Abstract
Background
Executive functioning (EF) deficits are frequently observed in preterms. EF development is linked to the prefrontal cortex and sleep-regulating homeostatic processes. As sleep is an essential driver of early brain maturation, curtailment of sleep in the NICU may be unfavorable. This study examined whether neonatal sleep behavior influences EF at 2 years corrected age in preterm children.
Methods
76 preterm infants ( < 34 weeks gestation and/or 1500 g) underwent overnight polysomnography before discharge. Sleep stages—Total Sleep Time, Active Sleep (AS), Quiet Sleep (QS) and Transitional Sleep—were quantified using an automated sleep algorithm. EF was assessed at 2 years corrected age, focusing on spatial working memory, cognitive flexibility, and inhibitory control. General linear models were used, adjusting for confounders.
Results
More Total Sleep Time was significantly associated with higher overall EF scores, and the subtest for spatial working memory. AS and QS durations were influenced by postmenstrual age. Longer AS bouts were linked to increased Total Sleep Time. However, individual AS or QS percentages were not directly associated with EF. Higher levels of Transitional Sleep were related to lower EF performance.
Conclusions
Neonatal sleep duration is positively associated with better EF outcomes at 2 years. Protecting sleep in the NICU may support early brain development and executive functioning.
Impact
-
This study links objective neonatal sleep measurements—using polysomnography and automated sleep staging—with later executive functioning development in preterm children.
-
Neonatal sleep duration, particularly Total Sleep Time, is positively associated with executive functioning at 2 years in children born preterm.
-
This study highlights neonatal sleep as a potential early marker of altered brain development.
-
The findings support the importance of protecting and improving sleep in the NICU as a possible modifiable factor that could enhance early brain maturation and neurodevelopment.
Similar content being viewed by others
Introduction
Despite a decline in neonatal death and severe cerebral palsy, there remains a high incidence of cognitive impairment, social and emotional challenges and altered executive functioning (EF) among children and young adults born preterm.1,2,3,4 The immature brain is vulnerable to both primary injury as well as secondary alterations in brain development5 and numerous studies have shown morphological alterations of the frontal and temporal brain regions associated with these deficits.6,7,8 Understanding the effect of early key factors during the period spent in the neonatal care unit (NICU), that may modulate early brain dysmaturation and their neurodevelopmental consequences, can improve early care practices and neuroprotective strategies.9 EF reflects the integrity of early brain networks and begins to emerge well before formal diagnoses of neurodevelopmental conditions are possible. Because EF can be impaired across a wide range of conditions (including autism spectrum disorder, attention deficit hyperactivity disorder, and learning disabilities)—all of which are more common in preterm populations), it serves as a valuable early marker of atypical development.10 Moreover, EF strongly predicts long-term functional outcomes, including behavioral and social competence and academic performance.11 Importantly, EF skills are amenable to improvement through early intervention,12 making EF a promising and modifiable indicator of neurodevelopmental risk—especially relevant for children born preterm.
Electroencephalography (EEG) is a crucial component of polysomnography (PSG), which is the golden standard to quantify sleep.13,14,15,16,17 EEG provides a non-invasive means of measuring early cortical activity. The natural emergence of sleep states in the newborn is dependent on the maturation of the central nervous system and is a reliable indicator of normal development of the brain. In turn, neonatal sleep plays a critical role in the development of neuronal pathways.18 Sleep-specific, endogenous motor activity (e.g. twitches and limb movements) in Active Sleep (AS) evokes somatotopic cortical activity in the preterm brain and formation of early neuronal networks. This endogenous neural (non-stimulus dependent) stimulation during AS will gradually decline and fully disappear by the end of gestation. This differs crucially from the later experience-dependent (environmental stimuli during wakefulness) fine tuning of cortical networks. The uniquely regulated, spontaneous neural activity during AS and subsequent consolidation by optimizing neuronal networks via mechanisms of synaptic homeostasis during Quiet Sleep (QS) lays the groundwork for early neuronal circuitry and underscores the fundamental importance of sleep over wakefulness in early life.19,20 By term equivalent age, distinct sleep states emerge with two AS (AS1 and AS2 or Low Voltage Irregular pattern, (LVI) and two QS (Quiet Sleep Trace Alternant (QS-TA) pattern and a Quiet Sleep High Voltage Slow Wave Sleep (QS-HVS)).16
Visual EEG analysis is time-consuming, requires expertise and subject to inter-rater variability. These factors limit the routine clinical use of EEG, despite its potential to provide valuable insights into neonatal brain health. Computational EEG analyses based on machine learning have been developed to automate and objectify brain function evaluation.21,22 Automated sleep staging can provide continuous, objective measurements that would otherwise be impractical for clinicians to obtain.23,24
Both in children and adults, the crucial role of sleep and the impact of inadequate and interrupted sleep on general health but also difficulties in learning, memory and behavior have been established.25,26 Curtailment of sleep by different neonatal perturbations such as noxious stimulation, cardiovascular instability, or exposure to the extra-uterine environment, may be detrimental for further brain maturation and neuronal plasticity.9,27,28
In this study, we investigated to what extent indices of prematurity and early sleep behavior are associated with altered EF at 2 years corrected age (CA). To provide continuous and objective measures, we quantified sleep using an automated sleep algorithm.21,29 We hypothesized that a higher percentage of Total Sleep Time was related to better EF at preschool age. We subsequently compared other sleep features, focusing on predefined sleep features of interest, in relation to the composite EF score and the subtests of EF.
