Abstract
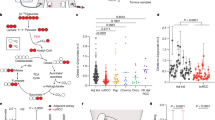
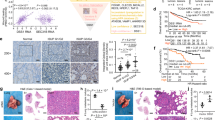
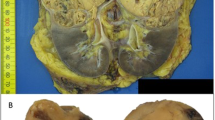
Renal cell carcinoma (RCC) is one of the most common malignancies in the urinary system. NSD2 is an H3K36-specific di-methyltransferase that has been reported to participate in diverse biological processes and human tumors. However, its role in RCC remains unclear. Here, we found that NSD2 is highly expressed in RCC, which is associated with poor survival in RCC patients. NSD2 facilitates the transition from Myc-induced polycystic kidney disease to tubulocystic renal cell carcinoma (TCRCC), which is a rare RCC subtype with distinctive clinicopathologic and genetic characterizations. The mice with kidney-specific overexpression of MYC and NSD2 (KMN) display severe cyst burden at only 6 weeks of age, and develop into TCRCC at 12 weeks of age. Mechanistically, NSD2 transcriptionally upregulates the expressions of integrins (Itga4 and Itga11), to further activate the FAK/AKT pathway. In addition, we found that NSD2 enhances cell proliferation on the stiff matrix of PEGDA hydrogel. Moreover, inhibition of FAK signaling relieves the symptoms of KMN mice, and significantly rescues the enhanced cell proliferation caused by NSD2 overexpression in vitro. Together, our findings highlight an epigenetic mechanism by which NSD2 regulates TCRCC tumorigenesis through the integrin/FAK/AKT pathway. This study may also pave the way for the development of targeted, patient-tailored therapies for TCRCC patients with NSD2 amplification or high expression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The sequencing data sets have been deposited in the National Center for Biotechnology Information (NCBI)’s Gene Expression Omnibus (GEO). RNA-Seq raw data have been deposited in GEO under accession number: GSE266614. CUT-Tag raw data have been deposited in GEO under accession number: GSE264634.
References
Sarungbam J, Mehra R, Tomlins SA, Smith SC, Jayakumaran G, Al-Ahmadie H, et al. Tubulocystic renal cell carcinoma: a distinct clinicopathologic entity with a characteristic genomic profile. Modern Pathol. 2019;32:701–9.
Amin MB, MacLennan GT, Gupta R, Grignon D, Paraf F, Vieillefond A, et al. Tubulocystic Carcinoma of the Kidney: Clinicopathologic analysis of 31 cases of a distinctive rare subtype of renal cell carcinoma. American J Surg Pathol. 2009;33:384.
Rao H, Li X, Liu M, Liu J, Feng W, Tang H, et al. Multilevel Regulation of β-Catenin Activity by SETD2 Suppresses the Transition from Polycystic Kidney Disease to Clear Cell Renal Cell Carcinoma. Cancer Res. 2021;81:3554–67.
Xing S, Liu A, Yang X, Chen L, Xu D. Tubulocystic renal cell carcinoma: Two-case report and literature review. Int J Immunopathol Pharm. 2021;35:205873842110029.
De Cubas AA, Rathmell WK. Epigenetic modifiers: activities in renal cell carcinoma. Nat Rev Urol. 2018;15:599–614.
Joosten SC, Smits KM, Aarts MJ, Melotte V, Koch A, Tjan-Heijnen VC, et al. Epigenetics in renal cell cancer: mechanisms and clinical applications. Nat Rev Urol. 2018;15:430–51.
Rao H, Liu C, Wang A, Ma C, Xu Y, Ye T, et al. SETD2 deficiency accelerates sphingomyelin accumulation and promotes the development of renal cancer. Nat Commun. 2023;14:7572.
Li Z, Zhang X, Xie S, Liu X, Fei C, Huang X, et al. H3K36me2 methyltransferase NSD2 orchestrates epigenetic reprogramming during spermatogenesis. Nucleic Acids Res. 2022;50:6786–6800.
Tanaka H, Igata T, Etoh K, Koga T, Takebayashi S, Nakao M. The NSD2/WHSC1/MMSET methyltransferase prevents cellular senescence-associated epigenomic remodeling. Aging Cell. 2020;19:e13173.
