Abstract
Study design
Prospective mixed methods implementation study.
Objectives
To: (1) implement a SCI-specific care pathway for body composition assessment (ATSCI-Nut); (2) pilot test the feasibility (reach, adoption, adherence, appropriateness, and acceptability) of the care pathway in patients with new traumatic SCI; (3) explore patient experiences with the care pathway and the effect of providing body composition information on dietary choices and (4) explore clinician experiences with the new care pathway.
Setting
Victorian Spinal Cord Service, Australia
Methods
Participants included individuals with acute SCI who received the ATSCI-Nut pathway and consented to data collection. Feasibility outcomes (reach, adoption and intervention fidelity) were collected from medical records. Acceptability and appropriateness were explored via patient semi-structured interviews and clinician focus groups.
Results
Twenty-three patients were eligible, 21 (91%) consented. Adherence to the ATSCI-Nut pathway initial assessment and review components during weeks 2–8 and >8 weeks was 86, 71 and 69%, respectively. Adherence to completing bioimpedance spectroscopy (BIS) measurements at specified time-points was 69%. However, only 43% of participants had all BIS measurements completed at specified time-points. Two themes were common to patients and clinicians: physiological and body composition changes directing focus of rehabilitation, and barriers and enablers to optimal care. One additional theme arose from patient interviews: impact of SCI on self-image and lifestyle.
Conclusions
The ATSCI-Nut pathway is a feasible and acceptable model to deliver body composition assessment despite mixed adherence to the pathway overall. However, barriers to optimal patient care and pathway adaptations need to be explored to improve adherence.

Similar content being viewed by others
Introduction
Increased adiposity and decreases in muscle mass and energy requirements occur following acute spinal cord injury (SCI), defined as the acute and rehabilitation hospital admissions immediately after injury [1, 2]. Body composition changes differ according to neurological level of injury and extent of sensory and motor impairment [1]. During the acute phase, a diet adequate in energy and protein is advised to avoid undernutrition, while during rehabilitation, a healthy lifestyle is encouraged to avoid weight gain [3]. However, body weight is a crude measure that doesn’t distinguish between muscle and fat. A 2022 review and clinical practice guidelines published in 2018 recommend assessing resting metabolism using indirect calorimetry (IC) [1, 4] or using SCI-specific energy prediction equations when IC is unavailable [1], and monitoring body composition [1, 4] at least annually [1]. Previous guidelines from 2009 recommend assessing body composition using bioimpedance or dual energy x-ray absorptiometry (DXA) [3]. However, the guidelines do not stipulate when and how often to repeat body composition measurements, nor how to interpret and use the results in clinical decision making during the acute and rehabilitation phases after SCI.
Care pathways describe activities, content and frequency of care that patients should receive throughout the care continuum [5]. Benefits of care pathways include translation of evidence into practice, standardised care, reduced practice variation and improved patient care, safety, and outcomes [5]. The Action, Actor, Context, Target and Time (AACTT) framework can assist in care pathway design by specifying who should do what, where, for whom and when and to help characterise actions that could be measured to assess uptake and/or adherence [6].
Bioimpedance is a valid method for assessing body composition [fat-free mass (FFM) and fat mass (FM)] during the acute and rehabilitation phases after SCI [7] and this FFM measure can be used in SCI-specific energy prediction equations to inform individual energy needs [8,9,10]. After SCI, we have shown that body fat changes over time in a U-shaped trajectory, initially decreasing over the first 3–4 months and returning to baseline by 7 months [11]. These key time points were used to develop an Acute Traumatic Spinal Cord Injury Nutrition (ATSCI-Nut) Care Pathway to standardize care and translate the use of bioimpedance and a SCI-specific energy predication equation [8] into clinical practice and to guide clinical decision making about when to change the dietary intervention focus from prevention of undernutrition to health promotion. This study aimed to: (1) implement a SCI-specific care pathway for patients with new traumatic-SCI; (2) test the adoption, feasibility, adherence, appropriateness and acceptability of implementing bioelectrical impedance spectroscopy (BIS) to assess body composition and a SCI-specific energy prediction equation into clinical practice; (3) explore patient experiences with the new care pathway and patient perceptions of the effect of providing information regarding body composition on dietary choices, and to (4) explore clinician experiences with the new care pathway.
