Abstract
Background
This study examined the effects of two doses of probiotics isolated from human milk on the incidences of necrotizing enterocolitis (NEC) and mortality in preterm infants.
Methods
This multicenter, randomized, double-blind study involved 583 very low birth weight preterm neonates (≤32 6/7 weeks, <1500 g) who started enteral nutrition within 72 h of birth (>5 ml/kg/day) and received either a high-dose (HD, 1 × 109 CFU/day per strain) or a low-dose (LD, 1 × 106 CFU/day per strain) of Limosilactobacillus fermentum CECT5716 (LC40) and Bifidobacterium breve CECT7263 (BfM26) until 36 6/7 weeks postmenstrual age or discharge.
Results
The probiotic combination was well tolerated with no adverse effects. The NEC (2.5% vs. 1.3%; p = 0.156) and mortality (3.9% vs. 3.6%; p = 0.685) rates were not significantly different between the groups (HD vs. LD). These rates were lower than those reported in a co-temporal cohort in the region (SEN1500 cohort).
Conclusion
No significant differences were found between the HD and the LD of the LC40 and BfM26 combination in the incidence of NEC and all-cause mortality. The incidence rates of death and NEC were lower in this study than in the SEN1500 cohort, suggesting that the low dose may suffice for preterm infants. This study supports the safety and tolerability of these probiotics in preterm infants.
Impact
-
The study responds to scientific societies’ request for more clinical trials to strengthen the currently low level of evidence on probiotics’ safety and benefits for preterm infants.
-
The study presents evidence supporting the safety and the effect of two different doses of a combination of two human milk-derived probiotic strains in preterm infants.
-
The study indicates that a low dose of probiotics may be sufficient to achieve beneficial effects in preterm infants. These findings will inform future research on optimizing probiotic dosing in this population.
Similar content being viewed by others
Introduction
Gut dysbiosis in preterm infants has been associated with necrotizing enterocolitis (NEC) and health care-associated sepsis,1two significant pathologies that premature newborns may experience during the postnatal hospitalization period. These conditions are associated with increased risks of morbidity and mortality in this patient group because of their severity and complexity.2,3 Over the past two decades, numerous studies have evaluated the use of probiotics in preterm infants, considering their potential utility in preventing NEC and sepsis. Probiotics may facilitate the establishment of a healthy intestinal microbiota, thereby preventing dysbiosis implicated in the pathogenesis of these conditions.4
To date, the published studies vary in their design, the strains or mixtures of probiotics used, age of initiation and end of administration, doses used and objective of the intervention (prevention of NEC, nosocomial infections, death, combined results).
According to recent recommendations from the European Society for Pediatric Gastroenterology Hepatology and Nutrition (ESPGHAN), only the use of certain strains or combinations of strains can reduce the risk of Grade II or III NEC.5 Based on the median dose used in the RCTs in preterm neonates, ESPGHAN suggests that a daily dose of 3 × 109 cfu/day may be appropriate for neonates of less than 32 weeks gestation.6 However, a recent systematic review to assess the effect of probiotic dose escalation on clinical outcomes and gut microbiota in preterm neonates evidences the lack of data and the urgent need of further studies to address this gap.7
Despite the numerous studies available, the level of evidence in published reviews is still low,8 so scientific societies such as the American Academy of Pediatrics (AAP) and ESPGHAN do not recommend the universal use of probiotics in high-risk newborns in neonatal intensive care units (NICUs), although they encourage continuing with clinical trials in which new strains or combinations of strains are tested.5,6
Meta-analyses on the administration of probiotics in preterm infants suggest that the most significant benefits are derived from combinations of Lactobacillus and Bifidobacterium.9,10 On the other hand, the presence of probiotic bacteria in human milk and their relationship with the beneficial effects of breastfeeding support their use during childhood.11
Limosilactobacillus fermentum CECT5716 (LC40) and Bifidobacterium breve CECT7263 (BfM26) are strains isolated in origin from breast milk that have different anti-infectious, bactericidal, and immune-modulating properties.12,13,14,15,16 Both species have demonstrated both safety and efficacy in various clinical studies carried out on full-term babies.17,18,19,20 These results, together with their human milk origin, pointed them out as good candidates to be evaluated in preterm infants. From the regulatory perspective, the probiotic preparations as dietary supplements have not received approval from the U.S. Food and Drug Administration (FDA) for administration in premature infants. Nonetheless, in the US Limosilactobacillus (formerly Lactobacillus) fermentum CECT5716 strain is Generally Recognized As Safe (GRAS) for its use in infant milk formula for term infants. and in Europe, the species L. fermentum and B. breve are included in the list of Qualified Presumption of Safety (QPS) bacteria released by the European Food Safety Authority (EFSA).21
The probiotic potential of these two strains derived from human milk (LC40 and BfM26) prompted us to explore the effect of a combination administered at two different doses (2 × 106 vs. 2 × 109 CFU) on the incidence of NEC or death in preterm patients ≤ 32 weeks gestation.
