Abstract
Background
Hypoxic-ischemic encephalopathy (HIE) remains a major cause of neonatal mortality and long-term neurodevelopmental disability. Intranasally delivered mesenchymal stem cells and neurotrophic factors from breast milk have been detected in the central nervous system and may have a role in neuroregenerative processes. F-NEO-BRIGHT (Feasibility- NEOnatal- intranasal BReast milk, Impact on brain Growth in HIE Therapy) is the first clinical study aimed to assess the feasibility and safety of intranasal fresh breast milk (iFBM) in neonates with HIE in both inpatient and home settings.
Methods
This prospective, single-center study involved ten patients diagnosed with moderate to severe HIE treated with therapeutic hypothermia. Own-mother’s fresh breast milk expressed within 4 h, was given intranasally twice daily, 0.4 ml in each nostril, initiated within 48 h of life and continued until day 28.
Results
Under continuous cardiorespiratory and neurointensive monitoring in neonatal intensive care, the therapy was well tolerated with no adverse events or side effects observed. The intervention proved to be feasible, treatment initiation was achieved within 48 h in all enrolled neonates and was successfully continued by parents in the home setting.
Conclusion
iFBM in neonates with moderate to severe HIE is safe and feasible in both inpatient and home settings.
Impact
-
F-NEO -BRIGHT Study is the first clinical trial where intranasal fresh breast milk is administered to neonates with neonatal encephalopathy.
-
Intranasal fresh breast milk administration during the first month of life appears to be feasible, safe and well-tolerated both in hospital and in the home settings.
-
The full therapeutic potential of this treatment could be revealed by further research into long-term neurodevelopmental outcomes.
Similar content being viewed by others
Introduction
Perinatal asphyxia, resulting in hypoxic-ischemic encephalopathy (HIE), is the most common cause of neonatal encephalopathy and a leading cause of neonatal mortality and disability.1 Improving long-term neurodevelopmental outcomes of newborns with HIE has been an intense area of research over the past decade.2 Based on our current knowledge, therapeutic hypothermia is the only therapy proven to reduce central nervous system damage in neonates with HIE.
Breast milk is a complex biological substance that contains a variety of bioactive components, including neurotrophic growth factors, cytokines, immunoglobulins, hormones and multipotent stem cells.3,4,5 Exclusive breastfeeding in the early stages of development has been shown to have a positive impact on cognitive outcomes.6,7 Studies also found that colostrum is the richest source of stem cells in breast milk.8 Animal studies demonstrated that mesenchymal stem cells and neurotrophic substances found in breast milk, when administered intranasally, enter the central nervous system and reduce the extent of neurological damage.7,9,10,11,12 Intranasal fresh breast milk (iFBM) has been shown to be safe and well-tolerated in preterm infants.13,14,15
In our F-NEO-BRIGHT study we aimed to evaluate the feasibility and safety of administering own mother’s iFBM to neonates with moderate to severe HIE receiving therapeutic hypothermia, initiated within the first 48 h of life and continued until the age of 28 days. The secondary objective was to evaluate the progression of enteral feeding and the duration of exclusive breastfeeding.
Methods
This prospective, open-label, single-center feasibility study was conducted in the tertiary Neonatal Intensive Care Unit of the Pediatric Center at Semmelweis University, Budapest, Hungary, treating out-born patients only. Written informed parental consent was obtained after a detailed verbal discussion. The study was performed in accordance with the Declaration of Helsinki and approved by the Hungarian National Center for Public Health and Pharmacy (NNGYK/40202-5/2024, SE-NEONAT-03/2024) and registered on ClinicalTrilals.gov (NCT06747260).
Subjects
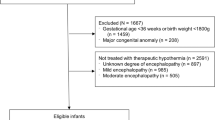
We enrolled 10 consecutive neonates born between December 2024 and February 2025 diagnosed with moderate to severe HIE (Supplemental Fig. S1). 16 Exclusion criteria were gestational age <35 weeks, postpartum asphyxia, congenital malformation, concurrent cerebral lesions, extracorporeal membrane oxygenation (ECMO), contraindication of lactation or other barriers to providing fresh breast milk. A multidisciplinary team consisting of nursing staff, physicians, lactation consultants, physiotherapists and social workers provided guidance and assistance for parents with expression, transport and administration of breast milk. Lactation and well-being support was available in-person and via telephone throughout the study period. Geographical distance was not considered as an exclusion criteria. Aid with transport was organized with the help of volunteers if requested by the parents.