Preterm born neonates were followed from birth and underwent multichannel PSG at term-equivalent age before discharge home. At 2 years CA, they returned for neurodevelopmental follow-up, including standardized BSID-III motor and cognitive testing, as well as early EF. EF tests were examined in relation to neonatal sleep behavior.
Methods
Participants
Inclusion/exclusion criteria
This study describes a cohort of children from the Resilience study, a prospective single-center cohort study with longitudinal follow-up, which was registered at Clinical Trials.gov (NCT02623400). The study was approved by the Ethical Committee of the University Hospital Leuven and was performed in accordance with the Guidelines for Good Clinical Practice and the Declaration of Helsinki.
Children born at University Hospitals Leuven between August 2016 and July 2018 were recruited if they were born before 34 weeks gestational age and/or had a birth weight lower than 1500 g. Parents were asked for written consent within the first week after birth. Exclusion criteria were the following: (a) maternal age less than 18 years, (b) the inability of a parent to speak and understand Dutch or English, (c) unstable medical (somatic and/or psychiatric) disease in the parent(s) and (d) the presence of a major congenital malformation or major central nervous system pathology (grade 3 or 4 intraventricular hemorrhage or periventricular leukomalacia detected on cranial ultrasonography during the neonatal phase) in the preterm infant.
Child demographics
Clinical data were collected prospectively daily from the NICU charts by a neonatal research nurse from NICU admission to discharge. Clinical data included were gestational age (GA), birth weight (BW), biological sex, postmenstrual age (PMA) at PSG, the total number of skin breaking procedures until day 5 (each invasive procedure and each attempt28), and the total days of ventilation (invasive/non-invasive). Maternal education was assessed according to the ISCED scale of the UNESCO Institute for Statistics30 by converting original classifications to ‘low education’ corresponding to mothers that obtained a secondary school degree or less, and ‘high education’ corresponding to mothers who obtained at least a bachelor’s degree. Mean daily amount of kangaroo care, defined as the total minutes of kangaroo care by the parents divided by the number of hospitalization days, was recorded.
Polysomnography and sleep features
EEG monitoring and sleep classification
Preterm infants had an overnight multichannel PSG before discharge home (between 3 pm and 8 am). PSG recordings were done with 9 EEG electrodes (Fp1-2, C3-4, T3-4, O1-2, reference electrode Cz) at a sampling frequency of 250 Hz, according to the modified international 10–20 standard. Recordings were done using a BRAIN RT EEG system, Micromed, NATUS, Rumst, Belgium. EEG signals were visually displayed using a 0.27–70 Hz filter. None of the infants received sedative drugs during the recording or less as 1 month prior to the recording.
For automated analysis, however, the raw EEG was processed through two independent, existing, validated pipelines: (i) automated artefact detection31 and (ii) automated sleep classification.21 Each pipeline implements its own preprocessing (including model-specific filtering and down sampling) to match the procedures used during model development.
Automated sleep classification was performed using a multi-step algorithm for which a previously published sleep classifier21 was extended with several pre- and postprocessing routines to improve robustness.29 This sleep classifier is a convolutional neural network and was developed based on sleep scoring in preterm infants according to André et al.16 and the American Academy of Sleep Medicine according to the proposed annotation rules for sleep scoring in full-term infants from birth to 2 months of age.13,32,33 This classifier has been subsequently validated on an extensive cohort of preterm and term-born neonates, recorded between 34 and 46 weeks.21
Prior to passing the EEG through the sleep classification pipeline, a novel preprocessing step was applied to improve robustness to noise. In this step, artefact locations are first automatically detected using a validated convolutional neural network, which labels each second of every channel as artefact or clean.31 The raw EEG was then cleaned by replacing a channel’s signal at artefact-marked time points with the average of the corresponding clean channels.31 Then, the sleep classifier is applied to the cleaned EEG data, which assigns a sleep stage to each 30 s of EEG data: AS1, LVI or AS2, QS-HVS, or QS-TA. Next, we apply novel postprocessing steps to improve robustness. First, the sequence of sleep states is smoothed based on a hidden Markov-model that optimizes the likelihood of the sleep state sequence, to reduce fast (erroneous) fluctuations in the sequence of sleep stages (i.e., the hypnogram).29
To further improve robustness, unreliable segments are automatically identified where the algorithm is likely to make errors. Here, a segment is marked as unreliable if 1) it contains too many artefacts; 2) the uncertainty of the sleep classifier is too high (i.e., probability of the predicted sleep stage is too low); 3) its temporal and spectral characteristics deviate too much from those of the training data used to develop the sleep classifier (i.e., novelty detection). The dataset originally used to train the sleep classifier was used to determine thresholds for each of these three categories. For more details, we refer to ref. 29
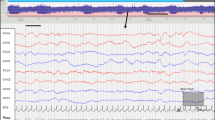
These unreliable segments are treated as gaps/unknowns in the hypnogram. Finally, a heuristic approach is applied that reduces gaps in hypnogram. This includes 1. ignoring unreliability flags for segments with enough reliable surrounding data, 2. interpolating unreliable segments if spanning a maximum of three consecutive minutes and surrounded by the same sleep stage, and 3. labelling unreliable segments (segments with low probability of the predicted sleep stage, meaning that the segment has features of two different sleep states) as transitional sleep, if spanning up to 6 minutes and if the segment lays in between two different sleep stages. This multi-step algorithm results in a hypnogram with the sleep state predicted for every 30 s segment, masking unreliable segments (Fig. 1).29 The Total Sleep Time is the total duration of all segments with reliable sleep states.
