Abstract
Background
In contrast to the extensive research on the effects of high-altitude exposure on adult neurological function, fetal and pediatric neurodevelopment in high-altitude environments has received relatively little attention. This study aimed to determine whether exposure to high-altitude exposure during gestation and infancy could influence neurodevelopmental outcomes compared with low-altitude infants.
Methods
A total of 50 high-altitude-born and 100 low-altitude-born infants were studied. Parents were followed longitudinally and instructed to complete the Ages and Stages Questionnaire, 3rd edition (ASQ-3), when their infants reached 6, 12, and 18 months of age. The primary outcome measures included neurodevelopmental scores and the proportion of infants classified as failing in the five ASQ-3 domains.
Results
High-altitude infants had lower gross motor scores and higher failure rates. Subgroup analysis revealed no significant differences attributable to confounding factors. Regression analysis identified high-altitude exposure as the only significant predictor of gross motor delay.
Conclusions
Prenatal and postnatal exposure to high-altitude exposure was found to be associated with an increased risk of gross motor neurodevelopmental delay in infants.
Impact
-
This is the first prospective cohort study to compare the neurodevelopmental outcomes of high-altitude born/raised infants with those of low-altitude controls.
-
Infants born and raised at high-altitude exhibited lower neurodevelopmental scores and a higher likelihood of failing in the gross motor domain of ASQ-3.
-
High-altitude exposure had no negative influence on infants’ physical growth and other neurodevelopmental domains assessed by the ASQ-3.
Similar content being viewed by others
Introduction
A high-altitude environment, defined as elevations of 2500 meters or more above sea level, presents extreme environmental stressors, including hypoxia, hypobaria, low temperatures, low humidity, and intense solar radiation, all of which impose significant physiological and neuropsychological challenges.1,2 Tibet, located in Southwest China, represents the highest plateau region globally. According to the 2020 national population census of China, more than 407,100 individuals had migrated to Tibet from lower-altitude areas for occupational or economic reasons, and this number continues to grow. In contrast to indigenous Tibetans, who have undergone extensive evolutionary adaptations to the plateau environment, newly arrived immigrants, primarily of Han ethnicity, exhibited a reduced capacity against hypoxic stress and are consequently more susceptible to its deleterious effects.3,4,5 The potential neurophysiological and cognitive ramifications of prolonged high-altitude exposure among these immigrants have remarkably garnered researchers’ attention, particularly among those considering relocation.
Optimal neurocognitive function is inherently dependent on the continuous and efficient delivery of oxygen to the brain.6 Among various environmental stressors associated with high-altitude exposure, hypoxia, induced by the reduction in oxygen partial pressure resulting from diminished atmospheric pressure, constitutes the principal physiological challenge to human neurocognitive integrity. A growing body of empirical research highlighted the detrimental impact of both acute and chronic high-altitude exposure on neurocognitive function across the lifespan, and some deficits could persist even after returning individuals to lower altitudes.7,8 Recent systematic investigations of high-altitude residents have consistently demonstrated impairments in motor speed, cognitive processing speed, cerebral blood flow velocity,9,10 and visual/verbal memory accuracy, alongside prolonged reaction time.11,12 Moreover, structural neuroimaging studies of high-altitude immigrants provided direct evidence of alterations in cerebral morphology.11,12,13,14 For instance, in soldiers stationed at high altitudes for two years, regional homogeneity (ReHo) increased in the sensorimotor cortex,13 while voxel-mirrored homotopic connectivity was elevated in the bilateral visual cortex.14 Furthermore, high-altitude immigrants exhibited a reduction in functional connectivity between the visual and motor cortices,15 as well as an increase in total brain volume, gray matter volume, and white matter volume.16 Additionally, Chen et al.11 identified gray matter loss and decreased ReHo in the putamen following chronic high-altitude exposure. Compared with pre-exposure conditions, the putamen exhibited attenuated functional connectivity with the superior temporal gyrus, anterior/middle cingulate gyrus, and other cognition-related brain regions, further substantiating the neuroimaging evidence linking psychomotor dysfunction.11
Early life represents a critical period for neurodevelopment, characterized by rapid synaptogenesis and establishment of foundational neural networks.17 While substantial research has advanced the understanding of high-altitude-induced neurological alterations in adults, investigations into its effects on pediatric neurodevelopment remain relatively scarce, and findings that are mainly incongruous.9,18,19 A seminal study by Saco-Pollitt20 on Peruvian neonates born at high altitude demonstrated diminished visual and auditory orientation, reduced activity and motor maturity, and impaired self-quieting abilities relative to low-altitude infants. Wehby18 presented compelling initial evidence linking altitude to an increased likelihood of neurodevelopmental risk over the first two years of life, based on a cohort of 2116 infants who aged 3–24 months across South American populations. Similarly, neurodevelopmental assessment of children and adolescents in Bolivia revealed a subtle but measurable decline in psychomotor speed with increasing altitude.9 Furthermore, Virués-Ortega et al.19 reported that despite physiological acclimatization, individuals born at high altitude exhibited mild cognitive and behavioral deficits. Consequently, these findings highlight the significant challenge that high altitude exposure pose to early neurodevelopmental outcomes. However, these neurodevelopmental effects may vary across populations inhabiting similar altitudes on different continents, as well as in genetically distinct subpopulations.9,20,21 Given the substantial demographic presence of Han immigrants in high-altitude regions and the crucial role of pregnancy and infancy in establishing the foundation for lifelong neurocognitive function, the impact of chronic high-altitude exposure on the neurodevelopment of immigrant offspring warrants escalated attention.
