Abstract
Background
While C-reactive protein (CRP) is a commonly used sepsis biomarker in neonates, studies in older patients propose procalcitonin (PCT) as potentially superior due to earlier rise in response to bacterial infection and shorter half-life.
Methods
We conducted a prospective cohort study of VLBW infants undergoing sepsis evaluations at a level IV neonatal intensive care unit (NICU) and combined the analysis with a retrospective data review from a different level III NICU. We adjusted all analyses for study site to account for potential heterogeneity. Two blood samples for each biomarker obtained within a 24-h period were analyzed. Diagnostic accuracy was assessed by sensitivity, specificity, predictive values, likelihood ratios, and the area under the receiver operating characteristic (ROC) curve (AUC).
Results
At literature-defined cut-offs, PCT demonstrated higher sensitivity, but lower specificity for sepsis compared to CRP. Using ROC-optimized cut-offs, CRP showed increased specificity (85%) but lower sensitivity (48%), whereas PCT maintained relatively high sensitivity (73%) but even lower specificity (60%). In multivariable modeling, only CRP was significantly associated with sepsis. Correlation between the biomarkers was weak.
Conclusions
PCT did not outperform CRP as a sepsis biomarker. CRP was more specific and remained the stronger independent predictor of sepsis.
Impact
-
Procalcitonin (PCT) shows higher sensitivity, but lower specificity compared to C-reactive protein (CRP) in very low birth weight infants with sepsis.
-
CRP emerges as the stronger independent predictor of sepsis in multivariable models.
-
This study constitutes one of the largest United States cohorts directly comparing CRP and PCT in this high-risk group.
Similar content being viewed by others
Introduction
Despite advances in care of neonates over the last decade, the burden of neonatal sepsis remains unchanged1,2 Very low birth weight (VLBW) infants are disproportionately affected with rates of 20/1000 for early-onset sepsis (EOS) and 200/1000 for late-onset sepsis (LOS).3,4 In clinical practice, blood culture-negative sepsis accounts for the majority of antimicrobial treatment courses in the neonatal intensive care unit (NICU), which is problematic because the clinical signs of sepsis are subtle and non-specific, and the definition of clinical sepsis is variable.5,6,7 A sensitive and specific biomarker of sepsis at an early stage of illness would be helpful in the diagnosis and management of neonates with suspected sepsis.
C-reactive protein (CRP) is an acute-phase reactant often used for evaluation and follow-up of neonatal infections. However, CRP can be elevated in neonates just after birth, increased relative to mode of delivery, birth weight and gestational age, and can be elevated in many non-infectious inflammatory processes such as birth injury, respiratory distress syndrome, meconium aspiration, intraventricular hemorrhage, and after surgery.8,9,10,11,12,13,14,15,16 A number of studies have suggested procalcitonin (PCT) as a potentially better biomarker for the diagnosis of neonatal sepsis.17,18,19,20,21 However, there have been very few studies of PCT in neonates in the United States and only few studies worldwide addressed VLBW infants. Given that VLBW infants comprise a sizable portion of the NICU population in the United States, and are the population of highest risk for sepsis, we sought to compare the performance of PCT with CRP in this group.
Methods
Study design and data collection
We conducted a prospective, observational study at the level IV multidisciplinary NICU at the Monroe Carell Jr. Children’s Hospital at Vanderbilt University Medical Center (VUMC) between December 1st, 2017, and February 28th, 2019. In addition, we included data from the level III Jackson-Madison County General Hospital (JMCGH) NICU, where CRP and PCT are measured concurrently as standard of care. That data was collected between April 5th, 2018, and January 21st, 2021. All neonates admitted to the NICU (2018–2021) with a birth weight of <1500 g and were evaluated for sepsis were eligible for inclusion in the study. Sepsis evaluations within 48 h of stopping antibiotics were not included as separate incidents but were considered part of the prior sepsis episode. Per local standard of care, initial PCT samples and CRP levels were drawn on admission in infants worked up for possible EOS, or at the time of clinical concern for possible LOS and followed by a repeat sample 24 h later. CRP levels were measured by the clinical laboratory by immunoturbidimetric assay (Abbott Architect c8000, Chicago, IL). PCT samples were frozen at −20 °C prior to batch analysis by enzyme-linked florescent assay (VIDAS Brahms, bioMérieux, France). Only CRP levels were available for clinical decision making to providers per standard of care. Decisions for sepsis evaluations were based on local clinical protocols and individual provider concerns.
