Abstract
From a neuroscience perspective, cancer neuroscience has emerged as a subfield of cancer research. Presumable mechanisms underlying cancer-related neuronal activity (termed neurosciences) include the induction and modulation of signaling pathways that govern cell fate determination and emotional responses (anxiety and stress), such as structural molecules (synaptic structures and current transduction) and secretory substances (neurotransmitters, cytokines, hormones and neuropeptides). In the past 3 years, these neuronal activities, which can either promote cancer growth or be hijacked by cancer cells to support tumor survival and invasion, have been widely demonstrated to be closely related to cancer progression. The molecular mechanisms are also being refined. Despite their great promise, translating neuroscientific discoveries into clinically actionable strategies for cancer diagnosis, prognosis, and treatment remains a formidable task. In this comprehensive review, we attempt to provide a full account of the intersection between neuroscience and cancer research. From the perspective of cancer neuroscience, we fully discuss the potential signaling molecules and their regulatory mechanisms, as well as targets and emerging therapeutic strategies that control tumor progression via multiomics approaches. Overall, cancer neuroscience may have unprecedented potential for understanding neuronal functions and cancer development, ultimately offering the significantly improved cancer treatment.
Similar content being viewed by others
Introduction
Neurons with approximately billion units are found throughout the body. The nervous system is the dominant regulatory system in the human body. In terms of function and location, the nervous system is categorically divided into two parts: the central nervous system (CNS) and the peripheral nervous system (PNS). Neural cell maturation, diversification, migration to specific anatomical locations, and functional circuit assembly, including axon pathfinding, synapse formation, and myelinated infrastructure establishment, are complex processes for nervous system circuit formation. Interestingly, cancerous brain cells exhibit certain features of the maturing nervous system, which is likely the initial driver of the early idea of cancer neuroscience.
In 2017, Venkatesh observed very rapid electrical activity in a population of human glioma cells.1 Cancerous brain cells can interact more actively with electrical communication between groups of brain cancer cells in a continuous and rapid manner.2,3 Similar approaches, such as the ability of tumors to recruit blood vessels to meet tumor tissues for their own feed and growth, cancer depends on the nervous system for almost everything to spread. Interestingly, neural involvement in previously neglected cancer research will exhibit unprecedented hidden regulatory effects. While deciphering which neurons and signaling pathways are involved, the immune system has emerged as a new participant in the interplay between oncology and neuroscience.4 Although this complexity poses challenges to unraveling biological mechanisms, the involvement of additional players may simultaneously open novel therapeutic avenues for patients.5,6,7,8
Ideas and research related to cancer neuroscience were documented a long time ago, but their clear concept did not emerge until 2022 (Fig. 1). Treatise De Tumoribus of Galen of Pergamon was the earliest documented theory that tumorigenesis is associated with black bile, and that worry promotes the production of black bile, thus linking negative emotions to tumorigenesis. Some scholars regard it as the very beginning of cancer neuroscience. The report of nerve involvement was that breast cancer spread after migrating into and around the facial nerve by Cruveilhier in 1835, which represents the aggressive stage during breast cancer progression. Similarly, Alaya et al. reported that perineural invasion is an active interaction between nerves and prostate cancer cells.9 When the density of prostate cancer cells increases to the greatest number, more directional outgrowth of neurites from the dorsal root ganglia can be recruited into prostate cancer cell colonies and establish contact.9 Once contact is established, cancer cells travel along the nerves until they reach the neuronal cell bodies. Prostate cancer cell colony growth in dorsal root ganglia cell cocultures is greater than that in prostate cancer cell-only control cultures. Neurite outgrowth was also increased in coculture.9,10 The discovery of autonomic neurogenesis in prostate cancer tissue represents a breakthrough, and similar infiltration phenomena are emerging in other cancers, which all gradually accelerate the system development of cancer innervation mechanisms.11,12 On the basis of currently available results, interactions can be divided into two categories: indirect interactions, which are usually mediated by the recruitment of immune inflammatory cells, and direct contact through neural-tumor links, such as synapses. Neural signals activate the ion channels on tumor cells, inducing inward currents and establishing an electrical network among tumor cells. Additionally, tumor cells secrete glutamate, which not only creates an autocrine loop but also enhances neuronal excitability, and the secretion of neurotrophic factors further encourages axonal growth in neurons (Fig. 2).6,13,14,15,16
Representative discoveries of the cancer neuroscience aligned with timeline of historic milestones. A summary of the currently available milestones in neuro-cancer crosstalk, including but not limited to: proposing a link between emotion and cancer, confirming that nerves are components of tumor tissue, proposing neural plasticity, confirming that nerves regulate cancer progression, revealing that there is electrical communication and synaptic structure between nerves and brain tumors, etc. PNI perineural invasion, NF1 neurofibromatosis type 1
Schematic of cancer neuroscience—the core concept a. b Widespread and strong communication occurs in the crosstalk of neurons/nerves, immune cells and cancer cells. These interactions control immune cell trafficking and function, antitumor immunity, immune inhibition, and pro-cancer inflammation. c Neuronal activity regulates tumor malignant progression through paracrine growth factors secretion, neuro-glioma synapses, and Ca2+ influx currents. In turn, glutamate and synaptogenic proteins derived from glioma cells promotes neural hyperexcitability and circuits functional remodeling, thus generating excitatory neuronal activity which enhances proliferation and invasion to complete the autocrine feedback loop. d The overview of cancer neuroscience. There is nerve input into solid tumors (both in glioma and extracranial tumors), indicating the neuron/nerves participating in the interaction between various cell types, including cancer cells, stromal cells, and neurons in tumor microenvironment. Paracrine factors together with synapses signaling mediated by AMPAR and NMDAR regulate tumor proliferation and invasion, and in turn, tumor-derived factors promote cancer cells perineural invasion (PNI) and also peripheral nerves in growth into tumor microenvironment
The nervous system has a vital impact on cancer, functioning as a cooperator, in both brain tumors and peripheral tumors, with dominant outcomes of cancer progression.15,17 Not only do nerves provide a highway for the spread of cancer, but they also seem to create a safe protected area. The regulation of cancer through the nervous system can permeate the entire malignant process of cancer (Fig. 3 and Table 1). Tumor recurrence is a multifaceted process involving intricate cellular interactions within the tumor microenvironment and currently remains a significant challenge in brain tumor treatment. In isocitrate dehydrogenase (IDH)-wild-type and IDH-mutant glioma patients, tumors recur in distinct manners, which rely on the IDH mutation status and are attributed to changes in the histological feature composition, somatic alterations, and microenvironment interactions.18 IDH-wild-type tumors are more invasive at recurrence, and neoplastic cells exhibit elevated neuronal activities and characteristics, reflecting a possible role for neuronal interactions in promoting glioma progression.18,19,20 We emphasize multiple signaling pathways connecting cancer to neuroscience. In this review, we hope to contribute to research on nervous system innervation in tumor tissue progression, which may shed new light on the design of therapeutic strategies for cancer patients.
Mechanisms underlying cancer cell fate determination. Neural signaling molecular pathways determine the fate of cancer cells. Normal cells constantly undergoing proto-oncogene and tumor suppressor gene signaling pathway alternation and environmental stress (physical or chemical) transform into cancer cells. Subsequently, the loss of contact inhibition and proliferation restriction of cancer cells allows them indefinite proliferation. Growing up as the tumor, tumor cells continue to secrete signaling inducing angiogenesis to supply for nutrients and oxygen required of tumor tissues. Meanwhile, the cancer cells can be heterogeneous mediated by CSCs, and recruit normal stromal cells to evade immune attack. Subsequently, cancer cells will compress nearby tissues, generate MMPs, break through the basement membrane, produce morphological changes that undergo EMT, achieve extravasation into blood vessels or lymphatic vessels, and metastasis with a bias. After CTCs survive in the blood circulation reach distant metastatic sites, they extravasate to the appropriate niche. Some choose to form secondary tumors, while others may become dormant, evade surveillance, or even reactivate after treatment, causing cancer recurrence. Further micrometastasis grows to form macrometastasis, but certain tumor cells have been shown to be regulated to differentiate into benign cell lineages. Throughout the course of carcinogenesis and development, cancer cells under stress conditions, fail to survive in the blood circulation, and are attacked by the immune system or eliminated by treatment
Crosstalk between the nervous system and cancer
The communication between the nervous system and cancer is a fascinating topic. In this section, we classify and discuss the neuro-cancer crosstalk that has been demonstrated thus far (Figs. 4 and 5). Moreover, on the basis of the roles and preferences of classified signaling molecules, we speculate on signaling pathways associated with the key molecules involved in neuro-cancer communication.
Interactions between nervous system and cancer. a Regulation of cancer cells by the CNS and PNS, respectively. In CNS, neurotransmitters from neuro-cancer synapses can induce depolarization of cancer cell membrane currents. Paracrine molecules can control tumor growth by manipulating cancer cell metabolism pathways. Astrocytes can release LCN2 to participate in chronic neuroinflammation by recruiting granulocytes, which promotes cancer brain metastasis. Further, the expression level of neurotransmitter receptors also modulates brain metastasis. In PNS, peripheral nerves (adrenergic nerve, cholinergic nerve, and sensory nerve) release neurotransmitters to modulate cancer malignant progression. b In turn, cancer can also act back on the nervous system, and this phenomenon occurs in both the CNS and PNS. In CNS, main manifestations include nervous system hyperactivity, synaptogenesis and neuronal connectivity. Glutamate secreted by tumor cells not only induces nerve excitability but also forms an autocrine loop to promote tumor tissue growth. In PNS, cancer cells can recruit new nerve fibers and promote innervation in the local TME
Molecules involved in nervous system-cancer interactions. The major molecules involved in the relationship between neural signaling and cancer cells‘ malignant progression include systemic signals (neurotransmitters and innervation) a, local signaling molecules (paracrine signaling) b, synapses (electrochemical signaling) c, transcription factors d, and immune mediators e
Secretory signals
Paracrine signal is one type of neuronal input. Neurons or nerve cells in the tumor microenvironment secrete neurotransmitters, neurotrophic factors, cytokines, and neuropeptides, and activate cancer-promoting pathways, such as the Wnt/β-catenin and STAT3 pathways, in tumor cells through receptors. Paracrine interactions in the tumor microenvironment have been described between neurons/nerves and cancer cells (Figs. 4 and 5).
Neuroligins (NLGN)
NLGN, a postsynaptic adhesion molecule that participates in synapse assembly and function, is cleaved and released into the tumor microenvironment to drive glioma cell proliferation.15 Secretion of NLGN3 by active neurons induces the PI3K-mTOR signaling pathway to promote glioma growth.21 An uncontrolled cell cycle is a condition in which cancer cells grow at an exceedingly fast pace. Certain cell cycle proteins are repurposed to serve functions outside of cell cycle control.22 CYY-1, a cyclin box-containing protein, drives synapse removal in this process. Moreover, CDK-5 facilitates new synapse formation by regulating the transport of synaptic vesicles to the sites of synaptogenesis. NLGN3, a Cdk5 substrate, is activated by Cdk5-mediated phosphorylation, which further augments NLGN3-mediated synaptic currents and synaptic transmission between neurons.23 CDK5 amplification may trigger the activation of STAT3, which is involved in tumor proliferation,24 suggesting that CDK5 may be at the critical crossroad between neuronal activity and tumor proliferation.
In addition to NLGN3, the neuronal activity-dependent signaling of BDNF and GRP78 promotes glioma proliferation and growth, acting as a neuronal activity-regulated glioma mitogen.21,25,26 NTRK2, a receptor of BDNF, is relevant to glioma proliferation and can be inhibited to decrease neuronal activity-induced proliferation.26 This function has also been demonstrated in neuroblastoma, as the overexpression of BDNF and GRP78 promotes proliferation, whereas their knockdown inhibits growth through the PI3K/AKT pathway.27 GRP78 can be regulated through the GRP78-FOXM1-KIF20A pathway to control proliferation and G2/M cell cycle arrest.28 Interestingly, neuronal re-entry into the cell cycle and cancer cell tumor suppression may activate the same p53-dependent signaling pathway to induce cell death. The cause of cancer cell proliferation likely involves alterations in the expression of cell cycle molecules. Abnormal activity of cyclin-CDK in stressed glial cells is also observed in glioma cells. This phenomenon suggests a considerable similarity between the mechanisms governing the normal division and differentiation of cells and those involved in carcinogenesis. An in-depth study of the processes of dendritic and axonal differentiation and maturation can provide clues for identifying neural communication targets in brain tumors.
NLGN3 transcription may also be regulated by the Wnt/β-catenin signaling pathway and surrounding secretions to transform neighboring cells into CSCs.29 These findings suggest that the function of Wnt/β-catenin is to regulate both NLGN3 expression and the acquisition of CSC properties in glioblastoma. The nuclear translocation of YBX1 has been shown to be closely linked to increased NLGN3 expression and glioblastoma progression.30 YBX1 binds to the NLGN3 promoter to increase its level through the nuclear import of NLGN3.30 In gastric cancer, YBX1 can regulate the transcriptional activity of the Wnt signaling pathway via long noncoding RNAs, which together form a positive feed-forward loop to promote gastric cancer malignancy.31 This phenomenon is not limited to the brain, as NLGN3 mutation and amplification have also been demonstrated to promote cancer growth-promoting functions via innervation.32 In peripheral cancers that exhibit YBX1-positive expression, NLGN3 expression can also be enhanced by YBX1 binding; however, it remains to be verified whether there is tissue specificity in the brain and peripheral cancers.
Nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF)
NGF and BDNF, which are neurotrophins secreted by pancreatic cancer cells, are tightly regulated by sympathetic innervation and enrichment of norepinephrine in the local microenvironment.33 These observations underscore the importance of cancer cell-derived neurotrophic growth factors as the main drivers of axonogenesis within the tumor and metastatic tumor program. Breast cancer has a brain metastasis bias through the ability of the abluminal vascular migration route to enter the CNS along the surface of emissary veins.34 Integrin α6, a laminin receptor expressed on the surface of breast cancer cells, helps them move along laminin-rich emissary veins to the leptomeninges. During this process, glial-derived neurotrophic factor (GDNF) plays an important role in reducing breast cancer cell death in nutrient-poor leptomeninges. GDNF is usually secreted by reactive CNS microglia and macrophages in response to brain injury, and it is located in the extracellular matrix (ECM) to block apoptotic neuronal stress responses. A majority of breast cancer cells colocalize with macrophages in meningeal leptomeninges, and stimulate macrophages to produce GDNF to promote the survival of brain metastatic cancer cells.34 Nerve secretion of GDNF and artemin promotes pancreatic cancer invasion.35,36 The PNS is highly relevant to the invasion and spread of primary peripheral tumors (Figs. 4 and 5). Perineural invasion (PNI), which has been observed for more than 100 years, is involved in cancer aggressiveness and poor survival rates.12 Sensory nerve innervation enhances breast cancer invasion and spread.37 β-adrenergic receptor-mediated sympathetic neural signaling can regulate cytoskeletal alterations and protease production in breast cancer to increase cancer invasion.38 In clinical samples, a new outcome emphasized that highly metastatic tumors in mouse models have higher densities of nerve fibers than do less metastatic tumors in tumor tissues, indicating that nerve densities are highly related to cancer progression and that cancer cells can induce the outgrowth of nerve fibers to increase the number of neural structures and nerve terminals in the local microenvironment.39 These findings suggest that tumor innervation and the vasculature are the major signals for tumors outside of brain metastases.
Insulin growth factor 1 (IGF1)
IGF1, a secreted paracrine signaling molecule stimulated by olfaction in an activity-dependent manner, mediates olfactory bulb gliomagenesis through sensory neuronal circuits,40 suggesting that specific sources of neuro-paracrine factors modulate the malignant characteristics of distinct cancer types. Several molecular markers of brain cancer have been shown to influence neurocognitive functions, such as psychomotor speed, memory performance, and executive function.41 These molecules influence cognitive performance via several possible mechanisms, such as the perturbation of neuronal communication. These findings may provide an entry point for a detailed discussion of the mechanisms by which cancer interferes with neural circuits to promote the malignancy.
Acetylcholine (ACh)
ACh, a classic neurotransmitter of axon terminals that is released by cholinergic nerves, can exert either activating or inhibiting signals and is also involved in tumor progression and spread. For example, denervation of cholinergic nerves or knockout of the receptor CHRM1 inhibits prostate tumor cell spread.42 Denervated stimulation of cholinergic nerves also leads to the progression of pancreatic cancer through the release of ACh. Ach activates the receptor CHRM1 in cancer stem cells, and CHRM1 signaling inhibits the downstream MAPK and PI3K/Akt pathways in pancreatic cancer cells.42 Sensory and sympathetic nerves promote the growth of pancreatic cancer cells through neurokinin receptors and adrenergic signaling, respectively, whereas parasympathetic nerves inhibit cancer stem cells through cholinergic signaling. These findings suggest that tumor innervation is not only very precise but also a balance of neuromodulation specificity. Additionally, ACh can suppress the expression of CCL5 in PDAC via histone deacetylation by histone deacetylase 1. Low levels of CCL-5 inhibit the immune effect of CD8+ T-cells, leading to an immunosuppressive TME.43 These findings suggest that endocrine signals can play a regulatory role in multiple fields of peripheral tumors.
Noradrenaline
Noradrenaline, known to trigger the angiogenic switch by ADRB2 (β-adrenergic receptor) in endothelial cells, can promote tumor growth.44 Adrenergic nerves indirectly regulate cancer cell survival through angiogenesis and metabolism for nutrient availability.44 Given that the initiation of angiogenesis is mediated by aerobic glycolysis in endothelial cells, sympathectomy and the conditional deletion of endothelial Adrb2, which encodes the β2 adrenergic receptor, can increase the expression of COA6, which is involved in the electron transport chain, and further mediate the metabolic shift from glycolysis toward oxidative phosphorylation to inhibit angiogenesis.44 Adrenergic nerve infiltration in the prostate cancer microenvironment leads to the release of norepinephrine from nerve terminals, which subsequently stimulates the expression of ADRβ2 in endothelial cells. The latter enables aerobic glycolysis in endothelial cells to control tumor angiogenesis and malignant progression. When Adrb2 is ablated or inhibited, it not only activates neovascularization but also impairs tumor progression. The activation of β-adrenergic receptors located at the surface of cancer cells has been shown to promote tumor tissue growth by recruiting and nourishing nearby blood vessels.45 This broadens the field of angiogenesis research, as some phenotypic characteristics of endothelial cells or pericytes may be involved in the regulatory switch of angiogenesis in tumor tissues. Capillary pericytes in the vasculature play a key role in controlling the blood flow and diameter of capillaries throughout the brain.46 After vasoconstriction and slower blood flow are induced by optogenetic stimulation, capillary pericytes also show a slower rate of expansion close to baseline. When optogenetic stimulation is removed, capillaries dilate locally, and capillary blood flow increases in the absence of pericytes. Whether neural signals can also regulate pericytes is an urgent scientific question in vascular biology and tumor biology. The expression characteristics of pericytes, metabolic characteristics, and cytoskeletal contractility are related to the molecular mechanisms of neurotransmitter receptor expression or neuronal differentiation. There are specific alterations in the vasculature of healthy adults and in patients with brain tumors.47 How CNS-specific mechanisms control angiogenesis, whether this specificity can be compared and analyzed by multiomics in tumors, and how it is related to embryonic development and differentiation processes are still unanswered questions.
5-Hydroxytryptamine (5-HT)
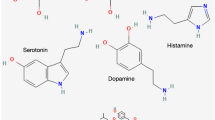
5-HT, also called serotonin, is released by enteric serotonergic neurons and combines with HTR1B/1D/1F receptors on the surface of colorectal cancer stem cells,48 which activates the Wnt/β-catenin signaling pathway to promote colorectal cancer stem cell self-renewal and tumorigenesis.48 In addition, the activity of serotonergic neurons can also regulate brain tumor formation by activating modifications to histones.49 After serotonylation on histones, the transcription factor ETV5 is enriched in ependymomas, which decreases overall survival and enhances tumor proliferation by downregulating the expression of neuropeptide Y.49 The overexpression of neuropeptide Y suppresses tumor progression by remodeling the brain microenvironment toward synaptic inhibition and, in turn, decreases brain hyperactivity and inhibits tumor progression.49 The activation of discrete subtypes of neurons suppresses ependymoma tumorigenesis, highlighting the need to decipher how neuronal-subtype and circuit-specific interactions affect brain-tumor progression.
Neuropeptides
Neuropeptides, important neural regulatory substances, are produced not only by the nervous system but also by the peripheral organs and are discussed in both the CNS and the PNS. Neuropeptides may function as neurotransmitters, neuromodulators, and endohormones. Paracrine signaling is also not limited to primary brain tumors.34 Breast cancer cells induce spontaneous calcium activity in sensory neurons and drive the release of the neuropeptide substance P from neurons. Elevated expression of substance P is associated with enhanced lymph node metastatic spread in patients. Substance P can act on cancer cell populations with high TACR1 expression, leading to the death of a small number of these cells. The dead cells then release single-stranded RNAs to act on TLR7 in neighboring tumor cells and thereby trigger the prometastatic gene expression program.39
Synapses and electrical signals
Glutamate
Synaptic connections, which require synapse signals (e.g., glutamate), are critically pivotal for communication between neurons and their environments, where electrical activity may be used to control various cellular processes and behaviors, including newly generated neuron migration to the appropriate location and axonal targeting.50,51,52 The glioblastoma cell subpopulation with invasive features exhibited migration patterns similar to those of immature neurons. Bona fide synapse formation between presynaptic neurons and postsynaptic glioma cells drives tumor growth and invasion by transmitting electrical and chemical signals.2,3,53 These synapses are oriented from neurons to glioma cells via AMPA receptor-mediated excitatory postsynaptic currents in glioma cells. The use of AMPA receptor inhibitors can effectively reduce glioma cell proliferation and invasion.3 Another kind of synapse, similar to the position of tripartite synapses mediated by astrocytes, has been shown to be involved in breast-to-brain metastasis.54 The key element of these two types of synapses is the glutamate signal. Glutamate, a paracrine secreted factor, potentially plays a role in increasing neuronal hyperexcitability and tumor growth if abnormally produced in GABAergic interneurons in the glioblastoma microenvironment.55,56 BDNF paracrine secretion regulates neuron-to-glioma synapse number and AMPA receptor trafficking to the glioma cell membrane via the BDNF-TrkB pathway, thus increasing the amplitude of glutamate-evoked currents.26 Abrogation of BDNF secretion or blockade of glioma TrkB expression effectively decreases neuron-to-glioma synaptic connections, reduces neuronal activity-induced glioma proliferation, and prolongs survival.26 The secretion of NLGN3 also participates in regulating the number of neuron-to-glioma synapses.2 The health adaptability of synaptic plasticity, which emphasizes the interaction between neurons and gliomas, can be strengthened via an activity-dependent regulatory mechanism.
Gamma-aminobutyric acid (GABA)
According to the latest results, GABAergic inhibitory synapses exist in the first frontal diffuse midline gliomas, not only early recognized glutamatergic excitatory synapses.57 GABAergic input increases the intracellular chloride concentration and a strong inward current in diffuse midline glioma cells via the NKCC1 chloride transporter, leading to depolarization of the diffuse midline glioma cell membrane and promoting diffuse midline glioma cell proliferation. This once again demonstrates the important role of ion concentration differences and potential changes on the membrane surface of brain tumors in tumor development. However, in contrast to the above results, the loss of GABAergic neurons leads to the proliferation of tumor cells. The high chloride concentration in diffuse midline gliomas can also lead to depolarization of the cancer cell membrane, thereby promoting the survival of tumor cells. The strong inward current induced by GABAergic input is due to the difference in Cl- concentration, which still leads to depolarization of the tumor cell membrane, thereby promoting brain tumor survival.57 We can speculate that membrane potential depolarization, rather than excitatory and inhibitory transmitters, is the main cause of cancer cell proliferation. The feature that tumor cells “hijack” neuronal features to support survival is worth exploring in peripheral tumors. An important breakthrough for combating and controlling malignant development and progression could be achieved if the brain metastasis of peripheral tumors mimics the hijacking of neural circuits and the generation and dissemination of electrical signals on the surface of the tumor cell membrane.
The overexpression of glutamic pyruvate transaminase (GPT2) elevates the concentration of GABA and then activates GABAA receptors to promote breast cancer metastasis.58 In breast-to-brain metastatic models, the GABAergic phenotype is similar to that of neuronal cells. Breast-to-brain metastases take up and catabolize GABA into succinate, with the resulting formation of nicotinamide adenine dinucleotide as a biosynthetic source to provide proliferative ability itself.59 As a neurotransmitter, GABA is also a product of amino acid metabolism, and its expression and metabolic pathways may also be related to the trend of brain metastasis of tumors.
