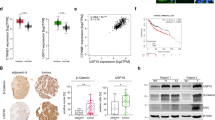
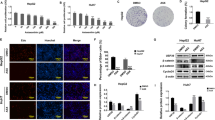
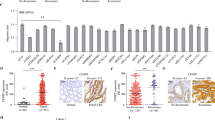
Abstract
Aberrant activation of the Wnt/β-catenin signaling pathway is a hallmark of colorectal cancer (CRC). Here, we identify the deubiquitinating enzyme USP17 as a critical regulator of β-catenin stability and activity in CRC. We demonstrate that USP17 directly interacts with and deubiquitinates β-catenin, preventing its degradation and enhancing its stability. CRISPR/Cas9-mediated knockdown of USP17 in CRC-derived cell lines significantly reduced β-catenin levels and suppressed epithelial-mesenchymal transition (EMT), as evidenced by distinct morphological changes and altered expression of classical EMT markers. USP17 depletion reduced the proliferation of CRC cell lines and impaired CRC tumor growth in vivo. Conversely, USP17 overexpression in immortalized rat enterocytes elevated β-catenin levels and enhanced KRAS-induced cell proliferation. RNA sequencing and quantitative proteomic analysis of USP17-depleted CRC cells revealed significant suppression of the transcriptional coactivator function of β-catenin, impacting key oncogenic-related pathways. Our findings establish USP17 as a key regulator of β-catenin signaling and highlight its potential as a candidate therapeutic target in CRC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting this study’s findings are available from the corresponding author upon reasonable request. The RNA sequencing (RNA-Seq) data generated in this study have been deposited in NCBI GEO (GSE298365). The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE [58] partner repository with the dataset identifier PXD065117 and 10.6019/PXD065117.
References
Brenner H, Kloor M, Pox CP. Colorectal cancer. Lancet. 2014;383:1490–502.
Nusse R, Clevers H. Wnt/beta-catenin signaling, disease, and emerging therapeutic modalities. Cell. 2017;169:985–99.
Cancer Genome Atlas N. Comprehensive molecular characterization of human colon and rectal cancer. Nature. 2012;487:330–7.
Schatoff EM, Leach BI, Dow LE. Wnt signaling and colorectal cancer. Curr Colorectal Cancer Rep. 2017;13:101–10.
He TC, Sparks AB, Rago C, Hermeking H, Zawel L, da Costa LT, et al. Identification of c-MYC as a target of the APC pathway. Science. 1998;281:1509–12.
Tetsu O, McCormick F. Beta-catenin regulates expression of cyclin D1 in colon carcinoma cells. Nature. 1999;398:422–6.
Maurice MM, Angers S Mechanistic insights into Wnt-beta-catenin pathway activation and signal transduction. Nat Rev Mol Cell Biol. 2025.
Burrows JF, Scott CJ, Johnston JA. The DUB/USP17 deubiquitinating enzymes: a gene family within a tandemly repeated sequence, is also embedded within the copy number variable beta-defensin cluster. BMC Genomics. 2010;11:250.
Burrows JF, McGrattan MJ, Rascle A, Humbert M, Baek KH, Johnston JA. DUB-3, a cytokine-inducible deubiquitinating enzyme that blocks proliferation. J Biol Chem. 2004;279:13993–14000.
Chen R, Zhang L, Zhong B, Tan B, Liu Y, Shu HB. The ubiquitin-specific protease 17 is involved in virus-triggered type I IFN signaling. Cell Res. 2010;20:802–11.
McFarlane C, Kelvin AA, de la Vega M, Govender U, Scott CJ, Burrows JF, et al. The deubiquitinating enzyme USP17 is highly expressed in tumor biopsies, is cell cycle regulated, and is required for G1-S progression. Cancer Res. 2010;70:3329–39.
McFarlane C, McFarlane S, Paul I, Arthur K, Scheaff M, Kerr K, et al. The deubiquitinating enzyme USP17 is associated with non-small cell lung cancer (NSCLC) recurrence and metastasis. Oncotarget. 2013;4:1836–43.
