Abstract
Background
Incidental prostate cancer (IPC) is not uncommon in patients undergoing surgery for benign prostate enlargement (BPE). However, the associated risk factors remain incompletely understood. This study aimed to evaluate potential clinical predictors of IPC.
Methods
A systematic search of MEDLINE, Embase, and Web of Science was conducted in January 2025 to identify studies assessing risk factors for IPC. Pairwise meta-analyses were performed using a random-effects model, pooling adjusted odds ratios (ORs) and 95% confidence intervals (CIs) from multivariable analyses. Risk of bias was evaluated using the ROBINS-I tool. (PROSPERO: CRD42024588776).
Results
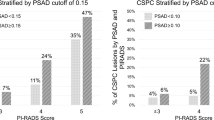
Twenty-five studies comprising 247,966 patients were included. IPC was identified in 6,748 patients, with reported rates ranging from 2.2% to 49.6%. Significant risk factors for IPC included: Elevated PSA level (OR: 1.03; 95% CI: 1.01–1.05; p = 0.01), PSA density >0.15 ng/mL/cc (OR: 3.02; 95% CI: 1.50–6.07; p = 0.01), Older age (OR: 1.05; 95% CI: 1.01–1.08; p = 0.01), and Higher body mass index (BMI) (OR: 1.10; 95% CI: 1.02–1.18; p = 0.01). Prostate volume, diabetes, and dyslipidemia were not associated with IPC risk. Notably, use of 5-alpha reductase inhibitors (5-ARIs) was linked to a significantly reduced risk (OR: 0.53; 95% CI: 0.33–0.85; p = 0.01). Study limitations included heterogeneity across studies, retrospective designs, and incomplete reporting of key variables.
Conclusions
Elevated PSA, high PSA density, older age, and increased BMI were associated with a higher risk of IPC in patients undergoing surgery for BPE. Conversely, preoperative use of 5-ARIs was associated with a reduced risk. These findings may support improved preoperative risk stratification and help identify patients in whom additional diagnostic evaluations could be considered or avoided. Further research is needed to better define predictors of clinically significant IPC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Jones JS, Follis HW, Johnson JR. Probability of finding T1a and T1b (incidental) prostate cancer during TURP has decreased in the PSA era. Prostate Cancer Prostatic Dis. 2009;12:57–60.
Zigeuner RE, Lipsky K, Riedler I, Auprich M, Schips L, Salfellner M, et al. Did the rate of incidental prostate cancer change in the era of PSA testing? A retrospective study of 1127 patients. Urology. 2003;62:451–5.
Abedi AR, Ghiasy S, Fallah-Karkan M, Rahavian A, Allameh F. The Management of Patients Diagnosed with Incidental Prostate Cancer: Narrative Review. Res Rep Urol. 2020;12:105–9.
Castellani D, Tramanzoli P, Chiacchio G, Cormio A, Rubino A, Nedbal C, et al. Reoperation Rate for Residual/Regrowth Adenoma Following Transurethral Interventions for Benign Prostatic Enlargement: Results from a Systematic Review and Meta-Analysis of Comparative Randomized Studies. J Endourol. 2024;38:605–28.
Pirola GM, Castellani D, Naselli A, Fong KY, Biligere S, Dellabella M, et al. Endoscopic enucleation of the prostate in men aged 80 years and older. Outcomes from a global, large, and multicenter series using different energy sources and techniques. World J Urol. 2025;43:344.
Leni R, Vertosick EA, van den Bergh RCN, Soeterik TFW, Heetman JG, van Melick HHE, et al. Oncologic Outcomes of Incidental Versus Biopsy-diagnosed Grade Group 1 Prostate Cancer: A Multi-institutional Study. Eur Urol Open Sci. 2024;68:10–7.
Markiewicz D, Hanks GE. Therapeutic options in the management of incidental carcinoma of the prostate. Int J Rad Oncol Biol Phys. 1991;20:153–67.
Perdana NR, Mochtar CA, Umbas R, Hamid AR. The Risk Factors of Prostate Cancer and Its Prevention: A Literature Review. Acta Med Indones. 2016;48:228–38.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
Shea BJ, Reeves BC, Wells G, Thuku M, Hamel C, Moran J, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008.