Neuronal excitability
Glioma cells have the ability to drive synaptogenesis60,61 and neuronal connectivity62 to further increase associated neuronal hyperexcitability. In brain tumor tissue, the tumor cells themselves exhibit neurodevelopmental features. Tumor microtubes (TMs) and tunneling nanotubes (TNTs), neurite-like membrane protrusions that mimic mechanisms of neurite pathfinding, are used to invade the brain and promote tumor colonization in glioma (Figs. 4 and 5).63,64,65 After neural stimulation, similar to immature neurons and oligodendrocyte precursor cells receiving message input from synapses, the invasion and the TMs genesis of glioma cells are increased.53 TMs act as routes for brain invasion and interconnection among tumor cells to form a resulting network in glioma.66 Single glioma cells are connected to functional and communication networks by TMs and gap junctions, which are essential for cancer metastasis, therapeutic resistance, and tumor homeostasis.63,64,66 Importantly, this type of connection is also found in the astrocytic network of the brain, which may provide resources for brain metastasis survival.53 Calcium transients and potassium currents that are spread by TMs and gap junction-mediated tumor networks have been shown to achieve electrical coupling, enable multicellular communication and drive brain tumor progression (Figs. 4 and 5).2,3 Indeed, accumulation of GAP43 and TTYH1 is observed at the tips of TMs, similar to the growth cones of neurites during neurodevelopment.63,67 In glioblastoma, a small number of tumor cells, called intrinsically rhythmic glioblastoma cells, exhibit autonomous activity, which generates rhythmic Ca2+ waves to influence tumor survival and growth.68 Along with the increased number of neuro-cancer synapses, the number of tumor-activating intercellular Ca2+ waves in glioma networks is also increased.2,3 These data suggest that electrical coupling allows electrical signals to propagate between cells and influence the behavior of tumor cells. In addition to considering synaptic structures, alterations in ion channels, neural plasticity, and cell connection coupling are also essential for broadening research directions. Some paracrine factors and synaptogenic factors are intermingled and result in glioma-induced neuronal hyperexcitability. In peripheral tumors, secreted neurotrophins released from nerves or secreted from tumor cells, have been shown to critically modulate tumor growth.69,70 The role of this peripheral nerve in the regulation of extrabrain tumors has been well described.69,71 Cancer outside of the brain can recruit new nerve fibers through axonogenesis to increase innervation and then remodel the neural microenvironment.10,71 The ability of peripheral tumors and recruited nerve fibers to develop electrical signal communication networks or metabolic networks similar to those of brain tumors may be a signal that triggers the survival and metastasis of tumor cell populations.
CGRP
Recent studies have demonstrated the existence of electrical signal transduction between peripheral nerves and peripheral tumors.72 Peripheral pain nerves (CGRP peptidergic neurons) are expanded in gastric cancer models in a manner dependent on tumor NGF expression, and there are anatomical and functional tight connections with tumor spheres. Through in vivo optogenetic and calcium imaging experiments, researchers have shown that activation of nociceptive nerves increases CGRP expression and release and the expression of the CGRP receptor RAMP1 on the tumor cell membrane. There is increased calcium flux between neurons and tumor cells.72 Bidirectional signaling between nerves and cancer cells promotes the proliferation of cancer cells and CAFs.
Nociceptive nerves also promote the metastasis of gastric cancer cells. Gastric cancer cells can migrate along the nerve, and then, circulating tumor cells can be detected in the portal vein.72 Transcription factors involved in neuronal reprogramming, such as NeuroD1 and ASCL1, are increased in cancer cells connected to neurons, suggesting that similar to glioma synapses, cancer cell-peptidergic neuronal circuits may induce neuron-like phenotypes in peripheral tumors promoting cancer metastasis. This finding highlights the importance of calcium communication in the peripheral tumor-nerve circuit and tumor cells with membrane potential depolarization and the upregulation of synaptophysin expression in cancer cells, suggesting that the communication mechanism between peripheral nerves and tumors is very similar to that in neuroglioma synapses. The mechanisms of sensory nerves and other types of peripheral tumors can also be further elucidated via similar approaches.
Neurotransmitters and other chemical messengers or modulators
Lactic acid
Lactic acid (lactate), a product of glucose metabolism in tumor cells, plays an important role in the tumor microenvironment, not only serving as a metabolic substrate to support tumor cell survival and proliferation, but also acting as a signaling molecule with various biological effects. One of the key functions of lactic acid is its ability to stimulate angiogenesis to provide nutrients and oxygen for tumor cells, and enhance the production of enzymes that digest the ECM for cancer invasion.73,74 In addition to overcoming nutrient deficiency by activating metabolic mechanisms, lactate has been proven to remodel histone acetylation and regulate gene expression to promote GBM cell survival and proliferation.75 In the CNS, glial cells promote axon regeneration after CNS injury via the lactate-GABAB receptor-cAMP signaling pathway.76 The lactate-GABAB receptor-cAMP signaling pathway can increase the stability and growth of axons. Similarly, axon regeneration has also been revealed in the PNS, in which lactate metabolism in neurons enhances axon stability mediated by Schwann cells.77 In summary, lactate might regulate neural signals to control the development of GBM because of the beneficial effects of lactic acid on axon regeneration in the CNS and PNS. However, it remains unclear whether lactate promotes GBM cell survival through mediating axon generation in local tumor microenvironment of GBM. One study demonstrated that anabolic and proliferating cells perform biomass synthesis via incomplete oxidation of glucose and high glucose consumption.78 The photoreceptor cells located on the outer retina still produce lactate in the presence of oxygen, enabling diurnal renewal of the photoreceptor outer segments.79 Under hypoxic conditions, dysregulation of oxidative phosphorylation and the tricarboxylic acid cycle (TCA) cycle in rods with an activated hypoxic response decelerates cellular anabolism, causing shortening of rod photoreceptor outer segments before the onset of cell degeneration. An intact TCA cycle does not exhibit these early signs of anabolic dysregulation and shows a slower course of degeneration, which highlights the importance of mitochondrial metabolism, especially the TCA cycle, for photoreceptor survival in conditions of increased hypoxia-inducible factor activity.80 Lactate produced by astrocytes is exported to neurons to fuel mitochondrial respiration to support synaptic activity.81 Overactivation of the kynurenine signaling pathway by indoleamine 2,3-dioxygenase 1 (IDO1) causes astrocytes to fail to produce sufficient lactate as an energy source for neurons, thereby disrupting healthy brain metabolism and damaging synapses.81 An IDO1 inhibitor rescues the ability of astrocytes to produce lactate, which is subsequently taken up by neurons to provide energy for mitochondrial respiration and synaptic activity.81 These reports suggest that glucose metabolic pathways are critical for ensuring nervous system homeostasis and are important mechanisms for neuro-cancer communication.
Dopamine
Dopamine, a monoamine neurotransmitter, affects the interplay between cancer progression, immunity, and the central nervous system.82 The treatment of prolactinomas in pituitary neuroendocrine tumors is unique in that they are the only first-line treatment, which is achieved with dopamine agonist drugs. Through transcriptome sequencing and single-cell sequencing analysis of drug-resistant and drug-sensitive prolactinomas, DA resistance was shown to be associated with upregulation of the adhesion plaque (FA) signaling pathway. Fagin can exert its antitumor effect by inhibiting the expression of FA pathway components. Fagin inhibits the proliferation of pituitary tumor cell lines and organoids and promotes the apoptosis of pituitary tumor cells. In in vivo experiments, genistein effectively inhibited the formation of subcutaneous tumors.83 Dopamine is produced not only by the brain, but also by peripheral organs, such as the spleen and pancreas.84,85 Detections in lung cancer patients indicate significant elevation of plasma dopamine in malignancy due to stress from the disease process, which can inhibit T-cell proliferation and cytotoxic capacity.86 Interestingly, one study revealed an unconventional role for dopamine receptor (DRD4) beyond its classic function as a neurotransmitter receptor.87 DRD4 expression is significantly upregulated in colorectal cancer clinical samples, which is associated with poor patient prognosis. In vitro experiments have shown that simple overexpressed DRD4 can interact with transforming growth factor β receptors (TGFBR1 and TGFBR2) to induce epithelial-mesenchymal transition (EMT) in cancer cells independent of dopamine. The regulatory role of dopamine in malignant tumor progression has been described in detail in a recent reference.85 In this section, we discuss the latest dopamine-based technological innovations. The use of MgO2 nanoparticles and dopamine-conjugated gelatin as the main components for the clinical treatment of osteosarcoma, which can release H2O2 via photothermal therapy to significantly inhibit tumor recurrence, and sustainable Mg2+ effectively promotes bone regeneration, has been reported.88 Similarly, dopamine-carrying nanomaterials have also shown effective therapeutic capabilities, on the one hand depleting essential nutrients to cancer cells and curbing cancer cell survival and, on the other hand, near-infrared lasers allowing real-time fluorescence imaging and guiding dopamine-mediated mild photothermal therapy to resist tumors together.89
Melatonin
Melatonin, an amino acid hormone, is produced by the ingestion of tryptophan in the body, is converted to 5-hydroxytryptamine (serotonin) and is then converted to melatonin by N-acetyltransferase and hydroxyindole-O-methyltransferase in the pineal gland. Melatonin is a major neurotransmitter and neurohormone involved in transferring information to the CNS to regulate brain function, such as circadian rhythm and sleep, as well as exerting antitumor effects on various cancer types.90,91 The effects of melatonin on cell fate determination-related mechanisms may have therapeutic potential and may be further explored to address this open question.
Transcription factors
Transcription factors are widely known to regulate gene expression. In our current understanding, they are more likely to reflect some similarity between neural component development and brain tumor progression (Fig. 5).
ZEB2
ZEB2 is a transcription factor that often functions in early germ layer specification and neocortical axon outgrowth, highlighting its importance in human neurodevelopment.92,93 In many aspects, the switch from neuroepithelial cells to transitioning neuroepithelial cells to radial glia resembles a partial EMT process with a progressive change in cell morphology, apical constriction, and a reduction in cell–cell junctions.94 Several reports have shown cross-regulation between ZEB2, the actin cytoskeleton and lipid metabolism.95,96 In glioma, ZEB has been confirmed to be involved in the regulation of its malignant progression, and there is an antagonistic effect with microRNAs to regulate the malignant invasion of glioma.97 Therefore, in other tumors with a propensity for brain metastasis, such as lung cancer, breast cancer, melanoma and colorectal carcinoma, investigating the molecular biological function of ZEB and epigenetic modulation molecules may reveal unknown effective metastasis regulatory pathways and targets for inhibiting cancer metastasis.
Studies have revealed the retinal cell development via single-cell RNA-seq and assays for transposase-accessible chromatin with high-throughput sequencing (ATAC-seq) analyses, indicating that the different subtypes of retinal neurons in vertebrates arise from distinct lineages.98 The discovery that a single transcription factor can efficiently reprogram neural precursor cells from a specific lineage into a particular type of retinal neuron represents a breakthrough in the field of cellular reprogramming.98
NeuroD1
NeuroD1 directly reprograms endogenous astrocytes into neurons, which can successfully develop and integrate into the visual cortical circuit after ischemic injury, leading to vision recovery.99 Reprogrammed astrocytes can even differentiate into spinal cord neurons and form synapses with host neurons upon transplantation, representing a major advance in reprogramming.100 NeuroD1 has also been shown to induce reprogramming from glioblastoma cells to glutamatergic neurons.101 Despite these advances, cross-lineage differentiation is controversial, and the differentiation in the nervous system is currently largely restricted to lineages.102,103 Reprogramming of cells into different lineages is likely difficult, and understanding the lineage-specific transcription factors and epigenetic modifications for reprogramming may be highly important. Astrocytes, for example, can reflect developmental cues because they are susceptible to reprogramming.104 Similarly, it is interesting to ask whether neurons that can be reprogrammed by astrocytes have conserved sequences in their genome and transcriptome sequences. It is not clear whether there are some program restrictions in lineages of benign intracellular transformation, and it is also unknown whether there are any characteristics of cancer cells that overcome this restriction.
Neuro-immune-cancer circuit
The communication between the nervous and immune systems was first identified more than three decades ago and is now well established. The tumor microenvironment can be subdivided into several different categories and related drug repurposing, with the main concerns being hypoxia and lactic acid metabolism, lipid metabolism, pH, and innervation.105 In addition, in primary and recurrent glioblastoma resections of patients, the expression of hallmark genes of glioblastoma does not significantly change, but tumor purity decreases over time.19 Moreover, this change is accompanied by increased expression of neuron and oligodendrocyte marker genes in tumors.19,106 PNS, especially the autonomic nervous system, mainly participates in the transmission of instructions and inflammatory regulation of the tumor immune response outside the brain tumor.107,108 Sympathetic nerves, or adrenergic signals, regulate the migration and responsiveness of immune cells to control the strength of the immune susceptibility.109 In response to psychological stress, stress networks in brain regions induce peripheral lymphocyte and monocyte migration to a suitable functional landscape to calibrate the response of the immune system to physical threats.110 In addition, stress upregulates the expression of the glucocorticoid-inducible factor TSC22D3 and subsequently subverts anticancer immune therapy.111 Close relationships have been demonstrated among plasma cortisol levels, negative psychological mood, and tumor progression in cancer patients.111,112 It is also not surprising that sensory nerves can influence the immune response of solid tumors.
The cancer cells in the tumor microenvironment might crosstalk with certain stromal cells, including CAFs, macrophages, MDSCs, and immune cells, as well as with vessels, nerves, and ECM.113,114 Cancer cells manipulate surrounding normal cells (cancer-associated fibroblasts, endothelial cells, and immune cells, etc.) or induce tumor microenvironment cells producing immunosuppressive cytokines, such as TGFβ, IL-10, or PGE2, to generate a growth-supportive microenvironment.115 Brain-gut axis is an essential pathway networks consisting of complex interdisciplinary fields including nervous system, immune system, and microbes, which actively participates in a variety of brain cancer types.116 Gut microbes, an important component of brain-gut axis, can secrete gut microbial neurotransmitter to mediate the malignant progress of tumor cells, which is similar the phenomenon in neuro-glioma in brain.117,118 The mechanisms, underlying cross-talk among immune cells, tumor cells and nervous system, profoundly influence tumor cells phenotype and behavior, as well as promote tumor proliferation, invasion, immune escape, and a more favorable microenvironment with angiogenesis for cancer development (Fig. 6).119
Crosstalk in neuron-immune-tumor microenvironment for cancer cell fate determination. The evolving tumor microenvironment is essential during all stages of cancer progression, supported by multiple key representative elements, including multiple immune cells (T-cell, Treg, NK cell, TAMs, neutrophil, and monocyte), CAFs, neurons, ECM, and cytokines. Normally, cancer cells are attacked to destruction by the immune system at the earliest stages of tumor initiation, while the immune system is gradually influenced along with the development of cancer to eventually provide pro-cancer functions. The hypoxic environment in which the cancer grows promotes immune cells and CAFs to secrete VEGF, IL-10, TGFβ, MMP-9, and integrin to support tumor growth, proliferation, angiogenesis, dormancy, EMT, invasion and metastasis. Meanwhile, neurons in tumor microenvironment are recruited to promote tumor malignant development, in turn, cancer cells secrete factors and EVs to regulate axongenesis, neuron reprogramming, and neurogenesis. (The arrows represent induct, and the horizontal lines represent inhibit)
Adrenergic receptor (AR) activation by norepinephrine (NE) and/or E increases the expression of vascular endothelial growth factor (VEGF), matrix metalloproteinases (MMPs) through AR-dependent cAMP-PKA pathway and further promotes angiogenesis, proliferation and invasion.120 On the other hand, tumors drive neurogenesis to initiate new nerve fibers by the secretion of neurotrophic factors.121 This dynamic interaction constitutes a nerve-tumor-immune microenvironment, and the involvement of neural signals also increases the complexity of tumor microenvironment components.
T-cells
T-cells (Table 2) are the main components of adaptive immune system to kill infected cells and the major targets of cancer immunotherapy. The activation of adrenergic signaling from sympathetic nervous system halted mobility of immune cells and impaired T-cell responses through reducing local blood flow and limiting immune cells access.109 Pain-initiating sensory neurons release the neuropeptide CGRP to promote CD8+ T-cell exhaustion through CGRP-RAMP1 axis inhibiting their immune capacity to eliminate melanoma and enabling melanoma cells survival.4 The similar mechanism of midkine secretion acting on T-cells also exhibits in gliomas. NF1 gene mutations enhance neuronal excitability acting on CD8+ T-cells to secrete CCL4, leading to the secretion of a mitogen CCL5 from microglia, which elicits cell proliferation signaling in cell cycle.122 Ventrolateral medulla (VLM) catecholaminergic (CA) neurons promote tumor growth particularly mediated by regulating adaptive immune through CD8+ T-cells.123 Whether other immune cells, such as Treg cells and macrophages, are involved in CA neuron-regulated tumor growth mechanisms in other tumor types maintain exploration.
Neuroendocrine-immune system is integral to controlling cancer cell fate. Cancer cells release neurotransmitters/neurohormones and mediators into the circulation to activate receptors on neuron membrane or circulating immune cells to affect body homeostasis and tissue function in a mode favoring the tumor’s progression. For example, skin cancer-derived neurohormones, including corticotropin releasing hormone (CRH), and pro-opiomelanocortin (POMC)-related downstream hormones, act on other endocrine glands and immune cells affecting circadian rhythm, stress responses, tumor progression and immune escape via hypothalamic-pituitary-adrenal (HPA) axis. The processing of cancer neuroendocrine involves circulatory system, immune system and peripheral tissues by cortisol, biogenic amine (BA) and neuropeptides to generate a permissive microenvironment for the tumor’s phenotype and behavior alternation as well as react on nervous system disrupting brain function.124 Similar to the HPA axis, mice with different subcutaneous tumors exhibit hypothalamus activation and increased production of α-melanocyte-stimulating hormone (α-MSH) encoded by proopiomelanocortin (POMC) via the hypothalamic-pituitary (HP) unit to enhance tumor-induced myelopoiesis and immunosuppression by melanocortin receptor MC5R. Of these, tumor-derived α-MSH inhibits the tumor-infiltration of CD8+ T, CD4+ T and NK cells as well as the expression of interferon-γ (IFN-γ) in CD8+ T-cells to resist tumor immunity leading to tumor cell survival.125 T-cells are often known to exhibit anti-tumor immunity; however, tumor microenvironment can hamper this function by altering metabolic and epigenetic programs in T-cells.126 Understanding these complex interactions is essential for developing novel therapeutic strategies that can modulate the neuro-immune axis to enhance anti-tumor immunity.
Tumor-associated macrophages (TAMs)
TAMs, one of the most important tumor-promoting inflammatory cells, can be categorized into two types (M1 and M2). TAMs produce massive growth factors, small molecule cytokines, and MMPs that drive uncontrolled proliferation, promote EMT, support angiogenesis, aid the degradation of ECM, foster invasion, and suppress the adaptive immune system to help cancer avoid immune recognition and destruction.127,128,129 Tumor necrosis factor (TNF) is a chemoattractant for macrophages. Adrenergic nerves, which innervate spleen activated by vagal stimulation, promote splenic T-cells with β2-adrenergic receptors to secrete Ach that in turn inhibits the production of TNF of macrophages to suppress immune responses.107,130 Vagotomy can remove the immunosuppression and elevate TNF levels.130 Similarly, a phenomenon about the increment of nerve-dependent TAMs has been observed in breast, prostate, and pancreatic cancer.131,132,133 However, the sympathetic nervous system and parasympathetic nervous system appear to have the opposite effects on TAMs recruitment just like sympathetic nervous system signals to promote TNF release and TAM recruitment whereas parasympathetic nervous system might suppress these processes.134 TAMs represent one of the most abundant tumor-infiltrating immune cell types in the tumor microenvironment and present at all stages of cancer progression. Targeting TAMs has become a promising immunotherapy strategy, and understanding cancer neuroscience deeper may significantly improve the breadth and precision of this strategy.
Tumor-infiltrating B cells
Tumor-infiltrating B cells, including plasma cells and regulatory B cell (Breg cell) subsets, perform multiple functions in cancer treatment. Tumor-infiltrating B cells produce antibody and also participate in activating T-cell anti-tumor effect via unique pattern of antigen presentation.135 The development of B cell-based immunotherapeutic strategies via B cell activation performs effective antitumor responses which might be attractive in future clinic application.136,137 These B cell-based strategies are classified into activating cytotoxic B cells and/or inhibiting downstream immunosuppressive pathways, leading to antitumor immune responses.138,139 GABA is an important inhibitory neurotransmitter in CNS, and the GABA receptors have been clearly identified in multiple tumor tissues exerting regulative effects in cancer progression.140,141 B cells-derived GABA inhibits CD8+ T-cell killing function and enhances the recruitment of macrophages that secret IL-10, leading to anti-tumor immunosuppression mediated by the GABA receptor.142 Considering the regulatory role of B cells on neural signaling, it would be possible to forge B cells to adjust neuron function to remodel tumor microenvironments to ultimately inhibit cancer development.
Myeloid-derived suppressor cells (MDSCs)
Myeloid-derived suppressor cells (MDSCs) have the ability to significantly inhibit immune cell responses. Adrenergic signaling stress drives the immune suppressive activity of MDSCs mediated by β2 adrenergic receptor through decreasing glycolysis and increasing oxidative phosphorylation and FAO.143 Norepinephrine from adrenergic nerves also target to β2 adrenergic receptor on CD8+ T-cells and inhibit the expression of glucose transporter GLUT1 to suppress T-cells glycolysis and activation.144 Current link between neuro-tumor mostly through direct or indirect (via immune cells) effects of neurohormones/transmitters on cancer cells.145 It is highly agreed that the nervous system and peripheral organs exert metabolic regulation.
As described earlier (section “CGRP”), CGRP is released from nociceptor neurons, which directly induced the exhaustion of cytotoxic T-cells to impede their immune capacity to eliminate melanoma cells.4 Similar neural pathways have mentioned that stress-activated ventral tegmental area in brain regulates sympathetic innervation of the bone marrow, to alter the functional profile of MDSCs, and suppress melanoma and lung cancer growth.146
Recent studies have demonstrated that leukemia inhibitory factor (LIF) and Galectin-3 (Gal3) are elevated in the plasma of patients bearing different types of cancers, and these two cytokines can activate specific brain regions after being secreted by cancer cells.147 There may be a potential communication mechanism within the CNS in both peripheral cancers and intracranial tumors. Blockade of LIF and Gal3 or sympathetic nerves significantly inhibits MDSCs production and assume an antitumor immune response. The mechanism may involve peripheral cancer-initiated signaling connecting to the CNS. However, the causes of increased levels of LIF and Gal3 in peripheral cancers are still unclear. There may also be potential signaling molecules that help cancer cells sense the presence or absence of nerve and its signals in the microenvironment.
Cancer-associated fibroblasts (CAFs)
CAFs are a heterogeneous population of tumor stromal cells.148 In many types of tumors, dense extracellular matrix deposition inhibits immune cell infiltration through physical and chemical barrier action, creating an immunologically advantageous microenvironment. At the same time, the extracellular matrix components are changed into the form of type I collagen, which forms a super-large molecular polymer that promotes blood vessel growth and helps the migration of new blood vessels and nerves. CAFs can secrete and produce several growth factors, cytokines and/or chemokines, as well as exosomes to cross-link stromal cells and cancer cells.149,150 While increased extracellular matrix density early on helps prevent immune cells from entering, in advanced cancer, matrix metalloproteinases are secreted to degrade the extracellular matrix, allowing cancer cells to migrate and spread, known as collagen remodeling. Noradrenaline signaling acts on CAFs and promotes the type I collagen and secretion of MMPs to remodel the ECM and support tumor cell invasion.151,152 In pancreatic cancer, adrenaline signaling can enhance the expression of MMPs in stromal cells and promote the neural invasion of pancreatic cancer cells.153 This conclusion was also confirmed in the breast cancer model.154 Among them, beta-adrenergic blockers can save malignant progression. Schwann cells, one of the most important cell types in PNS, have important function in injured axons repair and new axon regeneration as well as tumor progression. Schwann cells activate the downstream STAT3 pathway to directly promote pancreatic ductal carcinoma cells perineural invasion through plasma membrane proteins (NCAM1) and secretory proteins (L1CAM and TGF-β). Schwann cells secrete chemokine to recruit various cell types via extensive communications with inflammatory macrophages fibroblasts and mast cells, promoting PNI and immune escape in PDAC.155,156 Schwann cells can drive tumor cells and CAFs in the pancreatic ductal adenocarcinoma microenvironment to achieve more malignant subtypes: basal-like and inflammatory CAFs, respectively.155 It means that refining Schwann cells to forge CAFs can be promising in obstructing the cancer cell proliferation and migration.
Collectively, in the field of tumor neuroscience, the immune microenvironment plays an indispensable role; and the research on neuro-immune direction is a heated area for tumor treatment, given the fact that it has more scientific supports than other fields discussed up to date.
