Abstract
Background
Data on advanced prostate cancer (PCa) suggest more prior systemic therapies might reduce tumor immune responsiveness. In treatment-naïve primary PCa, recent work correlated intratumoral plasma cell content with enhanced tumor immune-responsiveness. We sought to identify features of localized PCa at a high risk of recurrence following local treatment with high plasma cell content to help focus future immune-based neoadjuvant trials.
Methods
We performed retrospective analyses of molecular profiles from three independent cohorts of over 1300 prostate tumors. We used Wilcoxon Rank Sum to compare molecular pathways between tumors with high and low intratumoral plasma cell content and multivariable Cox proportional hazards regression analyses to assess metastasis-free survival.
Results
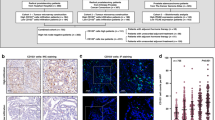
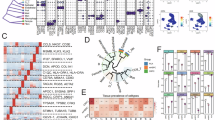
We validated an expression-based signature for intratumoral plasma cell content in 113 primary prostate tumors with both RNA-expression data and digital image quantification of CD138+ cells (plasma cell marker) based on immunohistochemisty. The signature showed castration-resistant tumors (n = 101) with more prior systemic therapies contained lower plasma cell content. In high-grade primary PCa, tumors with high plasma cell content were associated with increased predicted response to immunotherapy and decreased response to androgen-deprivation therapy. Master regulator analyses identified upregulated transcription factors implicated in immune (e.g. SKAP1, IL-16, and HCLS1), and B-cell activity (e.g. VAV1, SP140, and FLI-1) in plasma cell-high tumors. Master regulators overactivated in tumors with low plasma cell content were associated with shorter metastasis-free survival following radical prostatectomy.
Conclusions
Markers of plasma cell activity might be leveraged to augment clinical trial targeting and selection and better understand the potential for immune-based treatments in patients with PCa at a high risk of recurrence following local treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Kantoff PW, Higano CS, Shore ND, Berger ER, Small EJ, Penson DF, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363:411–22.
Antonarakis ES, Piulats JM, Gross-Goupil M, Goh J, Ojamaa K, Hoimes CJ, et al. Pembrolizumab for treatment-refractory metastatic castration-resistant prostate cancer: multicohort, open-label phase II KEYNOTE-199 study. J Clin Oncol. 2020;38:395–405.
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer J Clinicians. 2021;71:209–49.
Boettcher AN, Usman A, Morgans A, VanderWeele DJ, Sosman J, Wu JD. Past, current, and future of immunotherapies for prostate cancer. Front Oncol. 2019;9:884.
Subudhi SK, Vence L, Zhao H, Blando J, Yadav SS, Xiong Q, et al. Neoantigen responses, immune correlates, and favorable outcomes after ipilimumab treatment of patients with prostate cancer. Sci Transl Med. 2020;12:eaaz3577.
Gao J, Ward JF, Pettaway CA, Shi LZ, Subudhi SK, Vence LM, et al. VISTA is an inhibitory immune checkpoint that is increased after ipilimumab therapy in patients with prostate cancer. Nat Med. 2017;23:551–5.
Tewari A, Divine G, Chang P, Shemtov MM, Milowsky M, Nanus D, et al. Long-term survival in men with high grade prostate cancer: a comparison between conservative treatment, radiation therapy and radical prostatectomy—a propensity scoring approach. J Urol. 2007;177:911–5.
Sharonov GV, Serebrovskaya EO, Yuzhakova DV, Britanova OV, Chudakov DM. B cells, plasma cells and antibody repertoires in the tumour microenvironment. Nat Rev Immunol. 2020;20:294–307.
Helmink BA, Reddy SM, Gao J, Zhang S, Basar R, Thakur R, et al. B cells and tertiary lymphoid structures promote immunotherapy response. Nature. 2020;577:549–55.
Cabrita R, Lauss M, Sanna A, Donia M, Skaarup Larsen M, Mitra S, et al. Tertiary lymphoid structures improve immunotherapy and survival in melanoma. Nature. 2020;577:561–5.
Petitprez F, de Reyniès A, Keung EZ, Chen TW-W, Sun C-M, Calderaro J, et al. B cells are associated with survival and immunotherapy response in sarcoma. Nature. 2020;577:556–60.
Weiner AB, Vidotto T, Liu Y, Mendes AA, Salles DC, Faisal FA, et al. Plasma cells are enriched in localized prostate cancer in Black men and are associated with improved outcomes. Nat Commun. 2021;12:935.
Ross AE, Johnson MH, Yousefi K, Davicioni E, Netto GJ, Marchionni L, et al. Tissue-based genomics augments post-prostatectomy risk stratification in a natural history cohort of intermediate- and high-risk men. Eur Urol. 2016;69:157–65.
Kaur HB, Guedes LB, Lu J, Maldonado L, Reitz L, Barber JR, et al. Association of tumor-infiltrating T-cell density with molecular subtype, racial ancestry and clinical outcomes in prostate cancer. Mod Pathol. 2018;31:1539–52.