Methods
Study design and sampling pool
This was a single-site pilot prospective implementation study. A mixed methods approach guided by the theoretical framework of acceptability [12] and the implementation framework proposed by Peters et al., [13] was used to evaluate pathway implementation outcomes. The sampling pool included staff who delivered care and patients who received care according to the pathway. Evaluation included collection of implementation and outcome information, patient demographic and clinical data and patient semi-structured interviews, a staff survey and staff focus group.
Setting
This study was conducted across the acute and rehabilitation wards of the Victorian Spinal Cord Service, a statewide service for individuals who experience traumatic SCI. Care pathway components were delivered based on scheduled timeframes rather than ward location, as patients moved variably between acute and rehabilitation settings- including possible transfers back to acute care.
Ethics approval and consent
Ethics approval was received from the Human Research Ethics Committee (HREC/75733/Austin 2022). Patients gave verbal, witnessed informed consent to participate in the care pathway and additional verbal consent to participate and for audio recording of the interview. Clinician participants in the focus group provided written informed consent and completion and return of the survey implied clinicians’ consent. All methods were performed in accordance with the relevant guidelines and regulations.
Development of the ATSCI-nut pathway
The project team [dietitians, exercise physiologist (EP) and body composition experts] designed the ATSCI-nut care pathway based on clinical practice recommendations, guidelines [1, 3, 4] and recent research [7, 9,10,11]. The AACTT framework was used to help define behaviours and components of care within the pathway [6].
Components considered within the pathway to support initiation of appropriate clinical care processes and sustainable implementation included:
-
screening and referral processes;
-
setting specific actions or behaviours;
-
specifying who is to conduct each action and on whom the actions are performed;
-
timelines for clinical processes including screening, assessment and treatment and; definition of assessments to be administered when and by whom.
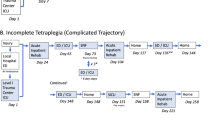
Local barriers and enablers to implementing the pathway were identified and considered. Supplementary Table 1 summarises clinical practice guidelines, and the recent literature mapped to the AACTT framework [6]. Figure 1 illustrates the ATSCI-nut care pathway.
Acute Traumatic Spinal Cord Injury (ATSCI-Nut) Pathway.
Implementation strategies
The implementation strategies utilised are reported according to the Expert Recommendations for Implementing Change (ERIC) study classifications [14].
Train and educate stakeholders
Dietitians and an EP completed an education program and a BIS competency package developed by the study lead. This included an overview of the care pathway and BIS methodology, observing the study lead conducting BIS, completing BIS with the study lead, followed by independently completing BIS with the study lead observing.
Support clinicians
The study lead was present for BIS measurements until the staff member felt confident and was competent to complete measurements independently.
Develop stakeholder interrelationships
The project team met monthly during the first six months of implementation. The multidisciplinary ward newsletter and Quality Improvement Committee were used to communicate project updates prior to study commencement, at the end of recruitment and study cessation.
Use evaluative and iterative strategies
A continuous quality improvement feedback cycle was used to make immediate adjustments to the care pathway processes.
Change infrastructure
The care pathway was included in a local nutrition clinical guideline. Additional strategies used after the initial implementation included development of a spreadsheet to track dates body composition measurements were due and clarification of processes when patients transferred between campuses.
Engage consumers/adapt and tailor the content
Plotting and printing body composition trends was introduced following patient reports of poor communication about body composition results.
Pilot test the feasibility of the ATSCI-nut care pathway
The care pathway was piloted between May 2023 and October 2024 without additional staffing resources. The duration of the pilot was determined by number of eligible patients and available staffing resources. Operational and clinical data were entered into the Research Electronic Data Capture (REDCap) secure web platform [15] by a project team member weekly.