Transitional Sleep (TS) and unreliable segments are automatically identified. The segments are marked as movement, artefacts, uncertainty and novelty (see color bars on the right side of the hypnogram).
Neonatal sleep analysis
Based on literature data regarding EF, we designated 6 variables a priori as primary explanatory sleep features to be evaluated.23,34,35 When analyzing the impact of sleep features on outcome, it is important to account for maturational changes occurring during the recording period (PMA at recording).16 We studied the percentage of Total Sleep Time (%TST), Transitional Sleep, AS1, QS-HVS, QS-TA and the number of unexpected transitions per hour.23 To define the characteristic duration of a sleep state within a recording, a survival analysis (Kaplan-Meier) was conducted.34 The survival analysis captures the probability that a sleep state will last for a specific duration and takes also into account censored bouts (e.g. for some bouts we don’t know the survival time exactly, such as the offset or onset of the sleep bouts at the start or end of the recording are not captured (censored data). A Cox regression model was applied on the Kaplan-Meier survival curves to test the effect of PMA.
Executive function
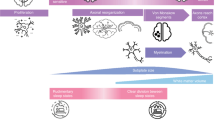
Preterm born infants were followed from birth to 2 years CA. Assessments, including BSID-III motor and cognitive scales, were conducted by a certified physiotherapist, psychologist, and a physician, blinded for the PSG results. Evidence indicates that preterm infants exhibit deficits in 3 core EFs: working memory, cognitive flexibility, and response inhibition. EF was assessed using three age-appropriate tasks as described earlier by our group36, with adherence to a predefined checklist and illustrated in Fig. 2.
Multisearch Multilocation Task (MSML), Reversed Categorization Task (RevCat) and Snack Delay Test (SDT) and scoring grid.
The Multisearch Multilocation Task mainly taps into spatial working memory, the Reversed Categorization Task in cognitive flexibility, and the Snack Delay Task provides a measure of inhibitory control.37,38,39,40 For the Multisearch Multilocation Task, three colored boxes were presented to the child: yellow, red, and blue. In the preswitch trial, a toy animal was placed in the red box. The child watched the toy animal being hidden and was encouraged to retrieve it. A trial was scored as successful if the child found the toy animal on the first attempt. Preswitch trials continued until the child achieved three consecutive correct trials or until four trials were attempted. One point was awarded for each correct attempt. The postswitch trial was introduced as a “silly game” and the child was encouraged to watch as the toy animal was now hidden in the yellow box. A 10-second delay was imposed before the child was presented with the boxes and encouraged to find the toy animal. The post switch trials continued until the child had correctly searched on two consecutive trials or until six post switch trials had been tried. The post witch trials were reverse scored with six points awarded for the first two searches, both being correct and thereafter penalizing for each error. As such, scores from preswitch searching ranged from 0 to 3 and post switch searching from 0 to 6, giving a maximum total score of 9 points. For the Reversed Categorization test: In the preswitch phase, the child was encouraged to put three yellow blocks in a yellow bucket and three blue blocks in a blue bucket. In a post switch phase, the child was told to put three yellow blocks in the blue bucket and three blue blocks in the yellow bucket. One point was awarded for each block correctly sorted. To determine the total score, children were allocated to groups based on the highest level they achieved, with the following possible outcomes: (1) preswitch score less than 5 and post switch score less than 5, (2) minimum preswitch score of 5 and post switch score less than 5, (3) minimum preswitch score of 5 and post switch score of 5, (4) minimum preswitch score of 5 and post switch score of 6. Three points were given for outcome 4, 2 points for outcome 3, 1 point for outcome two, and zero points for outcome one, yielding a maximum Reversed Categorization total score of 3 points. For the Snack Delay Task: A biscuit was placed in front of the child, on a plate underneath a transparent upturned glass. The experimenter had a bell in front of her, on the same table. The child was instructed to wait for the bell to be rung before retrieving the treat. Three consecutive trials with a delay of 5, 15, and 30 s were performed. Waiting for the ringing of the bell before touching and retrieving the treat was scored as a full wait, while lifting or touching the glass without eating the treat was defined as a partial wait. Eating the treat or ringing the bell by the child prior to the bell being rung by the experimenter was defined as a failed trial. The assessment continued until all three trials were completed or until the first failed trial. Snack Delay Task total score was the number of trials with a full wait. So, the total score ranged from 0 to 3. In line with this study, a summary EF composite score was determined by averaging the standardized (z) score on each of the three tasks, thereby giving equal weight to working memory, flexibility and inhibition.