Inflammation
Chronic inflammation has been proved to be one of the main causes of tumorigenesis and malignant progression, which strongly modulates the body’s immune responses and leads to anti-tumor treatment immunosuppression.157,158 LCN2 is central to induce neuroinflammation, functioning in the innate immune system, which acts as part of the inflammatory process to combat bacterial infections. Signals secreted from the primary tumor into the blood stimulate the proinflammatory activation of astrocytes in the brain. Astrocytes promote the recruitment of myeloid cells from the bone marrow (granulocytes) to the brain, and they in turn become the primary source of LCN2 signaling. The level of LCN2 in the blood is closely related to an existing tumor, and high LCN2 levels signal a trend of brain metastasis and poor survival.159 Some anti-inflammatory drugs can significantly reduce the incidence of cancer and the risk of death, and some proinflammatory cytokines or stimulants can promote the infiltration of immune cells into infected tissues, thereby significantly improving the efficacy of cancer treatment.160 Non-specific anti-inflammatory drugs can inhibit chronic inflammation in the early stage, thereby hindering the occurrence of tumors and increasing the sensitivity of tumor cells to treatment. For certain specific inflammatory mediators produced during tumor development and treatment, targeted inhibition of inflammatory signaling pathways may be helpful to improve the effect of anti-cancer therapies such as chemo/radiotherapy and immunotherapy.
Chronic stress
Chronic stress is widely considered one of the key promoters of cancer progression, especially in the context of neuro-immune-tumor interaction, which profoundly affects immune system function by activating the sympathetic nervous system and the hypothalamic-pituitary-adrenal axis, thereby promoting cancer occurrence, development and treatment resistance.
Chronic stress can induce stress-related hormones, catecholamines. Norepinephrine can stimulate the expression of hypoxia-inducible factor-1α, which is associated with VEGF secretion and tumor angiogenesis for metastasis.161 Using propranolol and galunisertib (Ly2157199) can block norepinephrine-stimulated cancer cell migration and invasion.161 In vivo, chemical consumption with 6-OHDA can reduce the release of catecholamines in tumor tissues, and inhibit the function of M2 macrophages, inhibit tumor growth by reducing tumor neovascularization. In vitro, catecholamine treatment can induce M2 polarization of macrophages and up-regulate VEGF expression, thus promoting tumor angiogenesis.162 At the same time, the adrenergic receptor antagonist propranolol can reverse the tumor-promoting effect of catecholamines under stress. A study uses an in-situ mouse model to simulate the complex interactions between pancreatic tumor cells and their microenvironment.152 in vivo optical imaging has been used to noninvasively track the growth and spread of primary pancreatic cancer. Stress-induced nerve activation increased the growth of the primary tumor and the spread of tumor cells to the normal adjacent pancreas, along with increased expression of invasive genes.152 At the same time, activation of β-adrenergic signaling induced a similar effect to chronic stress, and β-blocking reversed the effect of chronic stress on pancreatic cancer progression. These findings suggest that neural β-adrenergic signaling modulates pancreatic cancer progression under stress.
In addition, stress also increases the expression of MMP-2 and MMP-9 in tumor and stromal cells, prompting tumor cells to invade neighboring tissues.152 Recent work has shown that MMP-9 expression may drive neuronal damage in patients with colorectal cancer.163 In addition, in the inflammatory response of colorectal cancer, both the expression of MMP-9 and the expression of the pro-inflammatory factor IL-6 are upregulated, which may indicate that there are similar activation pathways in chronic stress and inflammatory response, leading to the spread of cancer cells and peripheral neuropathy, manifested by tumor metastasis and aggravated nerve pain.163 Chronic stress significantly increases the concentration of norepinephrine in serum and the expression of tyrosine hydroxylase in bone marrow. Norepinephrine activates the expression of pro-inflammatory factor IL-6, thereby activating the JAK/STAT3 signaling pathway, increasing the proportion of MDSCs in tumor tissue, and promoting breast cancer metastasis.164 β2-AR signaling modulates the expression of immunosuppressive molecules, such as arginase-I and PD-L1 and alters their ability to inhibit T-cell proliferation. The use of β2-AR antagonists can reduce MDSCs-induced immunosuppression.165
Chronic stress is also a predictor of recurrence in cancer patients. Norepinephrine can activate the ADRB2-CAMP-KA-CREB pathway to promote CLOCK transcription, leading to cancer stemness and resistance.166 Targeting this pathway, reducing the release of norepinephrine and inhibiting CLOCK expression, can counter the resistance of KrasLSL-G12D/WT lung cancer to olanzapine, an antipsychotic drug.
Psychological depression, as one kind of chronic stress, has been shown to dysregulate the immune system and promote tumor progression.133 Neuropeptide Y released from norepinephrine-treated Myc-CaP cells promotes macrophage trafficking and IL-6 releasing, which activates STAT3 signaling pathway in prostate cancer cells, thus remodeling the prostate cancer microenvironment.133 Clinical specimens from patients with prostate cancer with higher score of depression revealed higher CD68+ TAM infiltration and stronger neuropeptide Y and IL-6 expression. These suggest that chronic stress is a contributing factor in cancer, but detrimental to survival.
Therapeutic indication of cancer neuroscience
Discoveries in cancer neuroscience will optimize and even revolutionize oncological treatment.6,167 Targeting the interactions between the nervous system and cancer cells is most likely to become a supportive or alternative approach for oncological therapy, together with traditional therapeutic methods, including surgery, radiation, chemotherapy and immunotherapy.168,169,170 Neural signaling receptor or neurotrophin signaling pathways inhibition exhibits characteristics to either improve outcome and disease-free survival or provide immediate therapeutic benefit.171,172,173
Denervation strategies via blocking β-adrenergic receptors show potent effects in preventing metastasis.8 Surgical or chemical severing of sympathetic adrenergic nerve can inhibit the occurrence of prostate cancer, and blocking parasympathetic cholinergic nerve signal can reduce the spread of prostate cancer cells.174 However, different tumor tissues have different ways of innervation, such as secretory gland tumors (stomach, pancreas, and breast), and the degree of innervation is also different. Targeting neural input signals provides a promising approach for some specific tumors, but it also needs to be further explored in other tumors. For future efficient therapeutic exploration, it is crucial to decipher specific tumors-innervation crosstalk. However, a vital limitation of this approach lies on that the innervation of different tumor tissues may be specific, including the specificity of nerve species and the specificity of the degree of nerve signaling, as mentioned in a review written by Mancusi and Monje.15 In the era of targeted therapy, radiotherapy and chemotherapy combined with adjuvant treatment strategies, such as beta blocker, can provide a highly synergistic approach to controlling cancer progression. Considering the adverse effects of cancer in influencing the nervous system, such as seizures and nausea,175,176,177 improving the normal physiological activities of the body, such as sleep, anxiety and depression, and pain sensation, is also a valuable treatment choice.
It has long been suspected that biological behaviors, such as stress, depression, and social support, influence cancer development and disease progression, and this has indeed been demonstrated by research, and the molecular mechanisms of these effects are currently being explored.161,178 As we discussed above in the molecular mechanisms, recent findings in laboratory models suggest that biological behaviors can directly influence the functional activity of cancer cells through the neuroendocrine system. Stressful personalities, poor coping styles, negative emotional responses, and poor quality of life are associated with higher cancer incidence, poorer survival, and higher cancer mortality.
In order to visualize the existing therapeutic drugs, we summarized the current research status of neuro-drugs in anti-tumor in Table 3. Altogether, we believe that from the perspective of cancer, discussing the participation degree of cancer-related characteristics (gene expression patterns, morphological skeleton changes) in neuro-tumor interaction will help generate feasible research directions.
DNA level
Some oncogenes can act as important components-mediated neural signaling functions. A remarkable result showed that the recurrence of glioblastoma is related to B-raf proto-oncogene (BRAF) kinase activation and Wnt/PCP pathway, showing a molecular transition to neuronal state.106 Inhibition of the proto-oncogene BRAF significantly inhibits the transformation and migration of neuronal features in recurrent tumor cells.106 We will discuss that there may be other proto-oncogenes/suppressor genes that regulate the module state transition to neurons (neurodevelopment, synaptogenesis, etc.) and altering their DNA levels may contribute to innovative cancer treatments.
Myc
Myc is one of the well-characterized proto-oncogenes encoding nuclear transcription factors and includes three types: c-Myc, n-Myc, and l-Myc.179 Myc mediates multiple biological programs primarily as a transcription factor regulating 1000 gene expression (Fig. 7).180,181,182
Myc-centered network in cancer neuroscience. a Myc activation in cancer at genetic, transcriptional, and post-transcriptional levels. Chromosomal translocations and genomic amplifications promote Myc expression. The alternation of upstream signaling pathways regulates Myc increased or decreased expression. Serine 62 (S62) preferential phosphorylation on Myc protein prevents Myc degradation from threonine 58 (T58) and promotes stabilization. b These hallmarks regulated by MYC work together to drive cancer malignant progression, participating in controlling proliferation, differentiation, metabolism, dormancy, senescence, protein and ribosomal biosynthesis, apoptosis, autophagy, and so on (The arrows represent induce, and the horizontal lines represent inhibit)
Myc has oncogenic effect on nervous system-derived tumors, such as neuroblastoma and GBM,183,184 whose effects are not limited to these tumors but involved in development and repair of neurons and glial cells. In the context of nerve injury, specific overexpression of c-Myc in microglia induces microglial proliferation and neuropathic pain.185 In the injured retinal ganglion cells and peripheral nerve, both mRNA and protein levels of c-Myc are transiently upregulated to promote axon and nerve regeneration.186,187 From these data, c-Myc may have the potential for regulating neuroinflammatory homeostasis and repair, not just limited to cancer progression.
c-Myc acts as a component of Yamanaka factors can induce somatic cell reprogramming into iPSCs, and also n-Myc and l-Myc.188,189 Inducing differentiated mature or aged neurons back to a younger state through cellular state reprogramming is feasible, and the factors are also pluripotent and direct reprogramming factors, including c-Myc.190 Hence, Myc can act as a bridge for linking nervous system to cancer. Myc drives the alternation of small cell lung cancer subtypes progression along a single evolutionary trajectory by activating the Notch signaling pathway and then inducing neuroendocrine dedifferentiation.191 In orthotopic xenograft and Myc-driven genetic mouse models of prostate cancer, the expression of β2-adrenergic receptors of adrenergic signaling pathway on tumor stroma endothelial cells decreases oxidative phosphorylation, thus promoting angiogenesis in tumor progression.44 In a Myc-driven prostate cancer mouse model, doublecortin (DCX+) neural progenitors infiltrate and reside in prostate tumor tissue. These DCX+ neural progenitors subsequently generate new adrenergic neurons to promote tumor growth and metastasis.11 The density of DCX+ neural progenitors is strongly associated with the aggressiveness and recurrence of prostate adenocarcinoma. These findings underscore the multifaceted roles of CNS in both cancer biology and neuroscience, positioning oncogene-driven tumor and nervous system as a key player in the interface between these two fields.
Myc signaling targeting may be a potential therapeutic approach for cancers, but its trials are not quite successful due to the loose structure of MYC protein lacking suitable domain pockets for binding.192 Myc also drives embryonic development, tissue repair and chromosome remodeling in the nucleus, while abnormal regulation of Myc can promote malignant transformation and reprogram adult cells to a “reset” stem cell state.193,194,195 This makes targeting Myc extremely difficult due to its important noncancerous regulatory function in normal cells and tissues.
Ras
Ras is the most frequently mutated oncogene family found in numerous cancer types, with three isoforms K-Ras, N-Ras, and H-Ras.196,197 More recent studies revealed new druggable sites in the switch-II pocket of KRAS, offering a new potential direction for the development of inhibitors by directly binding to Ras.198,199 The overactivation of the RAS-MEK pathway can drive various clinical manifestations of the genetic syndrome neurofibromatosis type 1 (NF1).200 Blocking the downstream signaling pathway of Ras by MEK inhibitors has been shown to reduce the size of NF1-related nerve-derived tumors, such as neurofibromatosis, plexiform neurofibromas, and malignant peripheral nerve sheath tumor.201 Similarly, other inhibitors of downstream of Ras, such as mTOR inhibitors, have both the anti-tumor effects in tumor progression and emotional and cognitive improvement on hippocampal neurons (such as reducing depressive-like behavior) without causing tumor regression.202 These information suggests that mTOR inhibitors play different roles in tumors and nerves, highlighting the complexity of targeting Ras signaling pathways. Therefore, it may be difficult to find a drug that can act on both peripheral and intracranial tumors from peripheral chemotherapy drugs.
The Ras family also possesses a precise control during the whole stages of axonogenesis. This involves the dynamic alternation of actin and microtubule cytoskeletons, the interaction between growing axon and cells or extracellular matrix, and the transport of related protein.203 Oligodendrocyte myelination through multiple signaling pathways, such as PI3K/Akt/mTOR, Erk1/2-MAPK, and Wnt/β-Catenin, is responsible for protecting axons.204 Members of the Ras family, such as R-Ras1 and R-Ras2, are revealed as intermediaries to link myelination pathways.204,205 The above studies suggest that Ras may regulate the generation or differentiation of neuro-tumor synapses.
FMS
FMS (known as colony-stimulating factor receptor, CSFR, CSF-1R), which is activated by its ligands, CSF-1 and IL-34, is a type III tyrosine kinase receptor as a membrane-bound enzyme, which was initially described as an oncogene.206 FMS participates in myelinogenesis via regulating the activation of microglia, which are the most important immune cells in CNS.207 Under the conditions of NMDA-induced neurotoxicity and teratogen cyclophosphamide, increased expression of FMS causes microglia activation, cytokines release, enhanced phagocytosis, and migration to injured neuronal cultures, reducing neurotoxicity and teratogen-induced injury, preventing neurons from injury.207 Using FAM inhibitor can lead to microglia/macrophage depletion and repopulation to reduce neurodegeneration and foster synapse recovery after traumatic brain injury.208 Similar to the results discussed in c-Myc, neuroinflammation after nerve/brain injury shows changes in oncogene expression, which in turn regulates the activation and migration of microglia, and synaptic production. Glioma cells tend to migrate toward glioma-associated microglia/macrophages-conditioned media activated by GM-CSF in which CCL5 (inflammatory mediator produced by immune cells) was abundant.209 Most evidence supports that FMS (CSFR) -CSF axis is involved in tumor development and treatment, as well as neural development and repair pathways in the form of intermediates. It can also be seen that in the current summary of Myc and FMS oncogenes, neuroinflammation with microglia activation has a wide range of oncogenes regulating synaptic production, either as initial regulation or in the form of activation intermediates. This observation prompts further consideration of the mechanisms underlying neuro-tumor connection.
HER2
The β-adrenergic signaling pathway stimulates a variety of cancer-causing signaling pathways, including those of Src and HER2 (encoded by ERBB2). In HER+ cells, the β2-adrenergic receptor and HER2 form a positive feedback loop in human breast cancer cells, and the β-adrenergic receptor activates the signal transductor and transcriptional activator 3 (STAT3), which in turn activates the ERBB2 promoter to stimulate gene transcription.210 Catecholamines effectively antagonize the anti-proliferation effect of trastuzumab both in vitro and in vivo. Trastuzumab-resistant dependent PI3K/Akt/mTOR pathway is controlled by catecholamine-induced β2 adrenergic receptor activation. The β-blocker propranolol not only enhances the anti-tumor activity of trastuzumab, but also re-sensitizes resistant cells to trastuzumab.210
Moreover, β-adrenergic receptors mediate norepinephrine to induce SRC-S17 phosphorylation via the ADRB/cAMP/PKA axis and further activate Y419 phosphorylation, leading to tumor growth in breast. In human ovarian cancer samples, high tumor norepinephrine levels are associated with high levels of pSrcY419.211 Catecholamines stimulate the activation of ADAM10 expression and proteolytic activity, and catecholamines induce the activation of gamma-secretase, resulting in abscission of ADAM10 from the HER2 extracellular domain and subsequent intramembrane cleavage of the HER2 intracellular domain by presenilin-dependent gamma-secretase. Nuclear translocation of the HER2 intramembrane domain and transcriptional enhancement of COX-2, a gene associated with tumor metastasis. Nuclear localization of HER2 is significantly associated with overexpression of β2-adrenergic receptor in human breast cancer tissues, which plays a decisive role in tumor metastasis.212
Of these, ADAM10, a transmembrane zinc-dependent protease, has the function of proteolysis and can cleave a variety of transmembrane proteins (such as Notch, E-cadherin, HER2, VEGF, etc.) to regulate tumor-related signaling pathways. The high expression of ADAM10 is closely related to the enhancement of breast cancer metastasis and invasion ability and the decrease in the survival rate of patients.213 To target ADAM10, inhibition of HER2 cleavage by the ADAM inhibitor INCB7839 may enhance the clinical efficacy of trastuzumab in HER2+ breast cancer patients.214 As discussed above, ADAM10 promotes glioma cell proliferation and drug resistance.1 In addition, ADAM10 is screened as a target for adavivint (SM04690) to specifically regulate NOTCH2 protein expression, which mediates transcriptional regulation of the Wnt pathway. NOTCH2 directly activates transcription of TCF7L2 and Wnt target genes, such as MYC, JUN, and CCND1/2, rather than the downstream transcription factor critical for classical Wnt/β-catenin signal transduction, such as TCF7-like 2 (TCF7L2).215 ADAM10 inhibitor, GI254023X,216 can reverse anti-PD-1 resistance in human tumor-infiltrating lymphocytes, which necessitates effective clinical treatment.
Rb
Rb is the first identified tumor suppressor gene in retinoblastoma capable of regulating cell cycle, growth, and metabolism. Retinoblastomas are a cancer highly likely initiated by Rb mutation. A study has shown that mouse retinoblastomas has surprisingly high neuronal differentiation in a series of knockout mouse models.217 In these models, early-stage mouse retinoblastoma cells can form synapses and extending neurites, which exhibit similarity as amacrine or horizontal cells of the retina. However, as the disease progresses to the late stage, mouse retinoblastoma cells lose neuronal differentiated morphology, and become more invasive and aggressive. During the transition from early-stage to late-stage retinoblastoma cells, neurites and synapses can construct extensive cell-cell contacts.217 The validation consistent with the results of high neural involvement is that Rb protein was early found to widely express in adult sensory neurons and axons, and its downregulation can promote neuronal regeneration, including neurite growth and branch formation.218 Fully differentiated horizontal interneurons with neurites and synapses can reenter the cell cycle and proliferate to form cancer, while maintaining their differentiated state.219 One characteristic of retinoblastoma is the infiltration of optic nerve.220,221 These facts highlight the close connection between the loss-of-Rb-driven retinoblastoma and nervous system.
NF1
NF1 acts as a tumor suppressor gene, and its mutations are associated with the initiation of optic pathway gliomas derived from optic nerve glial cells.25 As mentioned above, NF1 mutations enhance the aberrant shedding of neuroligin 3 (NLGN3) from the postsynaptic membrane, to promote optic pathway glioma growth. The inhibition of NLGN3 shedding by knockdown or using sheddase inhibitor can impede NLGN3 production and subsequently decrease tumor growth.25 In NF1 mutation-driven optic pathway glioma mouse models, stimulating optic nerve activity can augment optic glioma growth, while reducing optic experience, such as light deprivation, can prevent tumor formation and maintenance.25 This raises a question of whether other sensory experiences besides visual experience, such as smell and taste experience, also have an impact on tumors within the corresponding neural pathways. Olfaction has been shown to directly regulate gliomagenesis.40 They all suggest that elements of functional sensory experience input circuits are key targets for potential treatment of tumor neuroregulation.
Neurofibroma type 1 can arise from any part of the visual system, including the optic nerve, optic chiasm, optic tract, optic radiation, and hypothalamus. Among them, as photoreceptors, rod cells are the first level neurons in the retina, which convert light into electrical signals. Retinal neurons transmit signals to the brain to form visual experience. NF1 mutations are associated with rod cell dysfunction, but there is no clear scientific evidence of a direct link between rod cells and optic glioma, so it cannot be considered as causal relationship. However, as a part of the eye, rod cells can be affected by optic neuropathy. If the optic nerve is compressed or invaded by the tumor, it may influence the function of the rod cells and thus affect the vision. Notably, one previous study revealed that NF1 deficiency can enhance cell proliferation while limit oxidative ATP production and energy flexibility.222 This phenomenon implies that NF1 mutations may indirectly influence rod energy metabolism. Mechanistic targets may also be explored to find link between metabolic properties of rod hypoxia to NF1 mutations. During state transition and totipotency acquisition in pluripotent stem cells, mitochondrial TCA cycle enzymes are translocated to the nucleus.223 If the translocation of mitochondrial TCA cycle enzymes to the nucleus occurs during rod development, this may provide a new perspective on the “NF1 mutation-rod metabolism-NLGN3 shedding” link. Although the study as mentioned above has shown the neuron development associated with GBM progression, there is limited evidence for the connection of lactate, axon biology, and brain tumor cells, which might be an emerging opportunity for exploring cancer target treatment.
p53
Various targeting therapeutic strategies in response to p53 expression levels have been used to regulate cancer cell cycle, energy metabolism, and autophagy, thus prohibiting tumor progression.224,225,226,227 RNA sequencing analysis of exosomal miRNAs shows that differentially expressed genes are highly correlated with neuronal growth, morphogenesis, synaptic formation, differentiation, dryness, and synaptic transmission. The expression of miR-34a and miR-141-5p in the exosomes of p53-deficient oropharyngeal carcinoma is significantly down-regulated, while inhibition of miR-34a and miR-141-5p in the exosomes of wild-type p53 cells could produce a p53-like effect.12 However, the effect of miR-141-5p is weak, suggesting that miR-34a and miR-141-5p in exosomes are involved in neural reprogramming, and miR-34a played a major role. In oral cavity squamous cell carcinoma, loss of p53 results in the secretion of cancer-derived extracellular vesicle (known as exosome) without miR-34a, which not only drives adrenergic neuritogenesis but also induces sensory nerve reprogramming into adrenergic neuron, and the adrenergic neuron-derived signals further promoting cancer cell growth.12,228 In vivo studies have shown that adrenalin secretion can be detected after injection of p53-deficient exosomes into tumors, suggesting that p53-deficient exosomes can induce neural reprogramming, and that the knock-out of Rab27A and Rab27B can inhibit this process. Rab27A and Rab27B are key proteins released by extracellular vesicles and are involved in intercellular communication and tumor microenvironment regulation. Interfering with these proteins can impair vesicle transport and selective payload, inhibiting cancer phenotype deterioration. The communication between neurons and p53 is involved in regulating synaptic plasticity, axon outgrowth, and neuron development.229 In addition, chronic stress- epinephrine decreased the stability of p53 protein by promoting p53 ubiquitination. It is suggested that the increase of sympathetic nervous system activity may promote tumorigenesis or chromosomal instability.230 Long-term activation of adrenaline signaling leads to ARRB1/AKT-mediated activation of MDM2, an E3 ligase, thus driving DNA damage and inhibiting p53 level.231 It is worth noting that nerve distribution varies significantly according to the physiological conditions of different tissues and cells, and the distribution of adrenergic receptors in different cancer cells also has its own characteristics. Considering the facts that current cancer treatment of targeting p53 is through stabilizing or preventing the degradation of p53 protein via acetylation, de-ubiquitination, and/or non-coding RNA, exploring the neurotransmitters and certain electrochemical signaling to regulate p53 stability and further inducing the death of neuron which is hijacked by cancer cell to inhibit tumor growth might be a direction for cancer treatment.
DNA therapeutics
Current understanding suggests that the mutation or loss of several tumor suppressor genes regulated by neural signals can influence neuron to glioma synapses, assuming that even no breakthrough can be made in inhibiting the malignant progression of tumors, researchers can look into its important regulatory role in development and differentiation. The combination of drugs targeting oncogene signaling with neural regulators may have potential applications in cancer treatment.
Using the CRISPR/Cas system, one can precisely modify the genomes of cancer cells and neurons, which helps to study specific signaling pathways as a current laboratory routine.232,233 In particular, CRISPR allows researchers to develop more accurate models of cancer and explore how tumors interact with the nervous system. For example, studies have found that certain neurotransmitter receptors or genes associated with tumor growth are mutated, which can cause tumors to become resistant to treatment. Given Cas9-induced safety risks, researchers need to pay special attention to long-term safety of gene therapy in the body.232
Notably, the study of intracranial tumors based on genomics and epigenomics should pay attention to the specificity of tumor type, and potential relationship between neuron origin and malignant molecules.49 The complexity of interactions between cancer cells and nervous system is the most significant challenge. The technological advances and innovations in genomics are mind-blowing.234 By combining information about neuronal signature proteins, it is possible to gain neuroregulatory preferences in pre-cancer, metastatic, and advanced stages, which will help us find ways to crack tumor metastasis and drug resistance. In addition, certain cancer types may have specific gene expression during metastasis, which has been demonstrated in several studies.235,236 Blood and cerebrospinal fluid (CSF) are the most commonly used biological fluids to collect circulating cells and biomolecules of tumor origin. There is great potential that in combination with cerebrospinal fluid biopsies and single-cell gene sequencing will help more quickly obtain markers of relevant neuro-tumor communication, which may contain some cancer therapeutic targets through gene regulation or editing.