This prospective cohort study aimed to compare neurodevelopmental outcomes between high-altitude and low-altitude Han infants at 6, 12, and 18 months of age while also examining the association between high-altitude exposure and early-life neurodevelopment. On the basis of prior research, it was hypothesized that infants born and raised at high altitude would exhibit a higher risk of neurodevelopmental delay versus low-altitude infants.
Methods
Study design
This longitudinal prospective cohort study recruited infants from two distinct geographical locations. The high-altitude group comprised infants born at Fokind Hospital for Women & Children in Lhasa (altitude: 3650 m) between June and September 2020, whereas the low-altitude group included those born at Northwest Hospital for Women & Children in Xi’an (altitude: 600 m) between August and September 2020. Newborns’ parents were invited to participate prior to hospital discharge.
Enrollment was dependent on meeting the following inclusion criteria: (1) both parents of Han ethnicity, (2) absence of severe perinatal complications, (3) no chronic or hereditary diseases, (4) no history of medication use that could impact neurological function, (5) normal maternal nutrition status (BMI ≥ 18 and ≤ 25 kg/m2) and (6) willingness to participate in the study and providing informed consent. Additional exclusion criteria were applied based on residence and exposure history. For the low-altitude group, exclusion criteria included (1) a permanent residence at an altitude exceeding 1000 m and (2) prior exposure to high-altitude environments (≥2500 m). For the high-altitude group, exclusion criteria included (1) maternal birth and upbringing at high altitude; (2) maternal absence from high-altitude regions for more than eight weeks during pregnancy, and (3) parental unwillingness to raise the child at high altitude. At baseline, parents completed a structured questionnaire to document parental demographic and health-related characteristics. Follow-up assessments, including neurodevelopmental evaluations, were conducted at 6, 12, and 18 months postpartum. During each follow-up visit, parents completed an infant characteristics questionnaire, and infants underwent comprehensive clinical examinations, including anthropometric measurements of body length and weight.
Neurodevelopmental assessment was conducted using the Ages & Stages Questionnaires, Third Edition (ASQ-3). Parents, guided by an ASQ-certified clinician, completed the assessment through structured home observations of their infants. To enhance participant retention, study reminders were sent via mobile text messages monthly, with an additional telephone reminder issued one week before each scheduled follow-up visit (at 6, 12, and 18 months). In cases of missed appointments, daily contact attempts were made over two consecutive weeks before classifying participants as lost to follow-up.
The study protocol was approved by the Ethics Committee of the Medical Faculty of Fourth Military Medical University (Approval No. KY20173352-1). All study procedures were conducted in strict adherence to the ethical guidelines for medical research involving human subjects, as outlined in the Declaration of Helsinki.
Sample size estimation
The high-altitude exposure group was matched to the low-altitude group based on sex, birth weight, gestational age, and maternal age.22,23 In the present study, given the constrained number of exposed subjects, a 1:n ratio matching strategy was implemented between the exposed and control groups to maintain sufficient statistical power in the analyses. The sample size calculation indicated a 20% difference (10% vs. 30%) in the proportion of infants falling below or near the predefined threshold for neurodevelopmental diagnoses. Assuming a sample size of 50 in the exposed group, a minimum of 90 controls (90.38 calculated by statistical power analysis) were required to ensure a 90% statistical power with a 5% significance threshold. The target enrollment was finally set at 50 exposed and 100 control subjects after incorporating a 10% buffer for potential loss to follow-up.