Participants
All patients >18 years with a new traumatic-SCI admitted between May 2023-January 2024 were eligible. Patients were excluded if they had a pacemaker, were pregnant or breastfeeding, in ICU or had a concurrent severe acquired brain injury (post-traumatic amnesia duration >7 days). Eligible patients were approached and gave verbal consent to receive clinical care, as per the care pathway. Twenty-one participants undertook individualised rehabilitation as per Clinical Practice Guidelines [16] and 30–60-min of exercise training (supported by an EP) 2–3 days per week including weights and/or wheelchair skills except for 3 patients who were also participants in an intensive rehabilitation trial. All patients received the diet prescribed by their treating dietitian. BIS measurements were scheduled on patient rehabilitation timetables. Individualised body composition feedback to patients was integrated into face-to-face dietitian reviews or goal setting and healthy lifestyle counselling delivered by the dietitian and/or EP.
Six clinicians (5 dietitians and 1 exercise physiologist) who delivered care according to the care pathway between May 2023 and October 2024 were eligible and participated in the study.
Patient demographic and clinical and body composition data
Participant demographics, clinical characteristics and admission American Spinal Injury Association (ASIA) Impairment Scale (AIS) criteria [17] were collected prospectively from the electronic medical record (EMR). Weight and supine length were obtained as described previously [7].
Body composition measurements were taken, either before scheduled exercise physiology sessions or in the morning whilst participants were in bed, using a four terminal BIS instrument (SFB7, ImpediMed. Brisbane, Australia) whilst the patient was supine as previously described [7]. The BIS instrument was set to selected frequency mode and a single whole-body measurement was taken on the right side of the body at 50 kHz. Participants were advised to eat and drink as usual, empty bladder if voiding on sensation and avoid caffeine and exercise two hours before the measurement. Body composition (FFM and FM, percentage body fat) were calculated in REDCap using an SCI-specific equation FFM equation [18]. Individual energy requirements were also calculated in REDCap using a SCI-specific energy prediction equation [8] and injury and/or activity factors [19,20,21] deemed clinically appropriate by the dietitian. Data on nutritional goal setting and healthy diet counselling were gathered retrospectively from the EMR.
Staff demographic data
Demographic data on age, sex, occupation, duration of working with patients with SCI and previous experience using bioimpedance were collected via a staff survey in REDCap [15].
Adoption
The study lead collected data on the number of patients (i) admitted during the pilot study (ii) who were eligible, approached and consented to receive care and (iii) received care according to the care pathway.
Intervention delivery and adherence
Data collected included number and proportion of:
-
priority one patients (new enteral or parenteral nutrition, nil/negligible intake <25% for >5 days, specialised mealplan, high refeeding risk, significant loss of weight ≥ 10%) seen within 24 h;
-
priority two patients (predicted increased nutritional needs, ≤ 50% intake for >5 days, loss of weight 5–9.9%) seen within 48 h;
-
patients assessed and reviewed at all timepoints as per pathway timeframes;
-
patients who had assessments, reviews and clinical assessment measures performed as per the care pathway, including FFM and FM using BIS and energy requirements calculated using the SCI-specific Buchholz et al., [8] energy predication equation;
-
patients where percentage body fat guided a change in dietary management.
Adherence was classified as high (80–100%), moderate (50–79.9%) and low (<50%) [22].
Participant acceptability and appropriateness
Patient acceptability and appropriateness of the care pathway was assessed using semi-structured interviews in a subset of participants before discharge from rehabilitation. Patients were invited to participate in an interview with the study lead, gave verbal consent to participate and for audio recording of the interview. Sampling was purposive and aimed to recruit 10–15 patients of diverse age, sex and injury severity to gain a broad understanding of acceptability and appropriateness. The interview guide was composed of predetermined semi-structured questions and accompanied by follow-up probes and questions that emerged from the interview (Supplementary Table 2). Interviews were conducted in person with only the interviewer (study lead - trained in qualitative research methods) and participant present (one patient’s spouse was present) and lasted 10–33 min.