Measurements
iFBM was defined as own mother’s fresh breast milk expressed within 4 h and transported at room temperature. Using a standard plastic 1 ml syringe, twice a day 0.4 ml iFBM was administered in each nostril over one minute, 15 min apart.13,14 For participants receiving non-invasive ventilation, the mask or nasal prong was removed during treatment. Gentle suctioning was applied prior to treatment if required to remove excessive nasal secretions. Intervention was initiated as soon as possible, but always within 48 h of age. iFBM administration was initially performed by nursing staff or the attending physician. Parents observed the first treatments, received training and then continued with the administration throughout the hospital stay and subsequently at home until day 28. Parents were trained to express, collect, transport and administer breast milk using an aseptic approach. To ensure protocol adherence iFBM administration was logged in the patient’s chart and a paper-based diary, which was kept by parents following discharge and collected at the time of their first follow-up appointment. Parents were contacted at least twice a week for ongoing support and confirmation of home administration times.
The maximum number of doses over 28 days was 56, however due to predicted challenges with breast milk transport during hospital stay, a pragmatically achievable target was set at 50 doses per patient.
Our goal was to administer 0.5-1.5×106/kg viable stem cells. In each study participant, we aimed to achieve 50 doses of 0.4-0.4 ml in each nostril. Based on an average weight of 3.28 kg,17 50 doses were estimated to deliver 1.4×106/kg stem cells (50 doses x 0.8 ml x 0.23×106/ml x 0.5 = 4.6×106 / 3.28 kg = 1.4×106/kg).14
Protocol
Whole-body cooling was induced as described in the Total Body Hypothermia for Neonatal Encephalopathy (TOBY) trial; initiated within the first 6 h of life and maintained for 72 h, followed by gradual rewarming.18 First-line sedation was morphine sulphate infusion (loading dose of 100 µg/kg, followed by continuous infusion of 10 µg/kg/h), adjusted based on response. Second line sedation was midazolam (boluses of up to 100 µg/kg/h followed by continuous infusion of 100 µg/kg/h if required) or dexmedetomidine (continuous infusion initiated at 0.2 µg/kg/h titrated up to a maximum dose of 1 µg/kg/h). No changes in sedation were made in relation to iFBM administration.
Cerebral activity was recorded using continuous 2-channel aEEG monitoring (Olympic Brainz CFM Neonatal monitor or Moberg CNS monitor, Natus Medical or EEG-1200K; Nihon Kohden). Recording was initiated as soon as possible, but in all cases before 6 h of age and continued for the duration of therapeutic hypothermia and rewarming, for a total of 84 h. aEEG background and seizure activity was scored for each 6-h interval by a neonatologist (UM), converting the Hellstrom–Westas criteria to a numeric value: continuous normal voltage (CNV; score 1), discontinuous normal voltage (DNV; score 2), burst suppression (score 3), continuous low voltage (score 4), and flat trace (score 5).19,20 The onset of sleep-wake cycling was also noted and scored (0, absent; 1, abnormal; 2, normal).21
First-line antiseizure medication was phenobarbital sodium, repetitive seizures were treated with a second loading dose of phenobarbital. Enteral feeding during hypothermia was initiated as early as possible using the mother’s own breast milk, starting at a dose of 10 ml/kg/day and increased as tolerated with a maximum daily increment of 10 ml/kg/day. During normothermic phase, the increment was increased to 30 ml/kg/day supplemented by donor breast milk if needed.
Post-rewarming brain MRI was performed at the Medical Imaging Center of Semmelweis University using a 3-T Philips Achieva scanner (Philips Medical Systems). T1-, T2-, diffusion-weighted MRI scans with apparent diffusion coefficient and proton magnetic resonance spectroscopy measurements were performed. Images were scored according to Weeke scoring system by a board-certificated radiologist specialized in neuroimaging (NN), blinded to the clinical data.22
Data analysis
Primary outcomes were feasibility and safety of iFBM therapy. Feasibility was determined by the ability to initiate treatment within 48 h, administering mother’s fresh milk expressed within 4 h and continuing treatment after discharge until day 28. Safety was based on clinical observations, cardiovascular monitoring and the absence of adverse events. Adverse events associated with iFBM administration were defined as desaturation (SpO2 < 80%), bradycardia (heart rate < 70% of baseline), apneic episode requiring bag and mask ventilation, or an increase in respiratory support (mode, setting, or FiO2 increase > 10%) within 1 h of the intervention. Secondary outcome was to evaluate the time to reach full enteral feeding and the duration of exclusive breastfeeding.
