Abstract
Early life adversity (ELA) is highly prevalent and has significant detrimental effects on children’s brain development and behavioral outcomes. However, associations of threat, unpredictability, and deprivation in dimensional models of ELA with corticolimbic circuits were unclear. Previous studies have highlighted the effects of proximity, especially the distinctions between interpersonal and socioeconomic ELA, on children’s neurodevelopment. This study first examined the associations between multiple ELA categories (i.e., interpersonal threat, socioeconomic threat, interpersonal unpredictability, socioeconomic unpredictability, interpersonal deprivation, and socioeconomic deprivation) and the developmental changes in resting-state functional connectivity (rsFC) of the frontolimbic and temporal-limbic circuits implicated in cognition and psychopathology, using data from the Adolescent Brain Cognitive Development (ABCD) study, which included four measurements from baseline (mean ± SD age, 119.13 ± 7.51 months; 2815 females) to 3-year follow-up (N = 5885). Controlling for concurrent ELA categories, interpersonal unpredictability and socioeconomic deprivation were associated with accelerated development of rsFC between CON/RTN-amygdala/hippocampus (Cingulo-opercular Network, Retrosplenial Temporal Network). Compared to girls, boys’ corticolimbic circuits development was more sensitive to interpersonal threat. Changes in rsFC mediated the associations between interpersonal unpredictability/socioeconomic deprivation with decreased crystallized cognition, fluid cognition, and internalizing problems. Additionally, socioeconomic deprivation was associated with decreased crystallized cognition through interpersonal unpredictability and changes in rsFC between CON and bilateral amygdala/right hippocampus. This study emphasized interpersonal unpredictability and socioeconomic deprivation as key environmental factors affecting corticolimbic circuit development implicated in cognition and psychopathology. Stable family environments and enriched neighborhoods are crucial for supporting optimal neural and behavioral development in children and adolescents.
Similar content being viewed by others
Approximately 45% children in the United States have experienced at least one type of early life adversity (ELA) [1], which has significant detrimental effects on children’s brain development and behavioral outcomes [2, 3], especially during critical periods of neural plasticity, such as childhood and adolescence [4].
ELA is a broad term that covers a wide range of experiences; therefore, recent studies have increasingly adopted dimensional models, which generally include threat, deprivation, and unpredictability based on their distinct effects on children’s neurodevelopment and psychopathology [5,6,7]. Threat indicates harm or harm threat resulting in extrinsic morbidity–mortality to children [5]. Deprivation involves limited or reduced social and cognitive inputs from the environment [6, 7]. Unpredictability reflects spatial-temporal variation in threat or deprivation [5]. Unpredictability has been studied less extensively than threat and deprivation, yet it plays an important and distinct role [8]. Although dimensional models significantly improve our understanding of the distinct effects of ELA on children’s and adolescents’ brain development, previous findings are highly inconsistent regarding the specific patterns of the influence on large-scale brain networks and frontolimbic system [9, 10]. For example, childhood violence threat, but not social deprivation, was associated with decreased connectivity within salience network (SN) [11]. However, both threat and deprivation were associated with increased connectivity within SN in another study [12]. Moreover, prior research found that unpredictability was associated with changes in connectivity of the cingulo-opercular network (CON) and default mode network (DMN) [13], while some studies have only identified its association with a weakened SN, but with no connection to the DMN or other networks [14, 15]. Even within the same ELA dimension (e.g., threat), studies investigating the associations between threat and resting-state functional connectivity (rsFC) of the emotion circuits have yielded mixed results: high-threat environments have been associated with lower rsFC [16], greater rsFC [17], and no significant associations [18]. It might suggest the need for a more nuanced analysis of ELA dimensions, taking into account other factors that contribute to adolescents’ altered neural and behavioral development. The integrated model of dimensions, which includes both proximal and distal cues to detect harshness (threat and deprivation) and unpredictability, proposed that individuals calibrate their development in response to both immediate rearing environments and broader ecological contexts [19]. This model highlighted that the proximity of ELA might be a key dimension in understanding the role of ELA.
The nature of ELA that occurs in hierarchical ecological contexts varying in proximity to the child, whether interpersonal or socioeconomic (with interpersonal factors being more proximal and socioeconomic factors being more distal), stands out as a significant ecological factor due to its robust and distinct effects on human’s development [20,21,22]. Interpersonal ELA refers to the ELA that involves at least one primary caregiver, including caregiving disruptions, caregiver psychopathology, maltreatment, and interpersonal trauma; whereas socioeconomic ELA, a non-interpersonal and distal adversity factor, refers to ELA related to socioeconomic or neighborhood disadvantages, including low family socioeconomic status, community crime/violence exposure, neighborhood trauma, and area deprivation [23]. Interpersonal and socioeconomic ELA may have diverse effects on individuals’ brain and behaviors [23]. Interpersonal ELA primarily affected affective brain regions (e.g., the amygdala, striatum, and hippocampus) [24,25,26,27]. Socioeconomic ELA, which may be orthogonal to interpersonal ELA [23], was suggested to affect the availability of material resources and cognitive enrichment opportunities, and therefore was more likely to influence cortical regions crucial for language, executive functions, and sensorimotor processing [28,29,30].