RNA level
In addition to genetic sequence mutations, it is crucial to pay close attention to epigenetic modifications that contribute to regulating carcinogenesis.237 The new finding identifies regulating bromodomain-containing protein 8 that occupies p53 target loci by enforcing a compact chromatin conformation to prevent p53 activation and promote GBM proliferation in a GBM-specific epigenetic mechanism.238 Epigenetic modifications, including those mediated by RNA-binding proteins (RBPs), noncoding RNAs (ncRNAs), and RNA modifications, play a crucial role in regulating gene expression and determining tumor cell fate, making them significant contributors to carcinogenesis. The epitranscriptome, an emerging component associated with human diseases, embeds RNA modifications that regulate RNA molecule activity. The RNA modifications have been proven to present in multiple RNA types, including coding RNA and non-coding RNA, all of which tightly link to carcinogenesis.239 A few reports have described the relationship between neural cells and tumor cells at RNA level during cancer malignant progression by single-cell RNA-seq.240,241 On the one hand, the developmental origin of ependymoma, a central system tumor, has been linked to neuronal-glial fate;240 and on the other hand, chemosensitivity of peripheral tumor cells and expression of ion signaling channels, are neuron-sensing Ca2+-influx channels.241 It is reasonable to understand that cell differentiation, axon formation, and ion transport channels in the nervous system may be important parts of the discussion of cancer neuroscience. There are great mysteries in the transcriptome, which serves as a worthwhile junction between cancer and neuroscience. We would discuss this in more detail below with a potential link for RNA-based therapy of tumors.
mRNAs and noncoding RNAs
Increasing studies have proven that aberrant mRNA translation widely participates in cancer initiation and progression.242,243,244 The treatment of tumors at the mRNA level is mostly immune direction, which control the expression of immune checkpoint molecules by the precise regulation of mRNA translation, to modulate the generation and presentation of aberrant peptides antigen, and affect the functional differentiation of tumor-associated immune cells.245,246,247,248,249,250,251 The toxicity and immune-related side effects of immune checkpoint inhibitors (ICI) can be seen in any organs, including the nervous system, providing the link between RNA and immunity to neuronal cancer. Toxic side effects on the nervous system include meningitis, neuroinflammation (peripheral nervous system myositis), and peripheral neuropathy.252,253 Alternation of the expression of neural signaling molecules that we discussed above in Section 2 would have a promoting effect on the cancer cells’ progression. Although they may directly function in the protein form, such as BDNF, NLGN3, β-adrenergic receptor, etc., they may produce similar effects through their counterparts if they can be inhibited at the mRNA stage, thereby inhibiting neural-tumor communication. However, our discussion is very limited by the fact that the major studies have not yet addressed the mechanism at the RNA stage. The histone methylation of BDNF is regulated by lysine demethylase 5C (KDM5C).254,255 In Alzheimer’s disease, KDM5C binds to a repressive element in the BDNF promoter region and reduces the nearby histone H3 lysine 4 di- and tri-methyl modification, thereby suppressing BDNF transcription.254
Only less than 2% of the human gene sequences can code proteins, while 90% are transcribed into RNA.256 Non-coding RNAs do not encode proteins but play essential roles in a variety of processes, including modulation of gene expression and responses to cellular pathway.257,258
MicroRNAs (miRNAs), a class of non-coding RNA molecules with a length of about 200 nt, regulate gene expression at the post-transcriptional level and participate in multiple biological processes (Table 4).259 miRNAs play an unprecedented role in nervous system development in which they can impact neuronal regeneration. TDP-43 is an RNA/DNA binding protein, which is critical for the development of neurodegeneration, and a number of TDP-43-associated miRNAs are involved in the progression of human cancers, such as lung cancer and triple-negative breast cancer.260,261 miR-34a in exosomes from p53 wild type cells inhibits the alternation of neuron and nerve reprogramming which further inhibits oral cavity squamous cell carcinoma cells survival.12 In brain tumor, miR-155 coated by virus-mimicking nucleic acid nanogel can reprogram microglia and macrophages that are infiltrated in GBM tumor microenvironment from tumor-supporting into anti-tumor phenotype and consequently inhibit tumor growth.262 These results prompt us to speculate that exploring miRNA regulation of neuronal differentiation, synapse formation and neurite generation might provide new therapeutic opportunities for intracranial tumors, such as targeting neuronal tumor synaptic marker molecules or tumor cells by designing exosomes coater by the membrane of microglia.
It is also worth elaborating on the types and splicing patterns of miRNAs in intracranial tumors, such as whether there are homologous sequences or similar splicing patterns between oncogenic/cancer-promoting miRNAs and neuronal differentiation-promoting miRNAs. Microglia and macrophages accumulate in GBM but not induce anti-tumor inflammation reactions.263 Of these, the anti-tumor capability of microglia is inhibited by epigenetic silencing with histone modifications. Considering the PNI characteristics of tumor cells, a previous review has drawn a conclusion of non-coding RNAs playing a major role in injured peripheral nerve repair.264 Given that the synapses can be mediated by NMDAR between breast-to-brain cancer and neurons, miRNA might drive breast cancer to undergo brain metastasis. However, whether different species or expression levels of miRNAs regulate the whole process of metastasis, and how the generation of synapses varies at different stages of metastasis require further exploitation.
Long non-coding RNA (lncRNA) with more than 200 nt as length, plays a major role in tumor-supporting and -suppressive effects265,266,267 (Table 4). Mechanistically, the regulatory locus of p53-induced lncRNA-p21 transcription activating the cis-regulation depends on the conserved regions in exon 1 of lncRNA-p21.268 lncRNAs can also direct protein binding partners to regulate gene expression to modulate neuronal development and function. lncRNAs control neuronal development via m6A modification and axon elongation via regulatory protein.269 Axon-enriched lincRNA ALAE binding with KHSRP can maintain Gap43 mRNA translation to locally enhance axon elongation of dorsal root ganglion neurons.270 This function of regulating synapse and axon elongation by lncRNA is important for neuro-glioma synapse formation and communication.
RNA-binding proteins
RNA-binding proteins and RNA modification involved in RNA splicing and translation are particularly relevant in neurogenesis and neuronal function as well as nervous system development.271,272 IGF2BP proteins, including IGF2BP1, -2, and -3, are well-known RNA-binding proteins that embody markedly important function in distinct aspects of oncogene expression associated with carcinogenesis, cell cycle, metastasis, metabolism, drug resistance, and relapse.273,274,275 IGF2BP protein is involved in the malignant process of cancer276,277,278 and also plays an important regulatory role in the nervous system. IGF2BP proteins act as key regulators in neuronal development, including synaptic terminal growth, neural cells differentiation and migration, neurogenic potential alternation, axons growth, dendritic branching, and nerve regeneration.279
Splicing factor polypyrimidine tract-binding protein 1 (PTBP1), acting as an RBP together with paralog PTBP2, controls the event of neural differentiation. The splicing co-factor SON that has the highest expression level in GBM can upregulate PTBP1-mediated oncogenic transcript splicing and suppress RBFOX2-mediated PTBP2 pro-neuronal splicing by forming a complex with hnRNP A2B1 in GBM.280 The RBP-mediated program markedly reduces GBM cell growth and eradicates the stemness of glioblastoma stem cells, while SON knockdown can significantly inhibit GBM cells growth as well as suppress intracranial tumor growth.280 Fragile X mental retardation protein (FMRP), a neuronal RNA-binding protein, controls synaptic plasticity in neurons via glutamate-stimulated NMDAR signaling and broadly governs protein translation and mRNA stability in multiple human solid tumors.5,281 The expression of FMRP promotes the secretion of cytokines to induce Treg cells and represses chemoattractant C-C motif chemokine ligand 7 to prevent CD8+ T-cells attack, ultimately achieving cancer immunosuppressive effects.282 High expression of RNA-binding protein SERBP1 is prevalent in GBMs, which correlates with poor survival of the cancer patients. SERBP1 regulates serine and one-carbon unit cycle metabolism to control methionine production, which is associated with genome methylation, further inhibiting the expression of genes associated with neurogenesis, synaptogenesis, and neuronal differentiation while promotes GBM stem cell phenotypes and radiation resistance. The function of SERBP1 that acts as a bridge linking metabolism and epigenetic regulation emerges as potential GBM therapeutic target.283 ERα, a receptor of hormone estrogen and also recognized as a novel RNA-binding protein, controls oncogenic mRNA alternative splicing and translation processes to promote breast tumor cells survival, preventing cells from multiple stressors like ER-stress, immune attack and so on.284
RNA-binding proteins are valuable biomarkers for a wide range of gene expression processes, and their regulation of certain oncogenes may have applicability to a class of cancer types driven by these oncogenes. However, RNA-binding proteins are nonenzymatic molecules that lack hydrophobic pockets capable of binding small-molecule inhibitors. Therefore, whether these proteins can become effective targets is yet to be explored.
RNA modifications in nervous systems
RNA modification (epitranscriptomics) plays a key role in nervous system development, neuroplasticity and tumorigenesis by dynamically regulating gene expression. For clinical applications, DNA methylation has been used to classify CNS tumors.285 RNA modification plays an important role in the interaction between nerves and tumors (rightly falling into the concept of cancer neuroscience), including the regulation of tumor-related neural invasion, neurotrophic signal transduction, and tumor microenvironment remodeling.
m6A is abundant and modulates various neural functions. m6A promotes translation of target transcripts by YTHDF1 in a neuronal-stimulus-dependent manner, and facilitates learning, memory, and synaptic transmission in the adult mouse hippocampus.286 There is a direct interplay between YTHDF1 and Fmr1 (the fly homolog of FMRP) to inhibit the translation of axonal growth transcripts, thus suppressing axonal growth.287 m6A eraser FTO is enriched in axons, which decreases m6A modification of GAP-43 mRNA and promotes its translation, thus giving rise to axon elongation to achieve neuronal development.288 m6A modification of lncRNA Dubr regulates the axonal elongation and migration of dorsal root ganglion neurons by stabilizing the YTHDF1/3 complex.269 The antagonistic function between YTHDF1 and FTO has been noticed during axon growth and development. It is worthy to explore the function between YTHDF1 and FTO in neural remodeling in the tumor tissue. Possibly, the growth of axons is perhaps controlled distinctly in different damage conditions or in different recovery mechanisms, or it may be due to tissue-specific existence, and all of these require further investigation to lead to some therapeutic clues. Based on a variety of database analysis, m6A is highly associated with the malignant development of head tumors.289 It is possible that tumor cells mimic this RNA modification mechanism to hijack the mechanisms of neural development and form neural infiltration and neural-tumor synapses, which promote tumor cells survival and invasion.
Higher m1A modification level has been observed in the major ganglia of simple defensive reflex trained animals than naïve animals, and correlated with increased polyglutamine synthesis and excitability in neurons.290 It indicates that RNA modifications may have the capability to establish synapses circuit to learning.
Ψ modified neurotrophin-3 (NT-3) mRNA is delivered into primary Schwann cells to enhance dorsal root ganglia neurite extension,291 indicating its potential capability of improving peripheral nerve regeneration. In the neuronal nuclei of the Drosophila brain, numerous RNA editing sites exist, each exhibiting distinct characteristics across different neuronal populations.292 These sites vary in editing levels for specific transcripts and often present as unique combinations of multiple editing sites. There may be a complex targeted regulation of editing levels in key transcripts, such as regulating the differentially expressed RNA-binding proteins and other regulators as well as molecular mechanisms.
To date, there is still limited information available on the role of RNA modifications in the progression of the nervous system-tumor axis, and the underlying signaling pathways are currently poorly understood.
RNA therapeutics
In view of the unparalleled characteristics of RNA, manipulating RNA biology can show great advantages within the scope of accessibility. RNA can induce protein expression process more rapidly than gene sequence. Meanwhile, RNA-based therapeutics can achieve a local delivery strategy to avoid the invasive body caused by systemic administration.
The structural diversity of RNA enables it to perform diverse biological functions. RNA editing, alternative splicing and RNA modifications expand their functional repertoire. Actually, the mRNA-based genome editing has also been successfully applied to stem cells for many diseases treatments.293 ZFN mRNA reserves multilineage potential and low cytotoxicity compared to TALEN mRNA and CRISPR/Cas9 editing, and such editing approach is used in a large animal model to modulate repopulating cells.294,295,296 The mRNA-based iPSC technology delivers Cas9 in the form of mRNA on the basis of the principle of CRISPR/Cas9 system to promote the production of protein molecules, and iPSCs can further differentiate into specific cell types for clinical application.297,298,299,300 For the current clinical treatment, whether iPSC-induced differentiation can help repair the diseased cells or damaged neurons present in cancer patients, thereby accelerating the recovery of cancer patients.
The limitations of RNA, including instability and off-target efficacy, are current major challenges in clinical application.301 We set out to summarize the central role of implementing mRNA-based therapeutics in potentially promising cancer therapy based on the following basic aspects:
(I) RNA is a promising diagnostic marker and therapeutic target: RNA molecules, whose expression levels change during tumor-nervous system interactions, can be used as potential biomarkers for early detection and prognostic assessment of cancer. Single-cell RNA sequencing technology can be used to analyze RNA profiles in tumor samples to provide specific medical treatment. Next, the tumor-nervous system interaction is blocked by silencing neurotrophic factors (such as BDNF) or neurotransmitter synthases (such as IDO1) by non-coding RNA.
(II) RNA can be a versatile therapeutic or preventive drug: mRNA vaccines, such as those against COVID-19, have demonstrated superior effectiveness in vaccine development. Encodes tumor-associated neuro-antigens (such as GDNF) that activate specific T-cells to clear the neuroinvasive microenvironment.
(III) mRNA-based therapies may have less genetic risk and are easy to deliver effectively: Generally, mRNA-based therapies do not introduce gene insertions, deletions, or mutations, thereby reducing genetic risk. Efficient transfection of mRNA transcripts is often easier to achieve than DNA, and entry into the nucleus is not a necessary step for most mRNA transcripts.
RNA vaccines and drugs
With the rise of cancer neuroscience, it is worth focusing on how RNA therapies can interfere with the interaction between tumors and the nervous system, including inhibiting tumor-associated neural infiltration, blocking neurotrophic signaling, and inhibiting perineural invasion.
Cancer vaccines acting as personalized cancer treatment almost deal with unique composition of mutations in each tumor, and the technology is continuously showcasing great progress towards maturity for clinical application.302,303,304 The development of mRNA-based therapeutics requires the following steps: mRNA design, synthesis, entrapment, pharmacodynamics, pharmacokinetics, safety evaluation, manufacturing, and clinical trials (Fig. 8).305,306,307 Given the characteristics of the cancer-nervous system interaction, it is worthy of discussion that RNA vaccines can intervene in this interaction. We hypothesize the following mechanisms that need to be demonstrated by research: (I) targeting tumor-associated neuro-antigens with vaccines to induce an immune response to clear tumor cells, thereby reducing the negative effects of tumors on the nervous system, (II) the vaccine directly targets nervous system signals in the tumor microenvironment, such as neurotransmitter receptors or related signaling pathways, (III) the above vaccines perhaps bear side effects on the normal nervous system, such as autoimmune reactions and neuroinflammation.
RNA-based cancer therapeutics and the application of in vitro transcribed (IVT) mRNA in antitumor treatment. a RNA therapy includes oligonucleotide therapeutics, such as ASOs, siRNAs, miRNAs, and RNA aptamers, which interact with RNA or proteins to regulate gene expression, and IVT mRNAs that translate into functional proteins to serve as an RNA drug/vaccine. b The components of IVT mRNA contain 7-methylguanosine (m7G) 5′ cap structure, 5′ untranslated region (UTR), open reading frame (ORF), 3′ UTR, and 3′ poly(A) tail. There are RNA structures in UTR, such as pseudoknots and hairpins; sequence binding sites including RNA-binding proteins (RBPs), RNA modification, and non-coding RNA-binding sites. Zip code regulates RNA localization, and CPE regulates RNA stability. ORF codes sequences to control cancer cell fate. c IVT mRNA is coated by delivery carriers and taken up by antigen-presenting cells, participating in CD8+ T-cell activation by MHC class I, and CD4+ T-cell activation by MHC class II. B cells activated by CD4+ T-cells produce antibodies, together with cytotoxic CD8+ T-cells to kill cancer cells (The arrows represent induce, and the horizontal lines represent inhibit)
The protein encoded by mRNA vaccine is regarded as an antigen after injecting into antigen-presenting cells, then activates antigen-specific cytotoxic immunity by CD4+ T-cells/CD8+ T-cells and also humoral immunity by B cells to achieve noticeable treatment.308,309 For example, DC-based mRNA vaccines have been shown to prolong GBM patient survival.310,311 mRNA vaccines encoding immunostimulants, such as cytokines and chemokines, have shown potential for promoting the maturation and activation of immune cells, thereby improving the tumor microenvironment.312,313,314 In central nervous system tumors, we can target at the level of neural cell differentiation. For example, gangliosides are key proteins in the central system that regulate the growth and protrusion of nerve cells. Among them, GD2, one kind of gangliosides, is highly expressed on the surface of various cancer cells and is a marker of glioblastoma.315 Existing clinical studies have shown that anti-GD2 monoclonal antibody drugs can effectively inhibit central nervous system recurrence of neuroblastoma after first complete remission.316 In addition, induced pluripotent stem cell (iPS) derived natural killer T (IPS-NKT) cells combined with anti-GD2 monoclonal antibody could effectively inhibit the growth of neuroblastoma.315 As potential molecules, mRNA vaccines encoding the GD2 antigen could be explored to determine whether they induce specific T-cell immune responses to eliminate tumor cells. A similar molecule (both responsible in the nervous system and as a tumor marker) is PSMA of prostate cancer cells, and also possesses therapeutic potential worthy of targeting.317
If RNA vaccines disrupt the synaptic connections between tumors and neurons by encoding neuron-related antigens specifically expressed on the surface of tumor cells, such as GDNF and NLGN3, it will be a great advance in RNA vaccines. RNA vaccines targeting glutamate metabolism, such as those encoding xCT transporter inhibitors, can block glutamate-cystine exchange resistance to ferroptosis in tumor cells.318 It also suggests that RNA vaccines targeting glutamate metabolism may reduce the excitotoxic damage caused by glutamate over-release and subsequent excitatory membrane receptor activation in neurons.319 Another valuable application of mRNA-based therapeutics is the delivery of mRNA molecules to regulate neuronal activity.320 Neuronal activity is closely linked to the growth and spread of cancer cells, thus modulating this activity could potentially inhibit tumor growth. However, few studies on the delivery of mRNA-encoded neural signaling molecules to treat tumor development have been documented at present. Ultimately, our understanding of the neural circuit-tumor mechanism is limited, and needs future attention and study.
RNA drugs can also include antisense oligonucleotides (ASOs), RNA interference, CRISPR/Cas systems, and RNA aptamers.321,322,323 miR-10b, a unique oncogenic miRNA with high expression in GBM. miR-10b ASO treatment results in targets’ depression, and inhibits GBM growth and progression.324 The ability of these RNA drugs to elicit emotional responses in the brain is noteworthy, and the method of successful delivery is also worth attention to promoting RNA therapy. Nanoparticle delivery system is also surprisingly suitable for ribonucleoprotein complexes based on CRISPR technology and aptamer for GBM therapy.325,326,327 Especially, CRISPR/Cas13-mediated RNA editing would dominate in RNA drug-based cancer therapy in future.
Notably, the Nobel Prize in Physiology or Medicine in 2023 was awarded for discovery of RNA modification that enabled the rapid development of successful mRNA vaccines against COVID-19, perhaps indicating the great promise of RNA prevention and therapy strategies for many other diseases including tumor and chronic inflammation. RNA-based therapeutics have also been practiced in CNS diseases. In diffuse midline gliomas (DMGs) that are pediatric high-grade brain tumors, 2’-O-methoxyethyl phosphorothioate ASOs have been demonstrated to inhibit tumor growth, enhance neural stem cell differentiation into neurons, oligodendrocytes, and astrocytes, and promote survival.328 TUG1-PRC2 complex inhibits the expression of neuronal differentiation genes, and ASOs targeting TUG1 induce glioma stem cells differentiation and in vivo growth.329 These results indicate that neuroscience plays a big part in RNA-based cancer therapy. If the complete production route of mRNA-based drugs can be generated, it is easy to scale up production through chemical synthesis.
In conclusion, future research needs to accurately screen the key molecules of tumor-nerve interaction (such as neurotransmitter receptors and synaptic adhesion proteins) to avoid unwanted damage to normal nerve tissues and interfere with their functions. At the same time, the blood-brain barrier-penetrating LNP or viral vectors should be developed to improve the delivery efficiency of mRNA in the central nervous system. Thus far, only a few phase I clinical trials (e.g., NCT04573140)330 have evaluated the safety of RNA vaccines in glioma, which may need further investigation with enlarged sample size and/or combination therapies (e.g., immune checkpoint inhibitors, denervated drugs). Since recent technological innovation has enabled mRNA to become a promising therapeutic tool in the field of cancer therapy, with strong confidence, we expect its application in cancer therapy.
Cytoskeletal changes
Mutations and abnormal expression of the cytoskeleton and cytoskeletal-associated proteins potentially regulate cancer metastasis and drug resistance. Catecholamines activate FAK signaling pathway via adrenergic receptors to inhibit ovarian cancer cells anoikis, one kind of cell death modes regarded as a type of apoptosis defined in terms of alternations in the cytoskeleton.331,332 Similar condition in the nervous system, efficient synaptic transmission is accomplished through a series of interactions with intracellular scaffold proteins, transmembrane helper subunits, adhesion proteins, and other extracellular elements.333 Cytoskeletal dynamics also control axon growth during development and axon regeneration.334 Next, we discuss the important regulatory roles of cytoskeleton components in cancer cells and in the nervous system, aiming to encourage breakthroughs in this area by extensively studying molecules with direct or indirect linkage to the cytoskeleton.
Microfilament
Cancer cells often exhibit aberrant actin cytoskeleton dynamics and altered expression of tropomyosin isoforms, which can contribute to their invasive phenotype. F-actin assists TNT-like protrusion growth.335 The actin-supporting TNTs act as open-ended conduits to directly connect cytoplasm of two cells for cargo transportation, including small ions, molecules, as well as large organelles, such as lysosomes and mitochondria.336 TNTs have been found in various cancer types including gliomas and non-brain-derived tumor and promote resistance against cytotoxic therapies, just like the function of tumor microtubules network in gliomas.337 In a recent report, mixed lineage kinase 3 (MLK3) has been shown to remodel actin cytoskeleton via the MLK3-EPS8 signaling pathway to promote GBM cells migration and invasion.338 Actin regulator protein EPS8 has early been proven to inhibit filopodia formation in neuron and promote TNT formation.339 Moreover, the upregulation of proteasomal target EPS8 hyperactivates RAC signaling in neurons, leading to actin cytoskeleton alternation and protein kinase JNK activation, which contributes to decreased lifespan.340 The protuberant-like morphological changes of these neurons should be an important physical scaffold for the neuro-glioma connection and worthy targets for glioma.
Tiam1, one kind of GEF, orchestrates actin cytoskeleton reorganization and synaptic receptor NMDAR stabilization to control synapse structure and plasticity in anterior cingulate cortex neurons.341 Probing whether Tiam1 can modulate neuropathic pain by regulating synaptic connections in the anterior cingulate cortex neurons could provide insights into the mechanisms of pain transmission and potential therapeutic targets. Similar to the role of visual experience in neurofibromas, nociceptive neural pathways may also serve as effective neuro-cancer circuits to suppress cancer. In functional synaptic transmission, receptors must be aggregated before the release site of neurotransmitters, indicating that receptor movement at synapses is a dynamic balance of interactions with synaptic, extra-synaptic, and intracellular receptors.333 The secreted BDNF increases synaptic current strength and thus, scientists evaluated whether the increased capture of BDNF by the diffusion of AMPAR receptors enhances the electrical signal.342 Assessing receptor movement may find a clue to describe the mechanism of neuro-glioma signal transmission. Ttfields, a non-wound damage device on brain surface, produces alternating electric fields with low intensity and medium frequency.7 The electric field force mainly acts on two proteins with high dipole moments (tubulin and septin) in tumor cells to inhibit tumor cell mitosis and DNA damage repair and increase DNA replication pressure. It has already been applied in the clinical treatment of GBM,7 demonstrating the important regulatory role of electrical signals in the brain.
However, receptor movement at synapses is a complex process, and the intracellular receptor recycling process extensively related to actin and molecular motors as well as ATP consumption, which constitute complex and discrete signaling pathways. Exploring how these precise molecular mechanisms by which paracrine signaling molecules (such as BDNF and NLGN3) regulate the synaptic transmission and localization of cytoskeletal proteins may define new therapeutic approaches for gliomas.
