Abstract
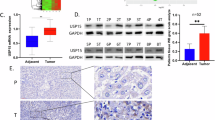
Hepatocellular carcinoma (HCC) is a leading cause of cancer-related mortality globally; however, the molecular drivers remain unclear. Dysregulated cholesterol metabolism is a hallmark of HCC and contributes to tumor progression. The Niemann-Pick type C1 protein (NPC1), a lysosomal cholesterol transporter, is overexpressed in cancers; however, its oncogenic mechanisms in HCC remain unclear. In this study, we identified NPC1 as a critical regulator of HCC progression through dual mechanisms involving p53 destabilization and modulation of cholesterol metabolism. Analysis of the clinical data revealed that NPC1 was significantly upregulated in HCC tissues and correlated with poor prognosis. Functional studies have demonstrated that NPC1 silencing suppresses HCC cell proliferation, both in vitro and in vivo. Mechanistically, NPC1 interacts with deubiquitinase ubiquitin-specific protease 7 (USP7), disrupting its binding to p53 and enhancing p53 ubiquitination and proteasomal degradation. Concurrently, NPC1 modulates cholesterol synthesis and distribution via the p53-SREBP2 axis, and p53 knockdown reverses the cholesterol reduction caused by NPC1 silencing. The pharmacological activation of p53 reversed the decrease in cholesterol levels mediated by the overexpression of NPC1. These findings reveal that NPC1 is a multifaceted oncoprotein in HCC, linking cholesterol metabolism to p53 regulation and highlighting its potential as a therapeutic target for HCC intervention.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Llovet JM, Kelley RK, Villanueva A, Singal AG, Pikarsky E, Roayaie S, et al. Hepatocellular carcinoma. Nat Rev Dis Primers. 2021;7:6.
Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229–63.
Rumgay H, Ferlay J, de Martel C, Georges D, Ibrahim AS, Zheng R, et al. Global, regional and national burden of primary liver cancer by subtype. Eur J Cancer. 2022;161:108–18.
Tsuchida T, Lee YA, Fujiwara N, Ybanez M, Allen B, Martins S, et al. A simple diet- and chemical-induced murine NASH model with rapid progression of steatohepatitis, fibrosis and liver cancer. J Hepatol. 2018;69:385–95.
Jiang Y, Sun A, Zhao Y, Ying W, Sun H, Yang X, et al. Proteomics identifies new therapeutic targets of early-stage hepatocellular carcinoma. Nature. 2019;567:257–61.
Hu N, Fu Y, Li WF, Yang XR, Cao M, Li FF, et al. Chemical mitochondrial uncouplers share a common inhibitory effect on NLRP3 inflammasome activation through inhibiting NFκB nuclear translocation. Toxicol Appl Pharmacol. 2021;414:115426.
Pauels L, De Waele M, Medart L, Debruche M. Two unusual variants of pulmonary intra-lobar sequestration. Interact Cardiovasc Thorac Surg. 2022;35:ivac189.
Che L, Chi W, Qiao Y, Zhang J, Song X, Liu Y, et al. Cholesterol biosynthesis supports the growth of hepatocarcinoma lesions depleted of fatty acid synthase in mice and humans. Gut. 2020;69:177–86.
Mamais A, Wallings R, Rocha EM. Disease mechanisms as subtypes: Lysosomal dysfunction in the endolysosomal Parkinson’s disease subtype. Handb Clin Neurol. 2023;193:33–51.
Gong X, Qian H, Zhou X, Wu J, Wan T, Cao P, et al. Structural insights into the Niemann-Pick C1 (NPC1)-mediated cholesterol transfer and Ebola infection. Cell. 2016;165:1467–78.
Xu J, Chen F, Zhu W, Zhang W. NPC1 promotes autophagy with tumor promotion and acts as a prognostic model for hepatocellular carcinoma. Gene. 2024;897:148050.
Li S, Yan L, Li C, Lou L, Cui F, Yang X, et al. NPC1 controls TGFBR1 stability in a cholesterol transport-independent manner and promotes hepatocellular carcinoma progression. Nat Commun. 2025;16:439.
Pfeffer SR. NPC intracellular cholesterol transporter 1 (NPC1)-mediated cholesterol export from lysosomes. J Biol Chem. 2019;294:1706–9.
Ahmad I, Fatemi SN, Ghaheri M, Rezvani A, Khezri DA, Natami M, et al. An overview of the role of Niemann-Pick C1 (NPC1) in viral infections and the inhibition of viral infections through NPC1 inhibitor. Cell Commun Signal. 2023;21:352.
Davis OB, Shin HR, Lim CY, Wu EY, Kukurugya M, Maher CF, et al. NPC1-mTORC1 signaling couples cholesterol sensing to organelle homeostasis and is a targetable pathway in Niemann-Pick type C. Dev Cell. 2021;56:260–276.e267.
Hussain SP, Schwank J, Staib F, Wang XW, Harris CC. TP53 mutations and hepatocellular carcinoma: insights into the etiology and pathogenesis of liver cancer. Oncogene. 2007;26:2166–76.
Meng X, Franklin DA, Dong J, Zhang Y. MDM2-p53 pathway in hepatocellular carcinoma. Cancer Res. 2014;74:7161–7.
