Abstract
Observational studies have shown an association between post-traumatic stress disorder (PTSD) and ischemic stroke (IS) but given the susceptibility to confounding it is unclear if these associations represent causal effects. Mendelian randomization (MR) facilitates causal inference that is robust to the influence of confounding. Using two sample MR, we investigated the causal effect of genetic liability to PTSD on IS risk. Ancestry-specific genetic instruments of PTSD and four quantitative sub-phenotypes of PTSD, including hyperarousal, avoidance, re-experiencing, and total symptom severity score (PCL-Total) were obtained from the Million Veteran Programme (MVP) using a threshold P value (P) of <5 × 10−7, clumping distance of 1000 kilobase (Mb) and r2 < 0.01. Genetic association estimates for IS were obtained from the MEGASTROKE consortium (Ncases = 34,217, Ncontrols = 406,111) for European ancestry individuals and from the Consortium of Minority Population Genome-Wide Association Studies of Stroke (COMPASS) (Ncases = 3734, Ncontrols = 18,317) for African ancestry individuals. We used the inverse-variance weighted (IVW) approach as the main analysis and performed MR-Egger and the weighted median methods as pleiotropy-robust sensitivity analyses. In European ancestry individuals, we found evidence of an association between genetic liability to PTSD avoidance, and PCL-Total and increased IS risk (odds ratio (OR)1.04, 95% Confidence Interval (CI) 1.007–1.077, P = 0.017 for avoidance and (OR 1.02, 95% CI 1.010–1.040, P = 7.6 × 10−4 for PCL total). In African ancestry individuals, we found evidence of an association between genetically liability to PCL-Total and reduced IS risk (OR 0.95 (95% CI 0.923–0.991, P = 0.01) and hyperarousal (OR 0.83 (95% CI 0.691–0.991, P = 0.039) but no association was observed for PTSD case-control, avoidance, or re-experiencing. Similar estimates were obtained with MR sensitivity analyses. Our findings suggest that specific sub-phenotypes of PTSD, such as hyperarousal, avoidance, PCL total, may have a causal effect on people of European and African ancestry’s risk of IS. This shows that the molecular mechanisms behind the relationship between IS and PTSD may be connected to symptoms of hyperarousal and avoidance. To clarify the precise biological mechanisms involved and how they may vary between populations, more research is required.
Similar content being viewed by others
Background
Post-Traumatic Stress Disorder (PTSD) has been described as an extreme response to traumatic conditions [1]. Several risk factors are implicated in the aetiology of PTSD, such as exposure to violence [1], childhood trauma [2], and educational attainment [3, 4]. However, not all individuals exposed to traumatic events end up developing PTSD [5]. The Diagnostic and Statistical Manual of Mental Disorders (DSM), DSM-IV [6], and DSM-5 [7] both employ the PTSD Checklist (PCL) to identify the symptoms of PTSD. A revised version of the PCL, known as the PCL-5, was designed to assess PTSD symptoms as defined in the DSM-5 whereas the original PCL was designed to evaluate PTSD symptoms as defined in the DSM-IV. The DSM-5 PTSD symptoms are represented by 20 items in the PCL-5. The symptoms categorization includes Exposure to actual or threatened death, serious injury, or sexual violence. Intrusive and distressing thoughts, memories, or dreams related to the traumatic event(s). Avoidance of people, places, or things that remind the person of the traumatic event(s). Negative changes in mood or cognition, such as persistent negative emotions, distorted thoughts about the event, and feelings of detachment or estrangement from others. Increased arousal or reactivity, including difficulty sleeping, irritability, anger outbursts, and hypervigilance.
The most common type of stroke, ischemic stroke (IS), is caused by a blockage in the circulatory arteries that carry blood to the brain. IS results in ~80% of all adult stroke cases [8]. The 10-year survival rate following an ischemic stroke is 25%, whereas the survival rate following a hemorrhagic stroke is 18% [9].
Observational studies support an association between PTSD and IS risk [10,11,12,13,14]. A growing body of research also indicates that PTSD predicts stroke. For example, in a longitudinal study, individuals with PTSD had a higher risk of any stroke (hazard ratio (HR) 3.37, 95% CI 2.44–4.67 and ischemic stroke (HR = 3.47, 95% CI 2.23–5.39) after controlling for demographic data and medical comorbidities [10]. The biological basis of the relationship between PTSD is poorly understood. However, a few viable explanations have been put forth suggesting possible biological underpinnings of stroke. One hypothesis is that the outcome of stroke can be attributed to the physiological impacts of long-term stress on the body, which can affect the cardiovascular system by altering immunological function, increasing inflammation and oxidative stress, and altering blood vessel response to stress [15, 16].