Gap junction
Gap junctions in glioma cells play a pivotal role in the communication between tumor cells and the surrounding neuronal environment, amplify potassium-evoked depolarizing currents in response to neuronal action potential, and promote neuronal activity-evoked calcium transients through neuron-to-glioma network. A subset of gap junction cells receives synaptic input, and the network integrates into brain through synapses and electrical signals.53 These unconnected glioma cells can hijack multiple traits of neuronal development via complex calcium signals from neurogliomal synapses by AMPAR, leading to the de novo formation of tumor microtubule network in glioblastoma sub-population, and then promoting cancer cells invasion as well as therapeutic resistance.53,343 Synaptic pathophysiology is not limited to primary brain cancer, which also exists in breast-to-brain metastatic tumors. The metastatic tumor to brain establishes glutamatergic synapses mediated by NADMR to integrate in neuronal circuits.54 Actin is an important component of cell cytoskeleton to form synaptic and protuberant structures, which is not only involved in the growth, metabolism and migration of normal cells and cancer cells, but also are critical for the connection of nervous system circuits. Altogether, gap junction and actin can be regarded as the key research object of cancer neuroscience.
Microtubule
Normally, the two cytoskeletal systems, including microfilaments and microtubules, regulate cytoskeletal structure to control cell motility and other cellular events with a large number of common or even overlapping signaling pathways, such as Rho GTPase pathways.344 Likewise, microtubules are also the major components of neuron cytoskeletal, and also essential for many fundamental neuronal and developmental processes, including neuronal migration, polarity, and differentiation, through guiding intracellular transport and inducing morphological changes.345 Inhibition of tumor survival by targeting tubulin responsible for axon guidance is validated. Overexpression of tubulin beta 2B class IIb (TUBB2B) in triple-negative breast cancer cells can activate astrocytes, thereby up-regulating the expression of TUBB2B in cancer cells.346 Eukaryotic translation elongation factor 1α1 (eEF1A1) is a mediator. TUBB2B eEF1A1 interacts with each other to promote protein synthesis by stabilizing this translation elongation factor. It suggests that there is a positive feedback interaction between TUBB2B and astrocytes in triple-negative breast cancer cells to promote the colonization of brain metastases.346 This provides a basis for exploring the mechanism between the expression of axon guidance genes and tumor brain metastasis.
Intermediate filament
Intermediate filaments formed by 40 different subunit proteins, including keratins, neurofilaments, desmin, laminin and vimentin, and intermediate filaments link from the ECM to the cytoplasmic interior surrounding the nucleus which allows intermediate filaments to relay information from cell surface to cellular compartments, thus coordinating cytoskeletal activities.347 Nestin, an intermediate filament protein, exhibits high expression levels in various cancer types, and is also correlated with aggressive growth, invasion, and poor prognosis.348,349 Meanwhile, nestin negatively regulates ACh-induced CDK5-dependent ACh receptor clusters dispersion of the neuromuscular junction synapse, which is essential for postsynaptic differentiation.350 In nestin-GFP transgenic mice model, the dentate gyrus of radial glia-like stem cells can ensheathe local synapses and vasculature in the inner molecular layer.351 These outcomes indicate that nestin plays a potential role in modulating neuro-tumor synapses formation and tumor migration. Vimentin is another intermediate filament protein that is involved in cancer progression.352,353,354 Loss of vimentin results in peripheral nerve hypermyelination mediated by axonal neuregulin-1 type III.355 Acting as a key component of intermediate filaments, vimentin is a fibrous component in both Schwann cells and neuron cytoskeleton, the expression of which is timely and spatially regulated during development and regeneration. Hence, above-discussed researches render sufficient evidence for multiple filament proteins that are involved in cancer neuroscience.
Targeting cytoskeletal proteins for therapy
It is now accepted that cytoskeletal components play important roles in the malignant development of cancer cells and the development and differentiation of neurons. Therapeutic strategies developed for brain tumors may have novel prompts for other cancer types. Some therapeutic modes have emerged from features of neuronal function and neurodevelopment, such as preventing neurite-like process growth, blocking neuronal inputs and neurotransmitter signals, and depletion of neuronal subsets in tumor microenvironment. The inhibition of neurite-like protrusions is indeed discovered to reduce tumor cells metastasis.356,357 Whether they play a role in the treatment of peripheral tumors remains to be established. This is mainly because abnormalities in neural subsets of extracranial tumors may lead to treatment failure.
Unlike the majority of cells in a single tumor, neuronal subsets of epithelial tumors are very diverse in gene expression and gene regulatory networks,173,358 which may require distinct and/or multiple therapeutic approaches. The presence of complex subgroups within the same tumor may require combination therapy targeting a single subgroup. Gap junction inhibitors might be used to disrupt tumor growth and resistance signals, and prevent the downstream effects of synaptic input.2,3 In combination with cytotoxic treatment might target effectively to gap junction in brain tumors. Gap junction inhibitors have been found in preclinical models to elevate sensitivity of radio- and chemo-therapy in brain tumor cells.359 Meclofenamate and tonabersat are regarded as promising candidate drugs in future clinical trials, which both inhibit the formation of heterocellular astrocyte-carcinoma gap junctions, then preventing brain metastasis and enhancing chemotherapy responses in mouse models.360 Synapses also act as the communication structure between neuron-tumor cells. Inhibitors of AMPAR and NMDAR are used to target neuro-glioma synapses in CNS.2,3 Perampanel, an AMPAR inhibitor acting as antiepileptic drug, together with radio- and chemo-therapy shows great tolerability and prolonged survival of glioma patients in the phase II clinical trial.361 Similarly, recent study finds NMDAR blocker, esmethadone, for the treatment of major depressive disorder with no neurotoxicity in rat models, as well as favorable safety and tolerability in phase 1 and phase 2 clinical trials.362 Extending clinical studies of these drugs into therapy of gliomas would be considered a great progress to send the drugs for application of cancer neuroscience knowledge as a core research and development.
Current challenges in cancer neuroscience
The field of cancer neuroscience is at its infancy, limited successes primarily aggregated on relatively easy parts, such as how the nervous system contributes to the tumorigenesis, growth, spread, recurrence and treatment resistance. Many more areas and related molecular mechanisms, such as how cancers respond to neuronal signals and how peripheral neurons may be widely related to tumors, require research attention.
Cancer heterogeneity is often manifested in gene, epigenetic, transcriptional, protein, and metabolic levels. Even if our current studies can improve the specific targeting of drugs and reduce side effects,363 there is still a lack of suitable heterogeneous biomarkers. Current research on tumor neurobiology chiefly relies on static cell models and mouse tumor transplantation experiments, which is difficult to dynamically capture the spatial-temporal evolution of neuro-tumor interactions, and species differences (such as the differences between mice blood-brain barrier and glial interactions with humans) limit the clinical transformation potential. In addition, most studies focus on a single signaling pathway and lack multiomics integration (such as joint analysis of spatial-temporal transcriptome and metabolome), which cannot resolve the spatial heterogeneity of nerve signals in the tumor microenvironment. Spatial multiomics approaches integrate spatial lipidomics and spatial transcriptomics sequencing technologies, achieving the accurate analysis in the areas of metabolism, lipid group and transcriptome data in a tumor tissue sample; associating the network of metabolites/lipids and upstream regulatory genes; characterizing the metabolic regulation and interaction of tumor cells, immune cells and stromal cells in the local tumor microenvironment in situ.364 The most important aim is that the combination of scRNA-seq and spatial multi-omics can analyze the distribution of neurotransmitter receptor subtypes. They provide innovative and effective methods, tools and new perspectives for the in-depth study of tumor metabolic heterogeneity.
Despite recent rapid and marvelous advancements, current technological techniques (e.g., neuroimaging) may not fully capture the dynamic changes occurring in the nervous system during cancer development and progression. Techniques to directly record and manipulate brain activity, such as deep brain stimulation (DBS) and optogenetics, have shown potential for treatment of certain diseases, but their applications are limited and carry certain risks. The high-resolution and direct imaging of neuronal activity has been proved as a noninvasive neuroimaging method, which enables direct imaging of neuronal activity with millisecond precision.365 ChR2/Halo, an optogenetic tool, enables accurate analysis and localization of neural activity within tumors.366 Changes in the expression of photosensitive proteins can visualize changes in neuronal activity and interaction. Moreover, calcium signal and axon dynamics were tracked in real time by living two-photon imaging.367 The breakthrough of technical limitations will benefit our research on monitoring the temporospatial dynamics of neural networks in cancer neuroscience.
Access to comprehensive clinical data, particularly concerning the neurobiological aspects of cancer progression and treatment response, remains restricted. Lacking comprehensive clinical data regarding the neurobiological aspects of cancer progression and treatment response hampers the ability to draw robust conclusions and develop effective interventions.
Existing models may not fully encapsulate the complexities of the tumor microenvironment and its influence on neural activity. Mice and nonhuman primate models have played an important role in studying the nervous system, but they differ physiologically and anatomically from humans, which may somewhat complicate the transferability of findings. Transplanting the human dorsal root ganglion into immunodeficient mice to establish a humanized neuro-tumor model may be more closely reflect the human-specific neuro-invasion mechanism, although its survival time will probably be limited by rejection. In order to overcome these limitations, a three-dimensional organoid co-culture system (integrating neurons, glial cells or immune cells and tumor cells) can be constructed in the future to simulate the interaction between synaptic formation and metabolism in vivo. 3D organoid co-culture technology may reduce the dependence on laboratory animals.368 The construction of a dorsal root ganglion-tumor organoid could simulate pain nerve signals and tumor growth.369 The development of in vitro technology will reduce the pressure of using laboratory animals to some extent, which is worth looking forward to the expansion direction. Given the interactive nature of cancer neuroscience, there is an urgent need to develop experimental models that characterize interactions between tumors, nerves, and other systems.
Conclusion and perspective
We have witnessed that over the recent decade cancer neuroscience has made steady progress and the hope is to connect the advances to clinical setting to benefit numerous patients with cancer. As such, tools used in neuroscience research and/or cancer therapy are also powerful for understanding cancer pathophysiology: whether the neuroscience-guided cancer therapy meets the target goal. Continued development of advanced neuroimaging techniques, such as functional MRI, diffusion tensor imaging (DTI), and positron emission tomography (PET), to better visualize and understand the dynamic changes in the nervous system associated with cancer progression. Integration of imaging data with other omics data can enhance the understanding of neurobiological mechanisms underlying cancer. In addition, utilizing multi-omics technologies, including genomics, transcriptomics, proteomics, and metabolomics, together with patch clamp technology, to comprehensively characterize the molecular signatures of cancer-neurobiology interactions. This holistic approach can provide insight into underlying mechanisms and identify novel therapeutic targets. Finding suitable biomarkers is critical, which ensure the connection between positive study results and the desired target engagement. Developing imaging and electrophysiological biomarkers may deepen our understanding of the molecular and cellular mechanisms underlying neuron-cancer crosstalk.
Furthermore, targeting the neural pathways and structures in tumor tissue requires careful validation of neurocognitive effects and behavioral assessment. Targeting neural-cancer interactions is highly likely to be used as a sensitizer for radiotherapy or chemotherapy. The mutation status of the genes may affect the characteristics of certain tumors, such as aggressive and recurrent tumors.18 Therefore, it is important to devise the optimal combination regimen with the concurrent use of cytotoxic, epigenetic, or immunotherapeutic therapies.
Moreover, artificial intelligence has already made progress in accelerating drug discovery and successfully using genomic and chemical data to predict drug behavior rather than using large-scale screening assays may accelerate drug reutilization.370 It is feasible and wise to take advantage of the rise of new technologies and apply them to research and practice. Leveraging bioinformatics tools and computational modeling approaches to analyze large-scale omics data, identify biomarkers, and predict patient outcomes. Integrating computational models of cancer-neurobiology interactions can facilitate hypothesis generation and accelerate drug discovery efforts. Exploring the adaptive response of the nervous system to cancer-related stressors, includes neuroplasticity mechanisms and resilience factors. Identifying neural pathways associated with resilience can help in the development of interventions to improve mental health and quality of life of patients during cancer treatment.
One may consider how peripheral drug delivery can enhance targeting, and how intracranial tumor drug delivery can cross the blood-brain barrier and decrease toxic side effects, which has been perplexing humans for a long time.371,372 When using drugs guided by neuroscience to act on tumors, screening the appropriate dose of drugs, such as reducing short-term side effects (vomiting, mood disorders, and suicidal tendencies) and long-term physical damage (hormone disorders, and organ failure), are the key factors to be aware of whether they can be used in the clinical practice. Under the philosophy of cancer neuroscience, it is expected that the incorporation of RNA-based therapeutics would provide new treatments for individuals suffering from cancer and improve the efficiency of existing treatments.
References
Venkatesh, H. S. et al. Targeting neuronal activity-regulated neuroligin-3 dependency in high-grade glioma. Nature 549, 533–537 (2017).
Venkatesh, H. S. et al. Electrical and synaptic integration of glioma into neural circuits. Nature 573, 539–545 (2019).
Venkataramani, V. et al. Glutamatergic synaptic input to glioma cells drives brain tumor progression. Nature 573, 532–538 (2019).
Balood, M. et al. Nociceptor neurons affect cancer immunosurveillance. Nature 611, 405–412 (2022).
Hanahan, D. & Monje, M. Cancer hallmarks intersect with neuroscience in the tumor microenvironment. Cancer Cell 41, 573–580 (2023).
Shi, D. D. et al. Therapeutic avenues for cancer neuroscience: translational frontiers and clinical opportunities. Lancet Oncol. 23, e62–e74 (2022).
Stupp, R. et al. Effect of tumor-treating fields plus maintenance temozolomide vs maintenance temozolomide alone on survival in patients with glioblastoma: a randomized clinical trial. Jama 318, 2306–2316 (2017).
Hiller, J. G. et al. Preoperative β-blockade with propranolol reduces biomarkers of metastasis in breast cancer: a phase II randomized trial. Clin. Cancer Res. 26, 1803–1811 (2020).
Ayala, G. E. et al. In vitro dorsal root ganglia and human prostate cell line interaction: redefining perineural invasion in prostate cancer. Prostate 49, 213–223 (2001).
Ayala, G. E. et al. Cancer-related axonogenesis and neurogenesis in prostate cancer. Clin. Cancer Res. 14, 7593–7603 (2008).
Mauffrey, P. et al. Progenitors from the central nervous system drive neurogenesis in cancer. Nature 569, 672–678 (2019).
Amit, M. et al. Loss of p53 drives neuron reprogramming in head and neck cancer. Nature 578, 449–454 (2020).
Monje, M. et al. Roadmap for the emerging field of cancer neuroscience. Cell 181, 219–222 (2020).
Pan, C. & Winkler, F. Insights and opportunities at the crossroads of cancer and neuroscience. Nat. Cell Biol. 24, 1454–1460 (2022).
Mancusi, R. & Monje, M. The neuroscience of cancer. Nature 618, 467–479 (2023).
Keough, M. B. & Monje, M. Neural signaling in cancer. Annu. Rev. Neurosci. 45, 199–221 (2022).
Zahalka, A. H. & Frenette, P. S. Nerves in cancer. Nat. Rev. Cancer 20, 143–157 (2020).
Varn, F. S. et al. Glioma progression is shaped by genetic evolution and microenvironment interactions. Cell 185, 2184–2199.e2116 (2022).
Hoogstrate, Y. et al. Transcriptome analysis reveals tumor microenvironment changes in glioblastoma. Cancer Cell 41, 678–692.e677 (2023).
Liu, J. et al. Multi-scale signaling and tumor evolution in high-grade gliomas. Cancer Cell 42, 1217–1238.e1219 (2024).
Venkatesh, H. S. et al. Neuronal activity promotes glioma growth through neuroligin-3 secretion. Cell 161, 803–816 (2015).
Park, M. et al. CYY-1/cyclin Y and CDK-5 differentially regulate synapse elimination and formation for rewiring neural circuits. Neuron 70, 742–757 (2011).
Jeong, J. et al. Regulation of NLGN3 and the synaptic Rho-GEF signaling pathway by CDK5. J. Neurosci. 43, 7264–7275 (2023).
Xu, N. et al. Integrated proteogenomic characterization of urothelial carcinoma of the bladder. J. Hematol. Oncol. 15, 76 (2022).
Pan, Y. et al. NF1 mutation drives neuronal activity-dependent initiation of optic glioma. Nature 594, 277–282 (2021).
Taylor, K. R. et al. Glioma synapses recruit mechanisms of adaptive plasticity. Nature 623, 366–374 (2023).
Li, Z. et al. NLGN3 promotes neuroblastoma cell proliferation and growth through activating PI3K/AKT pathway. Eur. J. Pharm. 857, 172423 (2019).
Wei, J. et al. Cucurbitacin B-induced G2/M cell cycle arrest of conjunctival melanoma cells mediated by GRP78-FOXM1-KIF20A pathway. Acta Pharm. Sin. B 12, 3861–3876 (2022).
Yun, E. J. et al. Targeting Wnt/β-catenin-mediated upregulation of oncogenic NLGN3 suppresses cancer stem cells in glioblastoma. Cell Death Dis. 14, 423 (2023).
Li, J. et al. Stabilization of KPNB1 by deubiquitinase USP7 promotes glioblastoma progression through the YBX1-NLGN3 axis. J. Exp. Clin. Cancer Res. 43, 28 (2024).
Wang, J. et al. LINC00665 activating Wnt3a/β-catenin signaling by bond with YBX1 promotes gastric cancer proliferation and metastasis. Cancer Gene Ther. 30, 1530–1542 (2023).
Stopczynski, R. E. et al. Neuroplastic changes occur early in the development of pancreatic ductal adenocarcinoma. Cancer Res. 74, 1718–1727 (2014).
Renz, B. W. et al. β2 adrenergic-neurotrophin feedforward loop promotes pancreatic cancer. Cancer Cell 34, 863–867 (2018).
Whiteley, A. E. et al. Breast cancer exploits neural signaling pathways for bone-to-meninges metastasis. Science 384, eadh5548 (2024).
He, S. et al. GFRα1 released by nerves enhances cancer cell perineural invasion through GDNF-RET signaling. Proc. Natl. Acad. Sci. USA 111, E2008–E2017 (2014).
Ceyhan, G. O. et al. The neurotrophic factor artemin promotes pancreatic cancer invasion. Ann. Surg. 244, 274–281 (2006).
Le, C. P. et al. Chronic stress in mice remodels lymph vasculature to promote tumor cell dissemination. Nat. Commun. 7, 10634 (2016).
Creed, S. J. et al. β2-adrenoceptor signaling regulates invadopodia formation to enhance tumor cell invasion. Breast Cancer Res 17, 145 (2015).
Padmanaban, V. et al. Neuronal substance P drives metastasis through an extracellular RNA–TLR7 axis. Nature 633, 207–215 (2024).
Chen, P. et al. Olfactory sensory experience regulates gliomagenesis via neuronal IGF1. Nature 606, 550–556 (2022).
van Kessel, E. et al. Tumor-related molecular determinants of neurocognitive deficits in patients with diffuse glioma. Neuro Oncol. 24, 1660–1670 (2022).
Faulkner, S. et al. Tumor neurobiology and the war of nerves in cancer. Cancer Discov. 9, 702–710 (2019).
Yang, M. W. et al. Perineural invasion reprograms the immune microenvironment through cholinergic signaling in pancreatic ductal adenocarcinoma. Cancer Res. 80, 1991–2003 (2020).
Zahalka, A. H. et al. Adrenergic nerves activate an angio-metabolic switch in prostate cancer. Science 358, 321–326 (2017).
Servick, K. War of nerves. Science 365, 1071–1073 (2019).
Hartmann, D. A. et al. Brain capillary pericytes exert a substantial but slow influence on blood flow. Nat. Neurosci. 24, 633–645 (2021).
Wälchli, T. et al. Shaping the brain vasculature in development and disease in the single-cell era. Nat. Rev. Neurosci. 24, 271–298 (2023).
Zhu, P. et al. 5-hydroxytryptamine produced by enteric serotonergic neurons initiates colorectal cancer stem cell self-renewal and tumorigenesis. Neuron 110, 2268–2282.e2264 (2022).
Chen, H.-C. et al. Histone serotonylation regulates ependymoma tumorigenesis. Nature 632, 903–910 (2024).
Silbereis, J. C. et al. The cellular and molecular landscapes of the developing human central nervous system. Neuron 89, 248–268 (2016).
Ohtaka-Maruyama, C. et al. Synaptic transmission from subplate neurons controls radial migration of neocortical neurons. Science 360, 313–317 (2018).
Ming, G. et al. Electrical activity modulates growth cone guidance by diffusible factors. Neuron 29, 441–452 (2001).
Venkataramani, V. et al. Glioblastoma hijacks neuronal mechanisms for brain invasion. Cell 185, 2899–2917.e2831 (2022).
Zeng, Q. et al. Synaptic proximity enables NMDAR signaling to promote brain metastasis. Nature 573, 526–531 (2019).
Buckingham, S. C. et al. Glutamate release by primary brain tumors induces epileptic activity. Nat. Med 17, 1269–1274 (2011).
Campbell, S. L. et al. GABAergic disinhibition and impaired KCC2 cotransporter activity underlie tumor-associated epilepsy. Glia 63, 23–36 (2015).
Barron, T. et al. GABAergic neuron-to-glioma synapses in diffuse midline gliomas. Nature 639, 1060–1068 (2025).
Li, N. et al. The delta subunit of the GABA(A) receptor is necessary for the GPT2-promoted breast cancer metastasis. Theranostics 13, 1355–1369 (2023).
Neman, J. et al. Human breast cancer metastases to the brain display GABAergic properties in the neural niche. Proc. Natl. Acad. Sci. USA 111, 984–989 (2014).
John Lin, C. C. et al. Identification of diverse astrocyte populations and their malignant analogs. Nat. Neurosci. 20, 396–405 (2017).
Yu, K. et al. PIK3CA variants selectively initiate brain hyperactivity during gliomagenesis. Nature 578, 166–171 (2020).
Krishna, S. et al. Glioblastoma remodelling of human neural circuits decreases survival. Nature 617, 599–607 (2023).
Osswald, M. et al. Brain tumour cells interconnect to a functional and resistant network. Nature 528, 93–98 (2015).
Gritsenko, P. G. et al. p120-catenin-dependent collective brain infiltration by glioma cell networks. Nat. Cell Biol. 22, 97–107 (2020).
Winkler, F. & Wick, W. Harmful networks in the brain and beyond. Science 359, 1100–1101 (2018).
Jung, E. et al. Tumor cell plasticity, heterogeneity, and resistance in crucial microenvironmental niches in glioma. Nat. Commun. 12, 1014 (2021).
Jung, E. et al. Tweety-homolog 1 drives brain colonization of gliomas. J. Neurosci. 37, 6837–6850 (2017).
Hausmann, D. et al. Autonomous rhythmic activity in glioma networks drives brain tumor growth. Nature 613, 179–186 (2023).
Winkler, F. et al. Cancer neuroscience: state of the field, emerging directions. Cell 186, 1689–1707 (2023).
Hayakawa, Y. et al. Nerve growth factor promotes gastric tumorigenesis through aberrant cholinergic signaling. Cancer Cell 31, 21–34 (2017).
Ayala, G. Neuroepithelial interactions in cancer. Annu Rev. Pathol. 18, 493–514 (2023).
Zhi, X. et al. Nociceptive neurons promote gastric tumor progression via a CGRP–RAMP1 axis. Nature 640, 802–810 (2025).
Gu, J. et al. Tumor metabolite lactate promotes tumorigenesis by modulating MOESIN lactylation and enhancing TGF-β signaling in regulatory T-cells. Cell Rep. 39, 110986 (2022).
Li, X. et al. Lactate metabolism in human health and disease. Signal Transduct. Target Ther. 7, 305 (2022).
Torrini, C. et al. Lactate is an epigenetic metabolite that drives survival in model systems of glioblastoma. Mol. Cell 82, 3061–3076.e3066 (2022).
Li, F. et al. Glial metabolic rewiring promotes axon regeneration and functional recovery in the central nervous system. Cell Metab. 32, 767–785.e767 (2020).
Jia, L. et al. Rheb-regulated mitochondrial pyruvate metabolism of Schwann cells linked to axon stability. Dev. Cell 56, 2980–2994.e2986 (2021).
Vander Heiden, M. G., Cantley, L. C. & Thompson, C. B. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324, 1029–1033 (2009).
Chinchore, Y. et al. Glycolytic reliance promotes anabolism in photoreceptors. Elife 6, e25946 (2017).
Todorova, V. et al. Deficits in mitochondrial TCA cycle and OXPHOS precede rod photoreceptor degeneration during chronic HIF activation. Mol. Neurodegener. 18, 15 (2023).
Minhas, P. S. et al. Restoring hippocampal glucose metabolism rescues cognition across Alzheimer’s disease pathologies. Science 385, eabm6131 (2024).
Zhang, X., Liu, Q., Liao, Q. & Zhao, Y. Potential roles of peripheral dopamine in tumor immunity. J. Cancer 8, 2966–2973 (2017).
Cheng, J. et al. Drug resistance mechanisms in dopamine agonist-resistant prolactin pituitary neuroendocrine tumors and exploration for new drugs. Drug Resist. Updat 73, 101056 (2024).
Eisenhofer, G. et al. Substantial production of dopamine in the human gastrointestinal tract. J. Clin. Endocrinol. Metab. 82, 3864–3871 (1997).