We reviewed medical records for results of microbiological cultures (blood, urine, endotracheal, and/or cerebrospinal fluid [CSF]), complete blood cell count (CBC) and differential, CSF analysis, and radiological evaluations. In addition, basal demographic data including birth weight, age, gestational age, and weight at time of sepsis evaluation were collected. The study was approved by Vanderbilt University’s Institutional Review Board.
Classification of sepsis
Only infants with a positive culture from a normally sterile body fluid (blood, CSF) were classified as septic. All others were included in the non-infected control group. Infants with necrotizing enterocolitis (NEC) or infants within 24 h of a surgical or otherwise invasive procedure were excluded.
Statistical methods
Descriptive summaries
Descriptive statistics were summarized by absolute and relative frequencies for categorical variables and median (interquartile range [IQR]) and mean (standard deviation) for continuous variables. The sample and patient counts reported in Study Population include all collected specimens, even if some were excluded from regression analyses due to incomplete observations.
CRP and PCT diagnostic performance
Cut-off values for both biomarkers were based on literature review (CRP 10.0 mg/L, PCT 0.5 ng/ml).20 Values of CRP and PCT not reported below the limit of detection (5.0 mg/L for CRP and 0.10 ng/ml for PCT) were imputed by using Regression on Order Statistics (ROS) for left-censored data.22,23,24 Logistic regression was used to evaluate the association of CRP, PCT, and study site with culture-proven sepsis diagnosed from specimens obtained at the initial evaluation (day 0) or within 24 h (day 1). Multiple samples per infant generally occurred within single suspected sepsis episodes (e.g., baseline and follow-up at ~24 h) (Supplementary Fig. S1). For regression and ROC analyses, we used the baseline measurement (first sample at evaluation) as representative. Incomplete observations of baseline CRP, baseline PCT, and sepsis event on day 0 or day 1 were dropped from the model. Diagnostic performance of the model was assessed using area under the curve (AUC) and receiver operating characteristic (ROC) curves. ROC curves were then generated for the combined model and for each biomarker individually. Optimal cutoffs for CRP and PCT were derived from their respective ROC curves using the Youden index.25,26 Various cutoff points were used to calculate sensitivity, specificity, positive and negative predictive values, as well as positive and negative likelihood ratios. Subsequent models were performed to determine the diagnostic performance of the biomarkers individually. Statistical significance was defined at a nominal level of α = 0.05. All analyses were performed using R version 4.1.1.
Results
Study population
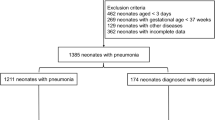
A total of 1195 samples were collected from 239 patients from both hospitals, of which 152 patients (63.6%) were VLBW infants. A total of 664 samples were analyzed among those 152 VLBW infants, of which 90 were from VUMC and 574 were from JMCGH. Of the 152 VLBW infants, 56 were from VUMC and 96 were from JMCGH. A total of 223 samples were from 35 septic infants based on the criteria defined above and 441 samples were from 117 non-septic infants (Table 1). The types of identified organisms are listed in Table 2. The correlation between CRP and PCT (all samples with complete observations of CRP and PCT aggregated across days) indicated a weak positive linear relationship (r = 0.22).
Diagnosing sepsis on day 0 and day 1
The model assessing sepsis on day 0 and day 1 included 118 complete observations (values of both PCT and CRP were available at baseline). In contrast to PCT, the adjusted odds ratio for CRP was statistically significant; for every mg/L change in CRP, the odds of sepsis increased by 1.06 (Table 3). The AUC was 0.85 (95% CI: 0.77, 0.92) indicating good discriminatory ability. The optimal cut-point resulted in a sensitivity of 96.0% and specificity of 58.1% (Fig. 1).
The area under the curve (AUC) was 0.846 (95% CI: 0.769–0.923), indicating good diagnostic performance.