A focus group was conducted with clinicians who provided clinical care following the care pathway using a semi-structured interview guide in May 2024 (Supplementary Table 3). Clinicians provided written and verbal recorded consent for interview participation. The staff focus group was conducted, recorded and transcribed via Microsoft Teams and lasted 51 min. The interviewer had a pre-existing clinical relationship with most patients and a pre-existing professional relationship as a senior colleague with all clinicians.
The interview and focus group guide questions were developed based on the study aims, existing literature [1, 3, 4, 9,10,11, 23] and the key dimensions of acceptability [13, 24]. Each interview and the focus group were electronically audio recorded, de-identified (using numerical coding) by the study lead who conducted the interview; and transcribed verbatim using artificial intelligence software (Otter.au Los Altos, CA). The transcriptions were verified for accuracy against the audio recording by the study lead and corrected as required. All participants were offered the opportunity to check transcripts for accuracy.
Data management and analysis
Demographic, clinical and operational data
After data entry into REDCap was completed, data were exported into Excel and/or Stata/BE 17.0 (College Station, TX) for analysis. Descriptive statistics including medians and interquartile ranges were used for continuous variables due to non-normal data distribution. Categorical variables and intervention delivery and adherence were reported as counts and proportions.
Patient interviews and staff focus groups
Interview and focus group transcripts were managed using NVivo 14.23.3 to facilitate thematic analysis [25]. Data collection and analysis for the patient interviews were completed simultaneously. Recruitment ceased when data saturation was reached. Five clinicians participated in the focus group. One researcher (KD) read and corrected interview and focus group transcripts in their entirety and completed initial open coding, which were reviewed by a second researcher (NK) after four interviews and at the completion of all interviews. Key concepts were identified from the data using an iterative and reflective process [25]. The two researchers then grouped the codes into themes.
Results
Staff characteristics
Six clinicians (1 male) comprising five dietitians (including study lead) and one EP completed BIS assessments. Clinician median age was 46 years (IQR 36, 58). Years of experience working with patients with SCI varied; < 1 year (n = 2),1–2 years (n = 1), 5–10 years (n = 2), and > 20 years (n = 1). Two staff had experience using bioimpedance.
Adoption
Thirty-four patients with a new acute traumatic-SCI were admitted and 23 were eligible for inclusion in the study. Eleven patients were ineligible: severe acquired brain injury (n = 6) and ventilator-dependent (n = 5). Ninety-one percent of the 23 new acute traumatic-SCI persons who were eligible consented (n = 21). One patient declined participation, and one patient was unable to be approached for consent.
Reach
Participants median age was 56 (IQR 54, 64) years, median time post-injury 12 days (IQR 6, 16), 76% were male and 81% had tetraplegia. Four participants with tetraplegia and one participant with paraplegia had motor complete SCI (AIS A-B) (Table 1). Median BMI at initial assessment was 24.8 (IQR 23.5, 29.1) kg/m2. 48% of participants were overweight or obese using WHO BMI cut-offs of >25 kg/m2 and >30 kg/m2 respectively [26].
Intervention delivery and adherence
Adherence to initial assessment timeframes outlined in the care pathway was high. Eighty-seven percent of priority 1 and 83% of priority 2 patients were assessed within 24 and 48 h of admission (Table 2). Participants had a mean of 3.00 ± 1.23 BIS measurements (range 1–5) completed. Four participants had one BIS measurement (2 discharged home, 1 transferred to general rehabilitation, 1 not participating in rehabilitation). The median time on the care pathway was 154 days (IQR 113, 179). Adherence for completing dietetic reviews as per pathway timeframes was moderate (71% reviewed fortnightly during weeks 2–8, 69% reviewed every 5 weeks until discharge from dietetics, discharge from or cessation of active rehabilitation). Adherence to completing BIS measurements when due was moderate (69%). Only 43% of participants had all BIS measurements completed within pathway timeframes, indicating low adherence to the pathway overall. The main reasons for non-completion of BIS measurements and estimation of energy requirements were patient unavailable (28%), BIS measurement inaccurate in one undernourished patient (18%), bed rest due to pressure injury (14%), clinician unplanned leave (11%) or patient unwell (11%). Goal setting and healthy diet counselling were initiated for 71% of participants. Percentage body fat guided a change in dietary management for 57% of participants.
