Abstract
The exposure to aversive experiences during early-life affects brain maturation and induces changes in behavior. Additionally, when these experiences coincide with subtle neurodevelopmental alterations, they may contribute to the emergence of psychiatric disorders, such as schizophrenia. Studies in patients and animal models have identified changes in parvalbumin (PV) expressing inhibitory neurons, highlighting their significance in the etiology of this disorder. Most studies have been focused on the cortex, but PV+ neurons also provide inhibitory input to diencephalic regions, particularly to the thalamus (through cells in the thalamic reticular nucleus, TRN) and the habenula. Remarkably, alterations in both nuclei have been described in schizophrenia. Some of these changes in PV+ cells may be mediated by perineuronal nets (PNN), specialized regions of the extracellular matrix that often surround them and regulate their synaptic input and activity. Interestingly, the physiological maturation and integration of PV+ neurons, which involves the assembly of PNN, occurs during early postnatal life. Plasticity molecules associated to inhibitory neurons, such as PSA-NCAM, or NMDA receptors (NMDAR) can also influence the structure and function of these cells. Growing evidence also indicates that glial cells regulate the physiology of PV+ neurons by influencing their maturation and modulating their synaptic connectivity. To explore the impact of early-life aversive experiences and concomitant subtle neurodevelopmental alterations on diencephalic PV+ cells, we analyzed adult male mice subjected to a double-hit model (DHM) of schizophrenia, combining a single injection of an NMDAR antagonist at P7 and post-weaning social isolation. We observed that exploratory behavior, PV+ neurons and their associated PNN, as well as PSA-NCAM and NMDAR expression and glial cells, in the TRN and the habenula were affected by the DHM or one of its factors. To our knowledge, this is the first report on such alterations in these diencephalic structures in an animal model combining neurodevelopmental alterations and early-life stress during adolescence. Our findings complement previous work on PV+ neurons in cortical regions and underscore the importance of studying diencephalic inhibitory networks and their intricate interactions with aversive experiences and neurodevelopmental alterations during early life in the context of schizophrenia.
Similar content being viewed by others
Introduction
Fast-spiking parvalbumin (PV) expressing GABAergic neurons play an important role in brain physiology by providing inhibitory control of cortical and subcortical circuits [1]. They are involved in cortical maturation through activity-dependent changes and the control of critical periods [2, 3]. These inhibitory neurons undergo a protracted period of physiological maturation and integration into cortical and thalamic circuitry during early postnatal life [4]. Consequently, it is unsurprising that alterations in their density, connectivity, and physiology have been found in different psychiatric disorders with a neurodevelopmental origin, such as schizophrenia [5, 6]. Indeed, numerous studies in patients with schizophrenia and animal models of the disease have found alterations in the subpopulation of cortical PV+ interneurons, especially in the prefrontal cortex (PFC) [7]. By contrast, the involvement of the PV+ cells of the diencephalon has been less explored.
In the thalamus, the TRN is the major source of GABAergic inhibition, and modulates the information between the cerebral cortex and the thalamic nuclei [8]. Interestingly, many inhibitory neurons in this nucleus express PV and, in rodents, the thalamic PV+ inhibitory neurons are almost exclusively located in the TRN [9]. The projections of these neurons contact with the rest of the thalamic nuclei, including the lateral posterior nucleus (LP), the homologous of the human pulvinar nucleus, and the mediodorsal nucleus (MD) [10]. These two higher-order nuclei establish connections with association cortices and are considered, therefore, very important in psychosis research. LP is involved in somatosensory and visual processing [11, 12], while MD is a key player in higher cognitive functions, like attention and decision-making [13]. Interestingly, dysfunction of these thalamic nuclei has been linked to schizophrenia [14]. Indeed, recent neuroimaging studies from our laboratory have described volumetric and shape alterations of the thalamus and some of its nuclei in these patients [15, 16]. There is also evidence showing the existence of locally targeting PV+ neurons in the habenula [17], another diencephalic structure. Due to its role in reward processing and the regulation of dopaminergic neurotransmission, alterations in the habenula have been also documented in schizophrenia [18]. Although diencephalic dysfunctions have been found in psychiatric disorders, little is known about specific impairments of PV+ neurons. There is only one report describing decreases in the density of PV+ neurons in the TRN of patients with schizophrenia and bipolar disorder [19]. These alterations are particularly interesting because there is growing evidence of the disruption of thalamocortical circuits involving PV+ inhibitory neurons in schizophrenia [20].
In the adult brain, the structure, connectivity and activity of PV+ inhibitory neurons are controlled by components of the extracellular matrix (ECM), such as perineuronal nets (PNN) [21] and adhesion molecules, such as the polysialylated form of the neural cell adhesion molecule (PSA-NCAM) [22]. Furthermore, these molecules may also control their final maturation and subsequent plasticity during early postnatal life. On one hand, PNN provide a physical barrier, maintain local ionic homeostasis, and protect neurons against oxidative stress [23]. They regulate the inhibitory input on cortical PV+ cells and gamma oscillations, which depend on these inhibitory neurons [24, 25]. Interestingly, PNN are remarkably altered in the amygdala, as well as in the entorhinal and prefrontal cortices of subjects diagnosed with schizophrenia and psychosis [26,27,28]. On the other hand, PSA-NCAM plays a key role in the structural remodeling and the connectivity of inhibitory neurons in the adult brain [29], regulating the perisomatic inhibition of medial PFC principal neurons by PV+ basket cells [22]. Moreover, previous studies have revealed alterations in PSA-NCAM expression in patients with schizophrenia, including decreases in the PFC [12] and increases in serum levels of PSA [30]. However, nothing is known about specific alterations of PSA-NCAM expression in diencephalic structures, such as the TRN or the habenula.
The physiology of PV+ inhibitory neurons is strongly influenced by N-methyl-D-aspartate receptors (NMDARs). They are critically involved in the regulation of the synaptic input that PV+ cells receive and participate in the generation of gamma oscillations by these inhibitory neurons [31]. Interestingly, alterations in these NMDARs in PV+ cells have been described in the cerebral cortex of patients with schizophrenia [32]. The NMDARs are tetramers that include two compulsory GluN1 subunits [33]. They are highly expressed in thalamic nuclei [34] and their expression is altered in the hippocampus [35], PFC [36] and dorsomedial thalamus [37] of patients with schizophrenia. The presence of NMDARs expressing GluN2B [38] and GluN2C [39, 40] is a remarkable feature of TRN neurons and has been considered a potential therapeutic target for treating schizophrenia. On the other hand, NMDAR antagonists reduce the activity of TRN neurons [41] and affect the functional connectivity between the PFC and multiple thalamic nuclei, including the TRN [42].
There is also increasing evidence that glial cells play an important role in regulating the physiology of PV+ neurons by influencing their maturation and modulating their synaptic connectivity [43]. Interestingly, all 4 glial cell types show dysregulated patterns of gene expression in postmortem tissue from patients with schizophrenia in both cortical and subcortical regions [44].