Morgan RL, Whaley P, Thayer KA, Schünemann HJ. Identifying the PECO: A framework for formulating good questions to explore the association of environmental and other exposures with health outcomes. Environ Int. 2018;121:1027–31.
Munn Z, Moola S, Lisy K, Riitano D, Tufanaru C. Methodological guidance for systematic reviews of observational epidemiological studies reporting prevalence and cumulative incidence data. JBI Evidence Implementat. 2015;13:147–53.
Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919.
Cumpston M, Li T, Page MJ, Chandler J, Welch VA, Higgins JP, et al. Updated guidance for trusted systematic reviews: a new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst Rev. 2019. 10. Ed000142.
Anract J, Klein C, Pinar U, Rouprêt M, Barry Delongchamps N, Robert G. Incidental Prostate Cancer in Patients Undergoing Surgery for Benign Prostatic Hyperplasia: A Predictive Model. Eur Urol Oncol. 2024;8:145–51.
Banno T, Nakamura K, Kaneda Y, Ozaki A, Kouchi Y, Ohira T, et al. Detection rate and variables associated with incidental prostate cancer by holmium laser enucleation of the prostate. Int J Urol. 2022;29:860–5.
Bendari A, Bendari A, Zhong X, Geetha SD, Al-Refai R, Sham S, et al. Hemoglobin-Albumin-Lymphocyte-Platelet (HALP) Score as a Predictive Value of Incidental Prostate Cancer for Patients Going for Transurethral Resection of the Prostate (TURP): A Single-Center Study. Cureus. 2024;16:e57736.
Bhojani N, Boris RS, Monn MF, Mandeville JA, Lingeman JE. Coexisting prostate cancer found at the time of holmium laser enucleation of the prostate for benign prostatic hyperplasia: predicting its presence and grade in analyzed tissue. J Endourol. 2015;29:41–6.
Bologna E, Licari LC, Franco A, Ditonno F, Manfredi C, De Nunzio C, et al. Incidental Prostate Cancer in Patients Treated for Benign Prostatic Hyperplasia: Analysis from a Contemporary National Dataset. Diagnostics. 2024;14:677.
Capogrosso P, Capitanio U, Vertosick EA, Ventimiglia E, Chierigo F, Oreggia D, et al. Temporal Trend in Incidental Prostate Cancer Detection at Surgery for Benign Prostatic Hyperplasia. Urology. 2018;122:152–7.
Elkoushy MA, Elshal AM, Elhilali MM. Incidental Prostate Cancer Diagnosis During Holmium Laser Enucleation: Assessment of Predictors, Survival, and Disease Progression. Urology. 2015;86:552–7.
Giampaoli M, Bianchi L, D’Agostino D, Corsi P, Romagnoli D, Mineo Bianchi F, et al. Can preoperative multiparametric MRI avoid unnecessary prostate biopsies before holmium laser enucleation of the prostate? Preliminary results of a multicentric cohort of patients. Minerva Urol Nefrol. 2019;71:524–30.
Gunda D, Kido I, Kilonzo S, Nkandala I, Igenge J, Mpondo B. Prevalence and Associated Factors of Incidentally Diagnosed Prostatic Carcinoma among Patients Who Had Transurethral Prostatectomy in Tanzania: A Retrospective Study. Ethiop J Health Sci. 2018;28:11–8.
Guo ZL, He JW, Pan J, Huang LJ, Cao JD, Bai ZG, et al. Prevalence and risk factors for incidental prostate cancer in patients after transurethral resection of the prostate with negative results on prostate biopsy: A retrospective study. Investig Clin Urol. 2022;63:201–6.
Herlemann A, Wegner K, Roosen A, Buchner A, Weinhold P, Bachmann A, et al. Finding the needle in a haystack”: oncologic evaluation of patients treated for LUTS with holmium laser enucleation of the prostate (HoLEP) versus transurethral resection of the prostate (TURP). World J Urol. 2017;35:1777–82.
Kim KH, Kim SW, Son HS, Kim DK, Jung DC, Kim HW, et al. Role of Holmium laser enucleation of the prostate to increase cancer detection rate in patients with gray-zone PSA level. Minerva Urol Nefrol. 2019;71:72–8.