However, observational studies are inherently limited in estimating causal relationships due to a vulnerability to confounders, amongst a number of other limitations [17,18,19,20]. Mendelian randomization (MR) can be used to circumvent this challenge and to date has not been leveraged to investigate this question. MR utilises genetic variants in an instrumental variable framework to investigate the causal relationship between a putative biological risk factor and a disease of interest [21, 22].
We therefore, used MR to investigate the relationship between genetic liability to PTSD and PTSD sub-phenotypes—hyperarousal, avoidance, re-experiencing, and PCL-Total (summation of the three quantitative phenotypes) as defined by Stein et al. [23], with risk of IS in European and African ancestry individuals.
Methods
Study populations
Genome-wide association study summary statistics for PTSD traits were obtained from the Million Veteran Program (MVP) for both African and European ancestry individuals (Supplementary ST1). The MVP comprises five PTSD phenotype consisting of a case-control phenotype and four quantitative PTSD phenotypes: re-experiencing, avoidance, hyperarousal, and total index of recent symptom severity (Table 1) [23]. The MVP obtained ethical approval from the Veteran Affairs Central Institutional Review Board in accordance with the principles outlined in the Declaration of Helsinki. Genetic association estimates for IS risk in African ancestry individuals were obtained from the Consortium of Minority Population Genome-Wide Association Studies of Stroke (COMPASS). COMPASS is a GWAS meta-analysis of 13 African ancestry cohorts including 3734 cases and 18,317 controls (Supplementary ST1). All participants provided written, informed consent, and institutional review boards approved each of the respective studies [24]. Genetic association estimates for IS and other stroke subtypes in European ancestry individuals were obtained from the MEGASTROKE consortium (34,217 cases and 406,111 controls) [25].
Statistical analyses
We constructed our genetic instrument for each PTSD trait in three steps. First, SNPs reaching a threshold of P < 5 × 10−7 were selected from the corresponding PTSD trait GWAS. This threshold was used because in several PTSD traits a portion of SNPs did not reach the standard genome-wide significance threshold of P < 5 × 10−8. To therefore increase the number of SNPs available for the primary and secondary analyses, we used P < 5 × 10−7. Secondly, we pruned these SNPs and selected independent SNPs by using a clumping window of 1000 kb and r2 < 0.01. Genetic association estimates for these SNPs were extracted from COMPASS and MEGASTROKE summary statistics for African and European ancestry, respectively. Third, we harmonised the effects by ensuring that the effect of the SNP on the exposure and the effect of the SNP on the outcome had the same effect allele. Palindromic SNPs with effect allele greater than 40% were not considered as viable instruments. In our current analysis, we made the necessary assumptions for MR analysis, i.e., instrumental variables (IVs) must be strongly associated with exposure of interest, and the IVs only influence the outcome phenotype via effects on risk factors [20, 26]. Any SNP that violated these assumptions was considered an invalid IV because it may cause bias in MR analysis. We used inverse-variance weighted (IVW) as the main MR method in this research. The causal estimate β was calculated with the equation wi (αi/γi). i refers to the IVs, αi connotes the association effect of IVs on IS, γi signifies the association effect of IVs with PTSD, and wi the weights of the causal effect of PTSD on IS. Where we have less than two instruments, Wald ratio was used as the main test. To corroborate the results of the MR-IVW estimates, we used MR-Egger and the weighted median MR method to determine the association of PTSD genetic liability with IS. MR-Egger accommodates the estimation of an intercept, a statistically significant intercept indicates the presence of unbalanced pleiotropy which subsequently suggests that the IVW MR estimates are more precise than the MR-Egger estimate [21]. Weighted median MR can compute causal estimates even if 50% IVs are invalid [22]. The weighted median method employs inverse-variance weight and bootstrapping in the estimations of confidence interval [22]. To estimate the strength of our instruments, we computed F- statistics for each SNP using method described by Burgess et al. [27].