PV+ cells appear to mature earlier in subcortical than in cortical areas, but they also complete their development during postnatal life [45]. This protracted development makes them extremely vulnerable to environmental changes and adverse experiences during early life [46]. Aversive experiences during childhood and adolescence disturb the maturation of neurons and alter the construction of neural circuits, causing functional and structural changes that last over time. Indeed, several neuroimaging studies have shown alterations in functional connectivity caused by early life stress [47, 48]. Therefore, it is considered a predisposing factor for psychiatric disorders such as schizophrenia [49]. Animal models confronted with adverse events during early life constitute a valid approach to study the influence of these experiences on the etiology of schizophrenia. One of these models is the post-weaning social isolation, which displays some of the behavioral, structural, and neurochemical changes observed in patients with schizophrenia [50,51,52]. Due to the importance of altered neurodevelopment on the etiology of schizophrenia, this paradigm has been lately combined with experimental interventions during early postnatal life, such as the single administration of an NMDAR antagonist at P7, which slightly alters the latest stages of neurodevelopment and produces certain cognitive and social impairments similar to those found in schizophrenia, as well as alterations in the inhibitory circuits of different brain regions [53]. Different studies in rats and mice, including our own [54,55,56,57], have demonstrated that this “double hit” model (DHM) combining these two approaches is robust and capable of inducing a wider spectrum of behavioral, functional and structural alterations and has also proven to be a robust and reliable developmental model of schizophrenia with predictive validity and potential as a translational mode [58]. Using this DHM we have already shown behavioral impairments, such as increases in locomotion and anxiety-related behaviors [55] and decreases in prepulse inhibition of the startle reflex (PPI) [56]. We also observed changes in the inhibitory/excitatory balance and alterations in the density of PV+ neurons and the expression of plasticity molecules in other regions in which PV+ circuits develop postnatally, such as the PFC, the hippocampus, and the retrosplenial cortex [54, 55]. However, we still do not have information on the impact of this model on the PV+ cells in the diencephalon. Therefore, based on our previous findings, we propose that the DHM model may also induce changes in the inhibitory circuits of the TRN and the habenula, indicating a potential association between these circuits and the etiopathology of schizophrenia. To this end, since the thalamus and the habenula play a crucial role in coordinating exploratory behavior [59,60,61,62], we have analyzed this parameter. Then, we have studied PV+ neurons and their associated PNN in the TRN, the habenula, and the projections of these neurons to the LP and MD. We have chosen these two nuclei because many studies have found structural and functional alterations in patients with schizophrenia and because we have previously observed effects of the DHM on the medial prefrontal cortex [51, 52] and the retrosplenial cortex [53], two association cortices that receive synaptic input from these nuclei. To have a readout of the putative effect on the excitatory/inhibitory balance, we studied the expression of the GABA and glutamate vesicular transporters (VGAT and VGLUT1). Additionally, we investigated the presence of alterations in the expression of PSA-NCAM, the constitutive subunit of NMDARs (GluN1), and the impact of the DHM on microglial and astroglial cells.
Materials and methods
Animals and experimental treatment
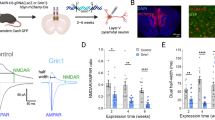
We used 32 adult male FVB mice. Three-month-old mice (1 male and 2 females) were maintained in breeding cages under standard conditions with a 12-hour light/dark cycle and ad libitum access to food and water. Following pregnancy in female mice, individual housing was implemented to avoid disturbances. At postnatal day 7 (P7), randomly selected male pups were injected intraperitoneally with MK-801 (1 mg/kg in 0.9% NaCl) or vehicle solution and after that, they were returned to their cages and stayed with their mothers until weaning at P21. At this point, from each group we randomly chose mice for isolation (n = 8) or social housing (n = 8) (Fig. 1A). All animals were sacrificed at P90. Details on the experimental procedure can be found in the supplementary methods section.
A Experimental procedure. B Scheme, based on Allen Brain Atlas, showing all studied regions: the thalamic reticular nucleus (TRN), lateral (LH) and medial (MH) subregions of habenula, the lateral posterior nucleus (LP), and the central (MDc), lateral (MDl) and medial (MDm) subregions of mediodorsal nucleus (C) Microphotograph showing the general distribution of PV+ neurons and PNN (WFA+) in a brain section, including the thalamic reticular nucleus (TRN). D High magnification of squared area in A showing the distribution of PV+ neurons and PNN in the TRN. Dotted squares indicate the 4 different ROIs used for analyses. Scale bar: 500 μm (A), 307 μm (B).
Ethics statements
All the experiments were conducted in accordance with the Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes and were approved by the Committee on Bioethics of the Universitat de València. Every effort was made to minimize the number of animals used and their suffering.
Behavioral analyses
The analysis of exploratory behavior is a continuation of a previous study from our laboratory [55]. Before their sacrifice (P90), all mice were tested in the hole-board apparatus (ANY-maze video tracking system v4.98; Stoelting Europe), and the time that animals spent exploring novel holes as well as the total time spent on exploration was measured. Details on the experimental procedure can be found in the supplementary methods section.
Perfusion and immunohistochemistry
Mice were sacrificed and brains were processed and cut as described before [55]. The tissue was processed free-floating for fluorescence immunohistochemistry using the primary and secondary antibodies, listed in Table S1 and Wisteria floribunda agglutinin (WFA) to mark PNN. Details on the experimental procedure can be found in the supplementary methods section.
Image acquisition and processing
All microphotographs were taken with a laser scanning confocal microscope (Leica, SPE, Leica Microsystems, Wetzlar, Germany). Then, we processed and analyzed them using FIJI-ImageJ Software [63]. For every analysis in the TRN, except from the study of PSA-NCAM, we selected 1 section containing the nucleus at bregma 1.70 mm and selected 4 different regions distributed dorso-ventrally (Fig. 1B, C). We used the same section for the analysis in medial and lateral habenula. Confocal z-stacks covering the whole depth of the sections were taken with 0.7 μm step size and only subsets of confocal planes with the optimal penetration level for each antibody were selected for analysis.
For every analysis, we used custom-made macros following a procedure described before [64]. In short, we subtracted the background with a rolling value of 50 converted them to 8-bit deep images, and binarized them using a determined threshold value, adjusted, and kept constant for every analysis. Details on the confocal microscopy and the macro used for each analysis are described in the supplemental methods section.
Analysis of immunohistochemical labeling
To analyze PV expressing neurons in the TRN, we estimated the density and fluorescence intensity (FI) of these cells; because of the packed organization of PV+ cells, we could not estimate their total number confidently. In addition, because PNN do not form singular structures around PV+ cells in the TRN, we decided to study only the FI of PNN immunostaining around every PV+ neuron in this region. The relationship between these parameters was also evaluated, because in a previous study in the prefrontal cortex [24] we observed a clear positive correlation between the intensities of PV and WFA immunolabelings. Since PV+ cells are only expressed in the lateral habenula, we estimated the total numbers of PV+ cells, PNN, PV+ cells surrounded by PNN, PV+ cells not being surrounded by PNN, and the ratio of PV+ cells surrounded by PNN in this subregion. To analyze PV expressing puncta in the LP and the central subregion of MD (MDc), we estimated the percentage of area covered by these structures and their FI. To analyze the expression of the GluN1 subunit of NMDARs on PV+ cells in TRN we calculated the FI of the immunoreactive puncta, as well as their density, and the percentage of area covered by these GluN1+ structures. We analyzed the density of puncta expressing inhibitory (VGAT) and excitatory (VGLUT1) markers in the neuropil of the TRN and the medial habenula where we observed a strong, homogeneous expression of both markers. Since the expression of PSA-NCAM was mainly restricted to the dorsal part of the TRN (see “results”), we decided to study the density of puncta expressing this molecule, the percentage of area covered by immunoreactive puncta and FI in this region. We also studied FI of PSA-NCAM in the lateral and medial habenula. Next, we analyzed GFAP+ astrocytes and Iba1+ microglia cells, studying the density, percentage of area covered by these markers and their FI in the TRN and medial habenula. All images were processed as described in the “Image Acquisition and Processing” section. Details on the analyses can be found in the supplementary methods section.