Pediatric neurodevelopmental outcomes
Pediatric neurodevelopmental outcomes were measured by the Chinese version of ASQ-3,24 a widely employed parent-reported instrument for developmental screening in both clinical and research contexts. This assessment tool comprises 30 age-calibrated items designed to be involved in five fundamental developmental domains: communication, involving verbal articulation, inquiry formulation, social interaction, and the exchange of personal information; gross motor proficiency, comprising coordination, balance, muscular strength, kinesthetic awareness, and reaction time; fine motor dexterity, including the manipulation of precision tools, such as scissors, keyboards, and rulers, alongside tasks requiring refined hand control (e.g., holding a pen, drawing, and producing legible handwriting); problem-solving aptitude, incorporating facets of creativity, decision-making, reliability, and collaborative functioning; and personal-social competencies, wherein personal attributes include adaptability, dependability, and intrinsic motivation, while social attributes involve cooperative behaviors, eye contact maintenance, active listening, spatial awareness, and demonstration of appropriate social etiquette. Each item was rated on a three-tier Likert scale: 10 (affirmative), 5 (intermittent), and 0 (not yet achieved). Domain-specific scores were computed through summation, and elevated scores were indicative of more favorable neurodevelopmental trajectories. Categorization of developmental status was implemented based on the ASQ-3 threshold values, whereby infants were classified as exhibiting normative development if their scores across all five domains exceeded one standard deviation (SD) below the mean, as presenting a suspected developmental delay if scores ranged between one and two SDs below the mean, and as demonstrating a definitive developmental delay if scores fell at or beyond two SDs below the mean.25
Clinical and demographic characteristics
The general information questionnaire was a self-designed questionnaire, including pediatric and parental characteristics. Parental characteristics comprised parental high-altitude exposure history, medical history, perinatal information (including parity and mode of delivery), and daily behavioral information (including maternal alcohol and tobacco use), as well as socioeconomic status (SES, including parental education and household income). Pediatric characteristics included infants’ high-altitude exposure, breast feeding, nurturer, and any medical events occurred during the study period.
Statistical analysis
The statistical analysis was performed using SPSS 26.0 (IBM, Armonk, NY), GraphPad Prism 9.5.0 (GraphPad Software Inc., San Diego, CA), and R 4.2.2 software. Multiple imputation by chained equations (MICE, m = 50) was conducted under the missing at random (MAR) assumption, incorporating maternal age, BMI, smoking/drinking, parental educational level, household income, parity, and mode of delivery as predictors. The multiple imputation of ASQ-3 scores was performed by predictive mean matching (PMM) using the ‘mic’ package in R. Both height and weight measurements were standardized using z-score transformation prior to analysis. Categorical variables were compared using Pearson’s χ² test, and the Fisher’s exact test was utilized when expected cell counts were below five. For continuous variables, the Student’s t-test, Mann-Whitney U test, and one-way analysis of variance (ANOVA) were employed as appropriate. Repeated-measures ANOVA was utilized to analyze longitudinal body length/height and weight data of infants. Logistic regression analysis was conducted at 6, 12, and 18 months of follow-up to identify risk factors associated with ASQ failure. A stepwise elimination approach was applied, sequentially removing variables with the highest p-value until only those with p < 0.10 remained in the final model.
Results
Infants’ and parents’ clinical and demographic characteristics
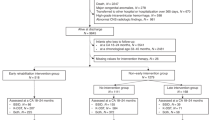
A schematic representation of the number of infants recruited and followed throughout the study is presented in Fig. 1. Between June and September, 150 infants (n high-altitude = 50; n low-altitude = 100) were recruited into this longitudinal prospective cohort study. Robust measures were implemented to maximize follow-up response rates. During the 18-month follow-up, 3 infants were lost to follow-up in the high-altitude group, while 15 were lost to follow-up in the low-altitude group.
LTFU loss to follow-up, M male, F female.
Parental characteristics at the baseline investigation are presented in Table 1. The results revealed that there were significant differences in paternal/maternal educational level and parity between the two groups, which aligned with the expected socioeconomic differences between the two cities and the study’s eligibility criteria. Mothers’ oxygen saturation level at high altitude was 89.33 ± 0.78% (measured 72 h postpartum), indicating the presence of systemic hypoxia among mothers residing at high-altitude environments. Multiple imputation with auxiliary variables was employed to reduce the risk of bias.
Details of high-altitude exposure among mothers and infants in the Lhasa group are presented in Supplementary Table S1. Among 50 mothers in this group, 31 (62.0%) had resided in a high-altitude region for more than two years, and 47 (94.0%) had spent less than four weeks away from high altitude during pregnancy. Follow-up investigations revealed that infants occasionally left the high-altitude area during parental vacations, while returned afterward. All infants had been continuously exposed to high-altitude for more than 16 weeks per 6-month period, with the exception of one infant who spent 11 weeks away from the high plateau between 6 and 12 months of age. These findings suggest that all infants in the high-altitude group experienced prolonged high-altitude exposure during both the prenatal period and early postnatal development.
Attrition was effectively mitigated throughout the study period. To validate the robustness of the statistical inferences, sensitivity analysis was conducted to determine the minimum detectable effect size (MDES) given the final sample sizes at each follow-up timepoint and a power of 0.80 (alpha = 0.05). The analysis revealed that the study was sufficiently sensitive to detect even small-to-moderate effects. Specifically, the study was powered to detect an effect size of Cohen’s d = 0.32 at the 6-month follow-up, which decreased to d = 0.28 at the 12-month follow-up and further to d = 0.21 at the 18-month follow-up. This is particularly notable for the 18-month assessment (effect sizes of d ≥ 0.25 are generally considered meaningful), where the detectable effect size is close to the threshold of meaningful effect, and the study still maintains adequate power to identify the key effects of interest.