Individual biomarker performance
When adjusted for site, baseline CRP demonstrated good diagnostic performance for sepsis, with an AUC of 0.83 (95% CI: 0.76–0.90), sensitivity of 71%, specificity of 80%, and an NPV of 90% at the optimal Youden’s index cut-point (Fig. 2). Baseline PCT showed comparable overall accuracy (AUC 0.83; 95% CI: 0.76–0.90), but with higher sensitivity (89%) and slightly lower specificity (66%), yielding a higher NPV of 94%. These findings suggest that while both markers perform similarly in overall discrimination, CRP may be more useful for ruling in sepsis, whereas PCT may offer greater utility in ruling it out.
The bold curve represents PCT, while the thinner curve corresponds to CRP. There was no difference in AUC between the two curves.
Diagnostic performance of biomarkers at specified cut-off points
Table 3b compares sensitivity, specificity, PPV, NPV, +LR, −LR, and AUC between PCT and CRP, only, using both literature-defined cut-off values and ROC-optimized thresholds. At the traditional literature-based cut-off of 10 mg/L for CRP, the sensitivity was 52% and specificity was 75%, with an AUC of 0.71. The cutoff for CRP at the optimized threshold, 17 mg/L, improved specificity to 85% and the positive likelihood ratio to 3.3, though sensitivity remained low at 48%. For PCT, the commonly cited cut-off of 0.5 ng/mL yielded a higher sensitivity of 86% but low specificity (37%), with an AUC of 0.66. Using a ROC-optimized threshold of 1.21 ng/mL slightly increased specificity to 60%, with lower sensitivity (73%). Notably, NPV remained high for both CRP and PCT across all cut-offs, ranging from 84 to 88%, suggesting both biomarkers are useful for ruling out sepsis.
Given the potential of contamination, we repeated the analyses after removing the 12 unique infants whose sepsis episode was attributed to Staphylococcus epidermidis. This reduced the analytic cohort to 110 infants with complete biomarker data. The updated findings are in Supplementary Table S1A. After removing these cases, the association between CRP and sepsis remained statistically significant and of similar magnitude to the original analysis. PCT continued to show no significant association with sepsis. Diagnostic performance characteristics improved slightly but remained consistent with original findings. CRP demonstrated moderate discrimination (AUC = 0.74) and PCT showed similar performance (AUC = 0.71). Sensitivity and specify estimates at relevant cutoffs also changed slightly (Supplementary Table S1B).
Discussion
Over the last two decades, several sepsis biomarkers have been evaluated as tools to improve diagnostic accuracy for neonatal sepsis.27,28,29 Among these, CRP and PCT have emerged as two of the most widely studied. There is a large array of proinflammatory cytokines upregulated in bacterial sepsis, some of which stimulate production of CRP and PCT. However, while CRP is solely stimulated by other cytokines, PCT can be directly stimulated by lipopolysaccharides in bacterial cell walls. This leads to an earlier rise in PCT concentration in the blood, detectable by 2 h after a bacterial exposure and peaking at 12–24 h, while CRP takes 4–6 h to rise in response to infection, and peaks at 36–48 h.21,29,30 Given these kinetics, PCT has been proposed as a potentially superior early marker for neonatal sepsis. Recent studies in neonates report sensitivity ranging between 52 and 100% and specificity ranging 54–89% compared to CRP with sensitivity ranging between 67 and 100% and specificity ranging between 67 and 77%.20,21,31,32,33,34 The few studies available in VLBW neonates show the range of PCT sensitivity and specificity between 88–97% and 72–84%, respectively, and CRP sensitivity and specificity between 12–88% and 70–100%, respectively.35,36,37 However, these studies were small, with 35–51 sepsis evaluations in each, with only one conducted in the United States.37 Here, we sought to compare CRP and PCT in a larger cohort of VLBW infants undergoing sepsis evaluations.