GraphPad Prism version 10.6.0 for macOS (GraphPad Software, Boston, Massachusetts) was used for data analysis. Descriptive statistics are presented as median (IQR) for continuous variables and as percentage for categorical variables.
Results
Eleven consecutive term neonates with moderate to severe HIE were assessed for eligibility, of whom ten were enrolled in the study. One patient who was not enrolled required ECMO therapy for severe meconium aspiration syndrome complicated with severe persistent pulmonary hypertension (Supplemental Fig. S1). Demographic and clinical characteristics of the population are shown in Table 1. and Supplemental Table S1. Based on clinical examination, aEEG assessment and MRI findings two patients were classified as severe and eight as moderate HIE. Of the two patients with severe HIE, one died at the age of 7 days due to multi-organ failure as a complication of perinatal asphyxia, unrelated to the study protocol. The other infant had a severe respiratory condition requiring prolonged intensive care.
All enrolled patients were already intubated and ventilated before the initiation of iFBM therapy. Four patients required non-invasive ventilatory support following extubation and 6 neonates received vasopressors and/or inotropes for circulatory support. The median aEEG score at 84 h was 2.0 (1.7–3.0), aEEG background activity normalized in 6 neonates at a median age of 35 h (24–69.5). Onset of sleep-wake cycling was observed in 8 neonates, at a median age of 25 h (7.5–45.5). Electrographic seizures were observed in 2 neonates before iFBM therapy was initiated. Both patients received a single loading dose of phenobarbital and did not require any further antiseizure medication. MRI was performed in all infants at a median age of 4.3 days with a median Weeke score of 2.0 (1.0–5.2).
iFMB feasibility
First dose of iFBM was administered at a median age of 24.7 h (14.8–40.2) and within our target of 48 h in all patients. The median duration of iFBM treatment in the surviving infants was 28 days (26.0–28.0). The summary of iFBM treatment is presented in Table 2. and Supplemental Table S1. The median of total doses administered in surviving infants was 51 (44.5–55.0). The primary reason for dose omission during hospital stay was the unavailability of fresh breast milk, as it had to be transported from a median distance of 28 km (10.4–92.1).
iFBM safety
There were no respiratory, cardiovascular or neurologic adverse events recorded in association with iFBM administration during hospital stay. All parents were trained to safely administer iFBM on the Neonatal Intensive Care Unit (NICU) with the intention to continue treatment after discharge to a different ward, hospital or home. Although home cardiorespiratory monitoring was not used, parents were advised to be vigilant, but did not report any complications such as choking, aspiration, nasal bleeding or any other symptoms requiring an emergency department visit. Figure 1. shows parental iFBM administration on the NICU.
One parent is supporting the infant’s head while the other is delivering 0.4 ml breastmilk intranasally via a small syringe to the right nostril. The photo is used with the permission of the parents.
Enteral feeding
Enteral feeding was initiated as early as possible using own mother’s breast milk at a median time of 27 h (20.5–54.5). The median time to full enteral feeds was day 8 (7–13).
Discussion
This first prospective clinical trial demonstrated that own mother’s iFBM administration for neonates with hypoxic-ischemic encephalopathy is safe, feasible and can be delivered by trained parents, paving the way for future large-scale research.
All parents approached for participation in the study consented to take part, including those who had not planned to breastfeed or had been unsuccessful in breastfeeding previously. Parental engagement was encouraged from an early stage to build the skills and confidence required for safe administration after discharge. Although all our patients were outborn, we believe the key strength of our study is that we were able to demonstrate that, through a multidisciplinary approach, it was feasible to initiate treatment in 100% of patients within the target age of 48 h, and 50% of the infants had their first treatment within 24 h of life.
No adverse events were observed in relation to the iFBM treatment. In intubated and ventilated patients the intranasal route of administration was readily accessible. Where non-invasive ventilation was required after extubation, ventilatory support was briefly discontinued during intranasal therapy. If a patient appeared uncomfortable, the dose was divided and administered in two parts, 2 minutes apart, without any observed adverse reactions. In the home setting, parents reported that the procedure was well tolerated and could be easily integrated into the infant’s normal daily routine.