Comparison of neurodevelopmental outcomes between high-altitude and low-altitude infants
At each timepoint (month at birth/6/12/18), there were no significant differences in body length/height and weight between the two groups (p length/height at birth = 0.525, p length/height at month 6 = 0.730, p length/height month 12 = 0.813, p length/height month 18 = 0.501, p weight at birth = 0.078, p weight at month 6 = 0.393, p weight month 12 = 0.426, p weight month 18 = 0.618), indicating that high-altitude exposure had no negative influence on infants’ physical growth.
ASQ-3 neurodevelopmental scores in the low-altitude exposure and high-altitude exposure groups at 6, 12, and 18 months are presented in Fig. 2 and Table 2. At each timepoint (months 6/12/18), the gross motor scores in the high-altitude exposure group were significantly lower than those in the low-altitude group (p month 6 < 0.001, p month 12 < 0.001 and p month 18 = 0.001). However, there were no significant differences in communication, fine motor, problem solving, and personal-social scores between the two groups, indicating that exposed to high-altitude in early life stage could mainly influence the motor-related neurological function.
a Neurodevelopmental scores at month 6 by study group. b Neurodevelopmental scores at month 12 by study group. c Neurodevelopmental scores at month 18 by study group. *** p < 0.001.
The ASQ-3 neurodevelopmental diagnoses for the low-altitude and high-altitude exposure groups at 6, 12, and 18 months are summarized in Table 3. Consistent with the neurodevelopmental scores, the proportion of gross motor failures (categorized as “Blow/Close to cut-off”) was significantly higher in the high-altitude group compared to the l group (p month 6 < 0.05, p month 12 < 0.01 and p month 18 < 0.001). This indicates a higher risk of motor developmental delay in infants exposed to high-altitude during early life. The consistent differences in gross motor scores and categories across all timepoints highlight a clear trend.
Characteristics associated with infants’ neurodevelopmental outcomes
Given the significant differences in paternal and maternal educational levels and parity between the two groups, subgroup analysis was conducted exclusively in the high-altitude exposure group to evaluate the potential impact of these factors on ASQ scores and neurodevelopmental classification (Supplementary Tables S2 and S3). Parental educational levels were categorized as junior middle school or below, senior middle school or equivalent, and college or above, while parity was classified as nulliparous or multiparous. The analyses revealed that neither parental educational level nor parity influenced the association between high-altitude exposure and ASQ failure, as no significant differences were identified in gross motor scores or failure rates across these stratifications.
Logistic regression analysis was performed at 6, 12, and 18 months to identify risk factors for ASQ gross motor failure, incorporating variables such as high-altitude exposure, parental educational level, smoking, alcohol consumption, maternal age, gestational age, parity, mode of delivery, and breastfeeding (Supplementary Table S4). When all variables were retained in the model, high-altitude exposure was associated with a significantly elevated risk of ASQ gross motor failure, with odds ratios (ORs) of 3.691 (95% CI: 1.180–12.030) at 12 months and 4.675 (95% CI: 1.504–14.528) at 18 months. None of the other variables, including parental education and parity, demonstrated statistically significant ORs. In the backward stepwise regression model, high-altitude exposure remained the only retained variable, yielding ORs of 2.852 (95% CI: 1.095–7.429, p < 0.05) at 6 months, 2.844 (95% CI: 1.076–7.517, p < 0.05) at 12 months, and 5.658 (95% CI: 2.173–14.732, p < 0.001) at 18 months. Collectively, these findings suggest that infants exposed to high-altitude exposure in utero and early life are at an increased risk of failing the gross motor ASQ category.
Discussion
China has the world’s largest high-altitude immigrant population, making the health effects of chronic high-altitude exposure a critical concern in public health and environmental medicine. This study aimed to compare the neurodevelopmental status of infants born and raised at high altitudes with those at low-altitude in the Han population. The findings revealed that while prenatal and early-life exposure to high-altitude elevated the risk of gross motor neurodevelopmental delay, no significant differences were identified in other domains of the ASQ-3. Moreover, the proportion of infants classified as “Below/Close to Cut-off” in gross motor function was significantly higher in the high-altitude exposure group compared with that in the control group, highlighting an escalated risk of motor developmental retardation associated with early-life high-altitude exposure. Notably, this is the first study to assess neurodevelopmental outcomes in high-altitude born and raised Han infants compared with low-altitude infants, providing valuable insights for decision-making regarding potential high-altitude migration.