Our findings suggest that in VLBW infants, the diagnostic performance of CRP and PCT varies depending on the cutoff levels used. At literature-defined thresholds (CRP ≥ 10 mg/L, PCT ≥ 0.5 ng/mL), PCT showed higher sensitivity (86%) than CRP (52%) but markedly lower specificity (37% vs 75%). However, when using ROC-optimized cut-points derived from our dataset, CRP achieved improved specificity (85%) with a modest sensitivity (48%), while PCT maintained high sensitivity (73%) and moderate specificity (60%). Importantly, CRP contributed significantly to multivariable models predicting sepsis, whereas PCT added little predictive value. This suggests that while both markers have value for ruling out sepsis (given high NPVs), CRP may be more useful for ruling in sepsis due to its higher specificity and stronger statistical association. However, neither biomarker can be used alone for sepsis diagnosis or to rule-out sepsis. They must be seen as complementary to a careful clinical evaluation, risk factor analysis and other laboratory data.
Several factors may explain why PCT did not outperform CRP in this VLBW population. These include natural physiologic rise of PCT after delivery, subclinical infections, or other disease processes that cause inflammation, such as respiratory distress syndrome (RDS).8 A study by Lee et al. showed increased PCT levels in non-infected patients with RDS, history of surfactant administration, ventilator use, and postnatal antibiotic therapy38. In case of VLBWs, PCT remained elevated for the first 7 days of life. In our cohort, inclusion of EOS cases may have exaggerated this variability and contributed to the lower specificity of PCT. However, a study evaluating PCT levels in VLBW infants after 7 days of life also reported lower specificity of PCT compared to CRP.37 A recent study by Goyal et al. using point-of-care methodology for equal numbers of early- and late-onset sepsis cases in mostly preterm infants found that CRP had the maximum diagnostic accuracy (AUC 0.73) followed by procalcitonin (AUC 0.65) with no significant difference between early and late-onset sepsis.39
Another contributing factor is the lack of well-established, gestational age–adjusted reference ranges for PCT in VLBW neonates. Reported normal values vary significantly by population and assay, complicating the selection of an optimal diagnostic threshold.40,41,42,43,44 This may introduce bias in studies comparing fixed cut-offs across heterogeneous neonatal populations.
Although this is the largest study directly comparing PCT and CRP in VLBW infants in the United States to date, several limitations deserve consideration. Our strict definition of sepsis (requiring culture-proven infection from a sterile site) may have excluded cases with true infection at non-sterile sites (e.g., urinary tract infections), potentially misclassifying infected infants as controls. Additionally, data were collected from two distinct sites with differing clinical practices and laboratory methodologies. The frequency of sepsis cases differed between the two study sites, and we cannot determine what contributed to this difference. Most documented blood stream infections at both sites were with coagulase-negative staphylococci (CONS), which may have been contaminants. However, we re-analyzed our data excluding infants with CONS and found a slight increase in performance metrices of both CRP and PCT, demonstrating that including potential skin contaminants would lower rather than falsely improving either sepsis marker. Furthermore, the average rate of sepsis in this population was similar to what has been described in the literature.4 However, given the general rarity of blood-culture positive sepsis in neonates, meaningful data on sepsis diagnosis can typically not be generated from single-center studies.
We also did not stratify results by early- vs. late-onset sepsis, although differential cut-offs may be more appropriate for these subgroups. A large European study found that normal serial CRP and PCT measurements within 36 h after the start of empiric antibiotic therapy can exclude the presence of neonatal EOS with a high probability.44 In that study, the AUCs for CRP and PCT were 0.986 and 0.921, respectively and with cutoff values at 16 mg/L for CRP and 2.8 ng/L for PCT sensitivity for diagnosing proven sepsis was 100%. Therefore, using the same cut-off value for EOS and LOS in our study may have contributed to the lower diagnostic accuracy of either marker in our study. Another limitation is that we did not account for infant sex in our analysis. Sex-based differences in immune responses and biomarker levels have been documented in both neonatal and pediatric populations, with male infants generally exhibiting higher baseline inflammatory markers and greater susceptibility to sepsis.45,46,47 The absence of this variable may have masked subtle performance differences in CRP or PCT between male and female infants. However, there are no published clinical guidelines to use different cut-off levels for male versus female infants for any sepsis marker. Lastly, for instances in which both biomarkers were not measured on the same day, these cases were dropped from analyses, which may have biased the estimates or limited the generalizability of the findings. A multicenter validation, with more culture-confirmed non-CONS sepsis cases, may be needed to better characterize PCT as a sepsis biomarker in VLBW neonates. Given the heterogeneity of PCT values in the non-infected cohort, a large healthy control cohort of VLBW infants will be necessary to determine normal values and optimal cut-off levels for sepsis.