Kim M, Song SH, Ku JH, Oh SJ, Paick JS. Prostate cancer detected after Holmium laser enucleation of prostate (HoLEP): significance of transrectal ultrasonography. Int Urol Nephrol. 2014;46:2079–85.
Kizilkan Y, Eroglu U, Senel S, Yildizhan M, Balci M, Aykanat IC, et al. Evaluation of factors associated with the detection of incidental prostate cancer after open prostatectomy for benign prostatic hyperplasia. Andrologia. 2022;54:e14261.
Klein C, Marquette T, Capon G, Yacoub M, Alezra E, Bernhard JC, et al. Incidental prostate cancer after holmium laser enucleation of the prostate: incidence and predictive factors for clinical progression. Int J Clin Oncol. 2022;27:1077–83.
Li EV, Lee MS, Guo J, Dean N, Kumar S, Mi XL, et al. Modern predictors and management of incidental prostate cancer at holmium enucleation of prostate. Prostate. 2024;84:1427–33.
Mohamed AH, Abdullahi IM, Warsame FF, Mohamud HA. Incidence and associated factors for incidental prostate cancer among patients who underwent surgery for benign prostatic hyperplasia: first report from Somalia. J Cancer Res Clin Oncol. 2023;149:4041–6.
Ohwaki K, Endo F, Shimbo M, Fujisaki A, Hattori K. Comorbidities as predictors of incidental prostate cancer after Holmium laser enucleation of the prostate: diabetes and high-risk cancer. Aging Male. 2017;20:257–60.
Porcaro AB, Tafuri A, Inverardi D, Amigoni N, Sebben M, Pirozzi M, et al. Incidental prostate cancer after transurethral resection of the prostate: Analysis of incidence and risk factors in 458 patients. Minerva Urol Nephrol. 2021;73:471–80.
Porto JG, Blachman-Braun R, Ajami T, Zarli M, Chen R, Furtado T, et al. Incidental prostate cancer after holmium laser enucleation of the prostate: Critical analysis of independent risk factors and impact on surgical outcomes. BJUI Compass. 2024;5:374–81.
Sakai A, Borza T, Antar A, Richmond E, Allen GO, Knoedler M, et al. Incidental Prostate Cancer Diagnosis Is Common After Holmium Laser Enucleation of the Prostate. Urology. 2024;183:170–5.
Sakamoto H, Matsumoto K, Hayakawa N, Maeda T, Sato A, Ninomiya A, et al. Preoperative parameters to predict incidental (T1a and T1b) prostate cancer. Can Urol Assoc J. 2014;8:E815–20.
Skrzypczyk MA, Dobruch J, Nyk L, Szostek P, Szempliński S, Borówka A. Should all specimens taken during surgical treatment of patients with benign prostatic hyperplasia be assessed by a pathologist?. Cent Eur J Urol. 2014;67:227–32.
Yang CH, Lin YS, Weng WC, Hsu CY, Tung MC, Ou YC. Incidental Prostate Cancer from Prostate with Benign Biopsies: A Predictive and Survival Analysis from Cohort Study. Int J Gen Med. 2022;15:2807–16.
Zhao Z, Ma W, Zeng G, Qi D. PSCA mRNA expression in preoperatively negative prostate biopsies predicts incidental prostate cancer in patients undergoing transurethral resection of the prostate for benign prostatic hyperplasia. J Surgical Oncol. 2011;104:672–8.
Guo Z, He J, Huang L, Wang Z, Hu P, Wang S, et al. Prevalence and risk factors of incidental prostate cancer in certain surgeries for benign prostatic hyperplasia: A systematic review and meta-analysis. Int Braz J Urol. 2022;48:915–29.
Wang Y, Li X, Yang H, Yin C, Wu Y, Chen X. Predictive factors of incidental prostate cancer in patients undergoing surgery for presumed benign prostatic hyperplasia: an updated systematic review and meta-analysis. Front Oncol. 2025. 15:1561675.
Cheng BK-C, Castellani D, Chan IS-H, Baker A, Gauhar V, Wroclawski ML, et al. Incidence, predictive factors and oncological outcomes of incidental prostate cancer after endoscopic enucleation of the prostate: a systematic review and meta-analysis. World J Urol. 2022;40:87–101.