Methods
Study design and subjects
A multicenter, randomized, double-blind study was conducted in thirteen hospitals in southern Spain.
Preterm infants with a gestational age between 24 0/7 and 32 6/7 weeks, birth weight less than 1500 g, and who initiated enteral nutrition at a minimum of 5 ml/kg/day within the first 72 h after birth were eligible.
Patients with chromosomal diseases, complex malformations, or those whose clinical situation did not allow the administration of enteral nutrition in the first 72 h of birth were excluded from the study.
The study was conducted in accordance with the Declaration of Helsinki, and the study protocol was approved by the Regional Ethics Committee of Andalusia (Spain) on 20 January 2015. The parents or caregivers signed informed consent forms before the infants were included in the study. The trial was conducted in adherence to CONSORT guidelines and was retrospectively registered in the US Library of Medicine (http://www.clinicaltrials.gov) as NCT06501404.
The Spanish Agency for Medicines and Health Products evaluated the study with the aforementioned probiotic strains (L. Fermentun CECT5716 (LC40) and B. breve CECT7263 (BfM26)) and classified it as an “intervention study without drugs”.
Randomization
The neonates in the study were randomized into two intervention groups. They were included in the study consecutively. The assignment to each group was made with specific software (SIGESMU®), which generated a sequence of random numbers.
Intervention
Each patient received a daily administration of a mixture of the probiotic strains LC40 and B. BfM26 at the dose corresponding to their randomized group. Patients in the high-dose (HD) group were given 1 × 109 CFU/day of each strain, whereas those in the low-dose (LD) group received 1 × 106 CFU/day of each strain. For ethical reasons, a placebo group was not included in the study. At the time of study initiation, existing evidence was deemed sufficient to support the beneficial effects of probiotics in this population. Consequently, withholding potentially effective treatment from a control group was considered ethically unjustifiable.
The probiotic strains were facilitated by Biosearch SAU, a Kerry Group company (Granada, Spain). The probiotic mixture was prepared in capsules containing 3 × 109 CFU of each strain for the HD group and in capsules containing 3 × 106 CFU of each strain for the LD group. The Department of Galenics of the Faculty of Pharmacy of the University of Granada (Spain) manufactured the product. The probiotic mixture was kept refrigerated at a temperature between 2 and 8 °C. Viability of both strains was tested after capsules production for each batch and annually by colony-count enumeration using an adaptation of the method described in ISO 20128:2006. Basically, for LC40 strain a step of rehydration for 45 min at 37 °C ± 1 °C was included in ISO protocol in the first dilution. For BFM26 cysteine 0.1% is added to peptone buffered in serial 1/10 dilutions step.
At the time of administration, the content of each capsule was reconstituted with 3 ml of sterile injection water, and 1 ml was administered via a nasogastric tube to each patient once daily. The patient subsequently received the corresponding enteral nutrition intake, and the remaining 2 ml of probiotic mixture was discarded.
The intervention commenced within the first 72 h of life if the patient was receiving enteral nutrition at a volume greater than 5 ml/kg/day. The intervention continued until the patient reached a postmenstrual age of 36 6/7 weeks or until hospital discharge, whichever occurred first.
Each participating center adhered to its enteral nutrition protocol, which included the option of administering donated human milk from the first day of life. The decision to interrupt the intervention for clinical reasons was at the discretion of the care team at each center. In cases of clinical deterioration necessitating the suspension of enteral nutrition, the responsible physicians temporarily discontinue the administration of the product. If the interruption of the enteral nutrition exceeded 7 days, the patient was excluded from the study.
Study outcomes and data collection
The primary outcomes of the study were the incidence of NEC (modified Bell classification stages II and III)22,23 and all-cause mortality during the hospital stay in both intervention groups.
The secondary outcomes studied were as follows:
-
Incidence of health care-associated sepsis (late-onset sepsis, LOS), diagnosed in accordance with the criteria used by the Castrillo Group for neonatal infections in Spain (compatible clinical symptoms, elevation of acute phase reactants and positive blood culture24).
-
Time to reach complete enteral nutrition (140 ml/kg/day).