Cervantes-Villagrana, R. D., Albores-García, D., Cervantes-Villagrana, A. R. & García-Acevez, S. J. Tumor-induced neurogenesis and immune evasion as targets of innovative anti-cancer therapies. Signal Transduct. Target Ther. 5, 99 (2020).
Saha, B., Mondal, A. C., Basu, S. & Dasgupta, P. S. Circulating dopamine level, in lung carcinoma patients, inhibits proliferation and cytotoxicity of CD4+ and CD8+ T cells by D1 dopamine receptors: an in vitro analysis. Int. Immunopharmacol. 1, 1363–1374 (2001).
Zhou, Y. et al. DRD4 interacts with TGF-β receptors to drive colorectal cancer metastasis independently of dopamine signaling pathway. Adv Sci. 12, 2413953 (2024).
Zhang, W. et al. Injectable and adhesive MgO(2)-potentiated hydrogel with sequential tumor synergistic therapy and osteogenesis for challenging postsurgical osteosarcoma treatment. Biomaterials 315, 122959 (2025).
Mei, L. et al. Self-propelling intelligent nanomotor: a dual-action photothermal and starvation strategy for targeted deep tumor destruction. Biomaterials 315, 122968 (2025).
Kong, X. et al. Melatonin: a potential therapeutic option for breast cancer. Trends Endocrinol. Metab. 31, 859–871 (2020).
Cheng, L. et al. Melatonin regulates cancer migration and stemness and enhances the anti-tumour effect of cisplatin. J. Cell Mol. Med. 27, 2215–2227 (2023).
Stryjewska, A. et al. Zeb2 regulates cell fate at the exit from epiblast state in mouse embryonic stem cells. Stem Cells 35, 611–625 (2017).
Srivatsa, S., Parthasarathy, S., Molnár, Z. & Tarabykin, V. Sip1 downstream effector ninein controls neocortical axonal growth, ipsilateral branching, and microtubule growth and stability. Neuron 85, 998–1012 (2015).
Pastushenko, I. & Blanpain, C. EMT transition states during tumor progression and metastasis. Trends Cell Biol. 29, 212–226 (2019).
Lin, J. et al. A positive feedback loop between ZEB2 and ACSL4 regulates lipid metabolism to promote breast cancer metastasis. Elife 12, RP87510 (2023).
Wiles, E. T. et al. ZEB2 represses the epithelial phenotype and facilitates metastasis in Ewing sarcoma. Genes Cancer 4, 486–500 (2013).
Zeng, Y. et al. Oncogenic ZEB2/miR-637/HMGA1 signaling axis targeting vimentin promotes the malignant phenotype of glioma. Mol. Ther. Nucleic Acids 23, 769–782 (2021).
Wang, M. et al. Different lineage contexts direct common pro-neural factors to specify distinct retinal cell subtypes. J. Cell Biol. 219, e202003026 (2020).
Tang, Y. et al. Restoration of visual function and cortical connectivity after ischemic injury through neurod1-mediated gene therapy. Front. Cell Dev. Biol. 9, 720078 (2021).
Xu, J. et al. Generation of neural organoids for spinal-cord regeneration via the direct reprogramming of human astrocytes. Nat. Biomed. Eng. 7, 253–269 (2023).
Wang, X. et al. Transcription factor-based gene therapy to treat glioblastoma through direct neuronal conversion. Cancer Biol. Med. 18, 860–874 (2021).
Rao, Y. et al. NeuroD1 induces microglial apoptosis and cannot induce microglia-to-neuron cross-lineage reprogramming. Neuron 109, 4094–4108.e4095 (2021).
Rivetti di Val Cervo, P. et al. Induction of functional dopamine neurons from human astrocytes in vitro and mouse astrocytes in a Parkinson’s disease model. Nat. Biotechnol. 35, 444–452 (2017).
Bocchi, R., Masserdotti, G. & Götz, M. Direct neuronal reprogramming: Fast forward from new concepts toward therapeutic approaches. Neuron 110, 366–393 (2022).
Jin, M. Z. & Jin, W. L. The updated landscape of tumor microenvironment and drug repurposing. Signal. Transduct. Target Ther. 5, 166 (2020).
Kim, K. H. et al. Integrated proteogenomic characterization of glioblastoma evolution. Cancer Cell 42, 358–377.e358 (2024).
Borovikova, L. V. et al. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 405, 458–462 (2000).
Li, Y. T., Yuan, W. Z. & Jin, W. L. Vagus innervation in the gastrointestinal tumor: current understanding and challenges. Biochim. Biophys. Acta Rev. Cancer 1878, 188884 (2023).
Devi, S. et al. Adrenergic regulation of the vasculature impairs leukocyte interstitial migration and suppresses immune responses. Immunity 54, 1219–1230.e1217 (2021).
Poller, W. C. et al. Brain motor and fear circuits regulate leukocytes during acute stress. Nature 607, 578–584 (2022).
Yang, H. et al. Stress-glucocorticoid-TSC22D3 axis compromises therapy-induced antitumor immunity. Nat. Med. 25, 1428–1441 (2019).
Xu, X. R. et al. Activation of dopaminergic VTA inputs to the mPFC ameliorates chronic stress-induced breast tumor progression. CNS Neurosci. Ther. 27, 206–219 (2021).
Hanahan, D. & Coussens, L. M. Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell 21, 309–322 (2012).
Giraldo, N. A. et al. The clinical role of the TME in solid cancer. Br. J. Cancer 120, 45–53 (2019).
Kim, S. K. & Cho, S. W. The evasion mechanisms of cancer immunity and drug intervention in the tumor microenvironment. Front. Pharm. 13, 868695 (2022).
Mehrian-Shai, R., Reichardt, J. K. V., Harris, C. C. & Toren, A. The gut-brain axis, paving the way to brain cancer. Trends Cancer 5, 200–207 (2019).
Leung, E. L.-H. et al. Novel anticancer strategy by targeting the gut microbial neurotransmitter signaling to overcome immunotherapy resistance. Antioxid. Redox Signal. 38, 298–315 (2022).
Jiang, S. H. et al. Neurotransmitters: emerging targets in cancer. Oncogene 39, 503–515 (2020).
Khanmammadova, N., Islam, S., Sharma, P. & Amit, M. Neuro-immune interactions and immuno-oncology. Trends Cancer 9, 636–649 (2023).
Thaker, P. H. et al. Chronic stress promotes tumor growth and angiogenesis in a mouse model of ovarian carcinoma. Nat. Med. 12, 939–944 (2006).
Wang, H. et al. Role of the nervous system in cancers: a review. Cell Death Discov. 7, 76 (2021).
Guo, X. et al. Midkine activation of CD8(+) T cells establishes a neuron-immune-cancer axis responsible for low-grade glioma growth. Nat. Commun. 11, 2177 (2020).
Zhang, Z. et al. VLM catecholaminergic neurons control tumor growth by regulating CD8(+) T cells. Proc. Natl. Acad. Sci USA 118, e2103505118 (2021).
Slominski, R. M., Raman, C., Chen, J. Y. & Slominski, A. T. How cancer hijacks the body’s homeostasis through the neuroendocrine system. Trends Neurosci. 46, 263–275 (2023).
Xu, Y. et al. Pituitary hormone α-MSH promotes tumor-induced myelopoiesis and immunosuppression. Science 377, 1085–1091 (2022).
Møller, S. H. et al. Metabolic programs tailor T cell immunity in viral infection, cancer, and aging. Cell Metab. 34, 378–395 (2022).
Cassetta, L. & Pollard, J. W. Targeting macrophages: therapeutic approaches in cancer. Nat. Rev. Drug Discov. 17, 887–904 (2018).
Pittet, M. J., Michielin, O. & Migliorini, D. Clinical relevance of tumor-associated macrophages. Nat. Rev. Clin. Oncol. 19, 402–421 (2022).
Kloosterman, D. J. & Akkari, L. Macrophages at the interface of the co-evolving cancer ecosystem. Cell 186, 1627–1651 (2023).
Rosas-Ballina, M. et al. Acetylcholine-synthesizing T cells relay neural signals in a vagus nerve circuit. Science 334, 98–101 (2011).
Sloan, E. K. et al. The sympathetic nervous system induces a metastatic switch in primary breast cancer. Cancer Res. 70, 7042–7052 (2010).
Partecke, L. I. et al. Chronic stress increases experimental pancreatic cancer growth, reduces survival and can be antagonised by beta-adrenergic receptor blockade. Pancreatology 16, 423–433 (2016).
Cheng, Y. et al. Depression-induced neuropeptide Y secretion promotes prostate cancer growth by recruiting myeloid cells. Clin. Cancer Res. 25, 2621–2632 (2019).
Renz, B. W. et al. Cholinergic signaling via muscarinic receptors directly and indirectly suppresses pancreatic tumorigenesis and cancer stemness. Cancer Discov. 8, 1458–1473 (2018).
Laumont, C. M. & Nelson, B. H. B cells in the tumor microenvironment: multi-faceted organizers, regulators, and effectors of anti-tumor immunity. Cancer Cell 41, 466–489 (2023).
Wang, S. S. et al. Tumor-infiltrating B cells: their role and application in anti-tumor immunity in lung cancer. Cell Mol. Immunol. 16, 6–18 (2019).
Wieland, A. et al. Defining HPV-specific B cell responses in patients with head and neck cancer. Nature 597, 274–278 (2021).
Michaud, D., Steward, C. R., Mirlekar, B. & Pylayeva-Gupta, Y. Regulatory B cells in cancer. Immunol. Rev. 299, 74–92 (2021).
Harris, R. J. et al. Tumor-infiltrating B lymphocyte profiling identifies IgG-biased, clonally expanded prognostic phenotypes in triple-negative breast cancer. Cancer Res 81, 4290–4304 (2021).
Sung, H. Y., Yang, S. D., Ju, W. & Ahn, J. H. Aberrant epigenetic regulation of GABRP associates with aggressive phenotype of ovarian cancer. Exp. Mol. Med. 49, e335 (2017).
Kanbara, K. et al. GABA(B) receptor regulates proliferation in the high-grade chondrosarcoma cell line OUMS-27 via apoptotic pathways. BMC Cancer 18, 263 (2018).
Zhang, B. et al. B cell-derived GABA elicits IL-10(+) macrophages to limit anti-tumour immunity. Nature 599, 471–476 (2021).
Mohammadpour, H. et al. β2-adrenergic receptor signaling regulates metabolic pathways critical to myeloid-derived suppressor cell function within the TME. Cell Rep. 37, 109883 (2021).
Qiao, G. et al. β-Adrenergic signaling blocks murine CD8(+) T-cell metabolic reprogramming during activation: a mechanism for immunosuppression by adrenergic stress. Cancer Immunol. Immun. 68, 11–22 (2019).
Zeng, W. et al. Interactions between central nervous system and peripheral metabolic organs. Sci. China Life Sci. 65, 1929–1958 (2022).
Ben-Shaanan, T. L. et al. Modulation of anti-tumor immunity by the brain’s reward system. Nat. Commun. 9, 2723 (2018).
Xu, Q. et al. Multiple cancer cell types release LIF and Gal3 to hijack neural signals. Cell Res. 34, 345–354 (2024).
Kalluri, R. The biology and function of fibroblasts in cancer. Nat. Rev. Cancer 16, 582–598 (2016).
Martinez-Outschoorn, U. E., Lisanti, M. P. & Sotgia, F. Catabolic cancer-associated fibroblasts transfer energy and biomass to anabolic cancer cells, fueling tumor growth. Semin. Cancer Biol. 25, 47–60 (2014).
Kobayashi, H. et al. Cancer-associated fibroblasts in gastrointestinal cancer. Nat. Rev. Gastroenterol. Hepatol. 16, 282–295 (2019).
Oben, J. A. et al. Norepinephrine and neuropeptide Y promote proliferation and collagen gene expression of hepatic myofibroblastic stellate cells. Biochem. Biophys. Res. Commun. 302, 685–690 (2003).
Kim-Fuchs, C. et al. Chronic stress accelerates pancreatic cancer growth and invasion: a critical role for beta-adrenergic signaling in the pancreatic microenvironment. Brain Behav. Immun. 40, 40–47 (2014).
Demir, I. E., Friess, H. & Ceyhan, G. O. Nerve-cancer interactions in the stromal biology of pancreatic cancer. Front. Physiol. 3, 97 (2012).
Yamazaki, S. et al. Quercetin-3-O-glucuronide inhibits noradrenaline-promoted invasion of MDA-MB-231 human breast cancer cells by blocking β₂-adrenergic signaling. Arch. Biochem. Biophys. 557, 18–27 (2014).
Xue, M. et al. Schwann cells regulate tumor cells and cancer-associated fibroblasts in the pancreatic ductal adenocarcinoma microenvironment. Nat. Commun. 14, 4600 (2023).
Li, J., Kang, R. & Tang, D. Cellular and molecular mechanisms of perineural invasion of pancreatic ductal adenocarcinoma. Cancer Commun. 41, 642–660 (2021).
Zhao, H. et al. Inflammation and tumor progression: signaling pathways and targeted intervention. Signal. Transduct. Target Ther. 6, 263 (2021).
Agirman, G., Yu, K. B. & Hsiao, E. Y. Signaling inflammation across the gut-brain axis. Science 374, 1087–1092 (2021).
Adler, O. et al. Reciprocal interactions between innate immune cells and astrocytes facilitate neuroinflammation and brain metastasis via lipocalin-2. Nat. Cancer 4, 401–418 (2023).
Pelly, V. S. et al. Anti-inflammatory drugs remodel the tumor immune environment to enhance immune checkpoint blockade efficacy. Cancer Discov. 11, 2602–2619 (2021).
Zhang, J. et al. Role of transforming growth factor-β1 pathway in angiogenesis induced by chronic stress in colorectal cancer. Cancer Biol. Ther. 25, 2366451 (2024).
Xia, Y. et al. Catecholamines contribute to the neovascularization of lung cancer via tumor-associated macrophages. Brain Behav. Immun. 81, 111–121 (2019).
Regalado, C. R. & Balogh, M. MMP9: Link between neuropathy and colorectal cancer? Front. Mol. Biosci. 11, 1451611 (2024).
An, J. et al. Chronic stress promotes breast carcinoma metastasis by accumulating myeloid-derived suppressor cells through activating β-adrenergic signaling. Oncoimmunology 10, 2004659 (2021).
Mohammadpour, H. et al. β2 adrenergic receptor-mediated signaling regulates the immunosuppressive potential of myeloid-derived suppressor cells. J. Clin. Investig. 129, 5537–5552 (2019).
Lu, J. et al. Olanzapine suppresses mPFC activity-norepinephrine releasing to alleviate CLOCK-enhanced cancer stemness under chronic stress. Cell Commun. Signal 22, 375 (2024).
Demir, I. E. et al. Future directions in preclinical and translational cancer neuroscience research. Nat. Cancer 1, 1027–1031 (2021).
Zhang, Y. et al. Versatile metal-phenolic network nanoparticles for multitargeted combination therapy and magnetic resonance tracing in glioblastoma. Biomaterials 278, 121163 (2021).
Zhao, M. et al. Nanocarrier-based drug combination therapy for glioblastoma. Theranostics 10, 1355–1372 (2020).
Meric-Bernstam, F., Larkin, J., Tabernero, J. & Bonini, C. Enhancing anti-tumor efficacy with immunotherapy combinations. Lancet 397, 1010–1022 (2021).
Zhao, C. M. et al. Denervation suppresses gastric tumorigenesis. Sci. Transl. Med. 6, 250ra115 (2014).
Lamkin, D. M. et al. Chronic stress enhances progression of acute lymphoblastic leukemia via β-adrenergic signaling. Brain Behav. Immun. 26, 635–641 (2012).
Lu, R. et al. Neurons generated from carcinoma stem cells support cancer progression. Signal Transduct. Target Ther. 2, 16036 (2017).
Magnon, C. et al. Autonomic nerve development contributes to prostate cancer progression. Science 341, 1236361 (2013).
Mogavero, M. P. et al. Sleep disorders and cancer: state of the art and future perspectives. Sleep. Med. Rev. 56, 101409 (2021).
Pitman, A., Suleman, S., Hyde, N. & Hodgkiss, A. Depression and anxiety in patients with cancer. BMJ 361, k1415 (2018).
Zheng, X. Q., Wu, Y. H., Huang, J. F. & Wu, A. M. Neurophysiological mechanisms of cancer-induced bone pain. J. Adv. Res. 35, 117–127 (2022).
Yu, S. et al. Depression decreases immunity and PD-L1 inhibitor efficacy via the hypothalamic-pituitary-adrenal (HPA) axis in triple-negative breast cancer. Biochim. Biophys. Acta Mol. Basis Dis. 1871, 167581 (2025).
Dalla-Favera, R. et al. Human c-myc onc gene is located on the region of chromosome 8 that is translocated in Burkitt lymphoma cells. Proc. Natl. Acad. Sci. USA 79, 7824–7827 (1982).
Kress, T. R., Sabò, A. & Amati, B. MYC: connecting selective transcriptional control to global RNA production. Nat. Rev. Cancer 15, 593–607 (2015).
Dhanasekaran, R. et al. The MYC oncogene—the grand orchestrator of cancer growth and immune evasion. Nat. Rev. Clin. Oncol. 19, 23–36 (2022).
Tang, X. et al. Lipid phosphate phosphatase-2 promotes tumor growth through increased c-Myc expression. Theranostics 12, 5675–5690 (2022).
Chen, L. et al. CRISPR-Cas9 screen reveals a MYCN-amplified neuroblastoma dependency on EZH2. J. Clin. Investig. 128, 446–462 (2018).
Chen, J. et al. Glioblastoma stem cell-specific histamine secretion drives pro-angiogenic tumor microenvironment remodeling. Cell Stem Cell 29, 1531–1546.e1537 (2022).
Jiang, B. C. et al. NFAT1 orchestrates spinal microglial transcription and promotes microglial proliferation via c-MYC contributing to nerve injury-induced neuropathic pain. Adv. Sci. 9, e2201300 (2022).
Belin, S. et al. Injury-induced decline of intrinsic regenerative ability revealed by quantitative proteomics. Neuron 86, 1000–1014 (2015).
Ma, J. J. et al. Telomerase reverse transcriptase and p53 regulate mammalian peripheral nervous system and CNS axon regeneration downstream of c-Myc. J. Neurosci. 39, 9107–9118 (2019).
Blelloch, R., Venere, M., Yen, J. & Ramalho-Santos, M. Generation of induced pluripotent stem cells in the absence of drug selection. Cell Stem Cell 1, 245–247 (2007).
Nakagawa, M. et al. Promotion of direct reprogramming by transformation-deficient Myc. Proc. Natl Acad. Sci. USA 107, 14152–14157 (2010).
Yang, S. G., Wang, X. W., Qian, C. & Zhou, F. Q. Reprogramming neurons for regeneration: the fountain of youth. Prog. Neurobiol. 214, 102284 (2022).
Ireland, A. S. et al. MYC drives temporal evolution of small cell lung cancer subtypes by reprogramming neuroendocrine fate. Cancer Cell 38, 60–78.e12 (2020).
Mullard, A. Climbing cancer’s MYC mountain. Nat. Rev. Drug Discov. 21, 865–867 (2022).
Sabò, A. et al. Selective transcriptional regulation by Myc in cellular growth control and lymphomagenesis. Nature 511, 488–492 (2014).
Luo, Z. et al. ZFP281 Recruits MYC to active promoters in regulating transcriptional initiation and elongation. Mol Cell Biol. 39, e00329–19 (2019).
Louis, S. F. et al. c-Myc induces chromosomal rearrangements through telomere and chromosome remodeling in the interphase nucleus. Proc. Natl. Acad. Sci. USA 102, 9613–9618 (2005).
Moore, A. R., Rosenberg, S. C., McCormick, F. & Malek, S. RAS-targeted therapies: is the undruggable drugged? Nat. Rev. Drug Discov. 19, 533–552 (2020).
Parada, L. F., Tabin, C. J., Shih, C. & Weinberg, R. A. Human EJ bladder carcinoma oncogene is homologue of Harvey sarcoma virus ras gene. Nature 297, 474–478 (1982).
Tanaka, N. et al. Clinical acquired resistance to KRAS(G12C) inhibition through a novel KRAS Switch-II pocket mutation and polyclonal alterations converging on RAS-MAPK reactivation. Cancer Discov. 11, 1913–1922 (2021).
Pantsar, T. The current understanding of KRAS protein structure and dynamics. Comput. Struct. Biotechnol. J. 18, 189–198 (2020).
Dombi, E. et al. Activity of selumetinib in neurofibromatosis type 1-related plexiform neurofibromas. N. Engl. J. Med. 375, 2550–2560 (2016).
de Blank, P. M. K. et al. MEK inhibitors for neurofibromatosis type 1 manifestations: clinical evidence and consensus. Neuro Oncol. 24, 1845–1856 (2022).
Costa, R. M. et al. Mechanism for the learning deficits in a mouse model of neurofibromatosis type 1. Nature 415, 526–530 (2002).
Hall, A. & Lalli, G. Rho and Ras GTPases in axon growth, guidance, and branching. CSH Perspect. Biol. 2, a001818 (2010).
Alcover-Sanchez, B., Garcia-Martin, G., Wandosell, F. & Cubelos, B. R-Ras GTPases signaling role in myelin neurodegenerative diseases. Int. J. Mol. Sci. 21, 5911 (2020).
Sanz-Rodriguez, M. et al. R-Ras1 and R-Ras2 are essential for oligodendrocyte differentiation and survival for correct myelination in the central nervous system. J. Neurosci. 38, 5096–5110 (2018).
Luo, J. et al. Colony-stimulating factor 1 receptor (CSF1R) signaling in injured neurons facilitates protection and survival. J. Exp. Med. 210, 157–172 (2013).
Mitrasinovic, O. M. et al. Microglia overexpressing the macrophage colony-stimulating factor receptor are neuroprotective in a microglial-hippocampal organotypic coculture system. J. Neurosci. 25, 4442–4451 (2005).
Wang, Y. et al. Early posttraumatic CSF1R inhibition via PLX3397 leads to time- and sex-dependent effects on inflammation and neuronal maintenance after traumatic brain injury in mice. Brain Behav. Immun. 106, 49–66 (2022).
Yu-Ju Wu, C. et al. CCL5 of glioma-associated microglia/macrophages regulates glioma migration and invasion via calcium-dependent matrix metalloproteinase 2. Neuro Oncol. 22, 253–266 (2020).
Liu, D. et al. β2-AR signaling controls trastuzumab resistance-dependent pathway. Oncogene 35, 47–58 (2016).
Armaiz-Pena, G. N. et al. Src activation by β-adrenoreceptors is a key switch for tumour metastasis. Nat. Commun. 4, 1403 (2013).
Liu, D. et al. β2-AR activation promotes cleavage and nuclear translocation of Her2 and metastatic potential of cancer cells. Cancer Sci. 111, 4417–4428 (2020).
Zhang, W. et al. A disintegrin and metalloproteinase domain 10 expression inhibition by the small molecules adenosine, cordycepin and N6, N6-dimethyladenosine and immune regulation in malignant cancers. Front Immunol. 15, 1434027 (2024).
Newton, R. C. et al. Clinical benefit of INCB7839, a potent and selective ADAM inhibitor, in combination with trastuzumab in patients with metastatic HER2+ breast cancer. J. Clin. Oncol. 28, 3025–3025.
Xiang, Z. et al. Targeting the NOTCH2/ADAM10/TCF7L2 axis-mediated transcriptional regulation of Wnt pathway suppresses tumor growth and enhances chemosensitivity in colorectal cancer. Adv. Sci. 12, 2405758 (2024).
Chen, C. et al. Soluble Tim-3 serves as a tumor prognostic marker and therapeutic target for CD8(+) T cell exhaustion and anti-PD-1 resistance. Cell Rep. Med. 5, 101686 (2024).
Johnson, D. A. et al. Neuronal differentiation and synaptogenesis in retinoblastoma. Cancer Res. 67, 2701–2711 (2007).
Christie, K. J. et al. Enhancing adult nerve regeneration through the knockdown of retinoblastoma protein. Nat. Commun. 5, 3670 (2014).
Ajioka, I. et al. Differentiated horizontal interneurons clonally expand to form metastatic retinoblastoma in mice. Cell 131, 378–390 (2007).
Jansen, R. W. et al. Magnetic resonance imaging can reliably differentiate optic nerve inflammation from tumor invasion in retinoblastoma with orbital cellulitis. Ophthalmology 129, 1275–1286 (2022).
Kaliki, S. et al. Defining high-risk retinoblastoma: a multicenter global survey. JAMA Ophthalmol. 140, 30–36 (2022).
House, R. R. J. et al. NF1 deficiency drives metabolic reprogramming in ER+ breast cancer. Mol. Metab. 80, 101876 (2024).
Li, W. et al. Nuclear localization of mitochondrial TCA cycle enzymes modulates pluripotency via histone acetylation. Nat. Commun. 13, 7414 (2022).