Han X, Piao L, Xu X, Luo F, Liu Z, He X. NSD2 promotes renal cancer progression through stimulating Akt/Erk Signaling. CMAR. 2020;12:375–83.
Nimura K, Ura K, Shiratori H, Ikawa M, Okabe M, Schwartz RJ, et al. A histone H3 lysine 36 trimethyltransferase links Nkx2-5 to Wolf–Hirschhorn syndrome. Nature. 2009;460:287–91.
Song D, Lan J, Chen Y, Liu A, Wu Q, Zhao C, et al. NSD2 promotes tumor angiogenesis through methylating and activating STAT3 protein. Oncogene. 2021;40:2952–67.
Mitra SK, Hanson DA, Schlaepfer DD. Focal adhesion kinase: in command and control of cell motility. Nat Rev Mol Cell Biol. 2005;6:56–68.
Eke I, Cordes N. Focal adhesion signaling and therapy resistance in cancer. Seminars Cancer Biol. 2015;31:65–75.
Poettler M, Unseld M, Braemswig K, Haitel A, Zielinski CC, Prager GW. CD98hc (SLC3A2) drives integrin-dependent renal cancer cell behavior. Molecular Cancer. 2013;12:169.
Seong J, Tajik A, Sun J, Guan J-L, Humphries MJ, Craig SE, et al. Distinct biophysical mechanisms of focal adhesion kinase mechanoactivation by different extracellular matrix proteins. Proc Natl Acad Sci USA. 2013;110:19372–7.
Hung T-W, Chen P-N, Wu H-C, Wu S-W, Tsai P-Y, Hsieh Y-S, et al. Kaempferol inhibits the invasion and migration of renal cancer cells through the downregulation of AKT and FAK Pathways. International J Med Sci. 2017;14:984–93.
Feng W, Niu N, Lu P, Chen Z, Rao H, Zhang W, et al. Multilevel regulation of NF-κB signaling by NSD2 suppresses Kras -Driven Pancreatic Tumorigenesis. Advanced Sci. 2024;11:2309387.
Feng W, Ma C, Rao H, Zhang W, Liu C, Xu Y, et al. Setd2 deficiency promotes gastric tumorigenesis through inhibiting the SIRT1/FOXO pathway. Cancer Lett. 2023;579:216470.
Markert CD, Guo X, Skardal A, Wang Z, Bharadwaj S, Zhang Y, et al. Characterizing the micro-scale elastic modulus of hydrogels for use in regenerative medicine. Journal Mech Behav Biomed Mater. 2013;27:115–27.
Beamish JA, Chen E, Putnam AJ. Engineered extracellular matrices with controlled mechanics modulate renal proximal tubular cell epithelialization. PLoS ONE. 2017;12:e0181085.
Guo M, Deng Y, Huang J, Huang Y, Deng J, Wu H. Fabrication and Validation of a 3D Portable PEGDA Microfluidic Chip for Visual Colorimetric Detection of Captured Breast Cancer Cells. Polymers. 2023;15:3183.
Doke T, Huang S, Qiu C, Liu H, Guan Y, Hu H, et al. Transcriptome-wide association analysis identifies DACH1 as a kidney disease risk gene that contributes to fibrosis. Journal Clin Investig. 2021;131:e141801.
Zhao M, Teng X, Ru G, Zhao Z, Hu Q, Han L, et al. Tubulocystic renal cell carcinoma with poorly differentiated foci is indicative of aggressive behavior: clinicopathologic study of two cases and review of the literature. Int J Clin Exp Pathol. 2015;8:11124–31.
Yang XJ, Shen S, Li R, Yang Y, Tretiakova MM Tubulocystic Carcinoma of the Kidney. Am J Surg Pathol. 2008; 32:177–187.
Azoulay S, Vieillefond A, Paraf F, Pasquier D, Cussenot O, Callard P, et al. Tubulocystic carcinoma of the kidney: a new entity among renal tumors. Virchows Arch. 2007;451:905–9.