-
Number of central vascular catheter days.
-
Length of hospital stay.
-
Incidence of bronchopulmonary dysplasia
-
Incidence of patent ductus arteriosus (PDA).
-
Growth at 36 6/7 weeks of postmenstrual age was evaluated in terms of the increase in body weight (g/kg per day), which was calculated using the exponential method, length (cm per week) and head circumference (cm per week).25 Similarly, the z scores were calculated for boys and girls for weight-for-age, length-for-age and circumference for age at birth, week 32 and week 37 of gestation using the Fenton curve as a ref.26
-
Due to the absence of a placebo group in this study, the protocol included a comparative evaluation to a co-temporal cohort to better understand the effect of the probiotic combination. Specifically, the incidence rates of NEC, death, LOS, and PDA in the study population were compared with those reported in the SEN1500 cohort from 2016 to 2020. The SEN1500 database collects data on newborns weighing less than 1500 grams at birth from 60 Spanish hospitals, with an average annual inclusion of approximately 2,500 patients,27
Statistical analysis
For the sample size calculation, we used incidence data on NEC and mortality from the SEN1500 cohort. According to this database, between 2009 and 2014, the incidence of NEC stage IIA or higher was 7.14%, while the combined outcome of NEC or death had an incidence of 19.92%. Assuming a relative risk of 0.53 in the HD probiotic group (based on published meta-analytic data28), the expected incidence in this group would be approximately 10.5%. To test the non-inferiority hypothesis (H₀: the difference between proportions is less than 5%) using a one-sided normal approximation test for independent samples, and assuming a 9% incidence in the LD group, we estimated that 258 infants per group would be required to achieve 80% power with a one-sided significance level of 5%. Accounting for an estimated 10% dropout rate, we planned to recruit 287 patients per group, totalling 574 participants in the study. The sample size calculation was performed using the ENE 3.0 program.
A descriptive study of the main variables was conducted, with quantitative variables being presented as the means and standard deviations (SDs). Qualitative variables were presented as counts and percentages.
To compare quantitative variables between treatment groups, the Mann‒Whitney U test was used, and for qualitative variables, the chi‒square test with Pearson’s correction was applied.
For dichotomous variables, logistic regression models were established with the treatment arm as the independent variable. Adjusted analyses included gestational age at birth and infant sex, as these are strong prognostic factors for NEC and mortality in preterm neonates., Stratified models by gestational age (<30 weeks/≥30 weeks) and birth weight (≤1000 g/>1000 g) were also run. Adjusted relative risk (aRR) for these variables was reported.
Mortality, LOS, PDA, and NEC rates were compared with those reported in the SEN1500 cohort from 2016 to 202027 using the Z test. Additionally, z scores for weight-for-age, length-for-age, and head circumference-for-age were calculated for boys and girls at birth, at 32 weeks, and at 37 weeks of gestation, using the Fenton growth chart as a reference.26
In all analyses, a significant level of 5% was predetermined. These analyses were conducted using SPSS 20.0 and the open-source statistical software R version 4.2.2.
Results
Between May 2016 and June 2020, 583 preterm infants weighing less than 1500 g, with a gestational age between 24 0/7 and 32 6/7 weeks, were recruited. A total of 279 preterm infants were included in the HD group, and 304 were included in the LD group.
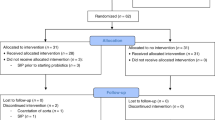
Throughout the study, 22 patients discontinued the administration of the product for various reasons. Ten patients in the HD group and 12 in the LD group were withdrawn from the study (Fig. 1).
Flow chart of the clinical study.
The perinatal characteristics of the study participants were similar between the two groups (Table 1), with no statistically significant differences in gestational age, birth weight, sex, use of medication, type of delivery, preeclampsia or infectious risk factors such as premature rupture of membranes and chorioamnionitis. The Apgar score was significantly different between the groups (p = 0.017), although no significant differences were found in terms of the need for or type of resuscitation. There were no significant differences in the timing of enteral nutrition initiation, the initial volume received, or the type of nutrition received at the start of the study (Table 1).
Additionally, it should be noted that there were no differences between the groups in the type of nutrition received at 32 weeks and at 36 6/7 weeks of postmenstrual age (Supplementary Table S1).
With respect to the primary outcomes, no significant differences were observed between the groups in terms of the incidence of grade II or III NEC according to the modified Bell’s classification or in the incidence of all-cause mortality (Table 2). The combined incidence of NEC and/or death was 5.7% (16/279) in the HD group and 4.3% (13/304) in the LD group, yielding an adjusted risk ratio (RR) of 0.67 (95% CI: 0.33–1.36; p = 0.270). The absolute risk difference (low dose minus high dose) was −1.45%, with the upper bound of the one-sided 95% confidence interval at 1.53%, remaining below the predefined non-inferiority margin of 5%.