Statistics
After checking the homoscedasticity and the normality of the data for every parameter, we performed a 2-way ANOVA considering treatment (vehicle and MK-801) and rearing (Social and Isolation) as between-subjects factors. For the analyses in which we obtained a significant interaction factor, we performed a post hoc analysis using a Tukey test. Details can be found in the supplementary methods section.
Results
Alterations in exploratory behavior in isolated mice
We observed several alterations in the exploratory behavior. The time that animals spent exploring new holes was shorter in isolated animals (F(1,60) = 7.250; p = 0.0093; Fig. 2A; Table S2). Likewise, the total time spent exploring all holes was also decreased in isolated animals (F(1,60) = 18.52; p = < 0.0001; Fig. 2B; Table S2), but was not affected by treatment or interaction between factors.
Changes observed in exploratory behavior. Histograms representing the effects of treatment, isolation, and their interaction on the time spent exploring novel holes (seconds) (A) and time spent in all holes (seconds) (B).
Isolated mice show differences in the FI of PV+ cells and PNN in the TRN, while MK-801 treated mice show lower numbers of PV expressing neurons in the habenula
We studied how our DHM affected PV+ cells and PNN in the TRN and the habenula (Figs. 3, 4). We did not find any significant alteration in the density of PV+ somata (Fig. 3A, B; Table S2) in the TRN. However, there was a significant interaction between treatment (vehicle vs. MK-801) and rearing (social vs. isolation) in the FI of PV (F(1,24) = 4.401; p = 0.0466; Fig. 3A, C; Table S2), as well as that of WFA (F(1,24) = 4.766; p = 0.0390; Fig. 3A, D; Table S2). When studying the ratio of FI between PV and WFA, we found a significant interaction between treatment and rearing (F(1,24) = 4.719; p = 0.0399; Fig. 3A, E; Table S2) and a trend for a decrease induced by rearing (F(1,24) = 3.278; p = 0.0828, Fig. 3A, E; Table S2). Interestingly, when performing post hoc comparisons, we found that isolated animals treated with vehicle had a higher PV/WFA ratio than social animals treated with vehicle (our control group) (Tukey HSD; p = 0.0430; Table S2). Regarding the analysis in the lateral habenula (Fig. 4) we found a significant effect of the treatment (decrease) on the total number of PV+ neurons (F(1,22) = 8.0860; p = 0.0095; Fig. 4A, B; Table S3). We also observed a trend towards a decrease in the total number of PNN induced by the treatment (F(1,22) = 3.2950; p = 0.0832; Fig. 4A, C; Table S3). These alterations seem to be caused by changes in PV+/PNN− cells, since we found that this cell population was also decreased due to the treatment (F(1,22) = 6.3402; p = 0.0196; Fig. 4A, D; Table S3). No differences were observed in the total number of PV + PNN+ cells (Fig. 4A, E; Table S3). Likewise, we did not find any significant effects of the treatment or the rearing when analyzing the ratio of PV+ cells surrounded by PNN in relation to the total number of PV+ cells (Fig. 4A, F; Table S3). In our study we did not observe any PV+ cells or PNN in the medial habenula.
A1–A4 Representative confocal planes of PV+ cells (red) and PNN (green, labeled with Wisteria floribunda agglutinin: WFA) in the TRN of animals from vehicle social (A1), vehicle isolation (A2), MK-801 social (A3) and MK-801 isolation (A4) groups. B–E Histograms representing the effects of treatment, isolation, and their interaction on the density of PV+ neurons (number of cells/mm2) (B), PV fluorescence intensity (values normalized to control) (C), WFA fluorescence intensity in PNN (values normalized to control) (D), and the ratio between the fluorescence intensities of PV and WFA (E). Horizontal black lines represent the statistically significant effect of rearing. Dashed lines indicate the presence of significant interactions in the two-way ANOVA. *p < 0.05, **p < 0.01. Scale bar: 70 µm.
A1–A4 Representative confocal planes of PV+ neurons (red) and PNN (green, labeled with Wisteria floribunda agglutinin [WFA]) in the habenula of animals from vehicle social (A1), vehicle isolation (A2), MK-801 social (A3) and MK-801 isolation (double-hit mice, A4) groups. The upper right panel shows a high magnification of WFA staining surrounding the cell soma and proximal dendrites of a cell lacking PV expression. B–F Histograms representing the effects of treatment, isolation, and their interaction on the total number of PV+ neurons (B), total number of PNN (C), total number of PV+ cells not surrounded by PNN (D), total number of PV+ cells surrounded by PNN (E) and the ratio of PV + PNN+ cells in relation to the total number of PV+ cells (F). Horizontal black lines represent statistically significant effects of MK-801 treatment in a two-way ANOVA. #0.1 > p > 0.05, *p < 0.05, **p < 0.01. Scale bar: 70 µm (A1–A4) and 10 µm (A5).
The DHM produces alterations in PV+ puncta in the LP and MD
We also studied how our DHM affected the expression of PV+ puncta in the LP and the MDc (Fig. S1). In the analysis of the area covered by PV+ puncta in the MDc (Fig. S1A1–4B), we observed a trend towards an interaction (F(1,18) = 3.851; p = 0.0654; Table S3) and a significant effect (decrease) of isolation (F(1,18) = 4.770; p = 0.0424; Table S3). We also observed a trend towards an interaction in the FI (F(1,18) = 3.564; p = 0.0753; Figs. S1A1–4C; Table S3), but no differences due to treatment or isolation. In the analysis of the area covered by PV+ puncta in the LP (Figs. S1A5–6D), we did not observe any significant effect of treatment, isolation or their interaction (Table S3). However, we found a significant impact of isolation (decrease) on the FI of PV+ puncta (F(1,18) = 5.456; p = 0.0313; Figs. S1A5–6E; Table S3).
Double-hit mice show a reduced density of GluN1 immunoreactive structures in the TRN
We next focused on how our DHM affects the expression of the NMDARs compulsory subunit GluN1 on PV+ expressing neurons of the TRN (Fig. 5A). The 2-way ANOVA analysis did not reveal any significant effects of treatment, rearing, or their interaction in the FI (Fig. 5A, B; Table S2). By contrast, the area covered by GluN1 immunoreactivity was significantly reduced by the treatment (F(1,20) = 5.749; p = 0.0264; Fig. 5A, C; Table S2), and we also observed a significant interaction between rearing and treatment in this parameter (F(1,20) = 8.736; p = 0.0078; Table S2). However, the post hoc comparisons only showed a trend towards a difference between the DHM animals and the control group (Tukey HSD, p = 0.060; Table S2). We did not observe a significant interaction between treatment and rearing for the density of GluN1+ puncta (Fig. 5A, D). Consequently, post hoc comparisons were not applicable (Table S2). However, the ANOVA showed a significant effect of the treatment (F(1,20) = 8.298; p = 0.0092; Table S2) and rearing (F(1,20) = 4.534; p = 0.0458; Table S2), both reducing this parameter.
A1–A4 Representative confocal planes of PV+ somata (green) and GluN1+ puncta (red) in the TRN of animals from vehicle social (A1), vehicle isolation (A2), MK-801 social (A3) and MK-801 isolation (double-hit mice, A4) groups. B–D Histograms representing the effects of treatment, isolation, and their interaction on GluN1 fluorescence intensity (values normalized to control) (B), percentage of area covered by GluN1+ puncta (C), and density of GluN1+ puncta (number of puncta/µm2) (D). Horizontal lines represent statistically significant effects of MK-801 treatment (black) or rearing (blue) in a two-way ANOVA. Dashed black lines indicate the presence of a significant interaction between the 2 factors in the two-way ANOVA. *p < 0.05, **p < 0.01. Scale bar” 10 μm.