Participant and clinician acceptability and appropriateness
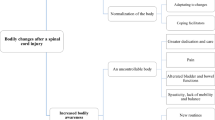
Twelve patients (4 female) were interviewed (Supplementary Table 4), and five clinicians (4 dietitians and 1 EP) participated in the focus group. Three key themes were identified from the interviews and focus group. One theme was unique to patients: (i) impact of SCI on self-image and lifestyle whist the other two themes were common to patients and clinicians; (ii) physiological and body composition changes directing focus of rehabilitation and (iii) barriers and enablers to optimal care. Table 3 summarises patient and clinician themes, codes and quotes. Interviews and the focus group suggested the care pathway was appropriate and acceptable albeit opportunities for improvement were identified.
Discussion
The findings demonstrate that many facets of body composition assessment using BIS are feasible, acceptable and appropriate to patients and clinicians and BIS has the potential to be a useful tool to support patient care and staff clinical decision making. However, low adherence to all components of the pathway suggests that adaptations are needed for improvement, and many such opportunities to improve overall pathway adherence were identified.
Implementing the care pathway into a real-world acute spinal unit presented numerous challenges. Adoption was moderate (62%) due to the high incidence of patients with concomitant acquired brain injury and requiring ventilation. Consequently, the eligibility criteria were modified to include ventilated patients. The 91% consent rate was higher than 80% reported in another study implementing cardiometabolic risk factor monitoring and receipt of health promotion activities in an inpatient rehabilitation SCI centre [27]. Adherence to the dietetic assessment and review components of the pathway was highest at initial assessment (86%) and declined progressively during weeks 2–8 (71%) and >8 weeks post injury (69%). This is likely a reflection of clinical demand exceeding capacity, with allocation of resources to higher acuity/priority patients and competing medical priorities such as patients requiring bedrest for pressure injury management. Similar adherence trends have been reported in another implementation study in head and neck cancer patients [28].
The proportion of patients who had all BIS measurements at all timepoints within pathway timeframes was low (43%) and reflects the complex clinical environment in terms of aligning patient and clinician availability, the need for patients to be supine, able to be weighed and weighed on the same day. The proportion of BIS measurements and energy requirements performed when due was moderate (69%). The main barriers which impeded pathway adherence included patient medical complications, patient and clinician availability, clinician priorities, frequency and number of BIS measurements per week and resources. Time constraints, available resources, insufficient staff and staff turnover have been described as implementation barriers [29]. Although not reflected in the adherence data, staff reported not providing timely body composition feedback to patients due to the multiple steps involved (obtaining weight, entering variables into the computer to calculate body composition, plotting the results, providing patient feedback). This was reflected in patients describing poor communication of their body composition results and the demands, fatigue and cognitive load of rehabilitation affecting their receptiveness to receiving the feedback. However, despite these challenges the results suggest it is feasible to implement body composition assessment using bioimpedance into clinical practice with ongoing support, teamwork, training and simple instructions. Use of a simpler, cheaper, single frequency bioimpedance device may increase uptake at other facilities.