Statistical analysis
Analyses were carried out using IBM SPSS Statistics Version 30.0.41 As part of an exploratory aim, we examined the associations between neonatal sleep and EF at 2 years CA using a general linear model (GLM). We analyzed the different sleep features in separate models. Our first aim was to address whether percentage of Total Sleep Time would predict 2-year EF in a basic model adjusting for clinical and experiential factors. These 7 covariates were: PMA, sex, birth weight, days of mechanical ventilation, neonatal skin breaking procedures, amount of Kangaroo Care and maternal education, with bootstrapping. Collinearity was checked with Variance Inflation Factor (VIF : < 5, ideally less than 2.5) and correlation <0.7.
We subsequently assessed if individual sleep features were related to EF. We tested in separate models the percentage transitional sleep, the number of unexpected transitions/hours, and 3 individual sleep states in relation to EF composite score. To estimate the unique contribution of each of these sleep features, standardized beta coefficients and respective p-values are calculated.
As this was an initial investigation in this area of research, we also explored in a subsequent analysis, whether those sleep parameters individually also predicted the subscales of the executive function tests. Since we had 3 subfields of outcome, all using a separate model, we set the alpha level at 0.0125 (p = 0.05/3) for multiple testing.
Results
Demographics and clinical characteristics
108 children underwent multichannel polysomnography and 76 (70%) returned for the neuropsychological assessment at 2 years CA and completed the EF battery. Missing assessments were mainly due to COVID-19 testing complications and since the EF battery was administered after the standard BSID-III cognitive and motor tests, some children were too fatigued to complete it.36 Clinical and demographic information is presented in Table 1.
Neonatal sleep parameters and executive function tests
Neonatal sleep characteristics and maturational trends
Preterm neonates recruited in this prospective observational study were completely ready to be discharged home, exclusively orally fed and nursed in a cot at the time of the PSG. Feeding and caregiving moments depend on the neonates’ feeding clues during the overnight recording. Sleep characteristics of this cohort are summarized in Table 2. The mean percentage of Total Sleep Time of the overnight recording was 10.6 h. Active Sleep was the dominant sleep state with 6.3 h, Quiet Sleep 3.8 h and transitional sleep 0.45 h. 20% Of the total recording time included EEG with artefacts not analyzable for sleep prediction, attributable to infant awakenings, cares and feeding. All preterm neonates displayed sleep cycling. AS bout durations which had offset to wakefulness (profuse movements) were shorter than AS bout durations prior to transitions to QS (Supplemental Fig. S1). The AS bouts which offset to wakefulness were 2 times more abundant than the AS bouts that offset to QS. The maturational trend of each sleep state’s contribution to the Total Sleep Time composition changes with PMA (Supplemental Fig. S2). The changes are larger for the QS states than for the AS states. A significant effect of PMA was found for both AS, QS and substates AS1, LVI, QS- HVS and QS- TA. The significant PMA effect means that the durations of AS, AS1 and QS-TA sleep bouts decrease with PMA, while duration of QS HVS bouts increase (Supplemental Fig. S3). Although influenced by PMA, the median AS time did positively correlate with the percentage Total Sleep Time (r = 0.52, p < 0.001) and sleep time per cycle (r = 0.52, p < 0.001).
Neonatal sleep and executive function
When examining the association between percentage Total Sleep Time before discharge and EF composite score, a significant relationship was found, indicating that preterm born children with lower EF composite scores experienced less Total Sleep Time (Fig. 3). These results were maintained, with percentage of Total Sleep Time (ß = 0.017, p = 0.006, CI 0.005–0.028, Table 3) positively predicting EF composite scores, when adjusting for birthweight, PMA, sex, total days of ventilation, number of skin breaking procedures, maternal education, and Kangaroo Care. This model explains 38% of the variation in EF with neonatal and experiential NICU factors (R2 = 0.38 p < 0.001, Table 4), including 10% of the variation in EF at 2 years explained through the amount of sleep of the Total Sleep Time (Fig. 3). The subtest ‘Multisearch Multilocation Task (Model 3 R² = 0.45 p < 0.001, Table 5) was also positively predicted by the percentage of Total Sleep Time, after adjustment for the same covariates (ß = 0.036, p < 0.001, CI 0.02–0.05). None of the other subtests were related to Total Sleep Time (Table 3). The residuals of the main model were assessed for normality using the Kolmogorov-Smirnov test.
Y-as: Total Sleep Time percentage of the recording X-as: Executive Function Composite Score.
The residual from 1 participant was considered an outlier (left lower corner Fig. 3). Removal of this participant’s data from the original model resulted in comparable effects sizes, indicating that the results were not unduly influenced by this participant.
EF test composite score (Model 2 R² = 0.35, p < 0.001, Table 4) and the subtest ‘Multisearch Multilocation Task’ (Model 4 R² = 0.33, p < 0.001, Table 5), respectively, were negatively predicted by the percentage of transitional sleep in a separate GLM.
The percentage of AS1, QS-HVS and QS-TA and the number of unexpected transitions did not yield significant results after adjusting for PMA and experiential factors (see Table 3).