The DHM affects the density of puncta expressing inhibitory and excitatory synaptic markers in the habenula but not in the TRN
In the TRN, we did not find any significant effects in the FI (Fig. S2A–C; Table S2), density (Fig. S2A, D, E; Table S2), or the area covered (Fig. S2A, F, G; Table S2) by VGAT and VGLUT1 expressing puncta. We also failed to observe significant changes in the E/I ratio (see “Material and methods”) (Fig. S2A, H). Therefore, post hoc comparisons were not applicable. When studying the medial habenula, we did not find any significant difference in the density of VGLUT1+ puncta (Fig. S3A, B; Table S3). However, we observed a significant effect of the interaction in the density of VGAT+ puncta (F(1,28) = 5.994; p = 0.0209; Fig. S3A, C; Table S3). The post hoc analysis showed an increment in animals that received MK-801 and had been kept together compared to control animals (Vehicle-Social group) (Tukey HSD; p = 0.0775; Table S3). We did not observe significant alterations in the area covered by VGLUT1+ or VGAT+ puncta (Fig. S3A, D, E; Table S3), but we observed a significant effect of the treatment (reduction) in the E/I ratio (F(1,27) = 8.067; p = 0.0085; Fig. S3A, F; Table S3).
Isolated mice show an increase in the area covered by PSA-NCAM in the TRN and in the habenula
Next, we studied how our DHM affected the expression of PSA-NCAM. Similar to what has been observed in rats [65], in mice the expression was mainly restricted to the dorsal region of the TRN, and its medial and lateral tires. We did not find any significant effect or interaction in PSA-NCAM FI (Fig. S4A, B; Table S2). By contrast, we observed a significant effect of rearing, with an increase in isolated mice, in the area covered by PSA-NCAM+ puncta in the dorsal TRN (F(1,18) = 4.700; p = 0.0438; Fig. S4A, C; Table S2). The interaction of rearing and treatment was not significant and, consequently, post hoc comparisons were not applicable (Table S2). The density of PSA-NCAM+ puncta in the TRN was not affected by treatment (Fig. S4A, D; Table S2), but we observed a trend towards an effect of rearing (F(1,18) = 3.732; p = 0.0693; Fig. S4A, D). PSA-NCAM expression in the medial and lateral habenular nuclei is characterized by a punctate staining that delineates cell contours, as described previously in rats [66]. Our analysis of the FI of PSA-NCAM immunostaining in this region did not reveal any significant effect of treatment, rearing, or their interaction (Fig. S5A, B; Table S3).
The DHM affects astroglial cells in the habenula
Our analysis of glial cells in the TRN (Fig. S6A) did not reveal any significant interaction between treatment and rearing for any of the parameters analyzed. Consequently, post hoc comparisons were not applicable (Table S2). We did not observe any significant alteration in the FI, the percentage of area covered by, and the density of Iba1+ cells (Fig. S6A–D; Table S2). Similarly, the FI and the percentage of area covered by GFAP+ were not affected by any factor or their interaction (Fig. S6A, E, F; Table S2). In the habenula, Iba1+ cells were uniformly distributed, while GFAP+ cells were predominantly located in the medial habenula, which was the focus of our analysis. The density, FI, and percentage of area covered by Iba1+ cells were not affected by any experimental condition (Fig. S7A–D; Table S3). By contrast, the analysis of GFAP+ cells showed a significant interaction between treatment and rearing for the FI (F(1,20) = 5.818; p = 0.0256; Fig. S7A & E; Table S3), and in the percentage of area covered by this intermediate filament (F(1,20) = 5.843; p = 0.0253; Fig. S7A, F; Table S3). Post hoc comparisons revealed a significant decrease in both parameters (Tukey HSD; FI: p = 0.0230, Percentage of the area covered: p = 0.0273; Table S3) between the control animals and the social animals treated with MK-801. We also found a significant effect of treatment and a trend towards a decrease in the animals that received MK-801 when analyzing the FI of (F(1,20) = 4.282; p = 0.0517; Fig. S7A, E) and the percentage of area covered by (F(1,20) = 3.802; p = 0.0654; Fig. S7A, F; Table S3) GFAP+ cells.
Discussion
In the present study, we have used a DHM, combining a MK-801 injection at P7 to disturb early postnatal development and post-weaning social isolation as an early life aversive experience, which have been considered animal models for schizophrenia [50, 67]. We have focused our attention on the diencephalon, studying in detail the thalamus and the habenula, and have found changes in PV+ cells and plasticity-related molecules that regulate their connectivity, structure, and physiology. We observed differential effects of MK-801 injection and social isolation on the two studied regions. In the habenula, most of the changes were caused specifically by the single injection of MK-801 at P7. This procedure disturbs acutely ongoing neurodevelopmental processes in this early postnatal period, which may particularly affect the maturation of inhibitory circuits in this region. On the other hand, in the TRN, we observed that both social isolation and MK-801 by themselves produced significant changes. Interestingly, the significant interaction and cumulative effect in DHM mice was only observed for the expression of NMDARs in the TRN. This contrasts with what we observed in different regions of the cerebral cortex, where both MK-801 injection and social isolation have a strong impact, often culminating in double-hit mice [54, 55, 57]. The results show the complexity of the responses of inhibitory circuits in different brain regions to social isolation and altered neurodevelopmental conditions. In Table S6, we present a summary of significant results observed in the current study, as well as the alterations observed in this model in our previous experiments focusing on PV+ cells, PNN, and plasticity-related molecules in the prefrontal and retrosplenial cortices, amygdala, and hippocampus.
Numerous studies have implicated the thalamus and habenula in the pathophysiology of psychiatric disorders, particularly in schizophrenia [18, 68]. Recent work from our laboratory using structural MRI has found changes in the shape of the thalamus [16] and in the volume of different thalamic nuclei in chronic patients with schizophrenia [15]. Other studies have also observed a significantly lower absolute volume of the bilateral habenula in patients with schizophrenia compared to healthy controls [69]. Additionally, these diencephalic regions play essential roles in neurotransmission and neural circuitry. The thalamus serves as a vital relay station for sensory information and participates actively in higher cognitive processes, motor control and arousal [70, 71], while the habenula as a modulator of monoaminergic systems influencing mood and motivation [72].
In our model, we found that postweaning social isolation decreases exploratory behavior. Although this behavior is not exclusively dependent on the TRN, a previous report in rats, also using the hole board test, has found that the lesion of this inhibitory nucleus induces a reduction in this trait [73]. Nevertheless, the reduced exploratory activity observed in the present study, should not be fully compared to the effect of specific lesions, due to its global effect on several cortical and subcortical circuits. On the other hand, we have previously demonstrated that DHM induces a reduction in PPI [56], and notably, the TRN is involved in the modulation of the response to this test [74]. One of the functions of the TRN is the control of the firing mode of excitatory neurons in the thalamus [75]. In fact, a burst firing mode takes place in the thalamus of rodents prior to and during exploratory behavior, which appears to be necessary to improve signal detection [76]. Consequently, modifications in the inhibitory circuits of the TRN, such as those described in the present study, may result in alterations in this behavior. The changes in exploratory behavior may be also influenced by the alterations that we observe in the habenula, because at least its lateral region is involved in the control of this type of behavior, at least partially through dopaminergic neurotransmission: bilateral lesions of this nuclei in rats increased locomotor activity, rearing, and hole-poke responses [61, 77].