The results were stratified on the basis of gestational age to assess the impact of the intervention according to this variable. Thus, infants born at less than 30 gestational weeks, a total of 312 infants, were included in one cohort, and those born between 30 and 32 6/7 gestational weeks, 272 infants, were included in another cohort. As expected, the rates of NEC and mortality were higher in the cohort of younger gestational age patients, but no significant differences were observed between the two intervention groups in either of the two cohorts (Table 3).
With respect to the secondary outcomes, no significant differences were found between the groups in any of them, including time to full enteral nutrition, days with a central catheter, average hospital stay, or the incidence of LOS and bronchopulmonary dysplasia (Table 4).
On the other hand, a lower incidence of PDA was detected in the HD group than in the LD group (16.1% vs. 22.7%, raw p value = 0.049). However, the significance did not persist when the analysis was adjusted for gestational age and sex (p value = 0.237, Table 4). Further analysis revealed that this difference was significant in infants whose birth weight was 1000 g or less (16.44% in the HD group vs. 39.76% in the LD group, p value = 0.033).
Since sepsis is a risk factor for PDA, the incidence of LOS plus PDA was evaluated. No significant differences were found between the groups (3.7% in the HD group vs. 7.2% in the LD group; p value = 0.203; Table 4). However, in infants with a gestational age less than 30 weeks, a significant difference was observed, with a lower incidence of LOS plus PDA in the HD group than in the LD group (4.2% vs. 11.8%; p value = 0.044).
The comparison to the co-temporal cohort SEN1500 was showed in the Supplementary Table S2. It was observed a significantly lower incidence of NEC in the study population that received the probiotic combination compared to the SEN1500 recorded data (1.89% in the study sample versus 5.33% in the SEN1500 cohort, p value < 0.001). Similarly, the incidence of mortality was significantly lower in this study compared to the reported in the SEN1500 regional cohort (3.77% vs. 10.38%, p value < 0.001). On the contrary, the incidence of LOS and PDA observed in the study sample were similar to that reported in the SEN1500 cohort (19.9% vs. 22.6%, p value = 0.134 and 22.7% vs. 25.1%, p value = 0.387, respectively). However, the HD probiotic group in the study cohort showed a significantly lower incidence of PDA than the SEN1500 cohort (16.1% vs. 24.9%, p value < 0.001) (Supplementary Table S2).
Growth during the intervention period was similar in both groups. No significant differences were found between the HD and LD groups regarding daily weight gain during the intervention (13.84 ± 2.30 g/kg/day vs. 14.06 ± 2.26 g/kg/day; p = 0.279, adjusted for sex and gestational age at birth). There were also no differences in length gain (0.97 ± 0.35 cm/week vs. 1.03 ± 0.35 cm/week; p = 0.104) or head circumference (0.82 ± 0.28 cm/week vs. 0.81 ± 0.28 cm/week; p = 0.862). Similarly, no differences were found between the groups in terms of z scores for body weight, length, or head circumference at birth or at 32 or 37 weeks of gestational age (Table 5).
No adverse effects attributable to the administration of the study product were recorded.
In cases of sepsis, the identified causal agents in blood samples were species not classified under the genera Lactobacillus or Bifidobacterium.
The administration of the study product was well tolerated in both intervention groups.
Discussion
The present clinical study showed the incidence of NEC and death in a cohort of preterm infants ≤ 32 weeks gestation that consumed a combination of the breastmilk-isolated probiotic strains LC40 and BfM26 at two different doses (2 × 109 and 2 × 106). No significant differences were found in the main outcomes between the two probiotic doses. However, the incidences of NEC and death observed in this study were significantly lower than the incidences recorded in the SEN1500 regional cohort (2016–2020), suggesting that the lower dose may be adequate to benefit preterm infants. Furthermore, this study provides evidence regarding the safety and tolerability of the strains in this vulnerable population.