Discussion
This study identifies a significant association between neonatal sleep behavior in preterm infants and EF assessed at preschool age. Preterm neonates with less Total Sleep Time and more Transitional Sleep before discharge from the NICU, had lower EF scores, sleep features which have been found to correlate with EF in older individuals as well.42,43 Whether this is cause (e.g. driver of abnormal development) or effect (e.g. a biomarker of current adverse neurodevelopment) is harder to determine and points out how entwined both processes are.44,45 Total Sleep Time was only associated with the subtest Multisearch Multilocation, mainly tapping into spatial working memory, one of the earlier EF to emerge.38
Compelling research suggests a specific link between EF and sleep in childhood. Mc Cann et al.46 investigated the relationship between EF and parent reported sleep problems in very preterm children and a control term group. The quality of nighttime sleep was found to adversely impact EF in the very preterm group, while no effect of sleep was found in the control group who slept poorly. Moreover, EF deficits are described in children with adversities of neurodevelopment, such as cerebral palsy and attention deficit disorder47,48,49 and sleep-disordered breathing. These studies link their results to the relative importance of the frontal areas of the brain in sleep, more specific to the sleep promoting neurons in the basal forebrain,50,51 with the prefrontal cortex particularly sensitive to changes in the homeostatic processes underlying sleep regulation.43,52 In adults, functional connectivity between thalamus and frontal and temporal gyri was decreased after sleep deprivation. Moreover, especially the prefrontal cortex which mediates functions such as working memory showed vulnerability to the effect of sleep deprivation.25
Together, these findings suggest that the restorative effect of sleep is especially relevant for the maintenance of functional connectivity of prefrontal brain regions.53 One of the hypotheses is that especially slow wave sleep during NREM sleep has a restorative effect on prefrontal areas.52 We could not find a relationship with QS-HVS (which can be seen as a ‘pre-cursor’ of this state) and EF, however QS-HVS is only beginning to emerge at term equivalent age, and the average PMA at EEG was 39 weeks. Nevertheless, a global higher level of Total Sleep Time might represent a better resilience to the ‘more’ awakening stimuli in preterm babies.54 Moreover, in line with Georgoulas et al., we showed nicely that longer AS bouts were more likely to offset into QS than wakefulness.34 We found a positive correlation between the median length of AS and the percentage of Total Sleep Time and sleep time per cycle. This means that for a neonate, to get to QS and to complete a sleep-wake cycle, AS should be preserved as well.
In this study, we describe neonatal EEG-determined sleep behavior in a fully automated and quantitative manner. Sleep characteristics were extracted from real-world data. Automated sleep analysis is a promising application and can help to advance neonatal sleep research. Quantitative sleep modelling in different neonatal cohorts can for example serve as a handle for understanding sleep architecture during the neonatal period.23,24 Extrapolated to the 24 h period, the preterm newborn infant around term equivalent age, sleeps around 16 h per day, which is on the lower side of the 16h-18h described in the full-term neonate, confirming that premature infants sleep less than full-term infants of the same post-conceptional age55 (observational data, manuscript in preparation). The preterm infant has been found to adapt to the NICU environment (with more extreme sensory stimuli) with longer bouts of wakefulness.34,55 The developmental trajectories of our sleep variables show a decrease in QS -TA and more QS-HVS (longer QS-HVS bouts). Furthermore, there is clear postmenstrual age influence on the heavy-tailed AS sleep bout distribution and arousability, similar as documented in polysomnographic studies by experts in the field.14,16,34,56,57
The higher amount of Transitional Sleep, an EEG characteristic which reflects less clear state distinction, might be a possible reflection of maladaptive brain maturity or dysmaturity.58
The literature demonstrates significant variability in the methodologies used to describe neonatal sleep, making it challenging to make direct comparisons. A consistent approach is necessary for evaluating sleep conditions, as well as for measuring and analyzing sleep, but can be offered with this automated method. Environmental factors may impact the duration and intensity of sleep,59 and the NICU is often considered as a hostile sleep environment. All preterm neonates in this study were cared for under similar environmental conditions (supine position, single room, ambient light and sound during the night and without disturbances for clinical purposes, caffeine use until 34-36 weeks PMA).
Several mediators may contribute to the variable findings on the effect of prematurity on sleep development, including the degree of prematurity, changed hormonal conditions compared to in utero, (epi)genetic links, pathologies (lung disease, sleep disordered breathing and comorbidities (growth restriction, hypoxia, neural injury and/or impaired neural maturation, systemic inflammation) and will occur in combination or additive and cause self-induced feedback loops.45,54
To deal with this inter-individual variation in functional brain maturation and sleep organization, different confounders have been considered in this exploratory study. We defined illness severity by the days of ventilation, number of skin breaking procedures and birthweight. Birthweight instead of GA has been used as covariate because of the link between intra-uterine growth restriction and sleep60 and previous literature reporting that lower birthweight is associated with lower inhibitory control and lower achievement on visuospatial skills in extremely preterm children.61 Studies have shown that EF is related to maternal intelligence,62 as well as parental socio-economic state.63 Of interest, more kangaroo care at the NICU was uniquely associated with higher EF scores in this study. The infant’s ability to regulate state of arousal is a development trait, where specific infant and parental factors interact in the development of self-soothing.64 Kangaroo care has been shown to influence the infant’s capacities to regulate sleep and wake states, and accelerated maturation of sleep states has been reported in preterm neonates.55 Although the amount of kangaroo care in this study was not correlated with the sleep features at discharge, this non-intrusive developmental style initiated during the NICU stay may further have its positive effects during the early months of life.65,66
None of the babies received sedative drugs during the PSG.67 In parallel with the effect of these drugs on anatomical brain growth and neurodevelopmental outcome,68,69,70 long lasting results of these drugs on the developmental aspect of sleep physiology is of interest, but out of scope of the present study.