Our results in the TRN have not shown any significant alteration in the density of PV+ neurons. However, there is an effect of the DHM, and particularly of social isolation, on the ratio of FI between PV+ cells and their associated PNN. This effect may be due to an increased activity in PV+ cells (PV expression is dependent on cell activity) or to decreases in PNN complexity. In this line, previous reports in patients with schizophrenia have shown reductions in the density of PNN and destabilization of these matricellular structures [19]. However, it must be noted that in the present study we have observed a reduction (also due to social isolation) in parameters related to PV+ puncta in the MD and LP, which may indicate a decrease in the activity or the density of the synaptic inputs from the TRN. These excitatory thalamic nuclei are particularly relevant because they have connections with association cortices, including the PFC, and have been implicated in the etiopathology of schizophrenia [68]. In fact, recent work from our laboratory has described volumetric alterations of these nuclei in chronic patients with schizophrenia [15]. It is possible, however, that some of the PV+ puncta in MD and LP did not belong to TRN axons, since the thalamus also receives PV+ synaptic input from extrathalamic inhibitory nuclei and, consequently, changes in their structure/activity may be also related to alterations in this inhibitory system [78]. In the habenula, we have observed a decrease in the number of PV+ cells and only a trend towards a decrease in PNN after treatment with MK-801. The observed changes appear to be caused by alterations in PV + /PNN− cells since we have found that the population of PV+ neurons not surrounded by PNN was also decreased due to the treatment. Although there is still no information on changes in habenular PV+ neurons in patients with schizophrenia, similar to our observations, a decrease in the number of PV+ cells has been observed in the lateral habenula of adult animals subjected to chronic stress from P10-20 [79], a period close to the perturbation induced by MK-801 at P7 in our model.
Our results describing a reduction in the expression GluN1 subunit of NMDAs in the PV+ cells of the TRN in the DHM are consistent with the NMDARs hypofunction hypothesis of schizophrenia, which is supported by evidence coming from different methodological approaches [80]. Our data align with previous studies that have shown reductions in the expression of the protein and mRNA of GluN1 in postmortem studies of the thalamus of patients with schizophrenia [81, 82]. It has to be mentioned, however, that there are also other studies describing upregulations of NMDARs in patients with schizophrenia [37] and that none of the postmortem studies of the thalamus studied specifically the TRN. Moreover, it has been suggested that a reduced GABA release by TRN neurons may underlie the psychosis induced by NMDAR antagonists and that this reduction would increase the firing rate of thalamic relay neurons to the cortex [83].
Despite the effects of the DHM on the expression of NMDARs and the PV+ puncta in the MD and LD, we have not found significant effects in any of the parameters related to VGAT and VGLUT1 expressing puncta or in the E/I ratio in the TRN, suggesting that the excitatory input coming from the cortex and the extrathalamic GABAergic afferents on PV+ neurons (the TRN of the adult mouse lacks intrinsic GABAergic connections [84]) may not be affected by the DHM. Although electrophysiological studies have to be performed to examine this in detail, it has to be noted that our results in the TRN align with previous ones that showed no changes in the expression of VGLUT1 and VGAT in the PFC of patients with schizophrenia [85]. However, when studying the habenula, we have observed significant MK-801-related effects on VGAT+ puncta density and the E/I ratio. This is in line with previous research that has emphasized the significant involvement of the activity of habenular circuits in mediating the effects of early-life stress and adult chronic stress [86, 87].
To our knowledge, this is the first study examining the expression of PSA-NCAM in the TRN in mice, but our results coincide with those of Mazzetti and colleagues in the rat TRN, who found that in adult animals the expression of this molecule persisted only in its dorsal head and in its medial and lateral tires [65]. In the habenula, we have not seen any significant effect of treatment, rearing, or their interaction. The changes in PSA-NCAM expression in the TRN may be related to plastic events: the presence of this molecule in cortical interneurons reduces their synaptic input and their structural features [88]. Although we have not detected changes in the density of puncta expressing synaptic markers in the TRN, more detailed studies should be done, exploring the relationship of PSA-NCAM expression in the TRN with the different synaptic inputs that the cells in this inhibitory nucleus receive. PSA-NCAM+ also regulates the output of PV+ interneurons in the cerebral cortex: a depletion of this molecule increases the density of perisomatic PV+ puncta on pyramidal neurons [22]. Future studies should explore specifically the effects of the DHM on the output of the dorsal region of the TRN, which mainly projects to the lateral geniculate nucleus [89, 90]. The study of rodent models of chronic stress has revealed alterations in the expression of PSA-NCAM, mainly in the cerebral cortex, which may suggest its involvement in the plasticity associated with this aversive experience, not only when the stressor occurred during adulthood [29, 91], but also in early life [92]. Moreover, alterations in PSA-NCAM expression have been detected in the amygdala and PFC of patients with schizophrenia [93, 94], and certain genetic variants of the enzymes that synthesize PSA have been associated with this disorder [95, 96].
Our study on the impact of the DHM on TRN glial cells found no significant alterations in the density and area covered by microglia and astrocytes (Iba1 and GFAP). This contrasts with reported increased microglia activity in patients with schizophrenia [97], although this study was mainly focused on the gray matter of the cerebral cortex and alterations in microglia may vary between regions. Notwithstanding, more detailed studies should be performed in our model to reveal alterations in microglial morphology as a readout of changes in the activity of these cells. In contrast with these negative results in the TRN, we found significant differences in parameters of astrocytes in the habenula. These results agree with a postmortem study in the dorsolateral PFC, which showed a decrease in the area fraction immunolabeled by GFAP and an increased astroglial clustering in schizophrenia and bipolar disorder samples when compared to healthy controls, with no significant alteration in the density or distribution of Iba1+ microglia [98].
An important limitation of our study is that only males were used. This is an important issue, particularly for understanding better women health, which fortunately is starting to be solved. Moreover, the studies available, including some of our lab [88], indicate in many cases a differential vulnerability to early life adverse experiences between sexes. We are currently replicating this double hit model in male and female mice.
Concluding remarks and future perspectives
The present results compile many alterations of inhibitory networks and plasticity associated molecules in the thalamus and habenula of a double-hit schizophrenia model in mice. These findings highlight these diencephalic regions as important targets of events altering the last stages of neurodevelopment occurring during early postnatal life. They also constitute a powerful resource for studying these early-life alterations on the adult brain and may help deepen further understanding of the neurobiological basis of mental disorders in which they seem to play a critical role, such as schizophrenia.
Data availability
Materials, data, and associated protocols will be available upon request.
References
Hu H, Gan J, Jonas P. Fast-spiking, parvalbumin + GABAergic interneurons: from cellular design to microcircuit function. Science. 2014;345:1255263.
Carulli D, Verhaagen J. An extracellular perspective on CNS maturation: perineuronal nets and the control of plasticity. IJMS. 2021;22:2434.
Reichelt AC, Hare DJ, Bussey TJ, Saksida LM. Perineuronal nets: plasticity, protection, and therapeutic potential. Trends Neurosci. 2019;42:458–70.
Reh RK, et al. Critical period regulation across multiple timescales. Proc Natl Acad Sci USA 2020;117:23242–51.
Lewis DA. Inhibitory neurons in human cortical circuits: substrate for cognitive dysfunction in schizophrenia. Curr Opin Neurobiol. 2014;26:22–6.
Ruden JB, Dugan LL, Konradi C. Parvalbumin interneuron vulnerability and brain disorders. Neuropsychopharmacol. 2021;46:279–87.