As expected, the present study found that infants born and raised at high-altitude had lower neurodevelopmental scores and were more likely to fail (below/close to cut-off) in the gross motor domain of ASQ-3, indicating that high-altitude exposure in pregnancy and infancy might lead to delay in the neurodevelopment of motor-related function. This finding confirms prior research, demonstrating the detrimental impact of high-altitude exposure on motor function in infants.26 The early postnatal period is characterized by rapid neurodevelopmental progression, wherein the acquisition of motor skills plays a remarkable role in the integrity of underlying neurological processes. Motor development in infancy constitutes a highly integrative phenomenon, necessitating the precise regulation of multiple neural circuits in conjunction with maturational refinements of musculoskeletal function.27 As previously documented, the putamen is fundamentally implicated in motor regulation and procedural learning, predominantly mediated through dopaminergic neurotransmission.28,29 Prolonged high-altitude exposure has been linked to neuronal attrition and dysregulated intrinsic neural activity in the putamen, resulting in attenuated functional connectivity with higher-order cognition-associated regions, including the superior temporal gyrus, anterior cingulate cortex, and hippocampus.11,12 Analogous to its pathophysiological effects on adults, chronic high-altitude exposure during early life stages may precipitate neurodevelopmental perturbations by inducing structural and functional maladaptations in the putamen, necessitating further elucidation through advanced neuroimaging methodologies. Despite the noticeable impairment in gross motor proficiency among high-altitude infants, no significant differences in somatic growth parameters were identified between the high-altitude and low-altitude groups. This difference highlights the selective vulnerability of neural substrates governing motor control to hypoxic insult, confirming the hypothesis that altitude-induced neurodevelopmental deficits primarily arise from central rather than peripheral mechanisms.
In contrast to findings from previous studies,30,31 no statistically significant associations were identified between parental characteristics (educational level, smoking status, alcohol consumption, maternal age, gestational age, and parity) and gross motor outcomes in infants, aside from the influence of high-altitude exposure. This finding highlights the predominant role of oxygen availability in shaping early neurodevelopmental trajectories, particularly in motor-related neurological domains. Cerebral hypoxia constitutes a fundamental pathophysiological mechanism underlying both neurodevelopmental perturbations in infants and neurodegenerative sequelae in adults. Postnatally, sustained exposure to high-altitude may further exacerbate neurodevelopmental vulnerabilities by impairing neonatal pulmonary gas exchange, thereby reducing arterial oxygen saturation and perpetuating cerebral oxygen insufficiency. Hypoxia-induced neuropathological alterations in functionally integral brain regions may subsequently manifest as persistent deficits in motor function and cognition throughout early childhood.
The mechanisms through which hypoxia disrupts fetal and infant neurodevelopment are complicated, involving aberrations in neural stem cell homeostasis and synaptic plasticity modulation. Neural stem cells play a remarkable role in modulating both embryonic neurogenesis and postnatal neural remodeling, mediating key processes, such as proliferation, lineage specification, neuronal migration, axonal outgrowth, and synaptic integration.32,33 While physiological hypoxia in a certain threshold can transiently enhance neural stem cell proliferation and differentiation, severe and prolonged high-altitude exposure has been demonstrated to induce cell cycle arrest, quiescence, and apoptotic degeneration in neural progenitor populations.34,35 Given the critical role of neural stem cells in early brain maturation, persistent hypoxia may dysregulate neurodevelopment by disrupting progenitor cell dynamics and neurogenic capacity during prenatal and early postnatal life. Another essential mechanism implicated in hypoxia-mediated neurodevelopmental impairment involves the disruption of synaptic plasticity, a fundamental morphofunctional process governing neuronal circuit refinement and network maturation. Synaptic plasticity perturbations have been implicated in both neurodevelopmental disorders and neurodegenerative conditions, and disruptions in synaptic connectivity and neurotransmission could serve as hallmarks of various neuropathological states.36,37 Previous studies have revealed that chronic neonatal intermittent hypoxia could induce pathological dysregulation of hippocampal synaptic plasticity, resulting in aberrant long-term potentiation and maladaptive synaptic remodeling.38 Similarly, neonatal exposure to hypercapnic conditions, characterized by the elevated carbon dioxide level, has exhibited to impair hippocampal synaptic plasticity, subsequently leading to cognitive dysfunction later in life.39 Despite accumulating evidence elucidating the neurobiological substrates of hypoxia-induced neurodevelopmental impairment, further research is warranted to comprehensively explore the correlation between altitude-associated high-altitude exposure and early-life neurological development, particularly in motor-related neural circuits and higher-order cognitive networks.