We acknowledge the relatively modest sample size of our study; this was partially due to loss to follow-up during the COVID-19 pandemic and test conditions. This study is uniquely in documenting neonatal sleep behavior through fully automated analysis by overnight polysomnography under similar environmental conditions; however one, single 24 h PSG before discharge may not be sufficient to represent sleep patterns in the NICU. Given the exploratory nature of this study, we aimed to include important confounders. Nonetheless, we acknowledge that the design and the sample size inherently limit our ability to account for all potentially relevant covariates, and some may not have been incorporated into the model. Consistent methodology to document sleep conditions as well as reproducible, quantitative sleep analysis is the way forward to design well conducted observational studies with repeated sleep measurements and interventional trials.
The results of this study highlight the need for enhanced neuroprotective strategies in the neonatal period as well as postnatal interventions that support extrauterine maturation and the neurodevelopment of preterm born neonates. Understanding the effect of early key factors during the period spent in the NICU, that may modulate early brain dysmaturation or maldevelopment and their neurodevelopmental consequences, can change care giving practices and organization of the NICU practices to promote sleep, especially in vulnerable infants. Whether and to what extent a “sleep protective” environment can be a buffer for harmful influences related to prematurity needs to be further investigated. Earlier recognition of the ‘sleep phenotype’, either in premature infants or high-risk infants35 may be a gateway for more focused interventional strategies to improve attention, cognition and social interactions.
Conclusion
The Total Sleep Time at term equivalent age is associated with early EF in 2-year-old children born preterm, mediated by other clinical and experiential factors. These results should encourage neonatal caregivers to further unravel the dynamics between prematurity, sleep and functional brain development. This enhanced understanding will gradually assist in customizing interventions to improve neonatal sleep, tailored to the developmental stage of each child, to maximize positive impacts on future neurodevelopmental outcomes.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Dai, D. W. T. et al. The contributions of intelligence and executive function to behaviour problems in school-age children born very preterm. Acta Paediatr. 110, 1827–1834 (2021).
Loe, I. M., Heller, N. A. & Chatav, M. Behavior problems and executive function impairments in preterm compared to full term preschoolers. Early Hum. Dev. 130, 87–95 (2019).
Aarnoudse-Moens, C. S., Weisglas-Kuperus, N., van Goudoever, J. B. & Oosterlaan, J. Meta-analysis of neurobehavioral outcomes in very preterm and/or very low birth weight children. Pediatrics 124, 717–728 (2009).
Van den Brande, A., Bollen, B., Boets, B., Naulaers, G. & Ortibus, E. Executive function assessment in 2-year-olds born preterm. Neuropediatrics (2023).
Inder, T. E., Volpe, J. J. & Anderson, P. J. Defining the neurologic consequences of preterm birth. N. Engl. J. Med 389, 441–453 (2023).
Nosarti, C. et al. Preterm birth and structural brain alterations in early adulthood. Neuroimage Clin. 6, 180–191 (2014).
Ostgard, H. F. et al. Executive function relates to surface area of frontal and temporal cortex in very-low-birth-weight late teenagers. Early Hum. Dev. 95, 47–53 (2016).
Schnider, B. et al. Altered brain metabolism contributes to executive function deficits in school-aged children born very preterm. Pediatr. Res. 88, 739–748 (2020).
Duerden, E. G. et al. Association of early skin breaks and neonatal thalamic maturation a modifiable risk? Neurology 95, E3420–E3427 (2020).
Sadozai, A. K. et al. Executive function in children with neurodevelopmental conditions: a systematic review and meta-analysis. Nat. Hum. Behav. 8, 2357–2366 (2024).
de Kieviet, J. F., Zoetebier, L., van Elburg, R. M., Vermeulen, R. J. & Oosterlaan, J. Brain development of very preterm and very low-birthweight children in childhood and adolescence: a meta-analysis. Dev. Med. Child Neurol. 54, 313–323 (2012).
Diamond, A. & Lee, K. Interventions shown to aid executive function development in children 4 to 12 years old. Science 333, 959–964 (2011).
Grigg-Damberger, M. M. The visual scoring of sleep in infants 0 to 2 months of age. J. Clin. Sleep. Med. 12, 429–445 (2016).
Scher, M. S. & Loparo, K. A. Neonatal EEG/sleep state analyses: a complex phenotype of developmental neural plasticity. Dev. Neurosci. 31, 259–275 (2009).