Ghosh, M. et al. Mutant p53 suppresses innate immune signaling to promote tumorigenesis. Cancer Cell 39, 494–508.e495 (2021).
Hu, J. et al. Targeting mutant p53 for cancer therapy: direct and indirect strategies. J. Hematol. Oncol. 14, 157 (2021).
Liu, F. et al. Whole-exome mutational landscape of neuroendocrine carcinomas of the gallbladder. Signal Transduct. Target Ther. 6, 55 (2021).
Lou, J. et al. Circular RNA CDR1as disrupts the p53/MDM2 complex to inhibit gliomagenesis. Mol. Cancer 19, 138 (2020).
Chen, J., Tan, Q., Yang, Z. & Jin, Y. Engineered extracellular vesicles: potentials in cancer combination therapy. J. Nanobiotechnol. 20, 132 (2022).
Gupta, R., Ambasta, R. K. & Pravir, K. Autophagy and apoptosis cascade: which is more prominent in neuronal death? Cell Mol. Life Sci. 78, 8001–8047 (2021).
Zhou, Z. et al. Stress-induced epinephrine promotes epithelial-to-mesenchymal transition and stemness of CRC through the CEBPB/TRIM2/P53 axis. J. Transl. Med. 20, 262 (2022).
Hara, M. R. et al. A stress response pathway regulates DNA damage through β2-adrenoreceptors and β-arrestin-1. Nature 477, 349–353 (2011).
Ge, W. et al. In vivo evaluation of guide-free Cas9-induced safety risks in a pig model. Signal Transduct. Target Ther. 9, 184 (2024).
Li, P. et al. Versatile and efficient mammalian genome editing with type I-C CRISPR system of Desulfovibrio vulgaris. Sci. China Life Sci. 67, 2471–2487 (2024).
Wang, D., Liu, B. & Zhang, Z. Accelerating the understanding of cancer biology through the lens of genomics. Cell 186, 1755–1771 (2023).
Ignatiadis, M., Sledge, G. W. & Jeffrey, S. S. Liquid biopsy enters the clinic—implementation issues and future challenges. Nat. Rev. Clin. Oncol. 18, 297–312 (2021).
Trivedi, R. & Bhat, K. P. Liquid biopsy: creating opportunities in brain space. Br. J. Cancer 129, 1727–1746 (2023).
Esteve-Puig, R., Bueno-Costa, A. & Esteller, M. Writers, readers and erasers of RNA modifications in cancer. Cancer Lett. 474, 127–137 (2020).
Sun, X. et al. BRD8 maintains glioblastoma by epigenetic reprogramming of the p53 network. Nature 613, 195–202 (2023).
Roundtree, I. A., Evans, M. E., Pan, T. & He, C. Dynamic RNA modifications in gene expression regulation. Cell 169, 1187–1200 (2017).
Gojo, J. et al. Single-cell RNA-Seq reveals cellular hierarchies and impaired developmental trajectories in pediatric ependymoma. Cancer Cell 38, 44–59.e49 (2020).
Takahashi, N. et al. Cancer cells co-opt the neuronal redox-sensing channel TRPA1 to promote oxidative-stress tolerance. Cancer Cell 33, 985–1003.e1007 (2018).
Li, W., Deng, X. & Chen, J. RNA-binding proteins in regulating mRNA stability and translation: roles and mechanisms in cancer. Semin. Cancer Biol. 86, 664–677 (2022).
Fabbri, L., Chakraborty, A., Robert, C. & Vagner, S. The plasticity of mRNA translation during cancer progression and therapy resistance. Nat. Rev. Cancer 21, 558–577 (2021).
Culjkovic-Kraljacic, B. & Borden, K. L. Aiding and abetting cancer: mRNA export and the nuclear pore. Trends Cell Biol. 23, 328–335 (2013).
Bartok, O. et al. Anti-tumour immunity induces aberrant peptide presentation in melanoma. Nature 590, 332–337 (2021).
Su, P. et al. Enhanced lipid accumulation and metabolism are required for the differentiation and activation of tumor-associated macrophages. Cancer Res. 80, 1438–1450 (2020).
Cerezo, M. et al. Translational control of tumor immune escape via the eIF4F-STAT1-PD-L1 axis in melanoma. Nat. Med. 24, 1877–1886 (2018).
Xu, Y. et al. Translation control of the immune checkpoint in cancer and its therapeutic targeting. Nat. Med. 25, 301–311 (2019).
Juneja, V. R. et al. PD-L1 on tumor cells is sufficient for immune evasion in immunogenic tumors and inhibits CD8 T cell cytotoxicity. J. Exp. Med. 214, 895–904 (2017).
Oh, S. A. et al. PD-L1 expression by dendritic cells is a key regulator of T-cell immunity in cancer. Nat. Cancer 1, 681–691 (2020).
Yin, Y. et al. Colorectal cancer-derived small extracellular vesicles promote tumor immune evasion by upregulating PD-L1 expression in tumor-associated macrophages. Adv. Sci. 9, 2102620 (2022).
Zhao, Z. et al. Immune checkpoint inhibitors and neurotoxicity. Curr. Neuropharmacol. 19, 1246–1263 (2021).
Lopes, S. et al. Central nervous system complications of immune checkpoint inhibitors: a comprehensive review. Crit. Rev. Oncol. Hematol. 206, 104595 (2024).
Han, J. et al. Suppression of KDM5C mitigates the symptoms of Alzheimer’s disease by up-regulating BDNF expression. Neurochem. Int. 186, 105975 (2025).
Ell, M. A., Schiele, M. A., Iovino, N. & Domschke, K. Epigenetics of fear, anxiety and stress - focus on histone modifications. Curr. Neuropharmacol. 22, 843–865 (2024).
Heery, R., Finn, S. P., Cuffe, S. D. & Gray, S. G. Long non-coding RNAs: key regulators of epithelial-mesenchymal transition, tumour drug resistance and cancer stem cells. Cancers. 9, 38 (2017).
Boccaletto, P. et al. MODOMICS: a database of RNA modification pathways. 2021 update. Nucleic Acids Res. 50, D231–d235 (2022).
Dai, L. et al. Systematic characterization and biological functions of non-coding RNAs in glioblastoma. Cell Prolif. 56, e13375 (2023).
Rupaimoole, R. & Slack, F. J. MicroRNA therapeutics: towards a new era for the management of cancer and other diseases. Nat. Rev. Drug Discov. 16, 203–222 (2017).
Chen, X. et al. TDP-43 regulates cancer-associated microRNAs. Protein Cell 9, 848–866 (2018).
Ke, H. et al. Loss of TDP43 inhibits progression of triple-negative breast cancer in coordination with SRSF3. Proc. Natl. Acad. Sci. USA 115, E3426–e3435 (2018).
Gao, X. et al. A virus-mimicking nucleic acid nanogel reprograms microglia and macrophages for glioblastoma therapy. Adv. Mater. 33, e2006116 (2021).
Maleszewska, M. et al. Sequential changes in histone modifications shape transcriptional responses underlying microglia polarization by glioma. Glia 69, 109–123 (2021).
Liu, M. et al. Role of non-coding RNAs in axon regeneration after peripheral nerve injury. Int. J. Biol. Sci. 18, 3435–3446 (2022).
Kopp, F. & Mendell, J. T. Functional classification and experimental dissection of long noncoding RNAs. Cell 172, 393–407 (2018).
Tan, Y. T. et al. LncRNA-mediated posttranslational modifications and reprogramming of energy metabolism in cancer. Cancer Commun. 41, 109–120 (2021).
McCabe, E. M. & Rasmussen, T. P. lncRNA involvement in cancer stem cell function and epithelial-mesenchymal transitions. Semin. Cancer Biol. 75, 38–48 (2021).
Winkler, L. et al. Functional elements of the cis-regulatory lincRNA-p21. Cell Rep. 39, 110687 (2022).
Huang, J. et al. m(6)A-modified lincRNA Dubr is required for neuronal development by stabilizing YTHDF1/3 and facilitating mRNA translation. Cell Rep. 41, 111693 (2022).
Wei, M. et al. Axon-enriched lincRNA ALAE is required for axon elongation via regulation of local mRNA translation. Cell Rep. 35, 109053 (2021).
Wang, E. et al. Targeting an RNA-binding protein network in acute myeloid leukemia. Cancer Cell 35, 369–384.e367 (2019).
Qin, H. et al. RNA-binding proteins in tumor progression. J. Hematol. Oncol. 13, 90 (2020).
Jiang, X. et al. EIF4A3-induced circARHGAP29 promotes aerobic glycolysis in docetaxel-resistant prostate cancer through IGF2BP2/c-Myc/LDHA signaling. Cancer Res. 82, 831–845 (2022).
Huang, X. et al. Insulin-like growth factor 2 mRNA-binding protein 1 (IGF2BP1) in cancer. J. Hematol. Oncol. 11, 88 (2018).
Ding, W. B. et al. HBV/pregenomic RNA increases the stemness and promotes the development of HBV-related HCC through reciprocal regulation with insulin-like growth factor 2 mRNA-binding protein 3. Hepatology 74, 1480–1495 (2021).
Pan, Z. et al. EWSR1-induced circNEIL3 promotes glioma progression and exosome-mediated macrophage immunosuppressive polarization via stabilizing IGF2BP3. Mol. Cancer 21, 16 (2022).
Zhao, B. et al. circPLIN2 promotes clear cell renal cell carcinoma progression by binding IGF2BP proteins and miR-199a-3p. Cell Death Dis. 13, 1030 (2022).
Yao, B. et al. CircEZH2/miR-133b/IGF2BP2 aggravates colorectal cancer progression via enhancing the stability of m(6)A-modified CREB1 mRNA. Mol. Cancer 21, 140 (2022).
Ravanidis, S., Kattan, F. G. & Doxakis, E. Unraveling the pathways to neuronal homeostasis and disease: mechanistic insights into the role of RNA-binding proteins and associated factors. Int. J. Mol. Sci. 19, 2280 (2018).
Kim, J. H. et al. SON drives oncogenic RNA splicing in glioblastoma by regulating PTBP1/PTBP2 switching and RBFOX2 activity. Nat. Commun. 12, 5551 (2021).
Monday, H. R. et al. Presynaptic FMRP and local protein synthesis support structural and functional plasticity of glutamatergic axon terminals. Neuron 110, 2588–2606.e2586 (2022).
Zeng, Q. et al. Aberrant hyperexpression of the RNA-binding protein FMRP in tumors mediates immune evasion. Science 378, eabl7207 (2022).
Kosti, A. et al. The RNA-binding protein SERBP1 functions as a novel oncogenic factor in glioblastoma by bridging cancer metabolism and epigenetic regulation. Genome Biol. 21, 195 (2020).
Xu, Y. et al. ERα is an RNA-binding protein sustaining tumor cell survival and drug resistance. Cell 184, 5215–5229.e5217 (2021).
Capper, D. et al. DNA methylation-based classification of central nervous system tumours. Nature 555, 469–474 (2018).
Shi, H. et al. m(6)A facilitates hippocampus-dependent learning and memory through YTHDF1. Nature 563, 249–253 (2018).
Worpenberg, L. et al. Ythdf is a N6-methyladenosine reader that modulates Fmr1 target mRNA selection and restricts axonal growth in Drosophila. EMBO J. 40, e104975 (2021).
Yu, J. et al. Dynamic m6A modification regulates local translation of mRNA in axons. Nucleic Acids Res. 46, 1412–1423 (2018).
Cong, P. et al. Identification of the role and clinical prognostic value of target genes of m6A RNA methylation regulators in glioma. Front. Cell Dev. Biol. 9, 709022 (2021).
Clark, K. D., Lee, C., Gillette, R. & Sweedler, J. V. Characterization of neuronal RNA modifications during non-associative learning in aplysia reveals key roles for tRNAs in behavioral sensitization. ACS Cent. Sci. 7, 1183–1190 (2021).
Puhl, D. L. et al. Electrospun fiber-mediated delivery of neurotrophin-3 mRNA for neural tissue engineering applications. Acta Biomater. 155, 370–385 (2023).
Sapiro, A. L. et al. Illuminating spatial A-to-I RNA editing signatures within the Drosophila brain. Proc. Natl. Acad. Sci. USA 116, 2318–2327 (2019).
Urnov, F. D. et al. Genome editing with engineered zinc finger nucleases. Nat. Rev. Genet 11, 636–646 (2010).
Hoban, M. D. et al. CRISPR/Cas9-mediated correction of the sickle mutation in human CD34+ cells. Mol. Ther. 24, 1561–1569 (2016).
Bjurström, C. F. et al. Reactivating Fetal hemoglobin expression in human adult erythroblasts through BCL11A knockdown using targeted endonucleases. Mol. Ther. Nucleic Acids 5, e351 (2016).
Peterson, C. W. et al. Long-term multilineage engraftment of autologous genome-edited hematopoietic stem cells in nonhuman primates. Blood 127, 2416–2426 (2016).
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007).
Miyoshi, N. et al. Reprogramming of mouse and human cells to pluripotency using mature microRNAs. Cell Stem Cell 8, 633–638 (2011).
Polo, J. M. et al. A molecular roadmap of reprogramming somatic cells into iPS cells. Cell 151, 1617–1632 (2012).
Katti, A. et al. CRISPR in cancer biology and therapy. Nat. Rev. Cancer 22, 259–279 (2022).
Winkle, M., El-Daly, S. M., Fabbri, M. & Calin, G. A. Noncoding RNA therapeutics—challenges and potential solutions. Nat. Rev. Drug Discov. 20, 629–651 (2021).
Sahin, U. & Türeci, Ö. Personalized vaccines for cancer immunotherapy. Science 359, 1355–1360 (2018).
Vormehr, M., Türeci, Ö. & Sahin, U. Harnessing tumor mutations for truly individualized cancer vaccines. Annu Rev. Med 70, 395–407 (2019).
Ren, F. et al. Shortwave-infrared-light-emitting probes for the in vivo tracking of cancer vaccines and the elicited immune responses. Nat. Biomed. Eng. 8, 726–739 (2023).
Sahin, U., Karikó, K. & Türeci, Ö. mRNA-based therapeutics-developing a new class of drugs. Nat. Rev. Drug Discov. 13, 759–780 (2014).
Boczkowski, D., Nair, S. K., Snyder, D. & Gilboa, E. Dendritic cells pulsed with RNA are potent antigen-presenting cells in vitro and in vivo. J. Exp. Med. 184, 465–472 (1996).
Kallen, K. J. & Theß, A. A development that may evolve into a revolution in medicine: mRNA as the basis for novel, nucleotide-based vaccines and drugs. Ther. Adv. Vaccines 2, 10–31 (2014).
Heine, A., Juranek, S. & Brossart, P. Clinical and immunological effects of mRNA vaccines in malignant diseases. Mol. Cancer 20, 52 (2021).
Chaudhary, N., Weissman, D. & Whitehead, K. A. mRNA vaccines for infectious diseases: principles, delivery and clinical translation. Nat. Rev. Drug Discov. 20, 817–838 (2021).
Lim, M., Xia, Y., Bettegowda, C. & Weller, M. Current state of immunotherapy for glioblastoma. Nat. Rev. Clin. Oncol. 15, 422–442 (2018).
Vik-Mo, E. O. et al. Therapeutic vaccination against autologous cancer stem cells with mRNA-transfected dendritic cells in patients with glioblastoma. Cancer Immunol. Immun. 62, 1499–1509 (2013).
Beck, J. D. et al. mRNA therapeutics in cancer immunotherapy. Mol. Cancer 20, 69 (2021).
Yang, J. et al. Intratumoral delivered novel circular mRNA encoding cytokines for immune modulation and cancer therapy. Mol. Ther. Nucleic Acids 30, 184–197 (2022).
Niu, D., Wu, Y. & Lian, J. Circular RNA vaccine in disease prevention and treatment. Signal Transduct. Target Ther. 8, 341 (2023).
Nishimura, K. et al. Antibody-dependent cellular cytotoxicity of iPS cell-derived natural killer T cells by Anti-GD2 mAb for neuroblastoma. Cancer Sci. 116, 884–896 (2025).
Kushner, B. H. et al. A phase II trial of Naxitamab plus stepped-up dosing of GM-CSF for patients with high-risk neuroblastoma in first complete remission. Clin. Cancer Res. 31, 1877–1884 (2025).
Li, Y. et al. Using mpMRI, PSMA, and GRPR PET to assess focal therapy eligibility and plan treatment in biopsy-proven low-/intermediate-risk prostate cancer: a whole-mount pathology-based study. Clin. Nucl. Med. 50, 410–418 (2025).
Koppula, P., Zhuang, L. & Gan, B. Cystine transporter SLC7A11/xCT in cancer: ferroptosis, nutrient dependency, and cancer therapy. Protein Cell 12, 599–620 (2021).
Lee, H.-G., Wheeler, M. A. & Quintana, F. J. Function and therapeutic value of astrocytes in neurological diseases. Nat. Rev. Drug Discov. 21, 339–358 (2022).
Deng, T. et al. Advances in mRNA nanomedicines for malignant brain tumor therapy. Smart Mater. Med. 4, 257–265 (2023).
Zhu, Y., Zhu, L., Wang, X. & Jin, H. RNA-based therapeutics: an overview and prospectus. Cell Death Dis. 13, 644 (2022).
Zamecnik, P. C. & Stephenson, M. L. Inhibition of Rous sarcoma virus replication and cell transformation by a specific oligodeoxynucleotide. Proc. Natl. Acad. Sci. USA 75, 280–284 (1978).
Paunovska, K., Loughrey, D. & Dahlman, J. E. Drug delivery systems for RNA therapeutics. Nat. Rev. Genet. 23, 265–280 (2022).
Teplyuk, N. M. et al. Therapeutic potential of targeting microRNA-10b in established intracranial glioblastoma: first steps toward the clinic. EMBO Mol. Med. 8, 268–287 (2016).
Ruan, W. et al. Brain-targeted CRISPR/Cas9 nanomedicine for effective glioblastoma therapy. J. Control Release 351, 739–751 (2022).
Wu, T., Liu, Y., Cao, Y. & Liu, Z. Engineering macrophage exosome disguised biodegradable nanoplatform for enhanced sonodynamic therapy of glioblastoma. Adv. Mater. 34, e2110364 (2022).
Su, J. et al. TfR aptamer enhanced blood-brain barrier penetration of biomimetic nanocomplexes for intracellular transglutaminase 2 imaging and silencing in glioma. Small 18, e2203448 (2022).
Zhang, Q. et al. Antisense oligonucleotide therapy for H3.3K27M diffuse midline glioma. Sci. Transl. Med. 15, eadd8280 (2023).
Katsushima, K. et al. Targeting the Notch-regulated non-coding RNA TUG1 for glioma treatment. Nat. Commun. 7, 13616 (2016).
Melnick, K. et al. Contemporary RNA therapeutics for glioblastoma. Neuromol. Med. 24, 8–12 (2022).
Sood, A. K. et al. Adrenergic modulation of focal adhesion kinase protects human ovarian cancer cells from anoikis. J. Clin. Investig. 120, 1515–1523 (2010).
Taddei, M. L., Giannoni, E., Fiaschi, T. & Chiarugi, P. Anoikis: an emerging hallmark in health and diseases. J. Pathol. 226, 380–393 (2012).
Groc, L. & Choquet, D. Linking glutamate receptor movements and synapse function. Science 368, eaay4631 (2020).
Blanquie, O. & Bradke, F. Cytoskeleton dynamics in axon regeneration. Curr. Opin. Neurobiol. 51, 60–69 (2018).
Ljubojevic, N., Henderson, J. M. & Zurzolo, C. The ways of actin: why tunneling nanotubes are unique cell protrusions. Trends Cell Biol. 31, 130–142 (2021).
Dahlstroem, C., Paraschiakos, T., Sun, H. & Windhorst, S. Cryo-EM structures of actin binding proteins as tool for drug discovery. Biochem. Pharm. 214, 115680 (2023).
Jung, E. et al. Neuronal signatures in cancer. Int. J. Cancer 147, 3281–3291 (2020).
Zhu, Y. et al. MLK3 is associated with poor prognosis in patients with glioblastomas and actin cytoskeleton remodeling in glioblastoma cells. Front. Oncol. 10, 600762 (2020).
Welsch, T. et al. Eps8 is increased in pancreatic cancer and required for dynamic actin-based cell protrusions and intercellular cytoskeletal organization. Cancer Lett. 255, 205–218 (2007).
Koyuncu, S. et al. Rewiring of the ubiquitinated proteome determines ageing in C. elegans. Nature 596, 285–290 (2021).
Ru, Q. et al. TIAM1-mediated synaptic plasticity underlies comorbid depression-like and ketamine antidepressant-like actions in chronic pain. J. Clin. Invest. 132, e158545 (2022).
Opazo, P. et al. CaMKII triggers the diffusional trapping of surface AMPARs through phosphorylation of stargazin. Neuron 67, 239–252 (2010).
Hevia, L. G. & Fanarraga, M. L. Microtubule cytoskeleton-disrupting activity of MWCNTs: applications in cancer treatment. J. Nanobiotechnol. 18, 181 (2020).
Vaughan, S. & Dawe, H. R. Common themes in centriole and centrosome movements. Trends Cell Biol. 21, 57–66 (2011).
Kapitein, L. C. & Hoogenraad, C. C. Building the neuronal microtubule cytoskeleton. Neuron 87, 492–506 (2015).
He, Q. et al. Targeting TUBB2B inhibits triple-negative breast cancer growth and brain-metastatic colonization. J. Exp. Clin. Cancer Res. 44, 55 (2025).
Jordan, M. A. & Wilson, L. Microtubules as a target for anticancer drugs. Nat. Rev. Cancer 4, 253–265 (2004).
Ishiwata, T., Matsuda, Y. & Naito, Z. Nestin in gastrointestinal and other cancers: effects on cells and tumor angiogenesis. World J. Gastroenterol. 17, 409–418 (2011).
Calderaro, J. et al. Nestin as a diagnostic and prognostic marker for combined hepatocellular-cholangiocarcinoma. J. Hepatol. 77, 1586–1597 (2022).
Yang, J. et al. Nestin negatively regulates postsynaptic differentiation of the neuromuscular synapse. Nat. Neurosci. 14, 324–330 (2011).
Moss, J. et al. Fine processes of Nestin-GFP-positive radial glia-like stem cells in the adult dentate gyrus ensheathe local synapses and vasculature. Proc. Natl. Acad. Sci. USA 113, E2536–E2545 (2016).
Satelli, A. & Li, S. Vimentin in cancer and its potential as a molecular target for cancer therapy. Cell Mol. Life Sci. 68, 3033–3046 (2011).
Fan, Y. et al. CircNR3C2 promotes HRD1-mediated tumor-suppressive effect via sponging miR-513a-3p in triple-negative breast cancer. Mol. Cancer 20, 25 (2021).
Zhang, N. et al. Isorhapontigenin (ISO) inhibits EMT through FOXO3A/METTL14/VIMENTIN pathway in bladder cancer cells. Cancer Lett. 520, 400–408 (2021).
Triolo, D. et al. Vimentin regulates peripheral nerve myelination. Development 139, 1359–1367 (2012).
Yang, D. et al. Axon-like protrusions promote small cell lung cancer migration and metastasis. Elife 8, e50616 (2019).
Venkataramani, V. et al. Disconnecting multicellular networks in brain tumours. Nat. Rev. Cancer 22, 481–491 (2022).
Cao, Y. Tumorigenesis as a process of gradual loss of original cell identity and gain of properties of neural precursor/progenitor cells. Cell Biosci. 7, 61 (2017).
Murphy, S. F. et al. Connexin 43 inhibition sensitizes chemoresistant glioblastoma cells to temozolomide. Cancer Res. 76, 139–149 (2016).
Chen, Q. et al. Carcinoma–astrocyte gap junctions promote brain metastasis by cGAMP transfer. Nature 533, 493–498 (2016).
Grossman, S. A. et al. Talampanel with standard radiation and temozolomide in patients with newly diagnosed glioblastoma: a multicenter phase II trial. J. Clin. Oncol. 27, 4155–4161 (2009).
Shram, M. J. et al. The novel uncompetitive NMDA receptor antagonist esmethadone (REL-1017) has no meaningful abuse potential in recreational drug users. Transl. Psychiatry 13, 192 (2023).
Krishnan, N. et al. A modular approach to enhancing cell membrane-coated nanoparticle functionality using genetic engineering. Nat. Nanotechnol. 19, 345–353 (2024).
Sun, C. et al. Spatially resolved multi-omics highlights cell-specific metabolic remodeling and interactions in gastric cancer. Nat. Commun. 14, 2692 (2023).
Toi, P. T. et al. In vivo direct imaging of neuronal activity at high temporospatial resolution. Science 378, 160–168 (2022).
Hamada, S. et al. An engineered channelrhodopsin optimized for axon terminal activation and circuit mapping. Commun. Biol. 4, 461 (2021).
Zhao, Z. et al. Two-photon synthetic aperture microscopy for minimally invasive fast 3D imaging of native subcellular behaviors in deep tissue. Cell 186, 2475–2491.e2422 (2023).
