Abstract
Background
Very preterm infants born before 32 weeks of gestation remain vulnerable to postnatal growth impairment. However, longitudinal data focusing on appropriate-for-gestational-age (AGA) infants are limited. Standard growth references may inadequately reflect the developmental trajectories of this population.
Methods
We constructed sex-specific growth curves for height, weight, body mass index, and head circumference from term-equivalent age to 6 years postmenstrual age in AGA infants born at <32 weeks of gestation without neurodevelopmental impairments. These trajectories were compared with Japanese national growth standards.
Results
Infants born before 28 weeks of gestation demonstrated significantly reduced growth relative to national standards, with the greatest deviation occurring between 40 and 60 weeks postmenstrual age. While gradual catch-up growth was observed thereafter, full alignment with standard curves was not achieved. Lower gestational age correlated with more pronounced deficits across all anthropometric parameters. Head circumference remained notably reduced among infants born before 25 weeks, raising concern for potential neurodevelopmental implications.
Conclusions
Extremely preterm AGA infants experience persistent growth deficits through early childhood. These findings underscore the importance of long-term growth surveillance and early intervention strategies to support optimal physical and neurological outcomes in this high-risk population.
Impact
-
Many extremely preterm infants grow more slowly than expected well into early childhood.
-
Babies born earlier in pregnancy show more delayed physical growth across all measures.
-
Infants born before 25 weeks may have a smaller head size, raising concerns for brain development.
Similar content being viewed by others
Introduction
Advancements in neonatal intensive care have markedly improved the survival of very preterm (VP) infants, defined as those born before 32 weeks of gestation.1,2 Recent data indicate survival rates of approximately 68% at 22 weeks and 89% at 24 weeks of gestation, reflecting significant improvements in perinatal care.3,4 Despite these improvements, postnatal growth impairment remains a persistent concern, with lasting implications for physical development, neurocognitive outcomes, and health-related quality of life.5,6
The growth trajectories of VPs and extremely preterm (EP) infants—particularly those born before 28 weeks of gestation, often diverge from reference standards derived from term-born populations, necessitating tailored, population-specific growth assessments.7,8,9,10 Accurate postnatal growth monitoring is essential for guiding clinical management, particularly nutritional strategies. Although widely adopted tools such as the Fenton growth charts11 and the World Health Organization (WHO) growth standards12 are used in neonatal and pediatric practice, they may not fully capture the unique growth patterns of extremely premature infants, particularly beyond term-equivalent age. Emerging evidence suggests that preterm growth remains distinct from term counterparts even after correction for gestational age, and that gestational age at birth plays a critical role in shaping long-term growth outcomes.7,8,9,10
In Japan, national growth reference curves have been developed;13 however, data characterizing the postnatal growth trajectories of VP or EP infants who are appropriate for gestational age (AGA) are limited. In particular, longitudinal analyses spanning from term-equivalent age through early childhood, stratified by gestational age subgroups, remain scarce.
This study aimed to construct postnatal growth curves for height, weight, body mass index (BMI), and head circumference in AGA infants born at less than 32 weeks of gestation without neurodevelopmental impairments. Furthermore, we sought to compare these trajectories, along with their corresponding standard deviation scores (SDS) and growth velocity curves, against Japanese national growth standards to elucidate deviations in growth patterns according to sex and gestational age subgroups.
Methods
Study design and participants
This multicenter, retrospective cohort study analyzed physical growth data from medical records obtained at 70 perinatal medical centers across Japan between May 1, 2022, and January 31, 2023. The study population included infants born between January 1, 2012, and December 31, 2016, with a birth weight of less than 2500 g who survived to hospital discharge. In addition to clinical records, longitudinal growth measurements were supplemented, when available, by data recorded in maternal and child health handbooks maintained by caregivers.
Eligible participants were defined as infants born at less than 32 weeks of gestation and classified as AGA, corresponding to a birth weight between the 10th and 90th percentiles based on Japanese neonatal anthropometric standards.14 Infants born at less than 22 weeks of gestation were excluded because of the inability to calculate SDS below this gestational threshold. As this was a retrospective analysis of all available eligible cases, no formal sample size calculation was performed. No imputation was performed for missing values; analyses were conducted using available data only.
Exclusion criteria included congenital anomalies known to affect physical growth, including trisomy 13, trisomy 18, trisomy 21, Turner syndrome, congenital hydrocephalus, cerebral malformations, spinal meningocele, and skeletal dysplasia. Infants undergoing growth hormone therapy were also excluded. Additionally, participants with neurodevelopmental impairments—defined as a diagnosis of cerebral palsy, developmental delay (developmental quotient <70), or a neurodevelopmental disorder diagnosed by 6 years postmenstrual age—were excluded from the analysis.