Lombroso, C. T. Neonatal polygraphy in full-term and premature infants: a review of normal and abnormal findings. J. Clin. Neurophysiol. 2, 105–155 (1985).
Andre, M. et al. Electroencephalography in premature and full-term infants. developmental features and glossary. Neurophysiol. Clin. 40, 59–124 (2010).
Dreyfus-Brisac, C. Neurophysiological studies in human premature and full-term newborns. Biol. Psychiatry 10, 485–496 (1975).
Blumberg, M. S., Dooley, J. C. & Sokoloff, G. The developing brain revealed during sleep. Curr. Opin. Physiol. 15, 14–22 (2020).
Graven, S. Sleep and brain development. Clin. Perinatol. 33, 693–706 (2006). vii.
Uchitel, J., Vanhatalo, S. & Austin, T. Early development of sleep and brain functional connectivity in term-born and preterm infants. Pediatr. Res. 91, 771–786 (2022).
Ansari, A. H. et al. A convolutional neural network outperforming state-of-the-art sleep staging algorithms for both preterm and term infants. J. Neural Eng. 17, 016028 (2020).
Pillay, K., Dereymaeker, A., Jansen, K., Naulaers, G. & De Vos, M. Applying a data-driven approach to quantify eeg maturational deviations in preterms with normal and abnormal neurodevelopmental outcomes. Sci. Rep. 10, 7288 (2020).
Hermans, T. et al. Functional brain maturation and sleep organisation in neonates with congenital heart disease. Eur. J. Paediatr. Neurol. 36, 115–122 (2022).
Scher, M. S., Turnbull, J., Loparo, K. & Johnson, M. W. Automated state analyses: proposed applications to neonatal neurointensive care. J. Clin. Neurophysiol. 22, 256–270 (2005).
Krause, A. The sleep-deprived human brain. Nat Rev Neurosci. 18, 404–418 (2017).
Bruni, O. et al. Executive functions in preschool children with chronic insomnia. J. Clin Sleep Med. 16, 231–241 (2020).
Ranger, M. et al. Neonatal pain-related stress predicts cortical thickness at age 7 years in children born very preterm. PLoS One 8 (2013).
Lavanga, M. et al. The effect of early procedural pain in preterm infants on the maturation of electroencephalogram and heart rate variability. Pain 162, 1556–1566 (2021).
Hermans, T. Improving Robustness and Reliability in Neonatal Brain Monitoring. PhD Thesis (2023)
Statistics, U. I. f. International Standard Classification of Education: Isced 2011. Comparat. Social Res. 30 (2012).
Hermans, T. et al. A multi-task and multi-channel convolutional neural network for semi-supervised neonatal artefact detection. J. Neural Eng. 20 (2023).
Berry, R. B. et al. The Aasm Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications, Version 2.0. (2014).
Grigg-Damberger, M. et al. The visual scoring of sleep and arousal in infants and children. J. Clin. Sleep. Med. 3, 201–240 (2007).
Georgoulas, A. et al. Sleep-wake regulation in preterm and term infants. Sleep 44 (2021).
Shellhaas, R. A., Burns, J. W., Barks, J. D. & Chervin, R. D. Quantitative sleep stage analyses as a window to neonatal neurologic function. Neurology 82, 390–395 (2014).
Van den Brande, A., Bollen, B., Boets, B., Naulaers, G. & Ortibus, E. Executive function assessment in 2-year-olds born preterm. Neuropediatrics 55, 16–22 (2024).
Ansell, J. M., Wouldes, T. A., Harding, J. E. & group, C. S. Executive function assessment in new zealand 2-year olds born at risk of neonatal hypoglycemia. PLoS One 12, e0188158 (2017).
Carlson, S. M. Developmentally Sensitive Measures of Executive Function in Preschool Children. Dev. Neuropsychol. 28, 595–616 (2005).
Kochanska, G., Murray, K. T. & Harlan, E. T. Effortful control in early childhood: continuity and change, antecedents, and implications for social development. Dev. Psychol. 36, 220–232 (2000).
Zelazo, P. D., Reznick, J. S. & Spinazzola, J. Representational flexibility and response control in a multistep multilocation search task. Dev. Psychol. 34, 203–214 (1998).
IBM SPSS Statistics for Windows, version 30.0 (IBM Corp., Armonk, N.Y., USA, 2024).
Bernier, A., Bordeleau, S., Carrier, J. & Carlson, S. M. Relations between physiological and cognitive regulatory systems: infant sleep regulation and subsequent executive functioning. Child Dev. 81, 1739–1752 (2010).
Jones, K. & Harrison, Y. Frontal lobe function, sleep loss and fragmented sleep. Sleep. Med. Rev. 5, 463–475 (2001).
de Groot, E. R., Dudink, J. & Austin, T. Sleep as a driver of pre- and postnatal brain development. Pediatr. Res. (2024).
Bennet, L., Walker, D. W. & Horne, R. S. C. Waking up too early - the consequences of preterm birth on sleep development. J. Physiol. 596, 5687–5708 (2018).
McCann, M. et al. The relationship between sleep problems and working memory in children born very preterm. Child Neuropsychol. 24, 124–144 (2018).