In one case the treatment was discontinued by the parents due to an upper respiratory tract infection, as they perceived an increase in coughing. Notably, despite the limited scientific evidence, intranasal administration of own mother’s fresh breast milk in the treatment of common cold is widely practiced. In a survey, conducted in Poland by Kracz et al., 86.1% of mothers reported a positive effect after using intranasal own mother’s breast milk in rhinitis.23 A recent study found that the ingestion of human milk reduces outpatient upper respiratory symptoms in premature infants during their first year of life. 24
We observed high adherence to the protocol with all surviving infants receiving iFBM for more than 3 weeks. 7 out of 9 surviving infants completed the full 28-day treatment course, one patient while still on NICU and the remaining six patients at home. Although geographical distance from the NICU was not considered as an exclusion criteria, breast milk was transferred from wide range of distances (6–128 km). In the early days of treatment, the primary causes of dose omissions were insufficient quantities of own mother’s fresh breast milk and logistical challenges related to transport.
The effective dose or ideal duration of treatment with intranasal breast milk therapy in neonates with hypoxic-ischemic encephalopathy is currently unknown. The daily dosing protocol was based on human and animal studies suggesting an effective treatment dose of 0.5–1.5 × 106/kg stem cells in neurological disorders.25,26,27 Human breast milk contains stem cells at concentrations ranging between 0.1–13 × 106 /ml. 28,29,30 A recent study by Hoban et al. found that the median live cell concentration in fresh breast milk was 0.23 × 106, with half of these cells being stem cells. They also observed a significant negative correlation between cell viability and day of age.14 Given that the specific stem cell content of each dose and the extent to which these cells reach the central nervous system remains unclear, the selected dose was based on theoretical considerations from the available literature.13,14
It has been suggested that total cell count and the proportion of stem-cell markers may be substantially higher in colostrum and transitional milk compared to mature milk.8,31 We hypothesized that with proactive engagement of our multidisciplinary team, we can maximize the potential benefits of the intervention by initiating treatment as early as possible, ideally within 48 h.
Previous clinical data support that intranasal administration of 0.1–0.4 ml per nostril is well tolerated in preterm infants, and a twice daily dosing regimen was feasible for most families.13,14 The period of 28 days was felt to be pragmatic and achievable by parents.13,14
Median NICU discharge time for infants with HIE undergoing therapeutic hypothermia was found to be 11 days in a UK cohort,32 so we anticipated that there would be challenges with the continuation of therapy beyond discharge, particularly in the home setting. It is a significant achievement that with excellent parental engagement we were able to demonstrate the successful continuation of treatment in all surviving infants following discharge from NICU, both on hospital wards and in the home environment.
These findings suggest that iFBM therapy is feasible and may have the potential for broader application in other conditions associated with adverse neurodevelopmental outcome, particularly in low-resource settings where access to advanced therapies is limited. Our results highlight the need for further, well-designed, randomized controlled studies, particularly in infants diagnosed with HIE, to clarify whether iFBM therapy can improve long-term neurodevelopmental outcomes.
The optimal enteral feeding strategy for infants with HIE undergoing therapeutic hypothermia remains a topic of debate. In our study enteral feeding was commenced within 48 h of life in 80% of the infants and the median time to full enteral feeding was 8 days. This is consistent with results from other studies that support early initiation of enteral feeding.33,34
Admittedly, our study has certain limitations. It was a small pilot trial conducted at a single center, limiting generalizability. As a feasibility and safety study, it was not powered to assess clinical or neurodevelopmental outcomes. Transporting fresh mother’s milk to the NICU for outborn neonates can be logistically challenging; however in our experience, families were generally highly motivated, and coordinated support from social workers and hospital volunteers enabled daily delivery of fresh milk. Although every effort was made to minimize handling and transport time, the impact of this on the viable stem cell content of the administered own mother’s breast milk could not be quantified. Existing literature suggests that stem cells can potentially reach the central nervous system through intranasal administration, however the extent to which this occurs in individual patients could not be directly measured in our study. Although it is unclear whether potential nasal microbial colonization through iFBM administration has any clinical relevance, it was not assessed in this trial. Finally, for practical purposes, monitoring of home administration of iFBM relied on regular parental telephone consultations only and follow-up visits were not scheduled during this period. However, in each case we placed a strong emphasis on the verbal confirmation of diary documentation. All diaries were subsequently collected from the patients when they attended their follow-up appointments at one month of age.