The thematic analysis highlights that patients and clinicians found body composition assessment acceptable and appropriate and helpful in directing the focus of rehabilitation. Care pathways clearly define care expectations for patients and are a means of monitoring progress [29]. This was reflected in patients having an awareness of expected body composition changes, their individual body composition progress and how to optimise their body composition. Patients and clinicians used body composition information for motivation and preferred visual presentation of body composition results (graphs). Patients found body composition results inspiring and clinicians used the detailed body composition information to provide positive reinforcement and to improve patient engagement in physical activity and weight management discussions. The study findings are aligned with Holm et al. [26] who reported that 30% of participants who had DXA measurements, found feedback on DXA motivating for weight management [27]. Dietitians felt that energy requirement calculations were more accurate than weight-based estimates and used the combination of body composition (FFM and % body fat) and energy requirement trends in their clinical decision making and to plan counselling. Similarly, patients described changing food choices and adjusting dietary patterns to minimise weight gain and to optimise muscle such as including additional protein. La Vela et al., [29] found similar nutrition beliefs and eating behaviours in a study of people living with long term SCI [30]. For example, calorie restriction, eating less and changing dietary patterns to manage or control weight; and a larger focus on including protein and fruits and vegetables [30]. Additional benefits of the care pathway included increased frequency of monitoring weight and patients proactively weighing themselves. However, staff acknowledged moral distress when providing body composition feedback to patients with high level tetraplegia due to patients limited ability to engage in physical activity and the role of food as a source of enjoyment. Clinicians felt BIS could be useful to monitor the impact of functional electrical stimulation (FES) cycling on body composition in these patients. Furthermore, body composition information was inaccurate in one undernourished patient. However, overall body composition assessment was considered to be acceptable and appropriate and had a positive impact on patients’ dietary choices.
Implementation and evaluation are a continuous process following introduction of a care pathway [30], hence further adaptations to the care pathway are required. Patients identified a need for more individualised management, detailed and specific advice about portion sizes, calories and more frequent monitoring and feedback. Aligned with the patient feedback, clinicians reported pathway sustainability would be enhanced by limiting body composition measurements to 1–2 patients per week. The adherence data also supports reducing the frequency of BIS assessments, however, further investigation is required to determine if this affects clinical management and outcomes. Other strategies to improve adherence include access to a mobile device to improve efficiency in calculating and graphing body composition results, hence enabling provision of real-time body composition feedback. Prioritising body composition measurements in patients with incomplete injuries and patients with high level tetraplegia interested in FES cycling was also suggested. Hence, there was clear support for ongoing body composition measurement albeit with adaptations to the care pathway.
Study strengths include the pragmatic study design which ensured implementation and evaluation of the care pathway in a genuine clinical setting and the mixed method approach which enabled triangulation of the results. Study limitations include the small sample size, predominantly including individuals with tetraplegia and implementation at a single health service, specialising in acute traumatic SCI. This limits generalisability to persons with non-traumatic SCI and paraplegia. Consequently, confirmation of these findings in a larger patient cohort comprising more individuals with paraplegia and non-traumatic SCI across multiple health care settings is warranted. Other limitations include the BIS measurements were not taken under standardised conditions due to the pragmatic nature of the study [31]. Furthermore, clinicians and patients may not have spoken freely during the interviews and focus group as the study lead was a senior colleague and had a clinical relationship with most patients. The study lead also coded the data therefore bias may have been introduced, however this was minimised during analysis by inclusion of a second coder who was unfamiliar with participants. Furthermore, staffing shortages post COVID-19 may have impacted the delivery and adherence to the care pathway.
This study demonstrates strong support for the feasibility of using BIS to assess body composition and as a useful tool to enable use of SCI-specific energy requirement prediction equations when IC is unavailable in clinical practice. Overall, patients and staff found body composition assessment acceptable and appropriate and beneficial to support patient care, inform dietary choices and staff clinical decision making. Adaptations to the care pathway to improve adherence and further research exploring which patients would most benefit from body composition measures are warranted.
Data availability
Deidentified data described in the manuscript will be made available upon reasonable request.
References
Farkas GJ, Sneij A, McMillan DW, Tiozzo E, Nash MS, Gater JrDR. Energy expenditure and nutrient intake after spinal cord injury: a comprehensive review and practical recommendations. Br J Nutr. 2022;128:863–87.