Lawrie CH, Armesto M, Fernandez-Mercado M, Arestín M, Manterola L, Goicoechea I, et al. Noncoding RNA Expression and Targeted Next-Generation Sequencing Distinguish Tubulocystic Renal Cell Carcinoma (TC-RCC) from Other Renal Neoplasms. Journal Mol Diagn. 2018;20:34–45.
Wu B, Liu D-A, Guan L, Myint PK, Chin L, Dang H, et al. Stiff matrix induces exosome secretion to promote tumour growth. Nat Cell Biol. 2023;25:415–24.
Integrin Signaling in Cancer. Mechanotransduction, Stemness, Epithelial Plasticity, and Therapeutic Resistance. Cancer Cell. 2019;35:347–67.
Sengupta D, Zeng L, Li Y, Hausmann S, Ghosh D, Yuan G, et al. NSD2 dimethylation at H3K36 promotes lung adenocarcinoma pathogenesis. Molecular Cell. 2021;81:4481–4492.e9.
Katoh K. FAK-dependent cell motility and cell elongation. Cells. 2020;9:192.
Herman SEM, Mustafa RZ, Jones J, Wong DH, Farooqui M, Wiestner A. Treatment with Ibrutinib Inhibits BTK- and VLA-4-Dependent Adhesion of Chronic Lymphocytic Leukemia Cells In Vivo. Clin Cancer Res. 2015;21:4642–51.
Piovan E, Yu J, Tosello V, Herranz D, Ambesi-Impiombato A, Da Silva AC, et al. Direct Reversal of Glucocorticoid Resistance by AKT Inhibition in Acute Lymphoblastic Leukemia. Cancer Cell. 2013;24:766–76.
Sunyer R, Trepat X. Durotaxis. Current Biol. 2020;30:R383–R387.
Lachowski D, Cortes E, Robinson B, Rice A, Rombouts K, Hernández AEDR. FAK controls the mechanical activation of YAP, a transcriptional regulator required for durotaxis. FASEB J. 2018;32:1099–107.
SenGupta S, Parent CA, Bear JE. The principles of directed cell migration. Nat Rev Mol Cell Biol. 2021;22:529–47.
Espina JA, Marchant CL, Barriga EH. Durotaxis: the mechanical control of directed cell migration. FEBS J. 2022;289:2736–54.
Lin F-Y, Li J, Xie Y, Zhu J, Nguyen TTH, Zhang Y, et al. A general chemical principle for creating closure-stabilizing integrin inhibitors. Cell. 2022;185:3533–3550.e27.
Grove RA, Shackelford S, Sopper S, Pirruccello S, Horrigan J, Havrdova E, et al. Leukocyte counts in cerebrospinal fluid and blood following firategrast treatment in subjects with relapsing forms of multiple sclerosis. Eur J Neurol. 2013;20:1032–42.
Miller DH, Weber T, Grove R, Wardell C, Horrigan J, Graff O, et al. Firategrast for relapsing remitting multiple sclerosis: a phase 2, randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2012;11:131–9.
Acknowledgements
This work was supported by funds from National Key R&D Program of China (2022YFA1302704 to LL and W-QG), National Natural Science Foundation of China (82372604, 82073104 to LL, U23A20441 to W-QG), Science and Technology Commission of Shanghai Municipality (21JC1404100 to W-QG) and 111 project (no. B21024). We thank Genefund Biotech (Shanghai, China) for assistance in the data analysis.
Author information
Authors and Affiliations
Contributions
LL designed experiment and interpreted data; WF and NL. performed most of the experiments; CL, HR, ZC, WZ, YX, RA. and ZW assisted in some experiments; W-QG assisted in some discussion; WF, NL and LL wrote the manuscript; LL provided the overall guidance.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Feng, W., Liu, N., Liu, C. et al. NSD2 promotes cell durotaxis and drives the transition from polycystic kidney disease to tubulocystic renal cell carcinoma through integrin/FAK/AKT signaling. Oncogene 44, 3437–3448 (2025). https://doi.org/10.1038/s41388-025-03505-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41388-025-03505-x