Our findings align with high-altitude studies demonstrating that constrained physical activity opportunities—whether from environmental hypoxia (muscle oxygenation), cultural practices (prolonged swaddling), or thermal stress (cold floors limiting tummy time) – disrupt typical movement experience essential for motor pathway development. High-altitude exposure and cultural practices also synergistically impair infant motor neurodevelopment through restricting physical activity. Chronic oxygen deprivation (SaO₂ 80–90% at ≥2500 m) reduces spontaneous movements by 22%40 due to muscle fatigue and CNS suppression. Additionally, traditional swaddling (>12 h/day) and thermal protection further limit activity, reducing prone time to only 8 ± 3 min versus the WHO’s recommended 30 min41. This “double-hit” effect may disrupt activity-dependent neurodevelopment beyond the direct impact of high-altitude exposure on the CNS.
Children residing at high altitudes encounter distinct physiological challenges that may profoundly impact their health and development. Investigating the effects of altitude on postnatal and infant health outcomes is crucial for understanding its long-term biological consequences and developing strategies to mitigate potential adverse effects. The findings of this study hold significant implications for public policies aimed at optimizing pediatric neurodevelopment, as interventions in this domain may yield substantial benefits for overall child health. Given that the neurodevelopmental impact of high-altitude exposure may be associated with complex biological mechanisms that remain largely uncharacterized, preventive strategies may be difficult to formulate. Nevertheless, an effective approach to mitigating these risks could involve early screening for neurodevelopmental deficits among high-altitude-residing infants, accompanied by the enhanced household investments in early childhood development to counterbalance any deficiencies. Additionally, local governments may consider allocating greater resources to support the neurodevelopment of immigrant offspring, particularly concentrating on infants exhibiting lower neurodevelopment.
Several limitations of the present study are noteworthy. Firstly, the sample size was relatively constrained. As of 2020, the Tibetan region was home to approximately 3,241,000 indigenous residents and 407,100 high-altitude immigrants from low-altitude areas. Due to the dispersed distribution of the immigrant population, recruitment of sufficient immigrant parents who had given birth at high altitude proved challenging. Moreover, a substantial proportion of mothers were excluded due to the prolonged absences from high-altitude environments during pregnancy or a preference for relocating their children to low-altitude regions postnatally. Future large-scale research will be essential to further validate and enhance the generalizability of these findings. Secondly, demographic-based differences were noted between the high-altitude and low-altitude groups, particularly regarding parental educational levels and parity distribution. These differences were, in part, attributable to socioeconomic differences between Lhasa and Xi’an. Although matching procedures were implemented to control for key confounders identified in the literature (pediatric sex, birth weight, gestational age, and maternal age),42 it was pragmatically unfeasible to achieve precise matching for parental education and parity status. To address this limitation, subgroup analysis and logistic regression models were employed to adjust for these confounding variables. The results confirmed that high-altitude exposure emerged as the predominant and statistically significant factor, contributing to neurodevelopmental differences between the high-altitude and low-altitude groups. Thirdly, the present study was conducted exclusively among Han ethnicity high-altitude immigrant population, and indigenous Tibetan communities were not included. This delimitation inherently constrains the generalizability of the findings. Han populations exhibited poorer physiological adaptation to high-altitude environments compared with Tibetans, as evidenced by their attenuated hypoxic ventilatory responses and lower hemoglobin oxygen affinity. Future investigations should incorporate Tibetan cohorts to enhance the epidemiological representativeness and potential clinical applicability of findings across diverse high-altitude adaptation phenotypes.
This research is motivated by a significant public health challenge in China: a large, yet understudied, population of migrant children who undergo critical stages of growth and development (i.e., the first 1000 days of life, from conception to age 2) in persistent high-altitude environments. Current research on high-altitude pediatric health is limited in two key ways: firstly, most studies examine either prenatal or postnatal high-altitude exposure in isolation, neglecting their cumulative effects; secondly, there is a lack of longitudinal data tracking the growth and development of migrant children over time. By addressing these gaps, this study provides novel insights into the unique physiological (e.g., adaptive changes in oxygen metabolism) and sociological (e.g., interactions between high-altitude exposure and socioeconomic factors, such as access to pediatric care) challenges faced by this vulnerable population.
In summary, targeted interventions for parents and infants living at high altitudes should adopt a multidimensional strategy concentrated on alleviating hypoxic stress, safeguarding maternal and infant health, and protecting neurodevelopment. For infants, priority measures include enhanced blood oxygen monitoring coupled with individualized oxygen supplementation, supplementation with key nutrients (e.g., iron and vitamins), and the implementation of early neurodevelopmental screening. For parents, interventions concentrate on strengthening guidance for altitude acclimatization and nutritional management during pregnancy, as well as providing training on the identification of infant developmental status and scientific childcare practices. Collectively, these interventions can not only mitigate the adverse physiological effects of the high-altitude environment on mothers and infants but also provide practical support for protecting early infant neurodevelopment and reducing long-term cognitive risks, thereby laying a scientific foundation for formulating maternal and infant health policies in high-altitude regions.