Participants were stratified into three gestational age subgroups: 22–24 weeks, 25–27 weeks, and 28–31 weeks. All growth data were analyzed separately by sex. Age in weeks is expressed as postmenstrual age, calculated as the time from the mother’s last menstrual period to birth (gestational age) plus the time since birth (chronological age). Corrected age refers to postmenstrual age minus 40 weeks.
Data collection and measurement points
Mixed longitudinal data were collected for height (or supine length prior to ambulation), weight, and head circumference, spanning term-equivalent age (40 weeks postmenstrual age) to 360 weeks (6 years post-term). The total number of measurements used for each parameter is summarized in Table S1. To minimize redundancy due to closely timed outpatient visits, only measurements obtained at intervals of ≥28 days were included for each individual. The median number of measurements per participant ranged from 9 to 13, varying by gestational age subgroup.
Growth curve modeling
Growth curves for height/length, weight, BMI, and head circumference were developed separately for each sex and gestational age subgroup (22–24 weeks, 25–27 weeks, and 28–31 weeks). Growth trajectories were modeled using the Generalized Additive Models for Location, Scale, and Shape (GAMLSS) framework,15 a flexible statistical approach suitable for complex longitudinal data. The centile curves were estimated using the LMS (lambda-mu-sigma) method,16,17 which assumes a cross-sectional structure while incorporating repeated measurements from individual participants. The LMS method models three key distributional parameters: μ (mu), representing the median (central tendency); σ (sigma), representing the coefficient of variation (dispersion); and λ (lambda), representing the skewness of the distribution. This approach enables the construction of smoothed centile curves and corresponding SDS lines at −2 SD, −1 SD, 0 SD, +1 SD, and +2 SD for each anthropometric variable. The 50th percentile of each smoothed LMS curve was defined as the 0 SD reference, and SDS values were computed relative to Japanese national growth standards. Growth velocity was defined as the annual rate of change in height, weight, and head circumference.
Clinical definitions and neonatal characteristics
Gestational age was determined using a combination of first-trimester ultrasonographic measurements and the date of the last menstrual period. Clinical conditions were defined using standardized criteria. Chronic lung disease (CLD) was diagnosed based on the need for supplemental oxygen or respiratory support at 36 weeks postmenstrual age. Sepsis was defined by the presence of a positive blood culture. Necrotizing enterocolitis (NEC) was diagnosed in accordance with Bell’s staging criteria, with Stage II or higher considered diagnostic. Periventricular leukomalacia (PVL) was identified via cranial ultrasonography and/or magnetic resonance imaging. Intraventricular hemorrhage (IVH) was classified based on Papile’s criteria, with Grade III or IV categorized as severe.
Statistical analysis
Descriptive statistics were used to summarize baseline characteristics of the study population. Continuous variables were reported as means with standard deviations (SD), while categorical variables were expressed as frequencies and percentages. Comparisons between male and female participants were conducted using the Student’s t-test or the Wilcoxon rank-sum test for continuous variables, and the chi-square test or Fisher’s exact test for categorical variables, as appropriate. Statistical significance was defined as a two-sided p-value < 0.05. All analyses were performed using SPSS software version 30.0 (IBM Corp., Armonk, NY) and R software version 4.43 (R Foundation for Statistical Computing, Vienna, Austria).
Results
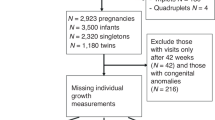
A total of 1,578 VP infants born between 22 and 31 weeks of gestation were included in the analysis, comprising 783 boys and 795 girls. Participants were stratified into three gestational age subgroups: 22–24 weeks, 25–27 weeks, and 28–31 weeks (Fig. 1). A comparison of baseline characteristics between infants with and without neurodevelopmental impairments is presented in Table S2. Infants with neurodevelopmental impairments tended to have lower gestational age and smaller birth size, were more likely to be male, and exhibited a higher incidence of perinatal complications compared with their neurodevelopmentally intact counterparts.
AGA appropriate for gestational age; GH growth hormone therapy; NI neurodevelopmental impairment; VP very preterm. NI was defined as the presence of cerebral palsy, developmental delay (developmental quotient [DQ] < 70), or a diagnosed neurodevelopmental disorder by 6 years postmenstrual age.
Table 1 summarizes the clinical characteristics of the eligible cohort. No statistically significant sex differences were observed in gestational age across any subgroup. However, girls had significantly smaller birth length, weight, and head circumference than boys. The prevalence of CLD was highest among infants born at 22–24 weeks of gestation (75.6% in boys and 71.7% in girls), with a marked decline in the 28–31 weeks group (10.0% in boys and 6.6% in girls). No significant sex-based differences were noted in the incidence of CLD, home oxygen therapy, tracheostomy, home ventilation, tube feeding, visual impairment, or hearing impairment.
As shown in Figs. S1a and S1b, the Japanese standard growth curve is approximately one standard deviation lower than the widely used Fenton + WHO growth reference across all anthropometric parameters (Fig. S1a, S1b). On the basis of this reference, we constructed longitudinal growth curves for AGA infants born at 22–24, 25–27, and 28–31 weeks of gestation, covering the period from 40 to 360 weeks postmenstrual age, and compared these trajectories with Japanese national growth standards.