Results
Genetic liability to PTSD phenotypes and stroke risk
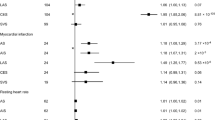
In African ancestry analysis the F-statistics of all the instrumental variables used were >10. Detailed information on the instruments is in supplementary ST2–ST6. We found no MR evidence of association between genetic liability to PTSD case-control, avoidance, re-experiencing, and IS. There was however evidence of association of genetic liability to PTSD PCL-Total and hyperarousal with decreased risk of IS. The OR of genetically predicted PTSD case-control against IS in African ancestry individuals was 1.05 (95% CI 0.794, 1.377; P = 0.750), PTSD hyperarousal against IS 0.83 (95% CI 0.691, 0.991; P = 0.039), PTSD avoidance against IS 0.98 (95% CI 0.925, 1.044; P = 0.569), PTSD re-experiencing against IS 0.88 (95% CI 0.747, 1.057; P = 0.182), and PTSD PCL-Total against IS 0.95 (95% CI 0.923, 0.991; P = 0.014) (Fig. 1, supplementary ST7–ST11).
Forest plot summarizing the effect of genetically predicted PTSD traits on IS risk in African and European populations.
In the European ancestry analysis five, 33, 30, 26, and 35 instrumental variables from PTSD case-control, hyperarousal, avoidance, re-experiencing, and PCL-Total respectively were selected from the MVP summary statistics (supplementary ST12–ST16). We found evidence of the association between genetic liability to PTSD avoidance, and PCL-Total with increased risk of IS. PTSD case-control, hyperarousal, and re-experiencing showed no statistical association with IS. The odds ratio (OR) of the genetically predicted exposure with IS [OR] per 1-standard deviation increase in PTSD traits were 1.19, 95% CI 0.992–1.441, P = 0.060 (PTSD case-control), 1.04, 95% CI 0.992–1.098, P = 0.093 (hyperarousal), 1.04, 95% CI 1.007–1.078, P = 0.017 (avoidance), 1.04, 95% CI 0.989–1.102, P = 0.117 (re-experiencing), and 1.02, 95% CI 1.010–1.040, P = 7.6 × 10−4 (PCL-Total) (Fig. 1, Supplementary ST17–ST21).
For sensitivity analysis, we used three different methods (simple median-based, weighted median-based, and MR-Egger). We did not detect any substantial horizontal pleiotropy in our analyses as indicated by the MR-Egger intercept P value (intercept close to zero and P > 0.05). The sensitivity analyses showed consistent results indicating robustness of the MR-Egger findings (Supplementary ST7–ST11, ST17–ST21). Similarly, no evidence of heterogeneity was observed (supplementary ST22) as evidenced by the non-significance of the test statistics (supplementary ST22) using MR-PRESSO. A detailed pleiotropy description of the genetic instruments is shown in supplementary ST22. In the reverse analysis, we found no evidence of significant causal relationship between IS liability against PTSD traits in either ancestral group (Supplementary ST23–ST42).
Discussion
Statement of findings
This MR study investigated the relationship between genetic liability to PTSD traits and IS risk in European and African ancestry individuals. In European ancestry individuals, we found evidence of an association between genetic liability to PTSD phenotypes including hyperarousal, avoidance, and PCL-Total, and an increased risk of IS. In evaluating the relationship between PTSD traits and the risk of other Stroke subtypes (LAS, CES, and SVS), we found no evidence of MR association between PTSD traits and stroke subtypes, except between PCL-Total with CES and between avoidance and SVS. This finding was further corroborated in pleiotropy-robust sensitivity analyses. In African ancestry individuals, no such association was found between the four PTSD phenotypes (case-control, avoidance, re-experiencing, and hyperarousal) and IS risk. However, PCL-Total was genetically associated with a reduced risk of IS in African ancestry individuals.
Our MR findings in European ancestry individuals are consistent with several observational studies that have reported that PTSD is a risk factor for IS [10, 28,29,30,31,32,33]. Several observational studies have found that PTSD is associated with higher stroke risk while adjusting for selected stroke risk factors [10, 28,29,30,31,32,33]. For example, after adjusting for depression, hypertension, dyslipidemia, and diabetes, a prospective study from the Taiwan National Health Insurance Research Database supported a threefold increase in IS in 5217 individuals with PTSD [34]. In another study, PTSD was shown to be associated with an increased risk of IS and transient ischemic attacks, the association remained consistent after further adjusting other stroke risk factors [28].