Nahar L, Delacroix BM, Nam HW. The role of parvalbumin interneurons in neurotransmitter balance and neurological disease. Front Psychiatry. 2021;12:679960.
Takata N. Thalamic reticular nucleus in the thalamocortical loop. Neurosci Res. 2020;156:32–40.
Lein ES, Hawrylycz MJ, Ao N, Ayres M, Bensinger A, Bernard A, et al. Genome-wide atlas of gene expression in the adult mouse brain. Nature. 2007;445:168–76.
Pinault D. The thalamic reticular nucleus: structure, function and concept. Brain Res Rev. 2004;46:1–31.
Allen AE, Procyk CA, Howarth M, Walmsley L, Brown TM. Visual input to the mouse lateral posterior and posterior thalamic nuclei: photoreceptive origins and retinotopic order. J Physiol. 2016;594:1911–29.
Viaene AN, Petrof I, Sherman SM. Properties of the thalamic projection from the posterior medial nucleus to primary and secondary somatosensory cortices in the mouse. Proc Natl Acad Sci USA 2011;108:18156–61.
Pergola G, Danet L, Pitel AL, Carlesimo GA, Segobin S, Pariente J, et al. The regulatory role of the human mediodorsal thalamus. Trends Cogn Sci. 2018;22:1011–25.
Mørch-Johnsen L, Jørgensen KN, Barth C, Nerland S, Bringslid IK, Wortinger LA, et al. Thalamic nuclei volumes in schizophrenia and bipolar spectrum disorders – associations with diagnosis and clinical characteristics. Schizophr Res. 2023;256:26–35.
Perez-Rando M, Elvira UKA, García-Martí G, Gadea M, Aguilar EJ, Escarti MJ, et al. Alterations in the volume of thalamic nuclei in patients with schizophrenia and persistent auditory hallucinations. NeuroImage: Clin. 2022;35:103070.
Perez-Rando M, García-Martí G, Escarti MJ, Salgado-Pineda P, McKenna PJ, Pomarol-Clotet E, et al. Alterations in the volume and shape of the basal ganglia and thalamus in schizophrenia with auditory hallucinations. Prog Neuro-Psychopharmacol Biol Psychiatry. 2024;131:110960.
Webster JF, Vroman R, Balueva K, Wulff P, Sakata S, Wozny C, et al. Disentangling neuronal inhibition and inhibitory pathways in the lateral habenula. Sci Rep. 2020;10:8490.
Fakhoury M. The habenula in psychiatric disorders: more than three decades of translational investigation. Neurosci Biobehav Rev. 2017;83:721–35.
Steullet P, Cabungcal JH, Bukhari SA, Ardelt MI, Pantazopoulos H, Hamati F, et al. The thalamic reticular nucleus in schizophrenia and bipolar disorder: role of parvalbumin-expressing neuron networks and oxidative stress. Mol Psychiatry. 2018;23:2057–65.
Delevich K, Jaaro-Peled H, Penzo M, Sawa A, Li B. Parvalbumin interneuron dysfunction in a thalamo-prefrontal cortical circuit in Disc1 locus impairment mice. eNeuro. 2020;7:ENEURO.0496–19.2020.
Carceller, H, Gramuntell, Y, Klimczak, P & Nacher, J. Perineuronal nets: subtle structures with large implications. Neuroscientist. 107385842211063. https://doi.org/10.1177/10738584221106346 (2022).
Castillo-Gómez E, Varea E, Blasco-Ibáñez JM, Crespo C, Nacher J. Polysialic acid is required for dopamine D2 receptor-mediated plasticity involving inhibitory circuits of the rat medial prefrontal cortex. PLoS ONE. 2011;6:e29516.
Bosiacki M, Gąssowska-Dobrowolska M, Kojder K, Fabiańska M, Jeżewski D, Gutowska I, et al. Perineuronal nets and their role in synaptic homeostasis. IJMS. 2019;20:4108.
Carceller H, Guirado R, Ripolles-Campos E, Teruel-Marti V, Nacher J. Perineuronal nets regulate the inhibitory perisomatic input onto parvalbumin interneurons and γ activity in the prefrontal cortex. J Neurosci. 2020;40:5008–18.
Wingert JC, Sorg BA. Impact of perineuronal nets on electrophysiology of parvalbumin interneurons, principal neurons, and brain oscillations: a review. Front Synaptic Neurosci. 2021;13:673210.
Alcaide J, Guirado R, Crespo C, Blasco-Ibáñez JM, Varea E, Sanjuan J, et al. Alterations of perineuronal nets in the dorsolateral prefrontal cortex of neuropsychiatric patients. Int J Bipolar Disord. 2019;7:24.
Markota M, Sin J, Pantazopoulos H, Jonilionis R, Berretta S. Reduced dopamine transporter expression in the amygdala of subjects diagnosed with schizophrenia. Schizophr Bull. 2014;40:984–91.
Pantazopoulos H, Woo T-UW, Lim MP, Lange N, Berretta S. Extracellular matrix-glial abnormalities in the amygdala and entorhinal cortex of subjects diagnosed with schizophrenia. Arch Gen Psychiatry. 2010;67:155.
Nacher J, Guirado R, Castillo-Gómez E. Structural plasticity of interneurons in the adult brain: role of PSA-NCAM and implications for psychiatric disorders. Neurochem Res. 2013;38:1122–33.
Müller-Miny L, Thiel K, Meinert S, Hahn T, Kircher T, Nenadić I, et al. Association of polysialic acid serum levels with schizophrenia spectrum and bipolar disorder-related structural brain changes and hospitalization. Sci Rep. 2023;13:2085.
Carlén M, Meletis K, Siegle JH, Cardin JA, Futai K, Vierling-Claassen D, et al. A critical role for NMDA receptors in parvalbumin interneurons for gamma rhythm induction and behavior. Mol Psychiatry. 2012;17:537–48.
Gonzalez-Burgos G, Cho RY, Lewis DA. Alterations in cortical network oscillations and parvalbumin neurons in schizophrenia. Biol Psychiatry. 2015;77:1031–40.
Hansen, KB, Yi, F, Perszyk, RE, Menniti, FS & Traynelis, SF. NMDA receptors in the central nervous system. in NMDA Receptors (eds. Burnashev, N & Szepetowski, P) vol. 1677 1–80 (Springer New York, New York, NY, 2017).
Vukadinovic Z. NMDA receptor hypofunction and the thalamus in schizophrenia. Physiol Behav. 2014;131:156–9.
Vrajová M, Stastný F, Horácek J, Lochman J, Serý O, Peková S, et al. Expression of the Hippocampal NMDA receptor GluN1 subunit and its splicing isoforms in Schizophrenia: postmortem study. Neurochem Res. 2010;35:994–1002.
Catts VS, Lai YL, Weickert CS, Weickert TW, Catts SV. A quantitative review of the postmortem evidence for decreased cortical N-methyl-d-aspartate receptor expression levels in schizophrenia: how can we link molecular abnormalities to mismatch negativity deficits? Biol Psychol. 2016;116:57–67.
Clinton SM, Haroutunian V, Meador-Woodruff JH. Up-regulation of NMDA receptor subunit and post-synaptic density protein expression in the thalamus of elderly patients with schizophrenia. J Neurochem. 2006;98:1114–25.
Astori S, Lüthi A. Synaptic plasticity at intrathalamic connections via Ca V 3.3 T-type Ca 2+ channels and GluN2B-containing NMDA receptors. J Neurosci. 2013;33:624–30.