Height/length trajectory
Sex-specific height/length growth curves for each gestational age subgroup are presented in Figs. S2a–S2f. Across all three gestational age groups, VP infants exhibited shorter stature compared with the Japanese standard population. However, as gestational age increased, height/length trajectories more closely approximated the national reference values.
Figure 2a–f illustrates the SDS derived from the median (0 SD) height/length growth curves, benchmarked against the Japanese standard. In all gestational age groups and both sexes, SDS values declined following term-equivalent age, reaching a nadir at approximately 60 weeks postmenstrual age. The magnitude of SDS decline was inversely proportional to gestational age, with more pronounced reductions observed in infants born at earlier gestations. Although a gradual recovery in SDS values was noted thereafter, none of the subgroups attained the standard 0 SD level.
Trajectories of height/length standard deviation scores (SDS) from term-equivalent age to 6 years, stratified by gestational age (GA) group and sex. The x-axis shows postmenstrual age in weeks (40 weeks postmenstrual age corresponds to term-equivalent age). The y-axis shows the SDS for height/length calculated using the Japanese growth reference. a, b, and c show boys born at GA 22–24, 25–27, and 28–31 weeks, respectively; d, e, and f show girls in the same GA groups. Solid lines (boys) and dashed lines (girls) represent the model-estimated mean SDS, and shaded areas indicate the 95% confidence intervals.
Figure 3a–f present a comparison of height/length growth velocity (cm/year) between this study’s median (0 SD) curves and the corresponding Japanese standard values. VP infants exhibited reduced growth velocity up to approximately 40–60 weeks postmenstrual age, with the most substantial deficits observed in the earliest gestational age subgroup. Between 60 and 80 weeks, infants born at lower gestational ages demonstrated less pronounced growth deceleration, with velocity temporarily exceeding that of the standard population. Following 80 weeks postmenstrual age, growth velocity in all subgroups progressively aligned with standard values.
Growth velocity curves for height/length from term-equivalent age to 6 years, stratified by gestational age (GA) group and sex. The x-axis shows postmenstrual age in weeks (40 weeks postmenstrual age corresponds to term-equivalent age), and the y-axis shows growth velocity (cm/year). a, b, and c show boys born at GA 22–24, 25–27, and 28–31 weeks, respectively; d, e, and f show girls in the same GA groups. Solid lines (boys) and dashed lines (girls) represent the model-estimated mean growth velocity, and shaded areas indicate the 95% confidence intervals.
Weight trajectory
Figure. S3a–f presents the sex-specific weight growth curves across the three gestational age groups. In all subgroups, VP infants exhibited lower body weight compared with the Japanese standard population. However, the degree of deviation diminished with increasing gestational age, and weight trajectories more closely approximated standard values in the later gestational subgroups.
Figure 4a–f depicts the SDS calculated by comparing the median (0 SD line) of the study’s weight growth curves with Japanese reference values. Similar to height SDS trends, weight SDS declined across all gestational age groups and both sexes, reaching a minimum at approximately 60 weeks postmenstrual age. The magnitude of SDS reduction was greatest among infants born at the lowest gestational ages. Notably, only infants in the 28–31 weeks gestational age group achieved SDS values near the standard 0 SD level; in contrast, earlier gestational age groups exhibited persistent deficits throughout the study period.
Trajectories of weight standard deviation scores (SDS) from term-equivalent age to 6 years, stratified by gestational age (GA) group and sex. The x-axis shows postmenstrual age in weeks (40 weeks postmenstrual age corresponds to term-equivalent age), and the y-axis shows the SDS for weight calculated using the Japanese growth reference. a, b, and c show boys born at GA 22–24, 25–27, and 28–31 weeks, respectively; d, e, and f show girls in the same GA groups. Solid lines (boys) and dashed lines (girls) represent the model-estimated mean weight SDS, and shaded areas indicate the 95% confidence intervals.
Figure 5a–f provides a comparison of weight growth velocity (kg/year) between this study’s median (0 SD) curves and the Japanese standard. In male infants, weight growth velocity remained below standard levels across all gestational age groups from 40 to 80 weeks postmenstrual age, eventually converging with reference values after 80 weeks.
Weight growth velocity curves from term-equivalent age to 6 years, stratified by gestational age (GA) group and sex. The x-axis shows postmenstrual age in weeks (40 weeks postmenstrual age corresponds to term-equivalent age), and the y-axis shows weight growth velocity (kg/year). a, b, and c show boys born at GA 22–24, 25–27, and 28–31 weeks, respectively; d, e, and f show girls in the same GA groups. Solid lines (boys) and dashed lines (girls) represent the model-estimated mean weight growth velocity, and shaded areas indicate the 95% confidence intervals.