Ko Hn-Y. Management and rehabilitation of spinal cord injuries. Singapore: Springer; 2022. Available from: https://doi.org/10.1007/978-981-19-0228-4. https://public.ebookcentral.proquest.com/choice/PublicFullRecord.aspx?p=6992139. https://www.vlebooks.com/vleweb/product/openreader?id=none&isbn=9789811902284.
Academy of Nutrition and Dietetics. Spinal Cord Injury (SCI) Evidence-Based Nutrition Practice Guideline 2009 October 2018. Available from: https://andeal.org/topic.cfm?menu=5292&pcat=3487&cat=5448.
Nash MS, Sabharwal S, Taylor AJ, Groah SL, Gater JrDR, Dyson-Hudson TA, et al. Identification and management of cardiometabolic risk after spinal cord injury: clinical practice guideline for health care providers. Lawrence, Kansas: Allen Press Publishing Services Inc. 2018;24:379–423.
Dewar SL, Porter J. The effect of evidence-based nutrition clinical care pathways on nutrition outcomes in adult patients receiving non-surgical cancer treatment: a systematic review. Nutr Cancer. 2018;70:404–12.
Presseau J, McCleary N, Lorencatto F, Patey AM, Grimshaw JM, Francis JJ. Action, actor, context, target, time (AACTT): a framework for specifying behaviour. Implementation Science. 2019;14:102.
Panisset MG, Desneves K, Ward LC, Rafferty J, Rodi H, Roff G, et al. Bedside quantification of fat-free mass in acute spinal cord injury using bioelectrical impedance analysis: a psychometric study. Spinal Cord. 2018;56:355.
Buchholz AC, McGillivray CF, Pencharz PB. Differences in resting metabolic rate between paraplegic and able-bodied subjects are explained by differences in body composition. Am J Clin Nutr. 2003;77:371–8.
Desneves KJ, Panisset MG, Rafferty J, Rodi H, Ward LC, Nunn A, et al. Comparison of estimated energy requirements using predictive equations with total energy expenditure measured by the doubly labelled water method in acute spinal cord injury. Spinal Cord. 2019;57:562–70.
Nevin AN, Atresh SS, Vivanti A, Ward LC, Hickman IJ. Resting energy expenditure during spinal cord injury rehabilitation and utility of fat-free mass-based energy prediction equations: a pilot study. Spinal Cord Ser Cases. 2024;10:70.
Desneves KJ, Kiss N, Daly RM, Abbott G, Ward LC. Longitudinal changes in body composition and diet after acute spinal cord injury. Nutrition. 2024;120:112345.
Sekhon M, Cartwright M, Francis JJ. Development of a theory-informed questionnaire to assess the acceptability of healthcare interventions. BMC Health Serv Res. 2022;22:1–12.
Peters DH, Adam T, Alonge O, Agyepong IA, Tran N. Implementation research: what it is and how to do it. BMJ. 2013;347:f6753.
Waltz TJ, Powell BJ, Matthieu MM, Damschroder LJ, Chinman MJ, Smith JL, et al. Use of concept mapping to characterize relationships among implementation strategies and assess their feasibility and importance: results from the expert recommendations for implementing change (ERIC) study. Implementation Science. 2015;10:109.
Harris PA, Taylor R, Minor BL, Elliott V, Fernandez M, O’Neal L, et al. The REDCap consortium: building an international community of software platform partners. J Biomed Inform. 2019;95:103208.
Glinsky JV, Harvey LA, and the Australian and New Zealand Physiotherapy Clinical Practice Guidelines consortium. Australian and New Zealand Clinical Practice Guideline for the physiotherapy management of people with spinal cord injury. 2022.
Kirshblum S, Snider B, Rupp R, Read MS. Updates of the international standards for neurologic classification of spinal cord injury: 2015 and 2019. Phys Med Rehabil Clin N Am. 2020;31:319–30.