Conclusion
The experience gained through this study represents an important preliminary step toward evaluating a novel therapeutic approach and informing the design of a larger randomized trial to assess the efficacy of iFBM therapy on long term neurodevelopmental outcome.
In infants with hypoxic-ischemic encephalopathy, intranasal breast milk administration twice daily appears to be feasible and safe both in the hospital and at home settings. Further randomized trials are warranted to explore the therapeutic potential of the treatment regarding long-term neurodevelopmental outcomes.
Data availability
Anonymized datasets collected and analyzed in the current study are freely available from the corresponding author on reasonable request.
References
Chakkarapani, E., de Vries, L. S., Ferriero, D. M. & Gunn, A. J. Neonatal encephalopathy and hypoxic-ischemic encephalopathy: the state of the art. Pediatr. Res. 98, 2444–2458 (2025).
Perez, A. et al. Long-term neurodevelopmental outcome with hypoxic-ischemic encephalopathy. J. Pediatr. 163, 454–459 (2013).
Ballard, O. & Morrow, A. L. Human milk composition: nutrients and bioactive factors. Pediatr. Clin. North Am. 60, 49–74 (2013).
Hassiotou, F. et al. Breastmilk is a novel source of stem cells with multilineage differentiation potential. Stem Cells 30, 2164–2174 (2012).
Ninkina, N. et al. Stem cells in human breast milk. Hum. Cell 32, 223–230 (2019).
Horta, B. L., Loret de Mola, C. & Victora, C. G. Breastfeeding and intelligence: a systematic review and meta-analysis. Acta Paediatr. 104, 14–19 (2015).
Strobel, K. M., Perez, K. M., Benjamin, H. & Hoban, R. Human milk as therapy: neurodevelopment and neonatal brain injury. Semin. Perinatol. 49, 152140 (2025).
Goudarzi, N. et al. Comparative phenotypic characterization of human colostrum and breast milk-derived stem cells. Hum. Cell 33, 308–317 (2020).
Donega, V. et al. Intranasal mesenchymal stem cell treatment for neonatal brain damage: long-term cognitive and sensorimotor improvement. PLoS ONE 8, e51253 (2013).
McDonald, C. A. et al. Intranasal delivery of mesenchymal stromal cells protects against neonatal hypoxic(-)ischemic brain injury. Int. J. Mol. Sci. 20, 2449 (2019).
.Kaps, J. et al. Human preterm colostrum stimulates outgrowth in neurogenic tissue. Pediatr. Res. 94, 1906–1910 (2023).
Alcala-Barraza, S. R. et al. Intranasal delivery of neurotrophic factors BDNF, CNTF, EPO, AND NT-4 TO THE CNS. J. Drug Target 18, 179–190 (2010).
Keller, T. et al. Intranasal breast milk for premature infants with severe intraventricular hemorrhage-an observation. Eur. J. Pediatr. 178, 199–206 (2019).
Hoban, R. et al. Feasibility of intranasal human milk as stem cell therapy in preterm infants with intraventricular hemorrhage. J. Perinatol. 44, 1652–1657 (2024).
Gallipoli, A. et al. Outcomes after intranasal human milk therapy in preterm infants with intraventricular hemorrhage. J. Perinatol. 45, 202–207 (2025).
Sarnat, H. B. & Sarnat, M. S. Neonatal encephalopathy following fetal distress. a clinical and electroencephalographic study. Arch. Neurol. 33, 696–705 (1976).
Hungarian Central Statistical Office. Demographic Yearbook (Központi Statisztikai Hivatal, 2024).
Azzopardi, D. V. et al. Moderate hypothermia to treat perinatal asphyxial encephalopathy. N. Engl. J. Med. 361, 1349–1358 (2009).
Meder, U. et al. Longitudinal analysis of amplitude-integrated electroencephalography for outcome prediction in hypoxic-ischemic encephalopathy. J. Pediatr. 246, 19–25 e15 (2022).
Hellström-Westas, L., Rosén, I. & Svenningsen, N. W. Predictive value of early continuous amplitude integrated eeg recordings on outcome after severe birth asphyxia in full term infants. Arch. Dis. Child. Fetal Neonatal Ed. 72, F34–F38 (1995).
