animal welfare regulation.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
You, L., Wang, Q., Zhang, T. et al. USP14-IMP2-CXCL2 axis in tumor-associated macrophages facilitates resistance to anti-PD-1 therapy in gastric cancer by recruiting myeloid-derived suppressor cells. Oncogene 44, 2413–2426 (2025). https://doi.org/10.1038/s41388-025-03425-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41388-025-03425-w
This article is cited by
-
The m6A modification in cancer: roles, implications, and its potential in therapy
Molecular Biomedicine (2025)
-
USP14-mediated stabilization of ACTN1 maintains mesenchymal characteristics in glioblastoma
Communications Biology (2025)
-
NFATC3 enhances osteosarcoma progression by increasing PD-L1 and CXCL2 levels
Medical Oncology (2025)