Kocina P, Heyward V. Validation of a bioimpedance equation for estimating fat-free mass of spinal cord injured adults 319. Med Sci Sports Exerc. 1997;29:55.
Farkas GJ, Gorgey AS, Dolbow DR, Berg AS, Gater DR. Caloric intake relative to total daily energy expenditure using a spinal cord injury-specific correction factor. Am J Phys Med Rehabil. 2019;98:947–52.
Long CL, Schaffel N, Geiger JW, Schiller WR, Blakemore WS. Metabolic response to injury and illness: estimation of energy and protein needs from indirect calorimetry and nitrogen balance. JPEN. 1979;3:452–6.
National Academies of Sciences E, Medicine. Applications of the Dietary Reference Intakes for Energy. Dietary Reference Intakes for Energy: National Academies Press (US); 2023.
Borrelli B. The assessment, monitoring, and enhancement of treatment fidelity in public health clinical trials. J Public Health Dent. 2011;71:S52–S63.
Felleiter P, Krebs J, Haeberli Y, Schmid W, Tesini S, Perret C. Post-traumatic changes in energy expenditure and body composition in patients with acute spinal cord injury. J Rehabil Med. 2017;49:579–84.
Sekhon M, Cartwright M, Francis JJ. Acceptability of healthcare interventions: an overview of reviews and development of a theoretical framework. BMC Health Serv Res. 2017;17:88.
Braun V, Clarke V Thematic analysis. APA handbook of research methods in psychology: Research designs: Quantitative, qualitative, neuropsychological, and biological, Vol 2, 2nd ed. Washington, DC: American Psychological Association; 2023.
Holm NJ, Møller T, Schou LH, Biering-Sørensen F. Monitoring outcome measures for cardiometabolic disease during rehabilitation and follow-up in people with spinal cord injury. Spinal Cord. 2024;62:125–32.
Findlay M, Rankin NM, Shaw T, White K, Boyer M, Milross C, et al. Best evidence to best practice: implementing an innovative model of nutrition care for patients with head and neck cancer improves outcomes. Nutrients. 2020;12:1465.
Evans-Lacko S, Jarrett M, McCrone P, Thornicroft G. Facilitators and barriers to implementing clinical care pathways. BMC Health Serv Res. 2010;10:1–6.
LaVela SL, Wu J, Nevedal AL, Frayne SM, Harris AHS, Arnow KD, et al. Nutrition and eating beliefs and behaviors among individuals with spinal cord injuries and disorders: Healthy or misconceived? Rehabil Psychol. 2024;70:1–14.
Kyle UG, Bosaeus I, De Lorenzo AD, Deurenberg P, Elia M, Gómez JM, et al. Bioelectrical impedance analysis—part I: review of principles and methods. Clin Nutr. 2004;23:1226–43.
Acknowledgements
The authors acknowledge the contributions of Dr Caroline McFarlane of the Victorian Spinal Cord Service for medical oversight of the study, Leonie Pearce for supporting the research, dietitians Angela Tremayne, Erin Mechelin, Ann Cassar and Aya Ikehiro for assistance with data collection, and the contributions of the participants and their families.
Funding
Open access funding provided by Austin Health, Victorian Health Libraries Consortium (VHLC).
Author information
Authors and Affiliations
Contributions
KJD designed the care pathway and study, collected and entered data, analysed results, and drafted the manuscript. BF and CE designed the care pathway, collected and entered data and helped write the manuscript. RMD and LCW conceived the project, provided feedback and oversight on all aspects of the project. NK conceived the project, assisted with data analysis, provided feedback and oversight on all aspects of the project.
Corresponding author
Ethics declarations
Competing interests
Author Ward consults to Impedimed Ltd. All other authors have no conflicts of interest to declare.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Desneves, K.J., Fittall, B., Elson, C. et al. Implementing body composition assessment into clinical practice in patients with acute spinal cord injury- a pilot feasibility study. Spinal Cord 64, 266–278 (2026). https://doi.org/10.1038/s41393-026-01169-2
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41393-026-01169-2