Karnes RJ, Bergstralh EJ, Davicioni E, Ghadessi M, Buerki C, Mitra AP, et al. Validation of a genomic classifier that predicts metastasis following radical prostatectomy in an at risk patient population. J Urol. 2013;190:2047–53.
Weiner AB, Liu Y, McFarlane M, Bawa PS, Li EV, Zhao X et al. A transcriptomic model for homologous recombination deficiency in prostate cancer. Prostate Cancer Prostatic Dis 2021. https://doi.org/10.1038/s41391-021-00416-2.
Abida W, Cyrta J, Heller G, Prandi D, Armenia J, Coleman I, et al. Genomic correlates of clinical outcome in advanced prostate cancer. PNAS. 2019;116:11428–36.
Yoshihara K, Shahmoradgoli M, Martínez E, Vegesna R, Kim H, Torres-Garcia W, et al. Inferring tumour purity and stromal and immune cell admixture from expression data. Nat Commun. 2013;4:1–11.
Chen S-H, Kuo W-Y, Su S-Y, Chung W-C, Ho J-M, Lu HH-S, et al. A gene profiling deconvolution approach to estimating immune cell composition from complex tissues. BMC Bioinforma. 2018;19:154.
Galaxy Tool Shed. https://testtoolshed.g2.bx.psu.edu/repository?repository_id=6e9a9ab163e578e0&changeset_revision=e3afe097e80a (2018) (accessed 15 Aug2020).
Bankhead P, Loughrey MB, Fernández JA, Dombrowski Y, McArt DG, Dunne PD, et al. QuPath: open source software for digital pathology image analysis. Sci Rep. 2017;7:16878.
Ayers M, Lunceford J, Nebozhyn M, Murphy E, Loboda A, Kaufman DR, et al. IFN-γ–related mRNA profile predicts clinical response to PD-1 blockade. J Clin Invest. 2017;127:2930–40.
Cristescu R, Mogg R, Ayers M, Albright A, Murphy E, Yearley J, et al. Pan-tumor genomic biomarkers for PD-1 checkpoint blockade-based immunotherapy. Science. 2018;362:eaar3593.
Spratt DE, Alshalalfa M, Fishbane N, Weiner AB, Mehra R, Mahal BA, et al. Transcriptomic heterogeneity of androgen receptor activity defines a de novo low ar-active subclass in treatment naïve primary prostate cancer. Clin Cancer Res. 2019;25:6721–30.
Zhao SG, Chang SL, Spratt DE, Erho N, Yu M, Ashab HA-D, et al. Development and validation of a 24-gene predictor of response to postoperative radiotherapy in prostate cancer: a matched, retrospective analysis. Lancet Oncol. 2016;17:1612–20.
Spratt DE, Yousefi K, Deheshi S, Ross AE, Den RB, Schaeffer EM, et al. Individual patient-level meta-analysis of the performance of the decipher genomic classifier in high-risk men after prostatectomy to predict development of metastatic disease. JCO. 2017;35:1991–8.
Charoentong P, Finotello F, Angelova M, Mayer C, Efremova M, Rieder D, et al. Pan-cancer immunogenomic analyses reveal genotype-immunophenotype relationships and predictors of response to checkpoint blockade. Cell Rep. 2017;18:248–62.
Carro MS, Lim WK, Alvarez MJ, Bollo RJ, Zhao X, Snyder EY, et al. The transcriptional network for mesenchymal transformation of brain tumours. Nature. 2010;463:318–25.
Lefebvre C, Rajbhandari P, Alvarez MJ, Bandaru P, Lim WK, Sato M, et al. A human B-cell interactome identifies MYB and FOXM1 as master regulators of proliferation in germinal centers. Mol Syst Biol. 2010;6:377.
Alvarez MJ, Shen Y, Giorgi FM, Lachmann A, Ding BB, Ye BH, et al. Functional characterization of somatic mutations in cancer using network-based inference of protein activity. Nat Genet. 2016;48:838–47.
Aytes A, Mitrofanova A, Lefebvre C, Alvarez MJ, Castillo-Martin M, Zheng T, et al. Cross-species regulatory network analysis identifies a synergistic interaction between FOXM1 and CENPF that drives prostate cancer malignancy. Cancer Cell. 2014;25:638–51.
McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM, et al. REporting recommendations for tumor MARKer prognostic studies (REMARK). Nat Clin Pract Oncol. 2005;2:416–22.
Shalapour S, Font-Burgada J, Di Caro G, Zhong Z, Sanchez-Lopez E, Dhar D, et al. Immunosuppressive plasma cells impede T-cell-dependent immunogenic chemotherapy. Nature. 2015;521:94–98.
Guo C, Crespo M, Gurel B, Dolling D, Rekowski J, Sharp A, et al. CD38 in advanced prostate cancers. Eur Urol. 2021;79:736–46.
Tedford K, Nitschke L, Girkontaite I, Charlesworth A, Chan G, Sakk V, et al. Compensation between Vav-1 and Vav-2 in B cell development and antigen receptor signaling. Nat Immunol. 2001;2:548–55.