Furthermore, although no prior MR studies have explored the association between PTSD traits and IS risk, the genetic association between genetically predicted liability to depression and stroke risk has been previously investigated [35, 36] Using an inverse-variance weighted method, Gill et al., found no evidence of genetically determined risk of depression on IS risk in European population [35]. Similarly, Cai et al. found no association between major depressive disorder and large artery stroke, cardioembolic stroke, and/or IS [36].
Several factors may explain the observed differences in the genetic associations between PTSD traits and IS risk in European versus African ancestry individuals. First, the prevalence and expression of different PTSD traits may differ between European and African ancestry individuals and thus the proportion of IS risk that is attributable to PTSD traits may differ between the two ethnicities [37,38,39,40]. For example, although Mainous et al. found no differences in symptoms severity between Europeans and African ancestry individuals, after adjusting for trauma type, Europeans reported more anguish from re-experiencing, hyperarousal, and avoidance symptoms in response to nonphysical trauma than African ancestry individuals [41]. Second, there may be ethnic differences in questionnaire response style, which is defined as an individual’s manner of answering questions such as psychological questionnaires [42]. These cultural differences are worth noting because they have the potential to bias PTSD diagnosis or may amplify or shroud the severity of the trauma. In cross-cultural situations, Western psychiatric diagnostic instruments for PTSD diagnosis may fall short of reflecting the full range of trauma reactions [43]. Instead, questionnaires that have undergone cross-cultural harmonization should be used to capture significant ancestral differences in individuals. Third, due to the dense linkage disequilibrium block within the African genome, it is possible the underlying interaction between different genetic and epigenetic differs in European versus African ancestry individuals.
The mechanisms underlying the effect of PTSD on IS may arise through behaviors such as smoking [44, 45], low exercise [45, 46], poor sleep [47, 48], and poor diet [47]. Another possible explanation is that lengthened exposure to psychological stress is connected to endothelial dysfunction, inflammation, platelet activation, and autonomic dysregulation, all of which have been shown to promote atherothrombotic incidents [45, 49,50,51].
Strengths
One major strength of our analysis is the reduced susceptibility of MR to confounding in comparison to traditional epidemiological studies. In our secondary analysis, we used different pleiotropy-robust MR methods that make different analytic assumptions, hence strengthening the credibility of this study. The data used in our analysis for both exposure and outcome phenotypes were strictly from African ancestries individuals, hence, there were no population biases in our analysis. Furthermore, there was no sample overlap between the exposure and outcome datasets.
Limitations
One limitation of this study is the number of instruments used in the African ancestry analysis, which were fewer (n = 21) than those used in the European ancestry analysis (n = 129). Rather than biological differences in the relationship between PTSD and stroke across ethnicities, it is possible that the comparatively smaller sample size of African ancestry individuals and subsequent lack of statistical power may partly explain differences between European and African ancestry individuals. We used a P value (P) of < 5 × 10−7 to select SNPs for our genetic instruments, this may have resulted in the inclusion of weak or invalid instruments, however, this was compensated by the F-statistics of >10. Other potential drawbacks of this study are that characteristic of MR analysis and assumptions [52]. As discussed above, another potential limitation is misclassification of PTSD across populations, especially in populations where scores are less validated, such as in African populations. This has necessitated a need to ensure continuous advocacy for people to report symptoms of PTSD. The MVP data is predominantly male while the COMPASS data is predominantly female, this is a potential limitation as it is known that sex and gender differences play roles in disease heterogeneity.
Conclusion
Our study showed that there may be differences in the genetic susceptibility to the development of PTSD and its associated risks for ischemic stroke between individuals of European and African ancestries. This highlights the importance of considering genetic and ancestral diversity when investigating the genetic basis of complex disorders such as PTSD and its related outcomes.
Data availability
All data used in this study are publicly available unless otherwise stated.
References
Nöthling J, Suliman S, Martin L, Simmons C, Seedat S. Differences in abuse, neglect, and exposure to community violence in adolescents with and without PTSD and depression. J Interpers Violence. 2019;34:4357–83.
McLaughlin KA, Koenen KC, Bromet EJ, Karam EG, Liu H, Petukhova M, et al. Childhood adversities and post-traumatic stress disorder: evidence for stress sensitisation in the World Mental Health Surveys. Br J Psychiatry. 2017;211:280–8.