Zhou B, Shu B, Xi T, Su N, Liu J. Dub3 expression correlates with tumor progression and poor prognosis in human epithelial ovarian cancer. Biomed Pharmacother. 2015;70:84–9.
Zhang S, Yuan J, Zheng R. Suppression of ubiquitin-specific peptidase 17 (USP17) inhibits tumorigenesis and invasion in non-small cell lung cancer cells. Oncol Res. 2016;24:263–9.
de la Vega M, Kelvin AA, Dunican DJ, McFarlane C, Burrows JF, Jaworski J, et al. The deubiquitinating enzyme USP17 is essential for GTPase subcellular localization and cell motility. Nat Commun. 2011;2:259.
Pereg Y, Liu BY, O’Rourke KM, Sagolla M, Dey A, Komuves L, et al. Ubiquitin hydrolase Dub3 promotes oncogenic transformation by stabilizing Cdc25A. Nat Cell Biol. 2010;12:400–6.
Hjortland NM, Mesecar AD. Steady-state kinetic studies reveal that the anti-cancer target Ubiquitin-Specific Protease 17 (USP17) is a highly efficient deubiquitinating enzyme. Arch Biochem Biophys. 2016;612:35–45.
Kim J, D’Annibale S, Magliozzi R, Low TY, Jansen P, Shaltiel IA, et al. USP17- and SCFbetaTrCP–regulated degradation of DEC1 controls the DNA damage response. Mol Cell Biol. 2014;34:4177–85.
Yang GF, Zhang X, Su YG, Zhao R, Wang YY. The role of the deubiquitinating enzyme DUB3/USP17 in cancer: a narrative review. Cancer Cell Int. 2021;21:455.
Nagasaka M, Inoue Y, Yoshida M, Miyajima C, Morishita D, Tokugawa M, et al. The deubiquitinating enzyme USP17 regulates c-Myc levels and controls cell proliferation and glycolysis. FEBS Lett. 2022;596:465–78.
Liu T, Yu J, Deng M, Yin Y, Zhang H, Luo K, et al. CDK4/6-dependent activation of DUB3 regulates cancer metastasis through SNAIL1. Nat Commun. 2017;8:13923.
Wu Y, Wang Y, Lin Y, Liu Y, Wang Y, Jia J, et al. Dub3 inhibition suppresses breast cancer invasion and metastasis by promoting Snail1 degradation. Nat Commun. 2017;8:14228.
Wen Y, Yang X, Li S, Huang L, Chen J, Tan L, et al. Targeting CDK4/6 suppresses colorectal cancer by destabilizing YAP1. MedComm. 2025;6:e70103.
Lin Y, Wang Y, Shi Q, Yu Q, Liu C, Feng J, et al. Stabilization of the transcription factors slug and twist by the deubiquitinase dub3 is a key requirement for tumor metastasis. Oncotarget. 2017;8:75127–40.
Song C, Liu W, Li J. USP17 is upregulated in osteosarcoma and promotes cell proliferation, metastasis, and epithelial-mesenchymal transition through stabilizing SMAD4. Tumour Biol. 2017;39:1010428317717138.
Ducker C, Chow LKY, Saxton J, Handwerger J, McGregor A, Strahl T, et al. De-ubiquitination of ELK-1 by USP17 potentiates mitogenic gene expression and cell proliferation. Nucleic Acids Res. 2019;47:4495–508.
Jia X, Li L, Wang F, Xue Y, Wu T, Jia Q, et al. DUB3/KLF4 combats tumor growth and chemoresistance in hepatocellular carcinoma. Cell Death Discov. 2022;8:166.
Delgado-Diaz MR, Martin Y, Berg A, Freire R, Smits VA. Dub3 controls DNA damage signalling by direct deubiquitination of H2AX. Mol Oncol. 2014;8:884–93.
Coulombe P, Rodier G, Pelletier S, Pellerin J, Meloche S. Rapid turnover of extracellular signal-regulated kinase 3 by the ubiquitin-proteasome pathway defines a novel paradigm of mitogen-activated protein kinase regulation during cellular differentiation. Mol Cell Biol. 2003;23:4542–58.