While earlier meta-analyses supported the benefits of probiotics in preterm infants,28 more recent systematic reviews are more cautious in their conclusions, particularly regarding the level of evidence for NEC and mortality.29 In our study, we compared the effects of a dose of the probiotic combination in line with the recommendation (2 × 109 CFU/day) with a much lower dose compared to other studies (2 × 106 CFU/day). A daily dose of 3 × 109 CFU/day based on the median dose used in RCTs has been recommended for preterm very low birth weight (VLBW) infants born at <32 weeks,30 so this was the dose selected as the high dose. The low dose (2 × 106 CFU) was chosen because it was the lowest recorded in some studies.31,32 On the other hand, various studies indicate that an infant consuming approximately 800 ml of breast milk per day could receive a bacterial load exceeding 105–107 CFU. Therefore, our low dose would provide a bacterial count comparable to that of an exclusively breastfed infant.33,34 Although only a few studies have evaluated the dose‒response effect of probiotic intervention in premature infants, their results showed no significant differences in the clinical response with respect to the incidence of NEC, sepsis or mortality.35 We drew similar conclusions, as no differences in these parameters were observed between the intervention groups.
A limitation of the clinical study is the lack of a placebo control arm, not allowed by the ethical committee that evaluated the study´s protocol, which considered that deprivation of treatment in control group was not ethical due to the existing evidence about probiotics in preterm infants. In order to better understand the effect of the probiotic combination, it was included in the protocol the comparison of the incidence rates of NEC, death, LOS and PDA in the studied population compared with those reported in the SEN1500 cohort from 2016 to 2020. The SEN1500 collects data on preterm infants from 60 hospitals in Spain, including 9 of the 13 hospitals participating in the present study.27 Between 2016 and 2020, during which the present study was conducted, the SEN1500 database collected data from 11,521 preterm infants born at more than 24 weeks of gestation. This database provides highly accurate information on the incidence rates of various conditions in preterm infants within the region where the study was conducted. The results showed very low incidence rates of NEC and of all-cause mortality in our study compared with those reported in the SEN1500 cohort, but similar incidence rates of LOS. Although the comparison with SEN1500 does not hold the same weight as a comparison with a control intervention group, the observed effects on NEC and mortality rates are consistent with expectations noted in the literature, particularly in clinical studies using multi-strain probiotic combinations. In the ProPrems Trial, which included 1099 preterm infants whose birth weights were less than 1500 g, Jacobs et al. reported a significant reduction in NEC from 4.4 to 2% in the intervention group compared to placebo. However, they found no significant differences in mortality (4.9% vs 5.1%) or LOS (13.1% vs 16.2%).36 They used a mixture of Bifidobacterium (2 different strains) and Streptococcus thermophilus. On the other hand, Costeloe et al., which tested the administration of B. breve BBG-001 versus placebo in 1315 patients under 30 weeks gestation (PiPS Trial) in different neonatal ICUs in southern England, found no significant differences between groups in the primary outcomes (NEC incidence rates were 9% and 10%, mortality rates of 8 and 9% and incidences of late-onset sepsis (LOS), 11% and 12%).37
The protective effect of probiotics against NEC has been associated with TLR4 signaling attenuation or TLR9 activation.38 In a model of human primary intestinal epithelial cells, L. fermentum CECT5716 was able to activate TLR9 at high concentrations. Accordingly, the probiotic strain induced a reduction in the level of IL6, one of the early interleukins and key regulators during inflammation.39 In a NEC mouse model, L. fermentum CECT5716 alleviated NEC symptoms via mechanisms related to intestinal epithelial regeneration.40 Although further studies will be needed, the results obtained in these pre-clinical models might partly explain the mechanism of action for the observed effects in NEC.
An intriguing observation is the reduced incidence of patent ductus arteriosus (PDA) in infants weighing under 1000 g who received the high dose of the probiotic combination. This effect might be linked to the ability of the LC40 strain to modulate nitric oxide (NO) through inducible nitric oxide synthase (iNOS) and reduce inflammatory cytokines, as observed in animal models of septic shock and inflammatory bowel disease.41,42 In septic conditions, the increased systemic inflammatory response can lead the ductus arteriosus to reopen, increasing the PDA risk. In our study, those infants under 30 weeks’ gestation receiving the high dose of probiotics had a lower incidence of LOS and PDA than those on the low dose, suggesting a stronger anti-inflammatory effect. However, this should be considered as preliminary results and further research is needed to confirm this observation.