Polimanti R, Ratanatharathorn A, Maihofer AX, Choi KW, Stein MB, Morey RA, et al. Association of economic status and educational attainment with posttraumatic stress disorder a mendelian randomization study. JAMA Netw Open. 2019;2:1–17.
Shalev AY, Gevonden M, Ratanatharathorn A, Laska E, van der Mei WF, Qi W, et al. Estimating the risk of PTSD in recent trauma survivors: results of the International Consortium to Predict PTSD (ICPP). World Psychiatry. 2019;18:77–87.
McFarlane AC. The long-term costs of traumatic stress: Intertwined physical and psychological consequences. World Psychiatry. 2010;9:3–10.
Bell CC. DSM-IV: diagnostic and statistical manual of mental disorders. JAMA. 1994;272:828–9.
Tandon R. Bipolar and depressive disorders in diagnostic and statistical manual of mental disorders-5: clinical implications of revisions from diagnostic and statistical manual of mental disorders-IV. Indian J Psychol Med. 2015;37:1–4.
Karch A, Thomas SL. Autoimmune thyroiditis as a risk factor for stroke: a historical cohort study. Neurology. 2014;82:1643–52.
Jia S, Liu Y, Shen L, Liang X, Xu X, Wei Y. Botulinum toxin type A for upper limb spasticity in poststroke patients: a meta-analysis of randomized controlled trials. J Stroke Cerebrovasc Dis J Natl Stroke Assoc. 2020;29:104682.
Chen M-H, Pan T-L, Li C-T, Lin W-C, Chen Y-S, Lee Y-C, et al. Risk of stroke among patients with post-traumatic stress disorder: nationwide longitudinal study. Br J Psychiatry. 2015;206:302–7.
Spitzer C, Barnow S, Völzke H, John U, Freyberger HJ, Grabe HJ. Trauma, posttraumatic stress disorder, and physical illness: findings from the general population. Psychosom Med. 2009;71:1012–7.
Brass LM, Page WF. Stroke in former prisoners of war. J Stroke Cerebrovasc Dis J Natl Stroke Assoc. 1996;6:72–78.
Coughlin SS. Post-traumatic stress disorder and cardiovascular disease. Open Cardiovasc Med J. 2011;5:164–70.
Akosile W, Young R, Lawford B, Voisey J, Colquhoun D. PTSD symptoms associated with myocardial infarction: practical clinical implications. Australas Psychiatry. 2018;26:60–64.
Edmondson D, von Känel R. Post-traumatic stress disorder and cardiovascular disease. Lancet Psychiatry. 2017;4:320–9.
Perkins JD, Wilkins SS, Kamran S, Shuaib A. Post-traumatic stress disorder and its association with stroke and stroke risk factors: a literature review. Neurobiol Stress. 2021;14:100332.
Skrivankova VW, Richmond RC, Woolf BAR, Davies NM, Swanson SA, VanderWeele TJ, et al. Strengthening the reporting of observational studies in epidemiology using mendelian randomisation (STROBE-MR): explanation and elaboration. BMJ. 2021;375:n2233.
Smith GD, Ebrahim S. Data dredging, bias, or confounding. BMJ. 2002;325:1437–8.
Lawlor DA, Davey Smith G, Kundu D, Bruckdorfer KR, Ebrahim S. Those confounded vitamins: what can we learn from the differences between observational versus randomised trial evidence? Lancet. 2004;363:1724–7.
Fatumo S, Karhunen V, Chikowore T, Sounkou T, Udosen B, Ezenwa C et al. Metabolic traits and stroke risk in individuals of african ancestry: mendelian randomization analysis. Stroke. 2021;52:2680–84.
Bowden J, Davey Smith G, Burgess S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol. 2015;44:512–25.
Bowden J, Davey, Smith G, Haycock PC, Burgess S. Consistent estimation in mendelian randomization with some invalid instruments using a weighted median estimator. Genet Epidemiol. 2016;40:304–14.
Stein MB, Levey DF, Cheng Z, Wendt FR, Harrington K, Pathak GA, et al. Genome-wide association analyses of post-traumatic stress disorder and its symptom subdomains in the Million Veteran Program. Nat Genet. 2021;53:174–84.
Keene KL, Hyacinth HI, Bis JC, Kittner SJ, Mitchell BD, Cheng Y-C, et al. Genome-wide association study meta-analysis of stroke in 22 000 individuals of african descent identifies novel associations with stroke. Stroke. 2020;51:2454–63.