Zhang Y, Buonanno A, Vertes RP, Hoover WB, Lisman JE. NR2C in the thalamic reticular nucleus; effects of the NR2C knockout. PLoS ONE. 2012;7:e41908.
Fernandez LMJ, Pellegrini C, Vantomme G, Béard E, Lüthi A, Astori S. Cortical afferents onto the nucleus Reticularis thalami promote plasticity of low-threshold excitability through GluN2C-NMDARs. Sci Rep. 2017;7:12271.
Troyano-Rodriguez E, Lladó-Pelfort L, Santana N, Teruel-Martí V, Celada P, Artigas F. Phencyclidine inhibits the activity of thalamic reticular gamma-aminobutyric acidergic neurons in rat brain. Biol Psychiatry. 2014;76:937–45.
Dawson N, Morris BJ, Pratt JA. Subanaesthetic ketamine treatment alters prefrontal cortex connectivity with thalamus and ascending subcortical systems. Schizophr Bull. 2013;39:366–77.
Gibel-Russo R, Benacom D, Di Nardo AA. Non-cell-autonomous factors implicated in parvalbumin interneuron maturation and critical periods. Front Neural Circuits. 2022;16:875873.
Bernstein H-G, Steiner J, Guest PC, Dobrowolny H, Bogerts B. Glial cells as key players in schizophrenia pathology: recent insights and concepts of therapy. Schizophr Res. 2015;161:4–18.
Majak K, Berdel B, Kowiański P, Dziewiatkowski J, Lipowska M, Moryś J. Parvalbumin immunoreactivity changes in the thalamic reticular nucleus during the maturation of the rat’s brain. Folia Neuropathol. 1998;36:7–14.
Canetta SE, Holt ES, Benoit LJ, Teboul E, Sahyoun GM, Ogden RT, et al. Mature parvalbumin interneuron function in prefrontal cortex requires activity during a postnatal sensitive period. Elife. 2022;11:e80324.
LeMoult J, Humphreys KL, Tracy A, Hoffmeister JA, Ip E, Gotlib IH, et al. Meta-analysis: exposure to early life stress and risk for depression in childhood and adolescence. J Am Acad Child Adolesc Psychiatry. 2020;59:842–55.
Philip NS, Valentine TR, Sweet LH, Tyrka AR, Price LH, Carpenter LL. Early life stress impacts dorsolateral prefrontal cortex functional connectivity in healthy adults: informing future studies of antidepressant treatments. J Psychiatr Res. 2014;52:63–9.
Read J, Os J, Morrison AP, Ross CA. Childhood trauma, psychosis and schizophrenia: a literature review with theoretical and clinical implications. Acta Psychiatr Scand. 2005;112:330–50.
Geyer MA, Wilkinson LS, Humby T, Robbins TW. Isolation rearing of rats produces a deficit in prepulse inhibition of acoustic startle similar to that in schizophrenia. Biol Psychiatry. 1993;34:361–72.
Gilabert-Juan J, Moltó MD, Nacher J. Post-weaning social isolation rearing influences the expression of molecules related to inhibitory neurotransmission and structural plasticity in the amygdala of adult rats. Brain Res. 2012;1448:129–36.
Wang Y-C, Ho U-C, Ko M-C, Liao C-C, Lee L-J. Differential neuronal changes in medial prefrontal cortex, basolateral amygdala and nucleus accumbens after postweaning social isolation. Brain Struct Funct. 2012;217:337–51.
Hickey AJ, Reynolds JN, Beninger RJ. Post-weaning social isolation and subchronic NMDA glutamate receptor blockade: effects on locomotor activity and GABA signaling in the rat suggest independent mechanisms. Pharmacol Biochem Behav. 2012;101:231–8.
Klimczak P, Rizzo A, Castillo-Gómez E, Perez-Rando M, Gramuntell Y, Beltran M, et al. Parvalbumin interneurons and perineuronal nets in the hippocampus and retrosplenial cortex of adult male mice after early social isolation stress and perinatal NMDA receptor antagonist treatment. Front Synaptic Neurosci 2021;13:733989.
Castillo-Gómez E, Pérez-Rando M, Bellés M, Gilabert-Juan J, Llorens JV, Carceller H, et al. Early social isolation stress and perinatal NMDA receptor antagonist treatment induce changes in the structure and neurochemistry of inhibitory neurons of the adult amygdala and prefrontal cortex. eNeuro. 2017;4:ENEURO.0034–17.
Garcia-Mompo C, Curto Y, Carceller H, Gilabert-Juan J, Rodriguez-Flores E, Guirado R, et al. Δ-9-Tetrahydrocannabinol treatment during adolescence and alterations in the inhibitory networks of the adult prefrontal cortex in mice subjected to perinatal NMDA receptor antagonist injection and to postweaning social isolation. Transl Psychiatry. 2020;10:177.
Gilabert-Juan J, Belles M, Saez AR, Carceller H, Zamarbide-Fores S, Moltó MD, et al. A “double hit” murine model for schizophrenia shows alterations in the structure and neurochemistry of the medial prefrontal cortex and the hippocampus. Neurobiol Dis. 2013;59:126–40.
Shangase KB, Luvuno M, Mabandla MV. Investigating the robustness of a rodent “Double Hit” (post-weaning social isolation and NMDA receptor antagonist) model as an animal model for schizophrenia: a systematic review. Brain Sci. 2023;13:848.
Orlowska-Feuer P, Ebrahimi AS, Zippo AG, Petersen RS, Lucas RJ, Storchi R. Look-up and look-down neurons in the mouse visual thalamus during freely moving exploration. Curr Biol. 2022;32:3987–99.e4.
Gummadavelli A, Motelow JE, Smith N, Zhan Q, Schiff ND, Blumenfeld H. Thalamic stimulation to improve level of consciousness after seizures: evaluation of electrophysiology and behavior. Epilepsia. 2015;56:114–24.
Lee EHY, Huang SL. Role of lateral habenula in the regulation of exploratory behavior and its relationship to stress in rats. Behav Brain Res. 1988;30:265–71.
Pandey S, Barson JR. Heightened exploratory behavior following chronic excessive ethanol drinking: mediation by neurotensin receptor type 2 in the anterior paraventricular thalamus. Alcohol Clin Exp Res. 2020;44:1747–59.
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, et al. Fiji: an open-source platform for biological-image analysis. Nat Methods. 2012;9:676–82.
Guirado R, Sanchez-Matarredona D, Varea E, Crespo C, Blasco-Ibáñez JM, Nacher J, et al. Chronic fluoxetine treatment in middle-aged rats induces changes in the expression of plasticity-related molecules and in neurogenesis. BMC Neurosci. 2012;13:5.
Mazzetti S, Ortino B, Inverardi F, Frassoni C, Amadeo A. PSA-NCAM in the developing and mature thalamus. Brain Res Bull. 2007;71:578–86.
Bonfanti L, Olive S, Poulain DA, Theodosis DT. Mapping of the distribution of polysialylated neural cell adhesion molecule throughout the central nervous system of the adult rat: an immunohistochemical study. Neuroscience. 1992;49:419–36.
Lim AL, Taylor DA, Malone DT. A two-hit model: behavioural investigation of the effect of combined neonatal MK-801 administration and isolation rearing in the rat. J Psychopharmacol. 2012;26:1252–64.
Steullet P. Thalamus-related anomalies as candidate mechanism-based biomarkers for psychosis. Schizophr Res. 2020;226:147–57.
Zhang L, Wang H, Luan S, Yang S, Wang Z, Wang J, et al. Altered volume and functional connectivity of the habenula in schizophrenia. Front Hum Neurosci 2017;11:636.