Another important point raised by some authors is the risk of systemic infections in highly vulnerable and immunosuppressed patients due to the administration of viable microorganisms. Most published studies scarcely reported any side effects of the administered products, and reports of health care-associated infections (HAIs) caused by probiotic strains were anecdotal.43,44 However, some researchers have been suggested that the possibility of an exaggerated proinflammatory response in the gastrointestinal tract and the risk of bacterial translocation (e.g., probiotic sepsis) would be increased at high doses of probiotics.35 Thus, in this context, these findings suggest that lower doses may offer a safer alternative, underscoring the need for more dose–response research in this vulnerable population. Importantly, the safety of the combination of the probiotics LC40 and BfM26 has been demonstrated in this group of preterm infants. No adverse effects attributable to the administration of the study product were recorded, and no infectious cases were attributed to the probiotic strains (Lactobacillus and Bifidobacterium) used as study product. Additionally, the growth of the infants was similar at both doses, with no significant differences in growth velocity or z scores for weight, length or head circumference. Moreover, the weight gain velocity was similar to that of the Spanish SEN1500 cohort45 and the Fenton cohort25 and comparable to that of the normal intrauterine growth rate (15 g/kg/d and 1 cm/week) according to the American Academy of Pediatrics.46
The main limitation of the study may be the requirement for patients to have initiated enteral feeding within the first 72 h of birth for inclusion, which would exclude the most severely ill patients in the early days of life. As previously discussed, an additional limitation of this study is the absence of a placebo-controlled group, which restricts the ability to rigorously assess the efficacy of the probiotic strains relative to a control group. Finally, some limitations in our comparative analysis should be acknowledged. The SEN1500 published data, while contemporaneous with our clinical study, represent aggregated outcomes across multiple centers, and do not account for center-specific practices or probiotic use. This limited our ability to control inter-center variability, which might influence NEC and mortality rates.
In conclusion, no significant differences were found in the main outcomes (NEC, all-cause mortality, and LOS) between the two probiotic doses. However, the incidence rates of death and NEC were lower in this study than in the SEN1500 cohort, suggesting the low dose (2 × 106 CFU/day) of the combination of the probiotic strains L. fermentum CECT5716 (LC40) and B. breve CECT7263 (BfM26) may be sufficient to benefit preterm infants. Additionally, this study provides evidence on the safety and tolerability of the strains used in this population. Further clinical studies controlled by placebo should be done to confirm the efficacy of the probiotic combination and define the optimal dosing strategy.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Campos-Martinez, A. M., Exposito-Herrera, J., Gonzalez-Bolivar, M., Fernandez-Marin, E. & Uberos, J. Evaluation of risk and preventive factors for necrotizing enterocolitis in premature newborns. A systematic review of the literature. Front. Pediatr. 10, 874976 (2022).
Jones, I. H. & Hall, N. J. Contemporary outcomes for infants with necrotizing enterocolitis-a systematic review. J. Pediatr. 220, 86–92 e83 (2020).
Fleischmann, C. et al. Global incidence and mortality of neonatal sepsis: a systematic review and meta-analysis. Arch. Dis. Child 106, 745–752 (2021).
Chang, H. Y. et al. Multiple strains probiotics appear to be the most effective probiotics in the prevention of necrotizing enterocolitis and mortality: an updated meta-analysis. PLoS ONE 12, e0171579 (2017).
van den Akker, C. H. P. et al. Probiotics and preterm infants: a position paper by the European Society for Pediatric Gastroenterology Hepatology and Nutrition Committee on nutrition and the European Society for Pediatric Gastroenterology Hepatology and Nutrition working group for probiotics and prebiotics. J. Pediatr. Gastroenterol. Nutr. 70, 664–680 (2020).
Poindexter, B., Committee On Fetus and Newborn. Use of probiotics in preterm infants. Pediatrics 147, e2021051485 (2021).
Olsen, R., Greisen, G., Schroder, M. & Brok, J. Prophylactic probiotics for preterm infants: a systematic review and meta-analysis of observational studies. Neonatology 109, 105–112 (2016).
Sharif, S., Meader, N., Oddie, S. J., Rojas-Reyes, M. X. & McGuire, W. Probiotics to prevent necrotising enterocolitis in very preterm or very low birth weight infants. Cochrane Database Syst. Rev. 7, CD005496 (2023).
Robertson, C. et al. Incidence of necrotising enterocolitis before and after introducing routine prophylactic lactobacillus and bifidobacterium probiotics. Arch. Dis. Child Fetal Neonatal Ed. 105, 380–386 (2020).
Wang, Q., Dong, J. & Zhu, Y. Probiotic supplement reduces risk of necrotizing enterocolitis and mortality in preterm very low-birth-weight infants: an updated meta-analysis of 20 randomized, controlled trials. J. Pediatr. Surg. 47, 241–248 (2012).
Martin, R. et al. Human milk is a source of lactic acid bacteria for the infant gut. J. Pediatr. 143, 754–758 (2003).