Conclusions
This prospective cohort study identified suboptimal neurodevelopmental outcomes among high-altitude immigrants’ offspring who experienced prolonged high-altitude exposure during both prenatal and early postnatal stages. Despite exhibiting no significant differences in physical growth parameters versus low-altitude infants, infants born and raised at high altitudes demonstrated remarkably lower gross motor neurodevelopmental scores and an increased likelihood of developmental failure in this domain. These findings highlight the medical necessity of implementing earlier neurodevelopmental screening for high-altitude infants, along with enhanced public health interventions and targeted investments in early childhood development to mitigate potential deficits.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Blumthaler, M., Ambach, W. & Ellinger, R. Increase in solar UV radiation with altitude. J. Photochem. Phoyobiol. Biol. 39, 130–134 (1997).
Gallagher, S. A. & Hackett, P. H. High-altitude illness. Emerg. Med Clin. North Am. 22, 329–355 (2004).
Li, C. et al. Genetic changes in the Epas1 gene between Tibetan and Han ethnic groups and adaptation to the plateau hypoxic environment. PeerJ 7, e7943 (2019).
Xing, J. et al. Genomic analysis of natural selection and phenotypic variation in high-altitude Mongolians. PLoS Genet. 9, e1003634 (2013).
Lorenzo, V. F. et al. Genetic adaptation to extreme hypoxia: study of high-altitude pulmonary edema in a three-generation Han Chinese fmily. Blood Cells Mol. Dis. 43, 221–225 (2009).
Hill, C. M. et al. Cognitive performance in high-altitude andean residents compared with low-altitude populations: from childhood to older age. Neuropsychology 28, 752–760 (2014).
Bucks, R. S., Olaithe, M. & Eastwood, P. Neurocognitive function in obstructive sleep apnoea: a meta-review. Respirology 18, 61–70 (2013).
Hogan, A. M., de Haan, M., Datta, A. & Kirkham, F. J. Hypoxia: an acute, intermittent and chronic challenge to cognitive development. Dev. Sci. 9, 335–337 (2006).
Hogan, A. M. et al. Development of aptitude at altitude. Dev. Sci. 13, 533–544 (2010).
Yan, X. Cognitive impairments at high altitudes and adaptation. High. Alt. Med. Biol. 15, 141–145 (2014).
Chen, X. et al. Cognitive and neuroimaging changes in healthy immigrants upon relocation to a high altitude: a panel study. Hum. Brain Mapp. 38, 3865–3877 (2017).
Chen, X. et al. Combined fractional anisotropy and subcortical volumetric abnormalities in healthy immigrants to high altitude: a longitudinal study. Hum. Brain Mapp. 40, 4202–4212 (2019).
Chen, J. et al. Increased intraregional synchronized neural activity in adult brain after prolonged adaptation to high-altitude hypoxia: a resting-state FMRI study. High. Alt. Med Biol. 17, 16–24 (2016).
Chen, J. et al. Long-term acclimatization to high-altitude hypoxia modifies interhemispheric functional and structural connectivity in the adult brain. Brain Behav. 6, e00512 (2016).
Zhang, X. et al. Resting-state neuronal activity and functional connectivity changes in the visual cortex after high altitude exposure: a longitudinal study. Brain Sci. 12, 724 (2022).
Fan, C. et al. Reversible brain abnormalities in people without signs of mountain sickness during high-altitude exposure. Sci. Rep. 6, 33596 (2016).
Chaudhury, S., Sharma, V., Kumar, V., Nag, T. C. & Wadhwa, S. Activity-dependent synaptic plasticity modulates the critical phase of brain development. Brain Dev. 38, 355–363 (2016).
Wehby, G. L. Living on higher ground reduces child neurodevelopment-evidence from South America. J. Pediatr. 162, 606–611.e601 (2013).
Virués-Ortega, J., Garrido, E., Javierre, C. & Kloezeman, K. C. Human behaviour and development under high-altitude conditions. Dev. Sci. 9, 400–410 (2006).
Kirkham, F. J. & Datta, A. K. Hypoxic adaptation during development: relation to pattern of neurological presentation and cognitive disability. Dev. Sci. 9, 411–427 (2006).
Beall, C. M. et al. Hemoglobin concentration of high-altitude Tibetans and Bolivian Aymara. Am. J. Phys. Anthropol. 106, 385–400 (1998).
Meher, S., Hernandez-Andrade, E., Basheer, S. N. & Lees, C. Impact of cerebral redistribution on neurodevelopmental outcome in small-for-gestational-age or growth-restricted babies: a systematic review. Ultrasound Obstet. Gynecol. 46, 398–404 (2015).
Figueras, F. et al. Small-for-gestational-age fetuses with normal umbilical artery doppler have suboptimal perinatal and neurodevelopmental outcome. Eur. J. Obstet. Gynecol. Reprod. Biol. 136, 34–38 (2008).