Liu, R. et al. From 2D to 3D co-culture systems: a review of co-culture models to study the neural cells interaction. Int. J. Mol. Sci. 23, 13116 (2022).
Lu, T. et al. Decoding transcriptional identity in developing human sensory neurons and organoid modeling. Cell 187, 7374–7393.e7328 (2024).
Elemento, O., Leslie, C., Lundin, J. & Tourassi, G. Artificial intelligence in cancer research, diagnosis and therapy. Nat. Rev. Cancer 21, 747–752 (2021).
Terstappen, G. C., Meyer, A. H., Bell, R. D. & Zhang, W. Strategies for delivering therapeutics across the blood–brain barrier. Nat. Rev. Drug Discov. 20, 362–383 (2021).
Ma, F. et al. Neurotransmitter-derived lipidoids (NT-lipidoids) for enhanced brain delivery through intravenous injection. Sci. Adv. 6, eabb4429 (2020).
Derakhshan, A., Chen, Z. & Van Waes, C. Therapeutic small molecules target inhibitor of apoptosis proteins in cancers with deregulation of extrinsic and intrinsic cell death pathways. Clin. Cancer Res. 23, 1379–1387 (2017).
Carneiro, B. A. & El-Deiry, W. S. Targeting apoptosis in cancer therapy. Nat. Rev. Clin. Oncol. 17, 395–417 (2020).
Morana, O., Wood, W. & Gregory, C. D. The apoptosis paradox in cancer. Int. J. Mol. Sci. 23, 1328 (2022).
Xu, D. et al. TBK1 suppresses RIPK1-driven apoptosis and inflammation during development and in aging. Cell 174, 1477–1491.e1419 (2018).
Park, G. et al. Caspase activation and caspase-mediated cleavage of app is associated with amyloid β-protein-induced synapse loss in Alzheimer’s disease. Cell Rep. 31, 107839 (2020).
Roberts, S. L. et al. Bim contributes to the progression of Huntington’s disease-associated phenotypes. Hum. Mol. Genet 29, 216–227 (2020).
Han, X. et al. Neuronal SH2B1 attenuates apoptosis in an MPTP mouse model of Parkinson’s disease via promoting PLIN4 degradation. Redox Biol. 52, 102308 (2022).
Gong, Y. et al. The role of necroptosis in cancer biology and therapy. Mol. Cancer 18, 100 (2019).
Yang, T. et al. Triggering endogenous Z-RNA sensing for anti-tumor therapy through ZBP1-dependent necroptosis. Cell Rep. 42, 113377 (2023).
Vucur, M. et al. Sublethal necroptosis signaling promotes inflammation and liver cancer. Immunity 56, 1578–1595.e1578 (2023).
Mitroshina, E. V., Saviuk, M. & Vedunova, M. V. Necroptosis in CNS diseases: focus on astrocytes. Front. Aging Neurosci. 14, 1016053 (2022).
Oñate, M. et al. The necroptosis machinery mediates axonal degeneration in a model of Parkinson's disease. Cell Death Differ. 27, 1169–1185 (2020).
Wang, Y. et al. A nuclease that mediates cell death induced by DNA damage and poly(ADP-ribose) polymerase-1. Science 354, aad6872 (2016).
Zhu, H. et al. PARP inhibitors in pancreatic cancer: molecular mechanisms and clinical applications. Mol. Cancer 19, 49 (2020).
Ghonim, M. A. et al. Targeting PARP-1 with metronomic therapy modulates MDSC suppressive function and enhances anti-PD-1 immunotherapy in colon cancer. J. Immunother. Cancer 9, e001643 (2021).
Fricker, M. et al. Neuronal cell death. Physiol. Rev. 98, 813–880 (2018).
Petrelli, F. et al. Poly(ADP-ribose) polymerase is not involved in the neuroprotection exerted by azithromycin against ischemic stroke in mice. Eur. J. Pharm. 791, 518–522 (2016).
Park, H. et al. PAAN/MIF nuclease inhibition prevents neurodegeneration in Parkinson’s disease. Cell 185, 1943–1959.e1921 (2022).
Mou, Y. et al. Ferroptosis, a new form of cell death: opportunities and challenges in cancer. J. Hematol. Oncol. 12, 34 (2019).
Yang, M. et al. COMMD10 inhibits HIF1α/CP loop to enhance ferroptosis and radiosensitivity by disrupting Cu-Fe balance in hepatocellular carcinoma. J. Hepatol. 76, 1138–1150 (2022).
Zhang, Y. et al. Loss of COPZ1 induces NCOA4-mediated autophagy and ferroptosis in glioblastoma cell lines. Oncogene 40, 1425–1439 (2021).
Yee, P. P. et al. Neutrophil-induced ferroptosis promotes tumor necrosis in glioblastoma progression. Nat. Commun. 11, 5424 (2020).
Moujalled, D., Strasser, A. & Liddell, J. R. Molecular mechanisms of cell death in neurological diseases. Cell Death Differ. 28, 2029–2044 (2021).
Rao, Z. et al. Pyroptosis in inflammatory diseases and cancer. Theranostics 12, 4310–4329 (2022).
Wu, J., Wang, L. & Xu, J. The role of pyroptosis in modulating the tumor immune microenvironment. Biomark. Res 10, 45 (2022).
Wang, B. et al. GSDMD in peripheral myeloid cells regulates microglial immune training and neuroinflammation in Parkinson’s disease. Acta Pharm. Sin. B 13, 2663–2679 (2023).
Moonen, S. et al. Pyroptosis in Alzheimer’s disease: cell type-specific activation in microglia, astrocytes and neurons. Acta Neuropathol. 145, 175–195 (2023).
Tang, D. et al. The molecular machinery of regulated cell death. Cell Res. 29, 347–364 (2019).
Jiang, X. et al. A RIPK3-independent role of MLKL in suppressing parthanatos promotes immune evasion in hepatocellular carcinoma. Cell Discov. 9, 7 (2023).
Xiang, H. et al. Targeting autophagy-related protein kinases for potential therapeutic purpose. Acta Pharm. Sin. B 10, 569–581 (2020).
Liu, X., Zhuang, L. & Gan, B. Disulfidptosis: disulfide stress-induced cell death. Trends Cell Biol. 34, 327–337 (2023).
Xie, J., Yang, Y., Gao, Y. & He, J. Cuproptosis: mechanisms and links with cancers. Mol. Cancer 22, 46 (2023).
Qin, Y. et al. Cuproptosis correlates with immunosuppressive tumor microenvironment based on pan-cancer multiomics and single-cell sequencing analysis. Mol. Cancer 22, 59 (2023).
Huang, M., Zhang, Y. & Liu, X. The mechanism of cuproptosis in Parkinson’s disease. Ageing Res. Rev. 95, 102214 (2024).
Song, J. et al. Cell-in-cell-mediated entosis reveals a progressive mechanism in pancreatic cancer. Gastroenterology 165, 1505–1521.e1520 (2023).
Erpenbeck, L. & Schön, M. P. Neutrophil extracellular traps: protagonists of cancer progression? Oncogene 36, 2483–2490 (2017).
Taifour, T. et al. The tumor-derived cytokine Chi3l1 induces neutrophil extracellular traps that promote T cell exclusion in triple-negative breast cancer. Immunity 56, 2755–2772.e2758 (2023).
Pietronigro, E. C., Della Bianca, V., Zenaro, E. & Constantin, G. NETosis in Alzheimer’s disease. Front Immunol. 8, 211 (2017).
Qiao, G. et al. Adrenergic stress regulates the exhausted phenotype of T cells in the tumor microenvironment. Trends Neurosci. 204, 165.136–165.136 (2020).
Cosentino, M. et al. Human CD4+CD25+ regulatory T cells selectively express tyrosine hydroxylase and contain endogenous catecholamines subserving an autocrine/paracrine inhibitory functional loop. Blood 109, 632–642 (2007).
Teratani, T. et al. The liver–brain–gut neural arc maintains the Treg cell niche in the gut. Nature 585, 591–596 (2020).
Zheng, C. et al. Tumor-specific cholinergic CD4(+) T lymphocytes guide immunosurveillance of hepatocellular carcinoma. Nat Cancer. 4, 1437–1454 (2023).
Wang, Y. N. et al. Vinblastine resets tumor-associated macrophages toward M1 phenotype and promotes antitumor immune response. J. Immunother Cancer 11, e007253 (2023).
Li, J. J. et al. The liver microenvironment orchestrates FGL1-mediated immune escape and progression of metastatic colorectal cancer. Nat. Commun. 14, 6690 (2023).
Xu, Z. et al. Otilonium Bromide acts as a selective USP28 inhibitor and exhibits cytotoxic activity against multiple human cancer cell lines. Biochem. Pharm. 215, 115746 (2023).
Hou, F. J. et al. Chelidonine enhances the antitumor effect of lenvatinib on hepatocellular carcinoma cells. Onco Targets Ther. 12, 6685–6697 (2019).
Li, J. et al. Rocuronium bromide suppresses esophageal cancer via blocking the secretion of C-X-C motif chemokine ligand 12 from cancer-associated fibroblasts. J. Transl. Med. 21, 248 (2023).
Fan, J. H. et al. Integrating network pharmacology deciphers the action mechanism of Zuojin capsule in suppressing colorectal cancer. Phytomedicine 96, 153881 (2022).
Sun, Y. et al. Jatrorrhizine inhibits mammary carcinoma cells by targeting TNIK-mediated Wnt/β-catenin signalling and epithelial-mesenchymal transition (EMT). Phytomedicine 63, 153015 (2019).
Yousef, E. H., Abo El-Magd, N. F. & El Gayar, A. M. Carvacrol enhances anti-tumor activity and mitigates cardiotoxicity of sorafenib in thioacetamide-induced hepatocellular carcinoma model through inhibiting TRPM7. Life Sci. 324, 121735 (2023).
Lerro, C. C. et al. Pesticide exposure and incident thyroid cancer among male pesticide applicators in Agricultural Health Study. Environ. Int. 146, 106187 (2021).
Hu, H. J. et al. Blockade of the forward Na(+) /Ca(2+) exchanger suppresses the growth of glioblastoma cells through Ca(2+) -mediated cell death. Br. J. Pharm. 176, 2691–2707 (2019).
Liu, Z., Cheng, Q., Ma, X. & Song, M. Suppressing effect of Na(+)/Ca(2+) exchanger (NCX) inhibitors on the growth of melanoma cells. Int. J. Mol. Sci. 23, 901 (2022).
Ahmed, E. A., Alkuwayti, M. A. & Ibrahim, H. M. Atropine is a suppressor of epithelial-mesenchymal transition (EMT) that reduces stemness in drug-resistant breast cancer cells. Int. J. Mol. Sci. 23, 9849 (2022).
Cui, X., Wang, Q., Liu, X. & Kang, C. Levetiracetam: a potent sword against microglia polarization in gliomas. Clin. Cancer Res. 30, 1073–1075 (2024).
Lv, S. et al. Integrated analysis reveals FOXA1 and Ku70/Ku80 as targets of ivermectin in prostate cancer. Cell Death Dis. 13, 754 (2022).
Zhang, C. et al. Methionine secreted by tumor-associated pericytes supports cancer stem cells in clear cell renal carcinoma. Cell Metab. 36, 778–792.e710 (2024).
Li, F. et al. Blocking methionine catabolism induces senescence and confers vulnerability to GSK3 inhibition in liver cancer. Nat. Cancer 5, 131–146 (2024).
Zhang, C. et al. Inhibition of GABAA receptors in intestinal stem cells prevents chemoradiotherapy-induced intestinal toxicity. J. Exp. Med. 219, e20220541 (2022).
Ali, O. M., Alotaibi, M. T., Zaki, Y. H. & Amer, H. H. Design, synthesis, and spectroscopic studies of some new α-aminophosphonate analogues derived from 4-hydroxybenzaldehyde with special reference to anticancer activity. Drug Des. Dev Ther. 16, 2589–2599 (2022).
Jamwal, A. et al. Glabridin plays dual action to intensify anti-metastatic potential of paclitaxel via impeding CYP2C8 in liver and CYP2J2/EETs in tumor of an orthotopic mouse model of breast cancer. Chem. Biol. Interact. 382, 110605 (2023).
Jeong, J. H. et al. Tumor-derived OBP2A promotes prostate cancer castration resistance. J. Exp. Med. 220, e20211546 (2023).
Zamora-Sánchez, C. J., Bello-Alvarez, C., Rodríguez-Dorantes, M. & Camacho-Arroyo, I. Allopregnanolone promotes migration and invasion of human glioblastoma cells through the protein tyrosine kinase c-Src activation. Int J. Mol. Sci. 23, 4996 (2022).
Drljača, J. et al. Diazepam diminishes temozolomide efficacy in the treatment of U87 glioblastoma cell line. CNS Neurosci. Ther. 28, 1447–1457 (2022).
Wen, X. et al. Evans blue-modified radiolabeled fibroblast activation protein inhibitor as long-acting cancer therapeutics. Theranostics 12, 422–433 (2022).
Yuan, D. et al. NMDAR antagonists suppress tumor progression by regulating tumor-associated macrophages. Proc. Natl. Acad. Sci. USA 120, e2302126120 (2023).
Vicente-Ruiz, S. et al. Poly-l-glutamic acid modification modulates the bio-nano interface of a therapeutic anti-IGF-1R antibody in prostate cancer. Biomaterials 301, 122280 (2023).
Chida, K. et al. The role of lidocaine in cancer progression and patient survival. Pharm. Ther. 259, 108654 (2024).
Cao, L. Q. et al. Exosomal miR-21 regulates the TETs/PTENp1/PTEN pathway to promote hepatocellular carcinoma growth. Mol. Cancer 18, 148 (2019).
Simeoli, R. et al. Exosomal cargo including microRNA regulates sensory neuron to macrophage communication after nerve trauma. Nat. Commun. 8, 1778 (2017).
Liu, X. et al. A functionalized collagen-I scaffold delivers microRNA 21-loaded exosomes for spinal cord injury repair. Acta Biomater. 154, 385–400 (2022).
Liu, Y. et al. miR-19a promotes colorectal cancer proliferation and migration by targeting TIA1. Mol. Cancer 16, 53 (2017).
Mak, H. K. et al. MicroRNA-19a-PTEN axis is involved in the developmental decline of axon regenerative capacity in retinal ganglion cells. Mol. Ther. Nucleic Acids 21, 251–263 (2020).
Liu, M. et al. Zinc-dependent regulation of ZEB1 and YAP1 coactivation promotes epithelial-mesenchymal transition plasticity and metastasis in pancreatic cancer. Gastroenterology 160, 1771–1783.e1771 (2021).
Abyadeh, M. et al. Key genes and biochemical networks in various brain regions affected in Alzheimer’s disease. Cells 11, 987 (2022).
Dou, R. et al. EMT-cancer cells-derived exosomal miR-27b-3p promotes circulating tumour cells-mediated metastasis by modulating vascular permeability in colorectal cancer. Clin. Transl. Med. 11, e595 (2021).
Chen, K. et al. Hypoxic pancreatic cancer-derived exosomal miR-30b-5p promotes tumor angiogenesis by inhibiting GJA1 expression. Int. J. Biol. Sci. 18, 1220–1237 (2022).
Moreau, M. P. et al. Altered MicroRNA expression profiles in postmortem brain samples from individuals with schizophrenia and bipolar disorder. Biol. Psychiatr. 69, 188–193 (2011).
Poon, V. Y. et al. miR-27b shapes the presynaptic transcriptome and influences neurotransmission by silencing the polycomb group protein Bmi1. BMC Genom. 17, 777 (2016).
Li, H. et al. miR-519a enhances chemosensitivity and promotes autophagy in glioblastoma by targeting STAT3/Bcl2 signaling pathway. J. Hematol. Oncol. 11, 70 (2018).
Zhuang, Z., Zhang, L. & Liu, C. SNHG14 upregulation was a molecular mechanism underlying MPP(+) neurotoxicity in dopaminergic SK-N-SH cells via SNHG14-miR-519a-3p-ATG10 ceRNA pathway. Neurotox. Res. 40, 553–563 (2022).
Qiu, S. et al. Gastric cancer-derived exosomal miR-519a-3p promotes liver metastasis by inducing intrahepatic M2-like macrophage-mediated angiogenesis. J. Exp. Clin. Cancer Res. 41, 296 (2022).
Hong, S. et al. Inhibition of tumor progression and M2 microglial polarization by extracellular vesicle-mediated microRNA-124 in a 3D microfluidic glioblastoma microenvironment. Theranostics 11, 9687–9704 (2021).
Jiang, D. et al. Neuron-derived exosomes-transmitted miR-124-3p protect traumatically injured spinal cord by suppressing the activation of neurotoxic microglia and astrocytes. J. Nanobiotechnol. 18, 105 (2020).
Li, Z. et al. M2 microglial small extracellular vesicles reduce glial scar formation via the miR-124/STAT3 pathway after ischemic stroke in mice. Theranostics 11, 1232–1248 (2021).
Ouyang, Q. et al. Brain-penetration and neuron-targeting DNA nanoflowers co-delivering miR-124 and rutin for synergistic therapy of Alzheimer’s disease. Small 18, e2107534 (2022).
Esteves, M. et al. MicroRNA-124-3p-enriched small extracellular vesicles as a therapeutic approach for Parkinson’s disease. Mol. Ther. 30, 3176–3192 (2022).
Namkung, H. et al. The miR-124-AMPAR pathway connects polygenic risks with behavioral changes shared between schizophrenia and bipolar disorder. Neuron 111, 220–235.e229 (2023).
Vermeire, S. et al. ABX464 (obefazimod) for moderate-to-severe, active ulcerative colitis: a phase 2b, double-blind, randomised, placebo-controlled induction trial and 48 week, open-label extension. Lancet Gastroenterol. Hepatol. 7, 1024–1035 (2022).
Ma, C. et al. miR-182 targeting reprograms tumor-associated macrophages and limits breast cancer progression. Proc. Natl. Acad. Sci. USA 119, e2114006119 (2022).
Roser, A. E. et al. miR-182-5p and miR-183-5p act as GDNF mimics in dopaminergic midbrain neurons. Mol. Ther. Nucleic Acids 11, 9–22 (2018).
Ding, W. et al. Astrocytes-derived exosomes pre-treated by berberine inhibit neuroinflammation after stroke via miR-182-5p/Rac1 pathway. Int. Immunopharmacol. 118, 110047 (2023).
Soheilifar, M. H. et al. Concomitant overexpression of mir-182-5p and mir-182-3p raises the possibility of IL-17-producing Treg formation in breast cancer by targeting CD3d, ITK, FOXO1, and NFATs: a meta-analysis and experimental study. Cancer Sci. 112, 589–603 (2021).
Shu, L. et al. Bile exosomal miR-182/183-5p increases cholangiocarcinoma stemness and progression by targeting HPGD and increasing PGE2 generation. Hepatology 79, 307–322 (2024).
Rigg, E. et al. Inhibition of extracellular vesicle-derived miR-146a-5p decreases progression of melanoma brain metastasis via Notch pathway dysregulation in astrocytes. J. Extracell. Vesicles 12, e12363 (2023).
Fan, C. et al. Microglia secrete miR-146a-5p-containing exosomes to regulate neurogenesis in depression. Mol. Ther. 30, 1300–1314 (2022).
Kim, H. K. et al. Plasma microRNA expression levels and their targeted pathways in patients with major depressive disorder who are responsive to duloxetine treatment. J. Psychiatr. Res. 110, 38–44 (2019).
Wu, C. S. et al. EZH2-mediated epigenetic silencing of tumor-suppressive let-7c/miR-99a cluster by hepatitis B virus X antigen enhances hepatocellular carcinoma progression and metastasis. Cancer Cell Int. 23, 199 (2023).
Yang, J. et al. MicroRNA-99a is a potential target for regulating hypothalamic synaptic plasticity in the peri/postmenopausal depression model. Cells. 8, 1081 (2019).
Hu, H. F. et al. The antipsychotic drug aripiprazole suppresses colorectal cancer by targeting LAMP2a to induce RNH1/miR-99a/mTOR-mediated autophagy and apoptosis. Adv. Sci. 11, e2409498 (2024).
Yang, L. et al. Long non-coding RNA HOTAIR promotes exosome secretion by regulating RAB35 and SNAP23 in hepatocellular carcinoma. Mol. Cancer 18, 78 (2019).
Zhao, J., Li, H. & Chang, N. LncRNA HOTAIR promotes MPP+-induced neuronal injury in Parkinson’s disease by regulating the miR-874-5p/ATG10 axis. Excli J. 19, 1141–1153 (2020).
Zhang, Y. et al. MALAT1-KTN1-EGFR regulatory axis promotes the development of cutaneous squamous cell carcinoma. Cell Death Differ. 26, 2061–2073 (2019).
Li, K. et al. Micro electrical fields induced MSC-sEVs attenuate neuronal cell apoptosis by activating autophagy via lncRNA MALAT1/miR-22-3p/SIRT1/AMPK axis in spinal cord injury. J. Nanobiotechnol. 21, 451 (2023).
Lukowski, S. W. et al. A single-cell transcriptome atlas of the adult human retina. EMBO J. 38, e100811 (2019).
Hussen, B. M. et al. A diagnostic panel for acquired immune-mediated polyneuropathies based on the expression of lncRNAs. Front. Immunol. 12, 643615 (2021).
Malakar, P. et al. Long noncoding RNA MALAT1 regulates cancer glucose metabolism by enhancing mTOR-mediated translation of TCF7L2. Cancer Res. 79, 2480–2493 (2019).
Lu, C. et al. DNA-methylation-mediated activating of lncRNA SNHG12 promotes temozolomide resistance in glioblastoma. Mol. Cancer 19, 28 (2020).
Sun, Y. et al. In vivo self-assembled small RNA targets H19 lncRNA for the treatment of colorectal cancer. J. Control Release 358, 142–160 (2023).
Wan, P. et al. LncRNA H19 initiates microglial pyroptosis and neuronal death in retinal ischemia/reperfusion injury. Cell Death Differ. 27, 176–191 (2020).
Safari, M. R. et al. Genomic variants within the long non-coding RNA H19 confer risk of breast cancer in Iranian population. Gene 701, 121–124 (2019).
Wen, S. et al. Long non-coding RNA NEAT1 promotes bone metastasis of prostate cancer through N6-methyladenosine. Mol. Cancer 19, 171 (2020).
Wang, C. et al. Stress induces dynamic, cytotoxicity-antagonizing TDP-43 nuclear bodies via paraspeckle LncRNA NEAT1-mediated liquid-liquid phase separation. Mol. Cell 79, 443–458.e447 (2020).
Park, M. K. et al. NEAT1 is essential for metabolic changes that promote breast cancer growth and metastasis. Cell Metab. 33, 2380–2397.e2389 (2021).
Huang, X. et al. Engineered exosomes as targeted lncRNA MEG3 delivery vehicles for osteosarcoma therapy. J. Control Release 343, 107–117 (2022).
Balusu, S. et al. MEG3 activates necroptosis in human neuron xenografts modeling Alzheimer’s disease. Science 381, 1176–1182 (2023).
Bayarmaa, B. et al. Association of LncRNA MEG3 polymorphisms with efficacy of neoadjuvant chemotherapy in breast cancer. BMC Cancer 19, 877 (2019).
Yan, H. et al. Cisplatin induces pyroptosis via activation of MEG3/NLRP3/caspase-1/GSDMD pathway in triple-negative breast cancer. Int. J. Biol. Sci. 17, 2606–2621 (2021).
Acknowledgements
This study was funded by the National Natural Science Foundation of China (Nos. 82201594, 81502582), National Key Research Project of MOST (2023YFA0915000), Fund of the Affiliated Xiangshan Hospital of Wenzhou Medical University, and Wenzhou Institute University of Chinese Academy of Sciences. Funding was also provided by the Fundamental Research Funds for the Central Universities (N182004002, N2220002), Natural Science Foundation of Liaoning Province (2021-MS-104, 2022-YGJC-39, 2022-MS-228), Fundamental Scientific Research Fund of Liaoning Provincial Education Department (LJKQZ2021002), and supported by the Open project of State Key Laboratory of Animal Biotech Breeding (No. 2025SKLAB6-13) and Construction Project of Liaoning Provincial Key Laboratory, China (2022JH13/10200026).
Author information
Authors and Affiliations
Contributions
S.Z., L.Y., M.W. and Y.H. participated in different parts of writing. S.Z., L.Y., P.L., G.Y., X.Z., J.X., M.W. and Y.H. composed and edited the manuscript. S.Z., L.Y. and Y.H. illustrated the figure and artwork in consultation with co-authors. The article has received approval from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests. M.W. is an editorial board member of Signal Transduction and Targeted Therapy, but he has not been involved in the process of manuscript handling.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, S., Yuan, L., Lin, P. et al. Cancer neuroscience: signaling pathways and new therapeutic strategies for cancer. Sig Transduct Target Ther 11, 66 (2026). https://doi.org/10.1038/s41392-025-02364-y
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41392-025-02364-y