Conclusion
In this large, multicenter cohort of very low birth weight infants, PCT did not outperform CRP as a diagnostic biomarker for neonatal sepsis. While PCT demonstrated higher sensitivity, CRP offered greater specificity and remained the stronger independent predictor of sepsis in multivariable modeling. These findings suggest that although both biomarkers have value in ruling out infection, CRP may be more reliable for confirming sepsis in this high-risk population. The diagnostic performance of both markers varied significantly depending on cutoff levels used, highlighting the need for context-specific thresholds rather than a one-size-fits-all approach. Our results also underscore the limitations of applying fixed cutoffs across heterogeneous neonatal populations. Future research should focus on establishing gestational age-, and postnatal age-specific reference ranges for PCT, incorporate serial biomarker measurements, and account for relevant covariates such as infant sex, which may influence inflammatory marker expression. A large, prospective validation cohort - including infected and non-infected VLBW infants - will be essential to refine the clinical utility of PCT in this population.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Bauserman, M. S. et al. Group B Streptococcus and Escherichia coli infections in the intensive care nursery in the era of intrapartum antibiotic prophylaxis. Pediatr. Infect. Dis. J. 32, 208–212 (2013).
Stoll, B. J. et al. Early-onset neonatal sepsis 2015 to 2017, the rise of Escherichia coli, and the need for novel prevention strategies. JAMA Pediatr. 174, e200593 (2020).
Gerios, L. et al. Late-onset sepsis in very low birth weight premature infants: a 10-year review of a Brazilian Tertiary University Hospital-the challenge remains. Am. J. Perinatol. 41, e1725–e1731 (2024).
Stoll, B. J. et al. Very low birth weight preterm infants with early onset neonatal sepsis: the predominance of gram-negative infections continues in the National Institute of Child Health and Human Development Neonatal Research Network, 2002-2003. Pediatr. Infect. Dis. J. 24, 635–639 (2005).
Stoll, B. J. et al. Late-onset sepsis in very low birth weight neonates: the experience of the NICHD Neonatal Research Network. Pediatrics 110, 285–291 (2002).
Cantey, J. B., Wozniak, P. S., Pruszynski, J. E. & Sánchez, P. J. Reducing unnecessary antibiotic use in the neonatal intensive care unit (SCOUT): a prospective interrupted time-series study. Lancet Infect. Dis. 16, 1178–1184 (2016).
Klingenberg, C., Kornelisse, R. F., Buonocore, G., Maier, R. F. & Stocker, M. Culture-negative early-onset neonatal sepsis - at the crossroad between efficient sepsis care and antimicrobial stewardship. Front Pediatr. 6, 285 (2018).
Thaver, D. & Zaidi, A. K. Burden of neonatal infections in developing countries: a review of evidence from community-based studies. Pediatr. Infect. Dis. J. 28, S3–S9 (2009).
Chiesa, C. et al. C-reactive protein, interleukin-6, and procalcitonin in the immediate postnatal period: influence of illness severity, risk status, antenatal and perinatal complications, and infection. Clin. Chem. 49, 60–68 (2003).
Assumma, M. et al. Serum procalcitonin concentrations in term delivering mothers and their healthy offspring: a longitudinal study. Clin. Chem. 46, 1583–1587 (2000).
Mjelle, A. B., Guthe, H. J. T., Reigstad, H., Bjorke-Monsen, A. L. & Markestad, T. Serum concentrations of C-reactive protein in healthy term-born Norwegian infants 48-72 h after birth. Acta Paediatr. 108, 849–854 (2018).
Kaapa, P. & Koistinen, E. Maternal and neonatal C-reactive protein after interventions during delivery. Acta Obstet. Gynecol. Scand. 72, 543–546 (1993).
Bolke, E. et al. Different acute-phase response in newborns and infants undergoing surgery. Pediatr. Res 51, 333–338 (2002).