Female infants exhibited a similar trend, although the decline in growth velocity was less pronounced. In females, convergence with the standard growth velocity occurred slightly earlier, at approximately 70 weeks postmenstrual age.
BMI trajectory
BMI-SDS trajectories were derived by calculating BMI from the median (0 SD line) values of height and weight curves generated in this study and comparing them with Japanese BMI reference standards, as shown in Fig. 6a–f.
Trajectories of body mass index (BMI) standard deviation scores (SDS) from term-equivalent age to 6 years, stratified by gestational age (GA) group and sex. The x-axis shows postmenstrual age in weeks (40 weeks postmenstrual age corresponds to term-equivalent age), and the y-axis shows the SDS for BMI calculated using the Japanese growth reference. a, b, and c show boys born at GA 22–24, 25–27, and 28–31 weeks, respectively; d, e, and f show girls in the same GA groups. Solid lines (boys) and dashed lines (girls) represent the model-estimated mean BMI SDS, and shaded areas indicate the 95% confidence intervals.
At 40 weeks postmenstrual age, both male and female infants exhibited BMI-SDS values greater than 0 across all gestational age groups. These values declined until approximately 60 weeks postmenstrual age and then stabilized throughout the remainder of the follow-up period. As gestational age increased, BMI-SDS values more closely approximated the 0 SD reference line; however, they remained below the standard in all groups except for those born at 28–31 weeks of gestation.
Head circumference trajectory
Sex-specific head circumference growth curves for each gestational age subgroup are presented in Figs. S4a–f. Among infants born at 22–24 weeks of gestation, the median head circumference was approximately −1 SD compared with the Japanese reference standard in both sexes. In the 25–27 weeks group, the 0 SD line for both male and female infants approximated the standard reference, while infants in the 28–31 weeks group demonstrated growth trajectories consistent with the national standard.
Figure 7a–f shows the SDS values calculated from the median (0 SD line) head circumference growth curves compared with Japanese standard values. At 40 weeks postmenstrual age, SDS values exceeded 0 across all gestational age groups and both sexes. These values declined between 40 and 60 weeks postmenstrual age and subsequently plateaued. As gestational age increased, head circumference SDS values approached the 0 SD reference, with normalization achieved only in the 28–31 weeks subgroup.
Trajectories of head circumference standard deviation scores (SDS) from term-equivalent age to 6 years, stratified by gestational age (GA) group and sex. The x-axis shows postmenstrual age in weeks (40 weeks postmenstrual age corresponds to term-equivalent age), and the y-axis shows the SDS for head circumference calculated using the Japanese growth reference. a, b, and c show boys born at GA 22–24, 25–27, and 28–31 weeks, respectively; d, e, and f show girls in the same GA groups. Solid lines (boys) and dashed lines (girls) represent the model-estimated mean head circumference SDS.
Figure 8a–f compares head circumference growth velocity (cm/year) derived from the study’s median (0 SD) curves with the Japanese standard growth velocity. Across all gestational age groups, growth velocity was reduced relative to the standard up to approximately 40–60 weeks postmenstrual age. Beyond this period, velocity trajectories gradually aligned with reference values in all subgroups.
Head circumference growth velocity curves from term-equivalent age to 6 years, stratified by gestational age (GA) group and sex. The x-axis shows postmenstrual age in weeks (40 weeks postmenstrual age corresponds to term-equivalent age), and the y-axis shows head circumference growth velocity (cm/year). a, b, and c show boys born at GA 22–24, 25–27, and 28–31 weeks, respectively; d, e, and f show girls in the same GA groups. Solid lines (boys) and dashed lines (girls) represent the model-estimated mean head circumference growth velocity, and shaded areas indicate the 95% confidence intervals.
Discussion
In this study, we developed longitudinal growth curves for AGA very preterm infants born at 22–24, 25–27, and 28–31 weeks of gestation, spanning from term-equivalent age (40 weeks postmenstrual age) to 360 weeks postmenstrual age (approximately 6 years). These trajectories were compared against the Japanese national growth standards. Across all gestational age groups, higher gestational age at birth was associated with physical stature more closely aligned with the reference population. Nonetheless, even at 6 years postmenstrual age, the median height of EP infants remained below that of the population median.
Height/length trajectories
Among EP infants, height/length SDS declined significantly between 40 and 60 weeks postmenstrual age, followed by a gradual recovery that did not reach the standard reference level. This trend is consistent with those observed in prior studies, including a longitudinal analysis of AGA infants born before 29 weeks of gestation, which reported a decline in height and weight SDS between 40 weeks and 60 weeks postmenstrual age.18 Similarly, investigations of extremely low birth weight (ELBW) and very low birth weight (VLBW) infants have demonstrated reductions in height and weight SDS from 40 weeks postmenstrual age to 1 year of corrected age.19,20 Another study evaluating 363 AGA infants born between 24 and 29 weeks with birth weights <1500 g found that the proportion of infants with z-scores <−2 SD peaked at 4 months of corrected age and subsequently declined at 8 and 21 months.21
Consistent with SDS findings, growth velocity in EP infants was lower than standard values up to 60 weeks postmenstrual age. After this period, the rate of deceleration was less pronounced in EP infants compared with reference norms, with a temporary elevation in growth velocity above standard levels before gradual convergence. These findings suggest that the most dynamic shifts in SDS occur within the first year of post-term (40–92 weeks postmenstrual age). Thereafter, SDS values stabilize, with limited further change observed.
