Abstract
Despite significant advances in colorectal cancer (CRC) diagnosis and treatment, drug therapy of CRC patients is still confronted with considerable challenges. Carbohydrate response element-binding protein (ChREBP), a glucose-responsive transcription factor regulating glycolysis and de novo lipogenesis, shows elevated expression in human CRC tissues and correlates with poor disease-free survival and overall survival. However, the in vivo role and mechanism of ChREBP in colorectal carcinogenesis remain unclear. We used ChREBP knockout mice, which were intraperitoneally injected with azoxymethane (AOM) followed by dextran sulfate sodium (DSS) in drinking water. In the AOM/DSS-induced colorectal cancer model, carcinogenesis was reduced in ChREBP null mice. In the initial phases of colorectal carcinogenesis, ChREBP deficiency was associated with diminished epithelial cell proliferation and a lower number of aberrant crypt foci, but it had no impact on DNA damage or the severity of colitis. The key transcription factor β-catenin and Wnt target gene expression were both decreased in the colons of ChREBP null mice and in ChREBP-knockdown Caco-2 colorectal cancer cells. In vitro studies demonstrated that ChREBP overexpression promoted β-catenin accumulation, nuclear translocation, and transcriptional activity by interacting with β-catenin, while ChREBP knockdown produced the opposite effects. These findings establish a novel mechanism whereby ChREBP drives CRC progression through Wnt/β-catenin pathway activation, positioning it as both a potential therapeutic target and prognostic biomarker for CRC.

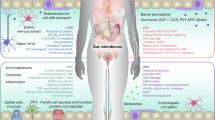
Working model of ChREBP in promoting Wnt signaling and colorectal carcinogenesis. [Figure created with BioRender.com].
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
RNA-seq data that support the findings of this study have been deposited in the Gene Expression Omnibus database with the following accession codes GSE310284 and are publicly available at the date of publication. Any additional information required to reanalyze the data reported in this paper and the materials generated and used in this study are available from the corresponding authors upon reasonable request.
References
Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA: A Cancer J Clin. 2024;74:12–49.
Siegel RL, Wagle NS, Cercek A, Smith RA, Jemal A. Colorectal cancer statistics, 2023. CA: A Cancer J Clin. 2023;73:233–54.
Liang X, Yao Y, Li X, Gao T, Dai X. Cancer mortality trends in China from 2013-2021 and projections to 2030. Cancer Biol Med. 2025;22:1223–39.
Han B, Zheng R, Zeng H, Wang S, Sun K, Chen R, et al. Cancer incidence and mortality in China, 2022. J Natl Cancer Cent. 2024;4:47–53.
West NR, McCuaig S, Franchini F, Powrie F. Emerging cytokine networks in colorectal cancer. Nat Rev Immunol. 2015;15:615–29.
Gupta AK, Pretlow TP, Schoen RE. Aberrant crypt foci: what we know and what we need to know. Clin Gastroenterol Hepatol. 2007;5:526–33.
Ullman TA, Itzkowitz SH. Intestinal inflammation and cancer. Gastroenterology. 2011;140:1807–16.
Markowitz SD, Bertagnolli MM. Molecular origins of cancer: molecular basis of colorectal cancer. N Engl J Med. 2009;361:2449–60.
Clevers H. Wnt/beta-catenin signaling in development and disease. Cell. 2006;127:469–80.
Nelson WJ, Nusse R. Convergence of Wnt, beta-catenin, and cadherin pathways. Sci (N Y, NY). 2004;303:1483–7.
Huber AH, Weis WI. The structure of the beta-catenin/E-cadherin complex and the molecular basis of diverse ligand recognition by beta-catenin. Cell. 2001;105:391–402.
Liu A, Geng X, Li X, Xi Y, Han Q, Wang X, et al. Atypical cadherin CELSR2 acts as a therapeutic target for glioma through WNT3A/β-catenin signaling. Cell death Dis. 2025;16:786.
Amit S, Hatzubai A, Birman Y, Andersen JS, Ben-Shushan E, Mann M, et al. Axin-mediated CKI phosphorylation of beta-catenin at Ser 45: a molecular switch for the Wnt pathway. Genes Dev. 2002;16:1066–76.
Liu C, Li Y, Semenov M, Han C, Baeg GH, Tan Y, et al. Control of beta-catenin phosphorylation/degradation by a dual-kinase mechanism. Cell. 2002;108:837–47.
MacDonald BT, Tamai K, He X. Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell. 2009;17:9–26.
Reya T, Clevers H. Wnt signalling in stem cells and cancer. Nature. 2005;434:843–50.
Schuijers J, Junker JP, Mokry M, Hatzis P, Koo BK, Sasselli V, et al. Ascl2 acts as an R-spondin/Wnt-responsive switch to control stemness in intestinal crypts. Cell Stem Cell. 2015;16:158–70.
Nusse R, Clevers H. Wnt/β-catenin signaling, disease, and emerging therapeutic modalities. Cell. 2017;169:985–99.