Bian, X. Y. et al. [Studies of the norm and psychometric properties of ages and stages questionnaires in Shanghai children]. Zhonghua Er Ke Za Zhi 48, 492–496 (2010).
Yao, G. Y., Bian, X. Y., Squires, J., Wei, M. & Song, W. Cutoff scores of the Ages and Stages Questionnaire-Chinese for screening infants and toddlers. Zhonghua Er Ke Za Zhi 48, 824–828 (2010).
Saco-Pollitt, C. Birth in the Peruvian Andes: physical and behavioral consequences in the neonate. Child Dev. 52, 839–846 (1981).
Bayley, N. Comparison of mental and motor test scores for ages 1-15 months by sex, birth order, race, geographical location, and education of parents. Child Dev. 36, 379–411 (1965).
Kreitzer, A. C. & Malenka, R. C. Striatal plasticity and basal ganglia circuit function. Neuron 60, 543–554 (2008).
Herrero, M. T., Barcia, C. & Navarro, J. M. Functional anatomy of thalamus and basal ganglia. Childs Nerv. Syst. 18, 386–404 (2002).
Shuffrey, L. C. & Fifer, W. P. in Encyclopedia of Infant and Early Childhood Development (Second Edition) (Benson, J. B. ed.) 608-620 (Elsevier, 2020).
Moore, L. G., Charles, S. M. & Julian, C. G. Humans at high altitude: hypoxia and fetal growth. Respir. Physiol. Neurobiol. 178, 181–190 (2011).
Galiakberova, A. A. & Dashinimaev, E. B. Neural stem cells and methods for their generation from induced pluripotent stem cells in vitro. Front Cell Dev. Biol. 8, 815 (2020).
Li, G., Liu, J., Guan, Y. & Ji, X. The role of hypoxia in stem cell regulation of the central nervous system: from embryonic development to adult proliferation. CNS Neurosci. Ther. 27, 1446–1457 (2021).
De Filippis, L. & Delia, D. Hypoxia in the regulation of neural stem cells. Cell Mol. Life Sci. 68, 2831–2844 (2011).
Fan, Y., Li, J. & Fang, B. A tale of two: when neural stem cells encounter hypoxia. Cell Mol. Neurobiol. 43, 1799–1816 (2023).
Mordelt, A. & de Witte, L. D. Microglia-mediated synaptic pruning as a key deficit in neurodevelopmental disorders: hype or hope? Curr. Opin. Neurobiol. 79, 102674 (2023).
Batool, S. et al. Synapse formation: from cellular and molecular mechanisms to neurodevelopmental and neurodegenerative disorders. J. Neurophysiol. 121, 1381–1397 (2019).
Goussakov, I., Synowiec, S., Aksenov, D. P. & Drobyshevsky, A. Occlusion of activity dependent synaptic plasticity by late hypoxic long term potentiation after neonatal intermittent hypoxia. Exp. Neurol. 337, 113575 (2021).
Tachibana, K. et al. Neonatal exposure to high concentration of carbon dioxide produces persistent learning deficits with impaired hippocampal synaptic plasticity. Brain Res 1507, 83–90 (2013).
Julian, C. G. High altitude during pregnancy. Clin. Chest Med 32, 21–31 (2011).
Weitz, C. A., Liu, J. C., He, X., Chin, C. T. & Garruto, R. M. Responses of Han migrants compared to Tibetans at high altitude. Am. J. Hum. Biol. 25, 169–178 (2013).
Peacock, J. L., Marston, L., Marlow, N., Calvert, S. A. & Greenough, A. Neonatal and infant outcome in boys and girls born very prematurely. Pediatr. Res 71, 305–310 (2012).
Acknowledgements
We thank all infants and their parents who participated in this study.
Funding
This study was supported by the National Key R&D Program of China (Grant No. 2023YFA1801200), National Science Foundation of China (Grant Nos. 82404215 and 82271920), and Natural Science Research Project of Shaanxi Province (Grant No. 2024JC-YBQN-0865).
Author information
Authors and Affiliations
Contributions
Y.Z. Investigation, Data curation, Formal analysis, Resources, Methodology, Funding acquisition; X.L. Investigation, Methodology; Q.Z. Investigation, Visualization; M.L. Investigation; S.Z. Writing, editing; Q.D. Investigation; S.M. Investigation; X.D. Investigation; Q.Z.X.: Investigation, Resources; D.L.: Resources; L.O.: Resources; X.G.: Resources; W.Z.: Validation, Writing—review & editing; K.D.: Supervision, Validation, Funding acquisition; X.C.: Writing, Software, Designing, Project administration, Funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Informed consent
The written informed consent for participation was obtained from infants’ parents.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhou, Y., Liu, X., Zhang, Q. et al. Neurodevelopmental outcomes of infants born at high altitude: evidence from Tibet. Pediatr Res (2026). https://doi.org/10.1038/s41390-026-04821-y
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41390-026-04821-y