Across all gestational age subgroups, the median height at 2 years postmenstrual age remained below the national standard. Notably, short stature at this age has been identified as a predictor of reduced final adult height.22,23 Given that SDS values tend to remain stable beyond age 2 years postmenstrual age and that the adolescent growth spurt contributes minimally to final stature in individuals born preterm,22,23 it is likely that EP infants will continue to exhibit shorter stature into adulthood.
The observed decline in SDS and growth velocity was inversely correlated with gestational age, indicating that longer gestational duration is associated with reduced deviation from normative growth patterns. Large-scale cohort studies have shown that gestational age, birth length, and birth weight independently influence final adult height, with earlier gestational age consistently linked to shorter adult stature.24,25 The period between 40 to 60 weeks of postmenstrual age corresponds to the “Infancy” phase in Karlberg’s infancy–childhood–puberty (ICP) growth model, during which the growth rate is at its lifetime peak.26,27,28 Because height gain beyond 2 years of age rarely exceeds 1 SD,28 preterm infants who fall behind during the infancy phase are unlikely to recover their growth deficit relative to term-born peers.
Weight and BMI trajectories
The 40th week of postmenstrual age generally corresponds to the timing of neonatal intensive care unit (NICU) discharge, by which point the majority of infants are capable of independent oral feeding. At this stage, body weight demonstrated an inverse relationship with gestational age; however, even among infants born at 22–24 weeks, the mean weight was approximately −0.5 SD relative to the population standard, suggesting only modest underweight status compared with that of term-born infants. Following 40 weeks postmenstrual age, a rapid decline in weight SDS was observed, reaching its lowest point between 70 and 80 weeks postmenstrual age, after which weight gain stabilized. Notably, the reduction in SDS was less pronounced in female infants, who exhibited higher SDS values than boys by 360 weeks postmenstrual age. Weight SDS remained below the reference median across all gestational age groups, with the exception of girls born at 28–31 weeks. These findings are in line with a large-scale U.S. cohort study of infants born at ≤28 weeks’ gestation and weighing <1250 g, which similarly reported higher post-discharge weight z-scores in girls than in boys.29
Weight growth velocity was lower than the Japanese standard across all groups until approximately 70–80 weeks postmenstrual age, after which it converged with the reference values. The observed sex-based differences in weight gain velocity were also consistent with previously reported trends.29
At 40 weeks postmenstrual age, BMI-SDS values were above zero across all gestational age groups, followed by a rapid decline. From 60 weeks onward, BMI-SDS remained persistently below zero throughout the observational period (up to 360 weeks postmenstrual age) in EP infants, indicating that these children were consistently thinner than their age-matched peers in the general population. This pattern of low BMI may reflect reduced muscle mass development,30 highlighting the importance of long-term monitoring of the balance between height and weight in this population.
Head circumference trajectories
Consistent with the patterns observed in height and weight, head circumference showed greater deviation from standard values with decreasing gestational age. No significant sex-based differences were identified in either SDS values or growth velocity for head circumference. Across all gestational age subgroups, SDS values declined until approximately 60 weeks postmenstrual age and then remained relatively stable throughout the remainder of the observation period. Growth velocity was consistently lower than the reference standard from 40 to 60 weeks postmenstrual age but converged with the normative trajectory thereafter. Preterm infants with low birth weight are more likely to exhibit reduced head circumference compared with term-born peers,31,32 and head circumference growth has been shown to correlate with later neurodevelopmental outcomes.31,33,34 In particular, head growth during NICU hospitalization is considered a critical predictor of subsequent neurological function.35,36 In this study, the degree of deviation in head circumference increased with decreasing gestational age, suggesting that infants born at earlier gestational ages may face a heightened risk of adverse neurodevelopmental outcomes.
Strengths
Although previous large-scale studies have examined the association between gestational age and somatic growth, the number of extremely preterm infants, particularly those born before 28 weeks of gestation, has often been limited. A major strength of the present study lies in the subdivision of the VP cohort into three distinct gestational age groups (22–24, 25–27, and 28–31 weeks), each with a sufficient sample size for independent analysis. Our findings demonstrate a clear positive association between gestational age and height/length growth, with trajectories approaching population standards as gestational age increased. Furthermore, by limiting the analysis to AGA infants, we minimized the confounding effects of intrauterine growth restriction, thereby isolating the specific influence of gestational age on postnatal growth trajectories.