Yamashita H, Takenoshita M, Sakurai M, Bruick RK, Henzel WJ, Shillinglaw W, et al. A glucose-responsive transcription factor that regulates carbohydrate metabolism in the liver. Proc Natl Acad Sci USA 2001;98:9116–21.
Iizuka K, Bruick RK, Liang G, Horton JD, Uyeda K. Deficiency of carbohydrate response element-binding protein (ChREBP) reduces lipogenesis as well as glycolysis. Proc Natl Acad Sci USA 2004;101:7281–6.
Stoeckman AK, Ma L, Towle HC. Mlx is the functional heteromeric partner of the carbohydrate response element-binding protein in glucose regulation of lipogenic enzyme genes. J Biol Chem. 2004;279:15662–9.
Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Sci (N Y, NY). 2009;324:1029–33.
Zhang P, Kumar A, Katz LS, Li L, Paulynice M, Herman MA, et al. Induction of the ChREBPβ isoform is essential for glucose-stimulated β-cell proliferation. Diabetes. 2015;64:4158–70.
Collier JJ, Zhang P, Pedersen KB, Burke SJ, Haycock JW, Scott DK. c-Myc and ChREBP regulate glucose-mediated expression of the L-type pyruvate kinase gene in INS-1-derived 832/13 cells. Am J Physiol Endocrinol Metab. 2007;293:E48–56.
Zhang Y, Tang D, Yang C, Lin C, Yuan Y, He M et al. Deficiency of SCAMP5 triggers pancreatic β-cell secretory dysfunction and apoptosis. Advanced science (Weinheim, Baden-Wurttemberg, Germany) 2025:e03072.
Sarrazy V, Sore S, Viaud M, Rignol G, Westerterp M, Ceppo F, et al. Maintenance of macrophage redox status by ChREBP limits inflammation and apoptosis and protects against advanced atherosclerotic lesion formation. Cell Rep. 2015;13:132–44.
Benichou E, Seffou B, Topçu S, Renoult O, Lenoir V, Planchais J, et al. The transcription factor ChREBP Orchestrates liver carcinogenesis by coordinating the PI3K/AKT signaling and cancer metabolism. Nat Commun. 2024;15:1879.
Zhang F, Wu Z, Xiang Y, He Q, Li W, Yang K, et al. SOX4 reprograms fatty acid metabolism through the CHREBP to inhibit ferroptosis in hepatocellular carcinoma. Cell Death Discov. 2025;11:246.
Yu A, Yu P, Zhu Y, Zhu R, Sun R, Ye D, et al. Glucose-induced and ChREBP: MLX-mediated lipogenic program promotes hepatocellular carcinoma development. Oncogene. 2023;42:3182–93.
Engeler M, Karim M, Gischke M, Willer F, Leiner H, Prey J, et al. Carbohydrate-responsive element-binding protein-associated metabolic changes in chemically induced hepatocarcinogenesis mouse model. Int J Mol Sci. 2025;26:6932.
Zhao J, Chen B, Deng Y, Fan L, Yin S, Yu H, et al. ChREBP-mediated choline deprivation and chemokine secretion shape tumor-associated macrophages to promote immune evasion. Cancer Res. 2025;85:4701–4717.
Tong X, Zhao F, Mancuso A, Gruber JJ, Thompson CB. The glucose-responsive transcription factor ChREBP contributes to glucose-dependent anabolic synthesis and cell proliferation. Proc Natl Acad Sci USA 2009;106:21660–5.
Lei Y, Zhou S, Hu Q, Chen X, Gu J. Carbohydrate response element binding protein (ChREBP) correlates with colon cancer progression and contributes to cell proliferation. Sci Rep. 2020;10:4233.
Li Y, Yang D, Tian N, Zhang P, Zhu Y, Meng J, et al. The ubiquitination ligase SMURF2 reduces aerobic glycolysis and colorectal cancer cell proliferation by promoting ChREBP ubiquitination and degradation. J Biol Chem. 2019;294:14745–56.
Lu Y, Tian N, Hu L, Meng J, Feng M, Zhu Y, et al. ERα down-regulates carbohydrate responsive element binding protein and decreases aerobic glycolysis in liver cancer cells. J Cell Mol Med. 2021;25:3427–36.
Neufert C, Becker C, Neurath MF. An inducible mouse model of colon carcinogenesis for the analysis of sporadic and inflammation-driven tumor progression. Nat Protoc. 2007;2:1998–2004.
Iizuka K, Miller B, Uyeda K. Deficiency of carbohydrate-activated transcription factor ChREBP prevents obesity and improves plasma glucose control in leptin-deficient (ob/ob) mice. Am J Physiol Endocrinol Metab. 2006;291:E358–364.
Herman MA, Peroni OD, Villoria J, Schön MR, Abumrad NA, Blüher M, et al. A novel ChREBP isoform in adipose tissue regulates systemic glucose metabolism. Nature. 2012;484:333–8.