Arkader, R. et al. Procalcitonin and C-reactive protein kinetics in postoperative pediatric cardiac surgical patients. J. Cardiothorac. Vasc. Anesth. 18, 160–165 (2004).
Celik, I. H. et al. The acute-phase response in differentiating sepsis from inflammation in neonates who require abdominal surgery. Acta Chir. Belg. 112, 292–296 (2012).
Neunhoeffer, F. et al. Serum concentrations of interleukin-6, procalcitonin, and C-reactive protein: discrimination of septical complications and systemic inflammatory response syndrome after pediatric surgery. Eur. J. Pediatr. Surg. 26, 180–185 (2016).
Pavcnik-Arnol, M., Bonac, B., Groselj-Grenc, M. & Derganc, M. Changes in serum procalcitonin, interleukin 6, interleukin 8 and C-reactive protein in neonates after surgery. Eur. J. Pediatr. Surg. 20, 262–266 (2010).
Simon, L., Gauvin, F., Amre, D. K., Saint-Louis, P. & Lacroix, J. Serum procalcitonin and C-reactive protein levels as markers of bacterial infection: a systematic review and meta-analysis. Clin. Infect. Dis. 39, 206–217 (2004).
Xu, L., Li, Q., Mo, Z. & You, P. Diagnostic value of C-reactive protein in neonatal sepsis: a meta-analysis. Eur. J. Inflamm. 14, 100–108 (2016).
Yu, Z. et al. The accuracy of the procalcitonin test for the diagnosis of neonatal sepsis: a meta-analysis. Scand. J. Infect. Dis. 42, 723–733 (2010).
Eschborn, S. & Weitkamp, J. H. Procalcitonin versus C-reactive protein: review of kinetics and performance for diagnosis of neonatal sepsis. J. Perinatol. 39, 893–903 (2019).
Kordek, A., Łoniewska, B., Podraza, W., Nikodemski, T. & Rudnicki, J. Usefulness of estimation of blood procalcitonin concentration versus C-reactive protein concentration and white blood cell count for therapeutic monitoring of sepsis in neonates. Postepy Hig. Med. Dosw 68, 1516–1523 (2014).
Canales, R. A., Wilson, A. M., Pearce-Walker, J. I., Verhougstraete, M. P. & Reynolds, K. A. Methods for handling left-censored data in quantitative microbial risk assessment. Appl. Environ. Microbiol. 84, e01203-18 (2018).
Hewett, P. & Ganser, G. H. A comparison of several methods for analyzing censored data. Ann. Occup. Hyg. 51, 611–632 (2007).
Helsel, D. R. Nondetects and Data Analysis: Statistics for Censored Environmental Data (John Wiley & Sons, 2005). https://pubs.usgs.gov/publication/70180734.
Youden, W. J. Index for rating diagnostic tests. Cancer 3, 32–35 (1950).
Hassanzad, M. & Hajian-Tilaki, K. Methods of determining optimal cut-point of diagnostic biomarkers with application of clinical data in ROC analysis: an update review. BMC Med. Res. Methodol. 24, 84 (2024).
Chauhan, N., Tiwari, S. & Jain, U. Potential biomarkers for effective screening of neonatal sepsis infections: an overview. Micro. Pathog. 07, 234–242 (2017).
Reinhart, K., Meisner, M. & Brunkhorst, F. M. Markers for sepsis diagnosis: what is useful?. Crit. Care Clin. 22, 503–519 (2006).
Monneret, G. et al. Procalcitonin and C-reactive protein levels in neonatal infections. Acta Paediatr. 86, 209–212 (1997).
Vouloumanou, E. K., Plessa, E., Karageorgopoulos, D. E., Mantadakis, E. & Falagas, M. E. Serum procalcitonin as a diagnostic marker for neonatal sepsis: a systematic review and meta-analysis. Intensive Care Med. 37, 747–762 (2011).
Pontrelli, G. et al. Accuracy of serum procalcitonin for the diagnosis of sepsis in neonates and children with systemic inflammatory syndrome: a meta-analysis. BMC Infect. Dis. 17, 302 (2017).