Notably, our findings demonstrated a marked deceleration in growth velocity, particularly in length and head circumference, between 40 and 60 weeks postmenstrual age across all gestational age groups. This period may represent a critical window during which nutritional demands exceed spontaneous intake, particularly among infants recently discharged from the NICU and transitioning to full oral feeding. Emerging evidence suggests that protein intake is a key determinant of lean body mass accretion and neurodevelopmental outcomes in preterm infants. Therefore, targeted nutritional interventions—such as protein-enriched human milk fortification or individualized fortification strategies—during the post-discharge period may help mitigate the observed growth faltering.37,38 Preterm infants have also been reported to accumulate more body fat than full-term peers.39,40 Further research is warranted to determine whether protein-focused nutritional support during this vulnerable phase can enhance linear and head growth without contributing to excess adiposity.
Limitations
This study was conducted exclusively among Japanese participants, a population in which mean body size has gradually decreased in recent decades. The Japanese standard growth curves used in this analysis are approximately 1 SD smaller than the internationally recognized Fenton+WHO curves, which may accentuate the apparent effects of preterm birth. As such, the generalizability of these findings to other populations or ethnic groups remains uncertain, warranting further research in diverse cohorts. Given that the fit of the WHO Child Growth Standards varies across countries, part of the approximately 1-SD difference from the Fenton+WHO curves may reflect the choice of reference chart and population-level anthropometry,41 rather than prematurity-related effects alone. Deviations from the WHO standards have been reported in both directions, even in high-income countries. For example, Danish national references indicate that children are generally taller and heavier (and have larger head circumferences) than the WHO standards, and Belgian and Norwegian data show fewer children below −2 SDs and more above +2 SDs than expected under the WHO standards.42,43 In contrast, reports from Japan and Hong Kong have described systematically lower height/length (and, in some analyses, lower weight) relative to the WHO standards.44,45 Accordingly, cross-chart comparisons should be interpreted cautiously, and the apparent magnitude of growth deficits following very preterm birth may differ depending on the reference charts used and the population to which they are applied.
The study did not include assessments of nutritional status and was therefore unable to examine the relationship between nutritional and physical growth during infancy, particularly within the first 2 years of life. Nutritional deficiencies during this critical developmental period—shaped by early postnatal nutritional status—have been associated with reduced adult height.40 As outlined in Karlberg’s ICP growth model, nutritional intake during infancy plays a pivotal role in determining linear growth. Future research should focus on delineating the influence of early-life nutrition on growth trajectories during this period.
Another limitation is the inability to predict final adult stature because the present study was restricted to growth outcomes up to 6 years postmenstrual age. Previous reports have shown that adolescents or adults born with ELBW tend to exhibit shorter final height compared with term-born peers.20,32,46,47 Additionally, parental height data were not available, limiting the ability to evaluate genetic target height. Notably, this study is the first to focus exclusively on AGA children, distinguishing it from previous studies that often included small-for-gestational-age infants. The long-term physical prognosis of AGA infants born extremely preterm remains incompletely understood. Moving forward, individualized evaluations of growth trajectories will be essential for predicting long-term outcomes.
Conclusion
Extremely preterm infants exhibited significantly slower growth between 40 and 60 weeks of postmenstrual age compared with standard population references, with marked deviations observed in height, weight, BMI, and head circumference. While partial catch-up growth occurred after this period, substantial disparities in physical growth persisted throughout early childhood. AGA infants born extremely preterm may continue to exhibit smaller body size relative to term peers well into childhood. Head circumference, in particular, failed to fully normalize in those born at earlier gestational ages, raising concern for potential adverse neurodevelopmental outcomes. These findings underscore the need for long-term, comprehensive follow-up for this population. Further longitudinal studies are warranted to elucidate the relationship between early postnatal growth and long-term neurological development.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Change history
16 March 2026
This article was originally published with the incorrect license CC by NC-ND; it should have been CC by 4.0.
References
Doshi, H. et al. National trends in survival and short-term outcomes of periviable births ≤24 weeks gestation in the United States, 2009 to 2018. Am. J. Perinatol. 41, E94–E102 (2024).
Norman, M. et al. Association between year of birth and 1-year survival among extremely preterm infants in Sweden during 2004–2007 and 2014–2016. JAMA 321, 1188–1199 (2019).
Miyazawa, T. et al. Mortality and morbidity of extremely low birth weight infants in Japan, 2015. Pediatr. Int. 65, e15493 (2023).
Kono, Y. et al. Neurodevelopmental outcomes at age 3 years of preterm infants born at 22–31 weeks’ gestation. J. Perinatol. 45, 1558–1567 (2025).