Malik R, Chauhan G, Traylor M, Sargurupremraj M, Okada Y, Mishra A, et al. Multiancestry genome-wide association study of 520,000 subjects identifies 32 loci associated with stroke and stroke subtypes. Nat Genet. 2018;50:524–37.
Didelez V, Sheehan N. Mendelian randomization as an instrumental variable approach to causal inference. Stat Methods Med Res. 2007;16:309–30.
Burgess S, Davies NM, Thompson SG. Bias due to participant overlap in two-sample Mendelian randomization. Genet Epidemiol. 2016;40:597–608.
Rosman L, Sico JJ, Lampert R, Gaffey AE, Ramsey CM, Dziura J, et al. Posttraumatic stress disorder and risk for stroke in young and middle-aged adults: a 13-year cohort study. Stroke. 2019;50:2996–3003.
Gaffey AE, Rosman L, Burg MM, Haskell SG, Brandt CA, Skanderson M, et al. Posttraumatic stress disorder, antidepressant use, and hemorrhagic stroke in young men and women: a 13-year cohort study. Stroke. 2021;52:121–9.
Beristianos MH, Yaffe K, Cohen B, Byers AL. PTSD and risk of incident cardiovascular disease in aging veterans. Am J Geriatr Psychiatry. 2016;24:192–200.
Sumner JA, Kubzansky LD, Elkind MSV, Roberts AL, Agnew-Blais J, Chen Q, et al. Trauma exposure and posttraumatic stress disorder symptoms predict onset of cardiovascular events in women. Circulation. 2015;132:251–9.
Gilsanz P, Winning A, Koenen KC, Roberts AL, Sumner JA, Chen Q, et al. Post-traumatic stress disorder symptom duration and remission in relation to cardiovascular disease risk among a large cohort of women. Psychol Med. 2017;47:1370–8.
Remch M, Laskaris Z, Flory J, Mora-McLaughlin C, Morabia A. Post-traumatic stress disorder and cardiovascular diseases: a cohort study of men and women involved in cleaning the debris of the world trade center complex. Circ Cardiovasc Qual Outcomes. 2018;11:e004572.
Goldfinger JZ. Post-traumatic stress disorder was associated with a threefold increased risk for stroke in a Taiwanese National Health Insurance Database. Evid Based Ment Health. 2015;18:e7.
Gill D, James NE, Monori G, Lorentzen E, Fernandez-Cadenas I, Lemmens R, et al. Genetically determined risk of depression and functional outcome after ischemic stroke. Stroke. 2019;50:2219–22.
Cai H, Cai B, Zhang H, Sun W, Wang Y, Zhou S, et al. Major depression and small vessel stroke: a Mendelian randomization analysis. J Neurol. 2019;266:2859–66.
Alim TN, Charney DS, Mellman TA. An overview of posttraumatic stress disorder in African Americans. J Clin Psychol. 2006;62:801–13.
Breslau J, Aguilar-Gaxiola S, Kendler KS, Su M, Williams D, Kessler RC. Specifying race-ethnic differences in risk for psychiatric disorder in a USA national sample. Psychol Med. 2006;36:57–68.
Marshall GN, Schell TL, Miles JNV. Ethnic differences in posttraumatic distress: Hispanics’ symptoms differ in kind and degree. J Consult Clin Psychol. 2009;77:1169–78.
Roberts AL, Gilman SE, Breslau J, Breslau N, Koenen KC. Race/ethnic differences in exposure to traumatic events, development of post-traumatic stress disorder, and treatment-seeking for post-traumatic stress disorder in the United States. Psychol Med. 2011;41:71–83.
Mainous AG 3rd, Smith DW, Acierno R, Geesey ME. Differences in posttraumatic stress disorder symptoms between elderly non-Hispanic Whites and African Americans. J Natl Med Assoc. 2005;97:546–9.
Franklin CL, Thompson KE. Response style and posttraumatic stress disorder (PTSD): a review. J Trauma Dissociation. 2005;6:105–23.
Patel AR, Hall BJ. Beyond the DSM-5 diagnoses: a cross-cultural approach to assessing trauma reactions. Focus (Madison). 2021;19:197–203.
Feldner MT, Babson KA, Zvolensky MJ. Smoking, traumatic event exposure, and post-traumatic stress: a critical review of the empirical literature. Clin Psychol Rev. 2007;27:14–45.