Kizilay Mancini O, Huynh DN, Menard L, Shum-Tim D, Ong H, Marleau S, et al. Ex vivo Ikkbeta ablation rescues the immunopotency of mesenchymal stromal cells from diabetics with advanced atherosclerosis. Cardiovasc Res. 2021;117:756–66.
Reissland M, Hartmann O, Tauch S, Bugter JM, Prieto-Garcia C, Schulte C, et al. USP10 drives cancer stemness and enables super-competitor signalling in colorectal cancer. Oncogene. 2024;43:3645–59.
van de Wetering M, Cavallo R, Dooijes D, van Beest M, van Es J, Loureiro J, et al. Armadillo coactivates transcription driven by the product of the Drosophila segment polarity gene dTCF. Cell. 1997;88:789–99.
Li T, Fu J, Zeng Z, Cohen D, Li J, Chen Q, et al. TIMER2.0 for analysis of tumor-infiltrating immune cells. Nucleic Acids Res. 2020;48:W509–W514.
Shtutman M, Zhurinsky J, Simcha I, Albanese C, D’Amico M, Pestell R, et al. The cyclin D1 gene is a target of the beta-catenin/LEF-1 pathway. Proc Natl Acad Sci USA. 1999;96:5522–7.
Katoh M. Multi‑layered prevention and treatment of chronic inflammation, organ fibrosis and cancer associated with canonical WNT/beta‑catenin signaling activation (Review). Int J Mol Med. 2018;42:713–25.
Herbst A, Jurinovic V, Krebs S, Thieme SE, Blum H, Goke B, et al. Comprehensive analysis of beta-catenin target genes in colorectal carcinoma cell lines with deregulated Wnt/beta-catenin signaling. BMC Genomics. 2014;15:74.
Zhao H, Ming T, Tang S, Ren S, Yang H, Liu M, et al. Wnt signaling in colorectal cancer: pathogenic role and therapeutic target. Mol Cancer. 2022;21:144.
Tran FH, Zheng JJ. Modulating the Wnt signaling pathway with small molecules. Protein Sci. 2017;26:650–61.
Lepourcelet M, Chen YN, France DS, Wang H, Crews P, Petersen F, et al. Small-molecule antagonists of the oncogenic Tcf/beta-catenin protein complex. Cancer Cell. 2004;5:91–102.
Wan C, Mahara S, Sun C, Doan A, Chua HK, Xu D, et al. Genome-scale CRISPR-Cas9 screen of Wnt/beta-catenin signaling identifies therapeutic targets for colorectal cancer. Sci Adv. 2021;7.
Zhong Y, Katavolos P, Nguyen T, Lau T, Boggs J, Sambrone A, et al. Tankyrase Inhibition Causes Reversible Intestinal Toxicity in Mice with a Therapeutic Index < 1. Toxicol Pathol. 2016;44:267–78.
Rosin-Arbesfeld R, Cliffe A, Brabletz T, Bienz M. Nuclear export of the APC tumour suppressor controls beta-catenin function in transcription. EMBO J. 2003;22:1101–13.
Yang J, Zhang W, Evans PM, Chen X, He X, Liu C. Adenomatous polyposis coli (APC) differentially regulates beta-catenin phosphorylation and ubiquitination in colon cancer cells. J Biol Chem. 2006;281:17751–7.
Ranes M, Zaleska M, Sakalas S, Knight R, Guettler S. Reconstitution of the destruction complex defines roles of AXIN polymers and APC in beta-catenin capture, phosphorylation, and ubiquitylation. Mol Cell. 2021;81:3246–61 e3211.
Novellasdemunt L, Foglizzo V, Cuadrado L, Antas P, Kucharska A, Encheva V, et al. USP7 Is a Tumor-Specific WNT Activator for APC-Mutated Colorectal Cancer by Mediating beta-Catenin Deubiquitination. Cell Rep. 2017;21:612–27.
Dominguez-Brauer C, Khatun R, Elia AJ, Thu KL, Ramachandran P, Baniasadi SP, et al. E3 ubiquitin ligase Mule targets beta-catenin under conditions of hyperactive Wnt signaling. Proc Natl Acad Sci USA. 2017;114:E1148–E1157.