Raju, T. N. K., Buist, A. S., Blaisdell, C. J., Moxey-Mims, M. & Saigal, S. Adults born preterm: a review of general health and system-specific outcomes. Acta Paediatr. 106, 1409–1437 (2017).
van der Pal, S., Steinhof, M., Grevinga, M., Wolke, D. & Verrips, G. Quality of life of adults born very preterm or very low birth weight: a systematic review. Acta Paediatr. 109, 1974–1988 (2020).
Hollanders, J. J., van der Pal, S. M., van Dommelen, P., Rotteveel, J. & Finken, M. J. J. Growth pattern and final height of very preterm vs. very low birth weight infants. Pediatr. Res. 82, 317–323 (2017).
Tuková, J., Kosek Krásničanová, H., Hladíková, M. & Marková, D. Novel growth reference Imagrow differed from existing charts for preterm children aged 0–2 years. Acta Paediatr. 113, 1818–1832 (2024).
Elmrayed, S., Dai, S., Lodha, A., Kumar, M. & Fenton, T. R. Preterm growth assessment: the latest findings on age correction. J. Perinatol. 45, 607–615 (2025).
Vinther, J. L. et al. Gestational age and trajectories of body mass index and height from birth through adolescence in the Danish National Birth Cohort. Sci. Rep. 13, 3298 (2023).
Fenton, T. R. & Kim, J. H. A systematic review and meta-analysis to revise the Fenton growth chart for preterm infants. BMC Pediatr. 13, 59 (2013).
de Onis, M. et al. The WHO multicentre growth reference study: planning, study design, and methodology. Food Nutr. Bull. 25, S15–S26 (2004).
Isojima, T., Kato, N., Ito, Y., Kanzaki, S. & Murata, M. Growth standard charts for Japanese children with mean and standard deviation (SD) values based on the year 2000 national survey. Clin. Pediatr. Endocrinol. 25, 71–76 (2016).
Itabashi, K., Miura, F., Uehara, R. & Nakamura, Y. New Japanese neonatal anthropometric charts for gestational age at birth. Pediatr. Int. 56, 702–708 (2014).
Rigby, R. A. & Stasinopoulos, D. M. Generalized additive models for location, scale and shape. J. R. Stat. Soc. C Appl. Stat. 54, 507–554 (2005).
Cole, T. J. & Green, P. J. Smoothing reference centile curves: the LMS method and penalized likelihood. Stat. Med. 11, 1305–1319 (1992).
Cole, T. J. Sample size and sample composition for constructing growth reference centiles. Stat. Methods Med. Res. 30, 488–507 (2021).
Niklasson, A., Engström, E., Hård, A. L., Wikland, K. A. & Hellström, A. Growth in very preterm children: a longitudinal study. Pediatr. Res. 54, 899–905 (2003).
Hack, M. et al. Growth of very low birth weight infants to age 20 years. Pediatrics 112, e30–e38 (2003).
Saigal, S. et al. Growth trajectories of extremely low birth weight infants from birth to young adulthood: a longitudinal, population-based study. Pediatr. Res. 60, 751–758 (2006).
Fenton, T. R., Samycia, L., Elmrayed, S., Nasser, R. & Alshaikh, B. Growth patterns by birth size of preterm children born at 24–29 gestational weeks for the first 3 years. Paediatr. Perinat. Epidemiol. 38, 560–569 (2024).
Luo, Z. C. & Karlberg, J. Critical growth phases for adult shortness. Am. J. Epidemiol. 152, 125–131 (2000).
Skogastierna, C. et al. Early life growth is related to pubertal growth and adult height—a QEPS-model analysis. Pediatr. Res. 98, 1339–1357 (2025).
Eide, M. G. et al. Size at birth and gestational age as predictors of adult height and weight. Epidemiology 16, 175–181 (2005).
Derraik, J. G. B., Lundgren, M., Cutfield, W. S. & Ahlsson, F. Association between preterm birth and lower adult height in women. Am. J. Epidemiol. 185, 48–53 (2017).
Karlberg, J. On the modelling of human growth. Stat. Med. 6, 185–192 (1987).
Karlberg, J., Engström, I., Karlberg, P. & Fryer, J. G. Analysis of linear growth using a mathematical model. I. From birth to three years. Acta Paediatr. Scand. 76, 478–488 (1987).
Karlberg, J. A biologically oriented mathematical model (ICP) for human growth. Acta Paediatr. Scand. Suppl. 78, 70–94 (1989).
Chou, F. S. & Yeh, H. W. Sex differences in postnatal weight gain trajectories of extremely preterm newborns. J. Perinatol. 41, 1835–1844 (2021).
Deprez, A. et al. Impact of preterm birth on muscle mass and function: a systematic review and meta-analysis. Eur. J. Pediatr. 183, 1989–2002 (2024).
Raghuram, K. et al. Head growth trajectory and neurodevelopmental outcomes in preterm neonates. Pediatrics 140, e20170216 (2017).