Halassa MM, Sherman SM. Thalamocortical circuit motifs: a general framework. Neuron. 2019;103:762–70.
McCormick DA, Bal T. Sensory gating mechanisms of the thalamus. Curr Opin Neurobiol. 1994;4:550–6.
Ables JL, Park K, Ibañez–Tallon I. Understanding the habenula: a major node in circuits regulating emotion and motivation. Pharmacol Res. 2023;190:106734.
El Boukhari H, Ouhaz Z, Ba‐M’hamed S, Bennis M. Early lesion of the reticular thalamic nucleus disrupts the structure and function of the mediodorsal thalamus and prefrontal cortex. Develop Neurobiol. 2019;79:913–33.
You Q, Luo ZC, Luo ZY, Kong Y, Li ZL, Yang JM, et al. Involvement of the thalamic reticular nucleus in prepulse inhibition of acoustic startle. Transl Psychiatry. 2021;11:241.
Kim U, Sanchez-Vives MV, McCormick DA. Functional dynamics of GABAergic inhibition in the thalamus. Science. 1997;278:130–4.
Nicolelis MAL, Fanselow EE. Dynamic shifting in thalamocortical processing during different behavioural states. Philos Trans R Soc Lond B. 2002;357:1753–8.
Casarrubea M, Davies C, Faulisi F, Pierucci M, Colangeli R, Partridge L, et al. Acute nicotine induces anxiety and disrupts temporal pattern organization of rat exploratory behavior in hole-board: a potential role for the lateral habenula. Front Cell Neurosci 2015;9:197.
Halassa MM, Acsády L. Thalamic inhibition: diverse sources, diverse scales. Trends Neurosci. 2016;39:680–93.
Nakamura T, Kurosaki K, Kanemoto M, Sasahara M, Ichijo H. Early-life experiences altered the maturation of the lateral habenula in mouse models, resulting in behavioural disorders in adulthood. jpn. 2021;46:E480–9.
Nakazawa K, Sapkota K. The origin of NMDA receptor hypofunction in schizophrenia. Pharmacol Ther. 2020;205:107426.
Ibrahim HM, Hogg AJ Jr, Healy DJ, Haroutunian V, Davis KL, Meador-Woodruff JH. Ionotropic glutamate receptor binding and subunit mRNA expression in thalamic nuclei in schizophrenia. AJP. 2000;157:1811–23.
Meador-Woodruff JH, Clinton SM, Beneyto M, McCullumsmith RE. Molecular abnormalities of the glutamate synapse in the thalamus in schizophrenia. Ann N. Y Acad Sci. 2003;1003:75–93.
Sharp FR, Tomitaka M, Bernaudin M, Tomitaka S. Psychosis: pathological activation of limbic thalamocortical circuits by psychomimetics and schizophrenia? Trends Neurosci. 2001;24:330–4.
Hou G, Smith AG, Zhang Z-W. Lack of intrinsic GABAergic connections in the thalamic reticular nucleus of the mouse. J Neurosci. 2016;36:7246–52.
Fung SJ, Webster MJ, Weicker CS. Expression of VGluT1 and VGAT mRNAs in human dorsolateral prefrontal cortex during development and in schizophrenia. Brain Res. 2011;1388:22–31.
Jacinto LR, Mata R, Novais A, Marques F, Sousa N. The habenula as a critical node in chronic stress-related anxiety. Exp Neurol. 2017;289:46–54.
Tchenio A, Lecca S, Valentinova K, Mameli M. Limiting habenular hyperactivity ameliorates maternal separation-driven depressive-like symptoms. Nat Commun. 2017;8:1135.
Gómez-Climent MÁ, Guirado R, Castillo-Gómez E, Varea E, Gutierrez-Mecinas M, Gilabert-Juan J, et al. The polysialylated form of the neural cell adhesion molecule (PSA-NCAM) is expressed in a subpopulation of mature cortical interneurons characterized by reduced structural features and connectivity. Cereb Cortex. 2011;21:1028–41.
Sherman SM. The thalamus is more than just a relay. Curr Opin Neurobiol. 2007;17:417–22.
Campbell PW, Govindaiah G, Masterson SP, Bickford ME, Guido W. Synaptic properties of the feedback connections from the thalamic reticular nucleus to the dorsal lateral geniculate nucleus. J Neurophysiol. 2020;124:404–17.
Sandi C. Stress, cognitive impairment and cell adhesion molecules. Nat Rev Neurosci. 2004;5:917–30.
Bueno-Fernandez C, Perez-Rando M, Alcaide J, Coviello S, Sandi C, Castillo-Gómez E, et al. Long term effects of peripubertal stress on excitatory and inhibitory circuits in the prefrontal cortex of male and female mice. Neurobiol Stress. 2021;14:100322.
Gilabert-Juan J, Varea E, Guirado R, Blasco-Ibáñez JM, Crespo C, Nácher J. Alterations in the expression of PSA-NCAM and synaptic proteins in the dorsolateral prefrontal cortex of psychiatric disorder patients. Neurosci Lett. 2012;530:97–102.
Varea E, Guirado R, Gilabert-Juan J, Martí U, Castillo-Gomez E, Blasco-Ibáñez JM, et al. Expression of PSA-NCAM and synaptic proteins in the amygdala of psychiatric disorder patients. J Psychiatr Res. 2012;46:189–97.
Arai M, Yamada K, Toyota T, Obata N, Haga S, Yoshida Y, et al. Association between polymorphisms in the promoter region of the sialyltransferase 8B (SIAT8B) gene and schizophrenia. Biol Psychiatry. 2006;59:652–9.
Gilabert-Juan J, Nacher J, Sanjuán J, Moltó MD. Sex-specific association of the ST8SIAII gene with schizophrenia in a Spanish population. Psychiatry Res. 2013;210:1293–5.
Gober R, Ardalan M, Shiadeh SMJ, Duque L, Garamszegi SP, Ascona M, et al. Microglia activation in postmortem brains with schizophrenia demonstrates distinct morphological changes between brain regions. Brain Pathol. 2022;32:e13003.
Hercher C, Chopra V, Beasley CL. Evidence for morphological alterations in prefrontal white matter glia in schizophrenia and bipolar disorder. J Psychiatry Neurosci. 2014;39:376–85.
Acknowledgements
This work was supported by the project PID2021-127595OB-I00 and financed by the Spanish Ministry of Science and Innovation/AEI/10.13039/501100011033/ (“FEDER Una manera de hacer Europa”) and the Generalitat Valenciana (CIPROM/2020/024 and CIPROM/2023/28). It is also supported by Red Española de Investigación en Estrés/Spanish Network for Stress Research RED2022-134191-T financed by MCIN/AEI /10.13039/501100011033.
Author information
Authors and Affiliations
Contributions
PK: formal analysis, investigation, writing original draft; JA: formal analysis, investigation; YG: formal analysis, investigation, Esther Castillo-Gómez: methodology, investigation; EV: writing-review & editing; MP-R: investigation, writing-review & editing; JN: conceptualization, writing-review & editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Klimczak, P., Alcaide, J., Gramuntell, Y. et al. Long-term effects of a double hit murine model for schizophrenia on parvalbumin expressing cells and plasticity-related molecules in the thalamic reticular nucleus and the habenula. Transl Psychiatry 14, 450 (2024). https://doi.org/10.1038/s41398-024-03166-6
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41398-024-03166-6
This article is cited by
-
Clozapine induces perineuronal net remodeling in a developmental mouse model exhibiting schizophrenia-relevant phenotypes
Neuropsychopharmacology (2025)