Osredkar, D. et al. Sleep-wake cycling on amplitude-integrated electroencephalography in term newborns with hypoxic-ischemic encephalopathy. Pediatrics 115, 327–332 (2005).
Weeke, L. C. et al. A Novel magnetic resonance imaging score predicts neurodevelopmental outcome after perinatal asphyxia and therapeutic hypothermia. J. Pediatr. 192, 33–40 e32 (2018).
Karcz, K., Walkowiak, M., Makuch, J., Olejnik, I. & Krolak-Olejnik, B. Non-nutritional use of human milk part 1: a survey of the use of breast milk as a therapy for mucosal infections of various types in Poland. Int. J. Environ. Res. Public Health 16, 1715 (2019).
Blaymore Bier, J. A., Oliver, T., Ferguson, A. & Vohr, B. R. Human milk reduces outpatient upper respiratory symptoms in premature infants during their first year of life. J. Perinatol. 22, 354–359 (2002).
Hachimi-Idrissi, S. Stem cell therapy in neurological disorders: promises and concerns. Explor. Neuroprot. Ther. 3, 346–362 (2023).
Ahn, S. Y. et al. Mesenchymal stem cells prevent hydrocephalus after severe intraventricular hemorrhage. Stroke 44, 497–504 (2013).
Park, W. S., Ahn, S. Y., Sung, S. I., Ahn, J. Y. & Chang, Y. S. Mesenchymal stem cells: The magic cure for intraventricular hemorrhage?. Cell Transpl. 26, 439–448 (2017).
Mane, S., Taneja, S., Madala, J. S., Agarkhedkar, S. & Khetan, M. Study of stem cells in human milk. Cureus 14, e23701 (2022).
Patki, S., Kadam, S., Chandra, V. & Bhonde, R. Human breast milk is a rich source of multipotent mesenchymal stem cells. Hum. Cell 23, 35–40 (2010).
Gleeson, J. P. et al. Profiling of mature-stage human breast milk cells identifies six unique lactocyte subpopulations. Sci. Adv. 8, eabm6865 (2022).
Tang, C., Zhou, Q., Lu, C., Xiong, M. & Lee, S. Comparison and culturing different types of cells from fresh breast milk with different culture medium. Pediatr. Med. 2, 5–5 (2019).
Gale, C. et al. Nutritional management in newborn babies receiving therapeutic hypothermia: two retrospective observational studies using propensity score matching. Health Technol. Assess. 25, 1–106 (2021).
Hu, Y. et al. Early versus delayed enteral nutrition for neonatal hypoxic-ischemic encephalopathy undergoing therapeutic hypothermia: a randomized controlled trial. Ital. J. Pediatr. 48, 146 (2022).
Costa, S. et al. Enteral nutritional strategy during therapeutic hypothermia: Who? When? What?. Front. Pediatr. 12, 1357831 (2024).
Acknowledgements
We wish to thank our medical and nursing team at Semmelweis University and our partner hospitals, care providers and volunteers for their professional care and invaluable support in facilitating this study. All phases of this study were supported by the TKP2021-EGA25 grant of the Hungarian National Research, Development and Innovation Office.
Funding
Open access funding provided by Semmelweis University. Open access funding provided by Semmelweis University.
Author information
Authors and Affiliations
Contributions
Eszter Tarjanyi conducted literature search, coordinated data collection, drafted the initial manuscript and assisted participants as lactation consultant. Agnes Jermendy and Miklos Szabo supervised data collection and critically reviewed and revised the manuscript for important intellectual contributions. Ferenc Adam Brandt and Barbara Szasz collected data and carried out the initial statistical analyses. Nora Nyilas interpreted and reported the MRI examinations. Unoke Meder performed literature search, conceptualized and designed the study, designed the data collection instruments, analyzed the data, and critically reviewed, revised and drafted the final manuscript. All authors approved the final manuscript and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors have no conflicts of interest to disclose. The Hungarian National Research, Development and Innovation Office had no role in the design and conduct of the study.
Consent statement
Written informed parental consent was obtained after a detailed verbal discussion.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tarjanyi, E., Jermendy, A., Szabo, M. et al. F-NEO-BRIGHT: feasibility and safety of intranasal fresh breast milk in neonatal encephalopathy. Pediatr Res (2026). https://doi.org/10.1038/s41390-026-04847-2
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41390-026-04847-2