Groner AC, Cato L, de Tribolet-Hardy J, Bernasocchi T, Janouskova H, Melchers D, et al. TRIM24 is an oncogenic transcriptional activator in prostate cancer. Cancer Cell. 2016;29:846–58.
Feng FY, Thomas S, Saad F, Gormley M, Yu MK, Ricci DS, et al. Association of molecular subtypes with differential outcome to apalutamide treatment in nonmetastatic castration-resistant prostate cancer. JAMA Oncol. 2021;7:1005–14.
Hamid AA, Huang H-C, Wang V, Chen Y-H, Feng F, Den R, et al. Transcriptional profiling of primary prostate tumor in metastatic hormone-sensitive prostate cancer and association with clinical outcomes: correlative analysis of the E3805 CHAARTED trial. Ann Oncol. 2021;32:1157–66.
Hedegaard J, Thorsen K, Lund MK, Hein AM, Hamilton-Dutoit SJ, Vang S, et al. Next-generation sequencing of RNA and DNA isolated from paired fresh-frozen and formalin-fixed paraffin-embedded samples of human cancer and normal tissue. PLoS One. 2014;9:e98187.
Smith X, Taylor A, Rudd CE. T-cell immune adaptor SKAP1 regulates the induction of collagen-induced arthritis in mice. Immunol Lett. 2016;176:122–7.
Kaser A, Dunzendorfer S, Offner FA, Ludwiczek O, Enrich B, Koch RO, et al. B lymphocyte-derived IL-16 attracts dendritic cells and Th cells. J Immunol. 2000;165:2474–80.
Skokowa J, Klimiankou M, Klimenkova O, Lan D, Gupta K, Hussein K, et al. Interactions among HCLS1, HAX1 and LEF-1 proteins are essential for G-CSF–triggered granulopoiesis. Nat Med. 2012;18:1550–9.
Fraschilla I, Jeffrey KL. The speckled protein (SP) family: immunity’s chromatin readers. Trends Immunol. 2020;41:572–85.
Zhang XK, Moussa O, LaRue A, Bradshaw S, Molano I, Spyropoulos DD, et al. The transcription factor Fli-1 modulates marginal zone and follicular B cell development in mice. J Immunol. 2008;181:1644–54.
Schaeffer E, Srinivas S, Antonarakis ES, Armstrong AJ, Bekelman JE, Cheng H, et al. NCCN guidelines insights: prostate cancer, version 1.2021. J Natl Compr Canc Netw. 2021;19:134–43.
Singh CK, Malas KM, Tydrick C, Siddiqui IA, Iczkowski KA, Ahmad N. Analysis of zinc-exporters expression in prostate cancer. Sci Rep. 2016;6:36772.
Kuruma H, Kamata Y, Takahashi H, Igarashi K, Kimura T, Miki K, et al. Staphylococcal nuclease domain-containing protein 1 as a potential tissue marker for prostate cancer. Am J Pathol. 2009;174:2044–50.
Acknowledgements
This work was supported in part by the National Institutes of Health grants 5U01CA196390 (EMS) and R01LM013236 (AM), the Prostate Cancer Foundation (EMS), Department of Defense grant W81XWH-15-1-0661 (EMS and TLL), American Cancer Society grant RSG-21-023-01-TBG (AM, EMS), and New Jersey Commission on Cancer Research grant COCR21RBG00 (AM).
Author information
Authors and Affiliations
Contributions
Conception and design: ABW, MD; ED, PhD; AM, PhD; TLL, MD; EMS, MD, PhD. Acquisition of data: ABW, MD; YL, PhD; ED, PhD; TLL, MD; EMS, MD, PhD. Analysis and interpretation of data: ABW, MD; CYY, PhD; YL, PhD. Drafting of Manuscript: ABW, MD; CYY, PhD; MK, MD; YL, PhD; ED, PhD; AM, PhD; TLL, MD; EMS, MD, PhD. Critical Revision of the manuscript for important intellectual content: ABW, MD; ED, PhD; AM, PhD; TLL, MD; EMS, MD, PhD. Statistical analysis: ABW, MD; CYY, PhD. Obtaining funding: AM, PhD; TLL, MD; EMS, MD, PhD. Administrative, technical, or material support: AM, PhD; TLL, MD; EMS, MD, PhD. Supervision: ED, PhD; AM, PhD; TLL, MD; EMS, MD, PhD. Other: none
Corresponding author
Ethics declarations
Competing interests
YL and ED are employees of Veracyte, Inc. The remaining authors declare no potential conflicts of interest.
Ethics approval and consent to participate
The study was performed in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Weiner, A.B., Yu, C.Y., Kini, M. et al. High intratumoral plasma cells content in primary prostate cancer defines a subset of tumors with potential susceptibility to immune-based treatments. Prostate Cancer Prostatic Dis 26, 105–112 (2023). https://doi.org/10.1038/s41391-022-00547-0
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41391-022-00547-0