Liu Y, Su Z, Tavana O, Gu W. Understanding the complexity of p53 in a new era of tumor suppression. Cancer Cell. 2024;42:946–67.
Huang Y, Che X, Wang PW, Qu X. p53/MDM2 signaling pathway in aging, senescence and tumorigenesis. Semin Cancer Biol. 2024;101:44–57.
Marine JC, Lozano G. Mdm2-mediated ubiquitylation: p53 and beyond. Cell Death Differ. 2010;17:93–102.
Li M, Brooks CL, Kon N, Gu W. A dynamic role of HAUSP in the p53-Mdm2 pathway. Mol Cell. 2004;13:879–86.
Li M, Chen D, Shiloh A, Luo J, Nikolaev AY, Qin J, et al. Deubiquitination of p53 by HAUSP is an important pathway for p53 stabilization. Nature. 2002;416:648–53.
Fang D, Hu H, Zhao K, Xu A, Yu C, Zhu Y, et al. MLF2 negatively regulates p53 and promotes colorectal carcinogenesis. Adv Sci. 2023;10:e2303336.
Moon SH, Huang CH, Houlihan SL, Regunath K, Freed-Pastor WA, Morris JPT, et al. p53 represses the mevalonate pathway to mediate tumor suppression. Cell. 2019;176:564–580.e519.
Fu L, Deng R, Huang Y, Yang X, Jiang N, Zhou J, et al. DGKA interacts with SRC/FAK to promote the metastasis of non-small cell lung cancer. Cancer Lett. 2022;532:215585.
Sankar K, Gong J, Osipov A, Miles SA, Kosari K, Nissen NN, et al. Recent advances in the management of hepatocellular carcinoma. Clin Mol Hepatol. 2024;30:1–15.
Brown ZJ, Tsilimigras DI, Ruff SM, Mohseni A, Kamel IR, Cloyd JM, et al. Management of hepatocellular carcinoma: a review. JAMA Surg. 2023;158:410–20.
Yang X, Yang C, Zhang S, Geng H, Zhu AX, Bernards R, et al. Precision treatment in advanced hepatocellular carcinoma. Cancer Cell. 2024;42:180–97.
Hu J, Cao J, Topatana W, Juengpanich S, Li S, Zhang B, et al. Targeting mutant p53 for cancer therapy: direct and indirect strategies. J Hematol Oncol. 2021;14:157.
Tavana O, Gu W. Modulation of the p53/MDM2 interplay by HAUSP inhibitors. J Mol Cell Biol. 2017;9:45–52.
Cummins JM, Rago C, Kohli M, Kinzler KW, Lengauer C, Vogelstein B. Tumour suppression: disruption of HAUSP gene stabilizes p53. Nature. 2004;428:1 p following 486.
Cummins JM, Vogelstein B. HAUSP is required for p53 destabilization. Cell Cycle. 2004;3:689–92.
Li X, Wang J, Coutavas E, Shi H, Hao Q, Blobel G. Structure of human Niemann-Pick C1 protein. Proc Natl Acad Sci USA. 2016;113:8212–7.
O’Neill KI, Kuo LW, Williams MM, Lind H, Crump LS, Hammond NG, et al. NPC1 confers metabolic flexibility in triple-negative breast cancer. Cancers. 2022;14:3543.
Fazliyeva R, Makhov P, Uzzo RG, Kolenko VM. Targeting NPC1 in renal cell carcinoma. Cancers. 2024; 16:517.
Millard EE, Srivastava K, Traub LM, Schaffer JE, Ory DS. Niemann-Pick type C1 (NPC1) overexpression alters cellular cholesterol homeostasis. J Biol Chem. 2000;275:38445–51.
Schwend T, Loucks EJ, Snyder D, Ahlgren SC. Requirement of Npc1 and availability of cholesterol for early embryonic cell movements in zebrafish. J Lipid Res. 2011;52:1328–44.
Funding
This study was supported by grants from the Shenzhen Medical Research Funds (A2303050), Shenzhen Science and Technology Innovation Commission Project (JCYJ20250604183742055), Science Technology and Innovation Commission of Shenzhen Municipality (RCBS20210706092253060), Sanming Project of Medicine in Shenzhen (SZSM202411034), Shenzhen Maternity and Child Healthcare Hospital 2022 Annual Research Fund Project (FYA2022011).
Author information
Authors and Affiliations
Contributions
RD, XA, XZ, Y Liu, and JG contributed to the conception and design of the study. RD, XA, FW, and Y Liu developed the methodology. RD, FL, and XA acquired the data. RD, XZ, FL and Y Li performed data analysis and interpretation. RD, XZ, XA, JY, Y Li, SW, and Y Liu contributed to writing, reviewing, and/or revising the manuscript. RD, JY, SW, FW, and Y Liu provided administrative, technical, or material support. RD, XZ, XA, Y Liu, and SW supervised the study.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Deng, R., Zheng, X., Liu, F. et al. The NPC1/USP7/p53 axis regulates cholesterol and promotes the proliferation of hepatocellular carcinoma. Oncogene 45, 1386–1397 (2026). https://doi.org/10.1038/s41388-026-03739-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41388-026-03739-3