Diaz-Ropero, M. P. et al. Two lactobacillus strains, isolated from breast milk, differently modulate the immune response. J. Appl. Microbiol. 102, 337–343 (2007).
Martin, R. et al. Probiotic potential of 3 lactobacilli strains isolated from breast milk. J. Hum. Lact 21, 8–17 (2005).
Olivares, M., Diaz-Ropero, M. P., Martin, R., Rodriguez, J. M. & Xaus, J. Antimicrobial potential of four Lactobacillus strains isolated from breast milk. J. Appl. Microbiol. 101, 72–79 (2006).
Algieri, F. et al. Intestinal anti-inflammatory effects of probiotics in DNBS-colitis via modulation of gut microbiota and microRNAs. Eur. J. Nutr. 60, 2537–2551 (2021).
de la Visitación, N. et al. Probiotics prevent hypertension in a murine model of systemic lupus erythematosus induced by toll-like receptor 7 activation. Nutrients 13, 2669 (2021).
Gil-Campos, M. et al. Lactobacillus fermentum cect 5716 is safe and well tolerated in infants of 1-6 months of age: a randomized controlled trial. Pharm. Res. 65, 231–238 (2012).
Maldonado, J. et al. Human milk probiotic Lactobacillus fermentum CECT5716 reduces the incidence of gastrointestinal and upper respiratory tract infections in infants. J. Pediatr. Gastroenterol. Nutr. 54, 55–61 (2012).
Maldonado, J. et al. Evaluation of the safety, tolerance and efficacy of 1-year consumption of infant formula supplemented with Lactobacillus fermentum Cect5716 Lc40 or Bifidobacterium breve Cect7263: a randomized controlled trial. BMC Pediatr. 19, 361 (2019).
Maldonado-Lobon, J. A. et al. Efficacy of Bifidobacterium Breve Cect7263 for infantile colic treatment: an open-label, parallel, randomised, controlled trial. Benef. Microbes 12, 55–67 (2021).
Hazards, E. P. O. B. et al. Scientific opinion on the maintenance of the list of QPS biological agents intentionally added to food and feed (2010 update). EFSA J. 8, 1944 (2010).
Bell, M. J. et al. Neonatal necrotizing enterocolitis. Therapeutic decisions based upon clinical staging. Ann. Surg. 187, 1–7 (1978).
Walsh, M. C. & Kliegman, R. M. Necrotizing enterocolitis: treatment based on staging criteria. Pediatr. Clin. North Am. 33, 179–201 (1986).
Lopez Sastre, J. B., Fernandez Colomer, B., Coto Cotallo, G. D. & de la Rosa Fraile, M. Sepsis En El periodo neonatal. Evid. Pediatr. 4, 68 (2008).
Fenton, T. R. et al. An attempt to standardize the calculation of growth velocity of preterm infants-evaluation of practical bedside methods. J. Pediatr. 196, 77–83 (2018).
Fenton, T. R. & Kim, J. H. A systematic review and meta-analysis to revise the fenton growth chart for preterm infants. BMC Pediatr. 13, 59 (2013).
SENEO. Sen1500 Morbimortalidad, https://www.seneo.es/index.php/comisiones/comisiones/redes-neonatales/sen1500/sen1500-morbimortalidad
AlFaleh, K. & Anabrees, J. Probiotics for prevention of necrotizing enterocolitis in preterm infants. Cochrane Database Syst. Rev. 10, CD005496 (2014).
Chi, C. et al. Effects of Probiotics in Preterm Infants: A Network Meta-Analysis. Pediatrics 147 (2021).
Deshpande, G. C., Rao, S. C., Keil, A. D. & Patole, S. K. Evidence-Based Guidelines for Use of Probiotics in Preterm Neonates. BMC Med 9, 92 (2011).
Fernandez-Carrocera, L. A. et al. Double-Blind, Randomised Clinical Assay to Evaluate the Efficacy of Probiotics in Preterm Newborns Weighing Less Than 1500 G in the Prevention of Necrotising Enterocolitis. Arch. Dis. Child Fetal Neonatal Ed. 98, F5–F9 (2013).
Lin, H. C. et al. Oral Probiotics Reduce the Incidence and Severity of Necrotizing Enterocolitis in Very Low Birth Weight Infants. Pediatrics 115, 1–4 (2005).
Heikkila, M. P. & Saris, P. E. Inhibition of Staphylococcus Aureus by the Commensal Bacteria of Human Milk. J. Appl Microbiol 95, 471–478 (2003).