Zhong W, Myers JS, Wang F, Wang K, Lucas J, Rosfjord E, et al. Comparison of the molecular and cellular phenotypes of common mouse syngeneic models with human tumors. BMC Genomics. 2020;21:2.
Nguyen HT, Kugler JM, Cohen SM. DUB3 deubiquitylating enzymes regulate hippo pathway activity by regulating the stability of ITCH, LATS and AMOT proteins. PLoS One. 2017;12:e0169587.
Huang S, Li J, Wu S, Zheng Z, Wang C, Li H, et al. C4orf19 inhibits colorectal cancer cell proliferation by competitively binding to Keap1 with TRIM25 via the USP17/Elk-1/CDK6 axis. Oncogene. 2023;42:1333–46.
Zhang Q, Zhang ZY, Du H, Li SZ, Tu R, Jia YF, et al. DUB3 deubiquitinates and stabilizes NRF2 in chemotherapy resistance of colorectal cancer. Cell Death Differ. 2019;26:2300–13.
Lepore Signorile M, Fasano C, Forte G, De Marco K, Sanese P, Disciglio V, et al. Uncoupling p38alpha nuclear and cytoplasmic functions and identification of two p38alpha phosphorylation sites on beta-catenin: implications for the Wnt signaling pathway in CRC models. Cell Biosci. 2023;13:223.
Li X, Huang J, Yu T, Fang X, Lou L, Xin S, et al. Fusobacterium nucleatum promotes the progression of colorectal cancer through Cdk5-activated Wnt/beta-catenin signaling. Front Microbiol. 2020;11:545251.
Prossomariti A, Piazzi G, Alquati C, Ricciardiello L. Are Wnt/beta-catenin and PI3K/AKT/mTORC1 distinct pathways in colorectal cancer? Cell Mol Gastroenterol Hepatol. 2020;10:491–506.
Xu X, Wu G, Han K, Cui X, Feng Y, Mei X, et al. Inhibition of OTUB2 suppresses colorectal cancer cell growth by regulating beta-Catenin signaling. Am J Cancer Res. 2023;13:5382–93.
Tong D, Yuan J, Wang Z, Yu E. EJ USP37 promotes angiogenesis and metastasis in colorectal cancer by facilitating beta-catenin stability. Am J Cancer Res. 2023;13:2323–41.
Shin JM, Yoo KJ, Kim MS, Kim D, Baek KH. Hyaluronan- and RNA-binding deubiquitinating enzymes of USP17 family members associated with cell viability. BMC Genomics. 2006;7:292.
Kim I, Kwak H, Lee HK, Hyun S, Jeong S. beta-Catenin recognizes a specific RNA motif in the cyclooxygenase-2 mRNA 3’-UTR and interacts with HuR in colon cancer cells. Nucleic Acids Res. 2012;40:6863–72.
Perez-Riverol Y, Bandla C, Kundu DJ, Kamatchinathan S, Bai J, Hewapathirana S, et al. The PRIDE database at 20 years: 2025 update. Nucleic Acids Res. 2025;53:D543–D553.
Acknowledgements
This work was supported by a Cancer Research Society (CRS) grant to MJS and a Canadian Institutes for Health Research (CIHR) Grant to MJS and SM.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: MJS, SM, MA, FD, PLT, LV, JS. Performed the experiments: MA, FD, FEM, PLT, LV, GL, DA, JF, EB. Analyzed the data: MJS, SM, MA, FEM, PLT, LV, TH, GL, DA, PPR, JS, SD, LW, EB. Contributed reagents/materials/analysis tools: PPR, JS, LW, EB. Wrote the first draft manuscript: MJS. Edited the manuscript: MJS, SM, MA, FD, FEM, JS, PPR, SD, EB.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Acevedo, M., Dô, F., El-Mortada, F. et al. The deubiquitinase USP17 regulates the expression and activity of the oncogenic driver β-catenin in colorectal cancer. Oncogene 45, 989–998 (2026). https://doi.org/10.1038/s41388-026-03695-y
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41388-026-03695-y