Zen AL, Whooley MA, Zhao S, Cohen BE. Post-traumatic stress disorder is associated with poor health behaviors: findings from the heart and soul study. Heal Psychol J Div Heal Psychol Am Psychol Assoc. 2012;31:194–201.
Godfrey KM, Lindamer LA, Mostoufi S, Afari N. Posttraumatic stress disorder and health: a preliminary study of group differences in health and health behaviors. Ann Gen Psychiatry. 2013;12:30.
Gavrieli A, Farr OM, Davis CR, Crowell JA, Mantzoros CS. Early life adversity and/or posttraumatic stress disorder severity are associated with poor diet quality, including consumption of trans fatty acids, and fewer hours of resting or sleeping in a US middle-aged population: a cross-sectional and prospective. Metabolism. 2015;64:1597–610.
Talbot LS, Rao MN, Cohen BE, Richards A, Inslicht SS, OʼDonovan A, et al. Metabolic risk factors and posttraumatic stress disorder: the role of sleep in young, healthy adults. Psychosom Med. 2015;77:383–91.
Koenen KC, Sumner JA, Gilsanz P, Glymour MM, Ratanatharathorn A, Rimm EB, et al. Post-traumatic stress disorder and cardiometabolic disease: improving causal inference to inform practice. Psychol Med. 2017;47:209–25.
Tawakol A, Osborne MT, Wang Y, Hammed B, Tung B, Patrich T, et al. Stress-associated neurobiological pathway linking socioeconomic disparities to cardiovascular disease. J Am Coll Cardiol. 2019;73:3243–55.
Rosman L, Sico JJ, Lampert R, Gaffey AE, Ramsey CM, Dziura J, et al. Posttraumatic stress disorder and risk for stroke in young and middle-aged adults. Stroke. 2019;50:2996–3003.
Burgess S, Davey Smith G, Davies NM, Dudbridge F, Gill D, Glymour MM, et al. Guidelines for performing Mendelian randomization investigations. Wellcome Open Res. 2020;4:186.
Acknowledgements
The authors thank Million Veteran Program (MVP) staff, researchers, and volunteers, who have contributed to MVP, especially participants who previously served their country in the military and now generously agreed to enroll in the study. (See https://www.research.va.gov/mvp/ for more details). The citation for MVP is Gaziano, J.M. et al. Million Veteran Program: A mega-biobank to study genetic influences on health and disease. J Clin Epidemiol 70, 214-23 (2016). This research is based on data from the Million Veteran Program, Office of Research and Development, Veterans Health Administration, and was supported by the Veterans Administration (VA) Cooperative Studies Program (CSP) award #G002. Data were accessed through approved dbGaP proposal #30287 entitled, “Genomic determinant of Complex Diseases in African ancestry individuals”. This work was supported by the National Institutes of Mental Health (U01MH115485). O.S. is supported by the Africa Research Excellence Fund (AREF-325-SORE-F-C0904). S.F. is supported by the Wellcome Trust grant 220740/Z/20/Z. T.C. is an international training fellow supported by the Wellcome Trust grant (214205/Z/18/Z).
Author information
Authors and Affiliations
Contributions
SF conceptualized the study and OS performed the primary analysis. All authors read and approved the final manuscript; meeting the ICMJE requirements for authorship.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Respective data/study used in this study obtained ethical clearance from different ethical boards as indicated in the method session.
Consent for publication
All authors give consent for publication
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Soremekun, O., Musanabaganwa, C., Uwineza, A. et al. A Mendelian randomization study of genetic liability to post-traumatic stress disorder and risk of ischemic stroke. Transl Psychiatry 13, 237 (2023). https://doi.org/10.1038/s41398-023-02542-y
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41398-023-02542-y
This article is cited by
-
Mendelian randomization analysis of blood metabolites and immune cell mediators in relation to GVHD and relapse
BMC Medicine (2025)
-
Mendelian randomization studies on ischemic stroke: a field synopsis and systematic review
Journal of Translational Medicine (2025)
-
A genetic exploration of the relationship between posttraumatic stress disorder and cardiovascular diseases
Translational Psychiatry (2025)
-
Shared genetic architecture of posttraumatic stress disorder with cardiovascular imaging, risk, and diagnoses
Nature Communications (2025)