Chaurasia, S. et al. Procalcitonin for detecting culture-positive sepsis in neonates: a prospective, multicenter study. Neonatology 120, 642–651 (2023).
Morad, E. A., Rabie, R. A., Almalky, M. A. & Gebriel, M. G. Evaluation of procalcitonin, C-reactive protein, and interleukin-6 as early markers for diagnosis of neonatal sepsis. Int. J. Microbiol. 2020, 8889086 (2020).
Bustos, B. R. & Araneda, C. H. Procalcitonin for the diagnosis of late onset sepsis in newborns of very low birth weight]. Rev. Chil. Infectol. 29, 511–516 (2012).
Distefano, G., Curreri, R., Betta, P., Romeo, M. G. & Amato, M. Procalcitonin serum levels in perinatal bacterial and fungal infection of preterm infants. Acta Paediatr. 93, 216–219 (2004).
Vazzalwar, R., Pina-Rodrigues, E., Puppala, B. L., Angst, D. B. & Schweig, L. Procalcitonin as a screening test for late-onset sepsis in preterm very low birth weight infants. J. Perinatol. 25, 397–402 (2005).
Lee, J., Bang, Y. H., Lee, E. H., Choi, B. M. & Hong, Y. S. The influencing factors on procalcitonin values in newborns with noninfectious conditions during the first week of life. Korean J. Pediatr. 60, 10–16 (2017).
Goyal, M., Mascarenhas, D., Rr, P. & Haribalakrishna, A. Diagnostic accuracy of point-of-care testing of C-reactive protein, interleukin-6, and procalcitonin in neonates with clinically suspected sepsis: a prospective observational study. Med. Princ. Pract. https://doi.org/10.1159/000536678 (2024).
Chiesa, C. et al. C reactive protein and procalcitonin: reference intervals for preterm and term newborns during the early neonatal period. Clin. Chim. Acta 412, 1053–1059 (2011).
Fukuzumi, N. et al. Age-specific percentile-based reference curve of serum procalcitonin concentrations in Japanese preterm infants. Sci. Rep. 6, 23871 (2016).
Hahn, W. H. et al. Reference intervals of serum procalcitonin are affected by postnatal age in very low birth weight infants during the first 60 days after birth. Neonatology 108, 60–64 (2015).
Turner, D. et al. Procalcitonin in preterm infants during the first few days of life: introducing an age related nomogram. Arch. Dis. Child Fetal Neonatal Ed. 91, F283–F286 (2006).
Stocker, M. et al. C-reactive protein, procalcitonin, and white blood count to rule out neonatal early-onset sepsis within 36 h: a secondary analysis of the neonatal procalcitonin intervention study. Clin. Infect. Dis. 73, e383–e390 (2021).
Biron-Shental, T. et al. Procalcitonin and C-reactive protein as early markers of neonatal sepsis in preterm neonates. J. Matern Fetal Neonatal Med. 33, 1673–1678 (2020).
Fischer, J., Jung, N., Robinson, N. & Lehmann, C. Sex differences in immune responses to infectious diseases. Infection 43, 399–403 (2015).
Klein, S. L. & Flanagan, K. L. Sex differences in immune responses. Nat. Rev. Immunol. 16, 626–638 (2016).
Acknowledgements
The project was supported by CTSA award No. UL1 TR002243 from the National Center for Advancing Translational Sciences. Its contents are solely the responsibility of the authors and do not necessarily represent official views of the National Center for Advancing Translational Sciences or the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
J.H.W. conceptualized the study. S.E., S.O.G., and J.H.W. acquired the data. S.E., T.S., G.D.A., and J.H.W. analyzed the data. E.A. T.S. G.D.A. D.C.D. and J.H.W. drafted the article. T.S., D.C.D., G.D.A. and J.H.W. revised it critically for important intellectual content. T.S., S.E., D.C.D., A.G.A., R.B., N.H., S.O.G., G.D.A., and J.H.W. approved the final version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Stopczynski, T., Eschborn, S., Dobson, D.C. et al. Comparing procalcitonin and C-reactive protein levels in very low birth weight infants with sepsis. Pediatr Res (2026). https://doi.org/10.1038/s41390-026-04867-y
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41390-026-04867-y