Ishii S, Iizuka K, Miller BC, Uyeda K. Carbohydrate response element binding protein directly promotes lipogenic enzyme gene transcription. Proc Natl Acad Sci USA 2004;101:15597–602.
Filhoulaud G, Guilmeau S, Dentin R, Girard J, Postic C. Novel insights into ChREBP regulation and function. Trends Endocrinol Metab: TEM. 2013;24:257–68.
Benhamed F, Denechaud PD, Lemoine M, Robichon C, Moldes M, Bertrand-Michel J, et al. The lipogenic transcription factor ChREBP dissociates hepatic steatosis from insulin resistance in mice and humans. J Clin Investig. 2012;122:2176–94.
Yu Y, Maguire TG, Alwine JC. ChREBP, a glucose-responsive transcriptional factor, enhances glucose metabolism to support biosynthesis in human cytomegalovirus-infected cells. Proc Natl Acad Sci USA 2014;111:1951–6.
Shivapurkar N, Huang L, Ruggeri B, Swalsky PA, Bakker A, Finkelstein S, et al. K-ras and p53 mutations in aberrant crypt foci and colonic tumors from colon cancer patients. Cancer Lett. 1997;115:39–46.
Takayama T, Miyanishi K, Hayashi T, Kukitsu T, Takanashi K, Ishiwatari H, et al. Aberrant crypt foci: detection, gene abnormalities, and clinical usefulness. Clin Gastroenterol Hepatol Assoc. 2005;3:S42–45.
Barker N, Ridgway RA, van Es JH, van de Wetering M, Begthel H, van den Born M, et al. Crypt stem cells as the cells-of-origin of intestinal cancer. Nature. 2009;457:608–11.
Calvisi DF, Wang C, Ho C, Ladu S, Lee SA, Mattu S, et al. Increased lipogenesis, induced by AKT-mTORC1-RPS6 signaling, promotes development of human hepatocellular carcinoma. Gastroenterology. 2011;140:1071–83.
Airley RE, McHugh P, Evans AR, Harris B, Winchester L, Buffa FM, et al. Role of carbohydrate response element-binding protein (ChREBP) in generating an aerobic metabolic phenotype and in breast cancer progression. Br J cancer. 2014;110:715–23.
Kaushik AK, Shojaie A, Panzitt K, Sonavane R, Venghatakrishnan H, Manikkam M, et al. Inhibition of the hexosamine biosynthetic pathway promotes castration-resistant prostate cancer. Nat Commun. 2016;7:11612.
Davies MN, O’Callaghan BL, Towle HC. Glucose activates ChREBP by increasing its rate of nuclear entry and relieving repression of its transcriptional activity. J Biol Chem. 2008;283:24029–38.
Chocarro-Calvo A, García-Martínez JM, Ardila-González S, De la Vieja A, García-Jiménez C. Glucose-induced β-catenin acetylation enhances Wnt signaling in cancer. Mol Cell. 2013;49:474–86.
Chen H, Wu L, Li Y, Meng J, Lin N, Yang D, et al. Advanced glycation end products increase carbohydrate responsive element binding protein expression and promote cancer cell proliferation. Mol Cell Endocrinol. 2014;395:69–78.
Acknowledgements
The authors thank Dr. Lin Li and Dr. Xiaomin Song for providing flag-β-catenin and HA-LEF1. We are grateful to Prof. Yong Zuo for kindly providing the NCM460 cells. This work was supported by the National Natural Science Foundation of China (82173002, 82330080, 82425042, 32371361, 324B200110, 31900562) and the National Key Research and Development Program of China (2019YFA0906100). We appreciate the support from Core Facility of Basic Medical Sciences, Shanghai Jiao Tong University School of Medicine and Shanghai Frontiers Science Center of Cellular Homeostasis and Human Diseases. The authors declare that they have not used AI-generated work in this manuscript.
Author information
Authors and Affiliations
Contributions
MF: Writing-original draft, Software, Methodology, Investigation, Data curation, Conceptualization, Funding acquisition. WH: Methodology, Investigation, Data curation. GJ: Methodology, Investigation, Data curation. LT: Methodology, Investigation, Data curation. ZC: Methodology, Investigation, Data curation, Funding acquisition. YZ: Software, Methodology, Data curation. YL: Software, Conceptualization, Data curation. NT: Conceptualization, Methodology, Investigation. QL: Conceptualization, Methodology, Investigation. PZ: Software, Methodology. LZ: Software, Methodology. YL: Software, Methodology, Investigation, Data curation, Conceptualization. XT: Writing-review & editing, Supervision, Resources, Project administration, Funding acquisition, Conceptualization. JM: Software, Methodology, Investigation, Data curation, Conceptualization. LW: Writing-original draft, Writing-review & editing, Software, Methodology, Investigation, Data curation, Funding acquisition, Conceptualization.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Feng, M., He, W., Ji, G. et al. Carbohydrate responsive element binding protein promotes colorectal carcinogenesis via Wnt/β-catenin pathway. Oncogene (2026). https://doi.org/10.1038/s41388-026-03779-9
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41388-026-03779-9