Omri N, Kamil M, Alexander K, Alexander K, Edmond S, Ariel Z, et al. Association between PSA density and pathologically significant prostate cancer: The impact of prostate volume. Prostate. 2020;80:1444–9.
Daskivich TJ, Fan KH, Koyama T, Albertsen PC, Goodman M, Hamilton AS, et al. Effect of age, tumor risk, and comorbidity on competing risks for survival in a U.S. population-based cohort of men with prostate cancer. Ann Intern Med. 2013;158:709–17.
Larsson SC, Spyrou N, Mantzoros CS. Body fatness associations with cancer: evidence from recent epidemiological studies and future directions. Metabolism. 2022;137:155326.
Muhammed AA, Eid RMHM, Mohammed WS, Abdel-Fadeil MR. An association between adropin hormone and total testosterone in obese men: a case-control study. BMC Endocrine Disorders. 2022;22:192.
Ahmed F, Hetty S, Laterveer R, Surucu EB, Mathioudaki A, Hornbrinck E, et al. Altered Expression of Aromatase and Estrogen Receptors in Adipose Tissue From Men With Obesity or Type 2 Diabetes. J Clin Endocrinol Metabol. 2025;110:e3410-e3424.
Giovannucci E, Michaud D. The role of obesity and related metabolic disturbances in cancers of the colon, prostate, and pancreas. Gastroenterology. 2007;132:2208–25.
Thompson IM Jr, Goodman PJ, Tangen CM, Parnes HL, Minasian LM, Godley PA, et al. Long-term survival of participants in the prostate cancer prevention trial. N Engl J Med. 2013;369:603–10.
Andriole GL, Bostwick DG, Brawley OW, Gomella LG, Marberger M, Montorsi F, et al. Effect of dutasteride on the risk of prostate cancer. N Engl J Med. 2010;362:1192–202.
Morote J, Picola N, Muñoz-Rodriguez J, Paesano N, Ruiz-Plazas X, Muñoz-Rivero MV, et al. Effect of 5-Alpha Reductase Inhibitors on Magnetic Resonance Imaging and Prostate Cancer Detection. Biomolecules. 2024;14:193.
Kim JK, Lee HJ, Hwang SI, Choe G, Kim HJ, Hong SK. The effect of 5 alpha-reductase inhibitor therapy on prostate cancer detection in the era of multi-parametric magnetic resonance imaging. Sci Rep. 2019;9:17862.
Acknowledgements
Marcin Miszczyk was supported by the European Urological Scholarship Programme (EUSP) Scholarship of the European Association of Urology (EAU).
Author information
Authors and Affiliations
Contributions
AC and AM contributed to protocol/project development, data collection and management, data analysis, creating Tables, and manuscript writing/editing. AD, ARA, MM, NR, ASA, SM, IT contributed to manuscript writing/editing. PR, UGF, FS contributed to manuscript editing. GMB, CB, GC, LC, ABG, DD, TK, SFS contributed to supervision and manuscript editing.
Corresponding author
Ethics declarations
Competing interests
Shahrokh F. Shariat received the following: Honoraria: Astellas, AstraZeneca, BMS, Ferring, Ipsen, Janssen, MSD, Olympus, Pfizer, Roche, Takeda Consulting or Advisory Role: Astellas, AstraZeneca, BMS, Ferring, Ipsen, Janssen, MSD, Olympus, Pfizer, Pierre Fabre, Roche, Takeda Speakers Bureau: Astellas, Astra Zeneca, Bayer, BMS, Ferring, Ipsen, Janssen, MSD, Olympus, Pfizer, Richard Wolf, Roche, Takeda. Pawel Rajwa is a paid consultant/advisor of Janssen. The other authors declare no conflicts of interest associated with this manuscript.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cormio, A., Matsukawa, A., Dematteis, A. et al. Risk factors for incidental prostate cancer following surgery for benign prostatic enlargement: A systematic review and meta-analysis. Prostate Cancer Prostatic Dis (2026). https://doi.org/10.1038/s41391-026-01108-5
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41391-026-01108-5