Raaijmakers, A. et al. Catch-up growth in the first two years of life in extremely low birth weight (ELBW) infants is associated with lower body fat in young adolescence. PLoS One 12, e0173349 (2017).
Ong, K. K. et al. Postnatal growth in preterm infants and later health outcomes: a systematic review. Acta Paediatr. 104, 974–986 (2015).
Selvanathan, T. Head growth trajectories as a window into neurodevelopment in preterm infants. J. Pediatr. 100, 458–459 (2024).
Sammallahti, S. et al. Infant growth after preterm birth and neurocognitive abilities in young adulthood. J. Pediatr. 165, 1109–1115.e3 (2014).
Cockerill, J., Uthaya, S., Doré, C. J. & Modi, N. Accelerated postnatal head growth follows preterm birth. Arch. Dis. Child. Fetal Neonatal Ed. 91, F184–F187 (2006).
Fenton, T. R. et al. Effect of enteral protein amount on growth and health outcomes in very-low-birth-weight preterm infants: phase II of the Pre-B Project and an Evidence Analysis Center systematic review. J. Acad. Nutr. Diet. 121, 2287–2300 (2021).
Sanchez-Holgado, M. et al. Systematic review and meta-analysis of enteral protein intake effects on growth in preterm infants. Pediatr. Res. 98, 1696–1710 (2025).
Roggero, P. et al. Is term newborn body composition being achieved postnatally in preterm infants? Early Hum. Dev. 85, 349–352 (2009).
Stein, A. D. et al. Growth patterns in early childhood and final attained stature: data from five birth cohorts from low- and middle-income countries. Am. J. Hum. Biol. 22, 353–359 (2010).
Natale, V. et al. Worldwide variation in human growth and the World Health Organization growth standards: a systematic review. BMJ Open 4, e003735 (2014).
Tinggaard, J. et al. The 2014 Danish references from birth to 20 years for height, weight and body mass index. Acta Paediatr. 103, 214–224 (2014).
Júlíusson, P. B. et al. Growth of Belgian and Norwegian children compared to the WHO growth standards: prevalence below −2 sd and above +2 SD and the effect of breastfeeding. Arch. Dis. Child. 96, 916–921 (2011).
Inokuchi, M. et al. WHO 2006 child growth standards overestimate short stature and underestimate overweight in Japanese Children. J. Pediatr. Endocrinol. Metab. 31, 33–38 (2018).
Hui, L. L. et al. Are universal standards for optimal infant growth appropriate? Evidence from a Hong Kong Chinese birth cohort. Arch. Dis. Child. 93, 561–565 (2008).
Peralta-Carcelen, M. et al. Growth of adolescents who were born at extremely low birth weight without major disability. J. Pediatr. 136, 633–640 (2000).
Hack, M. et al. Trajectory and correlates of growth of extremely-low-birth-weight adolescents. Pediatr. Res. 75, 358–366 (2014).
Acknowledgements
We thank Edanz (https://jp.edanz.com/ac) for editing a draft of this manuscript.
Funding
This work was supported by the Children and Families Sciences Research Grant (Grant No. 23DA1001) and the Health Labor Sciences Research Grant (Grant No. 25FC2001).
Author information
Authors and Affiliations
Contributions
A.M. conceptualized the study, curated the data, conducted the statistical analyses, interpreted the results, and drafted and revised the manuscript. Y.K. interpreted the results and critically reviewed and revised the manuscript. K.T. contributed to data collection, interpretation of results, and critical revision of the manuscript. Y.I. and K.C. supervised the statistical analyses, interpreted the results, and provided critical review for intellectual content. K.M., Y.K., R.K., N.I., and Y.Y. contributed to data collection and critically reviewed the manuscript for intellectual content. T.Y. and K.H. provided critical review of the manuscript for intellectual content. All authors approved the final manuscript as submitted and agreed to be accountable for all aspects of this work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study was conducted in accordance with the principles outlined in the Declaration of Helsinki. Ethical approval was obtained from the Clinical Research Ethics Review Committee of the Affiliated Hospital of Jichi Medical University (Approval No. 21-106, February 4, 2022) and the Ethics Review Committee of the National Center for Child Health and Development (Approval No. 2022-053, June 29, 2022).
Patient consent statement
This study was approved by the Clinical Research Ethics Review Committee of the Affiliated Hospital of Jichi Medical University (Approval No. 21-106, February 4, 2022) and the Ethics Review Committee of the National Center for Child Health and Development (Approval No. 2022-053, June 29, 2022). Patient consent was obtained using an opt-out process approved by the ethics committee in accordance with national guidelines.
Data deposition compliance
No datasets were deposited in a public repository.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Moriichi, A., Kono, Y., Toyoshima, K. et al. Postnatal growth trajectories of very preterm appropriate-for-gestational-age infants from term-equivalent age to 6 years. Pediatr Res (2026). https://doi.org/10.1038/s41390-026-04862-3
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41390-026-04862-3