Duncan, A. F. & Maitre, N. L. Sleep, cognition and executive functioning in young children with cerebral palsy. Adv. Child Dev. Behav. 60, 285–314 (2021).
Hulst, R. Y. et al. Sleep problems in children with cerebral palsy and their parents. Dev. Med Child Neurol. 63, 1344–1350 (2021).
Shetty, M. et al. The effects of sleep disordered breathing on sleep spindle activity in children and the relationship with sleep, behavior and neurocognition. Sleep. Med. 101, 468–477 (2023).
Jones, B. E. From waking to sleeping: neuronal and chemical substrates. Trends Pharm. Sci. 26, 578–586 (2005).
Hong, J., Lozano, D. E., Beier, K. T., Chung, S. & Weber, F. Prefrontal cortical regulation of rem sleep. Nat. Neurosci. 26, 1820–1832 (2023).
Achermann, P., Dijk, D. J., Brunner, D. P. & Borbely, A. A. A model of human sleep homeostasis based on eeg slow-wave activity: quantitative comparison of data and simulations. Brain Res. Bull. 31, 97–113 (1993).
Huber, R., Ghilardi, M. F., Massimini, M. & Tononi, G. Local sleep and learning. Nature 430, 78–81 (2004).
Guyer, C. et al. Very preterm infants show earlier emergence of 24-hour sleep-wake rhythms compared to term infants. Early Hum. Dev. 91, 37–42 (2015).
Holditch-Davis, D., Scher, M., Schwartz, T. & Hudson-Barr, D. Sleeping and waking state development in preterm infants. Early Hum. Dev. 80, 43–64 (2004).
Scher, M. S., Steppe, D. A., Dahl, R. E., Asthana, S. & Guthrie, R. D. Comparison of Eeg sleep measures in healthy full-term and preterm infants at matched conceptional ages. Sleep 15, 442–448 (1992).
Scher, M. S., Steppe, D. A., Banks, D. L., Guthrie, R. D. & Sclabassi, R. J. Maturational trends of EEG-sleep measures in the healthy preterm neonate. Pediatr. Neurol. 12, 314–322 (1995).
Dereymaeker, A. et al. Review of sleep-EEG in preterm and term neonates. Early Hum. Dev. 113, 87–103 (2017).
Ryan, M. A. J. et al. Sleep state organisation of moderate to late preterm infants in the neonatal unit. Pediatr. Res. 93, 595–603 (2023).
Whitehead, K. Co-developing sleep-wake and sensory foundations for cognition in the human fetus and newborn. Dev. Cogn. Neurosci. 71, 101487 (2025).
Fernandez-Baizan, C., Alcantara-Canabal, L., Solis, G. & Mendez, M. The association between perinatal and neonatal variables and neuropsychological development in very and extremely low-birth-weight preterm children at the beginning of primary school. Appl Neuropsych. Chil. 10, 348–358 (2021).
Ribner, A., Devine, R. T., Blair, C. & Hughes, C. Mothers’ and fathers’ executive function both predict emergent executive function in toddlerhood. Dev. Sci. 25, e13263 (2022).
Lawson, G. M., Hook, C. J. & Farah, M. J. A meta-analysis of the relationship between socioeconomic status and executive function performance among children. Dev. Sci. 21 (2018).
Geva, R. & Feldman, R. A neurobiological model for the effects of early brainstem functioning on the development of behavior and emotion regulation in infants: implications for prenatal and perinatal risk. J. Child Psychol. Psyc 49, 1031–1041 (2008).
Davis, M., Bilms, J. & Suveg, C. In sync and in control: a meta-analysis of parent-child positive behavioral synchrony and youth self-regulation. Fam. Process 56, 962–980 (2017).
Diamond, A. Executive functions. Annu Rev. Psychol. 64, 135–168 (2013).
Thewissen, L. et al. Cerebral autoregulation and activity after propofol for endotracheal intubation in preterm neonates. Pediatr. Res. 84, 719–725 (2018).
Duerden, E. G. et al. Midazolam dose correlates with abnormal hippocampal growth and neurodevelopmental outcome in preterm infants. Ann. Neurol. 79, 548–559 (2016).
Tataranno, M. L. et al. Morphine affects brain activity and volumes in preterms: an observational multi-center study. Early Hum. Dev. 144, 104970 (2020).
Schiller, R. M. et al. Analgesics and sedatives in critically ill newborns and infants: the impact on long-term neurodevelopment. J. Clin. Pharmacol. 58, S140–S150 (2018).
Funding
C3- KU Leuven Financial Support.
Author information
Authors and Affiliations
Consortia
Contributions
A.D. designed, analysed, interpreted, and drafted the initial manuscript together with TH. A.D, B.B. and E.O. contributed to the acquisition of the data. A.D., T.H., B.B., K.J., E.O., M.D.V. and G.N. guided the drafting of the initial article and reviewed it critically for important intellectual content. All authors approved the definitive version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent Statemen
Patient consent was obtained and fulfilled accordingly.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dereymaeker, A., Hermans, T., Bollen, B. et al. Neonatal sleep physiology and early executive functioning in preterm children. Pediatr Res (2026). https://doi.org/10.1038/s41390-026-04784-0
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41390-026-04784-0