Hunt, K. M. et al. Characterization of the Diversity and Temporal Stability of Bacterial Communities in Human Milk. PLoS One 6, e21313 (2011).
Rath, C., Athalye-Jape, G., Rao, S. & Patole, S. Effect of Probiotic Dose Escalation on Gut Microbiota and Clinical Outcomes in Preterm Infants-a Systematic Review. Children (Basel) 10 (2023).
Jacobs, S. E. et al. Probiotic Effects on Late-Onset Sepsis in Very Preterm Infants: A Randomized Controlled Trial. Pediatrics 132, 1055–1062 (2013).
Costeloe, K. et al. Bifidobacterium Breve Bbg-001 in Very Preterm Infants: A Randomised Controlled Phase 3 Trial. Lancet 387, 649–660 (2016).
Hackam, D. J. & Sodhi, C. P. Toll-Like Receptor-Mediated Intestinal Inflammatory Imbalance in the Pathogenesis of Necrotizing Enterocolitis. Cell Mol. Gastroenterol. Hepatol. 6, 229–238.e221 (2018).
Hedegger, K., Hommel, T., Schaubeck, M., Gimpfl, M. & Dahlhoff, M. Different Infant Formulas Can Activate Toll-Like Receptor 9 in Vitro and Inhibit Interleukin 6 in Human Primary Intestinal Epithelial Cells. Eur. J. Nutr. 64, 16 (2024).
Zhang, F., Yan, X., Chen, X., Li, S. & Han, S. Human Milk-Derived Lactobacillus Fermentum Drives Intestinal Stem Cell Regeneration to Repair Intestinal Injury in Necrotizing Enterocolitis. JPGN Rep. 5, s1319 (2024).
Arribas, B. et al. Evaluation of the Preventative Effects Exerted by Lactobacillus Fermentum in an Experimental Model of Septic Shock Induced in Mice. Br. J. Nutr. 101, 51–58 (2009).
Peran, L. et al. Lactobacillus Fermentum, a Probiotic Capable to Release Glutathione, Prevents Colonic Inflammation in the Tnbs Model of Rat Colitis. Int J. Colorectal Dis. 21, 737–746 (2006).
Kulkarni, T. et al. Probiotic Sepsis in Preterm Neonates-a Systematic Review. Eur. J. Pediatr. 181, 2249–2262 (2022).
Manzoni, P. et al. Prevention of Nosocomial Infections in Neonatal Intensive Care Units. Am. J. Perinatol. 30, 81–88 (2013).
Garcia-Munoz Rodrigo, F., Figueras Aloy, J., Saavedra Santana, P. & Garcia-Alix, A. [Postnatal Growth at Hospital Discharge in Extremely Premature Newborns in Spain]. Pediatr. (Barc.) 87, 301–310 (2017).
American Academy of Pediatrics Committee on Nutrition: Nutritional Needs of Low-Birth-Weight Infants. Pediatrics 75, 976-986 (1985).
Acknowledgements
We would like to thank all the staff of the Hospitals for their support, and all parents who agreed to participate in this study.
Funding
The authors received no specific funding for this work. Kerry group provided the combination of probiotics tested in the study and the funding for the open access.
Author information
Authors and Affiliations
Contributions
J.A.H.S. conception and design of the study; A.A.O., T.S.T., E.M.A., L.M.P., J.D.D.R., M.G.A., L.S.L., J.A.M.L., D.M.N., M.T.B.G., J.A.R.M., C.M.D., J.M.L.G., and M.P.C.: acquisition of data; R.B.R. and M.E.S.: analysis of the data; J.A.H.S., M.O. and R.B.R.: interpretation of the data; J.A.H.S., E.M.A., L.S.L., M.P.C., M.O, and R.B.R.: drafted the article and JA.H.S., A.A.O., T.S.T., E.M.A., M.E.S., M.O., and R.B.R. revised it critically for important intellectual content. All the authors have read and approved the last version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
J.A.H.S., M.O. and R.B.R. are inventors of a filed patent (pending) based on the study results. M.E.S., J.A.M.L., M.O. and R.B.R. are workers of Kerry Group P.L.C., owner of the filed patent (pending) based on the study results. The rest of authors declare that they have no competing interests.
Consent for publication
The parents or caregivers signed informed consent forms before the infants were included in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hurtado Suazo, J.A., Alonso Ojembarrena, A., Sánchez Tamayo, T. et al. L.Fermentum CECT5716 and B.Breve CECT7263 on premature infants morbidities: a randomized clinical trial. Pediatr Res (2025). https://doi.org/10.1038/s41390-025-04633-6
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41390-025-04633-6