Importantly, dimensional models and the proximity of ELA are not mutually exclusive; they can be integrated to refine the categorization of ELA. Threat and deprivation can both occur in close caregiver-child proximity [31]. A recent study employing mixed graphical models to obtain ELA categories revealed that deprivation could be split into parental neglect (interpersonal deprivation) and financial difficulties (socioeconomic deprivation), which had diverse effects on neurodevelopment [32]. Consistent with this finding, caregiver neglect (interpersonal deprivation) was associated with greater fractional anisotropy (FA) in the left uncinate, while food insecurity (socioeconomic deprivation) was associated with reduced FA in the left anterior limb of the internal capsule and greater FA in the left uncinate [33]. Moreover, violence occurring at home (interpersonal threat) was associated with multiple internalizing and externalizing problems, whereas violence exposure in the community (socioeconomic threat) was not [34]. Unpredictability is also frequently experienced both in interpersonal context (e.g., child-care) and socioeconomic context (e.g., income) [35]. Income fluctuations (socioeconomic unpredictability) were associated with children’s cognitive development [35,36,37], while variations in interpersonal factors, such as child-care (interpersonal unpredictability), were more likely to be related to children’s emotional responses [38]. Furthermore, in a recent meta-analysis, interpersonal ELA (including both threat and deprivation) demonstrated dissimilarity from socioeconomic ELA (also including both threat and deprivation) in the effects on the development of the brain structure in children from birth to adulthood; specifically, interpersonal ELA was associated with altered volumes in frontolimbic regions, whereas socioeconomic ELA was associated with altered volumes in temporal-limbic regions [23]. The distinction between interpersonal and socioeconomic factors may be one of reasons underlying the inconsistent associations between ELA dimensions and frontolimbic circuits. Additionally, to ascertain specificity, it is indispensable to concurrently measure diverse dimensions of ELA and incorporate them into a unified statistical model [39]; however, limited studies have examined the relative importance and distinct neurobehavioral effects of ELA dimensions while controlling for concurrent ELA, limiting our understanding of these effects within their interconnected nature [40]. In this study, therefore, we leveraged multiple ELA types in the Adolescent Brain Cognitive Development Study (ABCD) and integrated six categories of ELA, characterized by proximity (interpersonal and socioeconomic) and dimensions (threat, unpredictability, and deprivation), to elucidate their associations with brain and behavioral development and to identify the adverse factors that had the most significant influence on the development of children and adolescents.
Moreover, socioeconomic ELA may affect children indirectly via interpersonal ELA [28, 41]. Unpredictable parental signals (interpersonal unpredictability) represented a pathway accounting for the association between established forms of ELA (e.g., poverty; socioeconomic deprivation) and development [35]. Therefore, we further explored the transmission from distal ecological contextual factors (e.g., socioeconomic ELA) to the proximal hierarchy (e.g., interpersonal ELA). We conducted serial mediation models to examine whether the effects of socioeconomic ELA on adolescent behavioral outcomes were mediated by interpersonal ELA and adolescent brain development.
RSFC provides a robust approach to examine the neurobiological pathways linking ELA and child development, which has greater test-retest reliability and a stronger signal-to-noise ratio compared to task-based magnetic resonance imaging (MRI) [42]. Previous work has mainly focused on rsFC of the frontolimbic circuitry (i.e., CON-amygdala/hippocampus) [43, 44], though more cortical regions may be associated with ELA, such as the temporal cortex (e.g., the retrosplenial-temporal network; RTN). The CON, encompassing the dorsal anterior cingulate cortex and bilateral anterior insula, consistently activates during tasks involving error detection and ongoing task management [45]. The frontolimbic circuitry mainly functions in emotional processing and regulation [25]. The RTN, a community of retrosplenial and ventral temporal cortex [46], which was connected to amygdala/hippocampus (i.e., temporal-limbic circuits), was also associated with emotional and executive functions; for example, greater connectivity was found between the right amygdala and temporal areas during angry face processing compared to happy faces [47]. Additionally, the RTN is closely linked to structures in the medial temporal lobe (e.g., parahippocampal gyrus), which plays a unique role in memory functions, encompassing episodic and working memory as well as spatial navigation [48, 49].
Moreover, the CON, medial temporal cortex, and limbic regions including amygdala and hippocampus, all undergo dynamic reorganization in childhood and adolescence [50, 51]. There is a tendency for segregation in the rsFC between CON and the limbic regions (i.e., amygdala and hippocampus) [43]; however, the normative developmental trajectories of RTN-amygdala/hippocampus remain unclear. The stress acceleration theory suggests that ELA accelerates neural development, marked by faster maturation of the frontolimbic circuits [25, 52]. Although being evolutionarily adaptive in the short term, deviations from the typical neural development may be detrimental to various cognitive and emotional processes in the long run [12, 53, 54]. The existing work of ELA, however, has mostly relied on cross-sectional designs [25], which hinders our understanding of how ELA alters the normative developmental patterns of brain and its implications for subsequent cognitive impairments and psychopathology; therefore, longitudinal studies with larger sample sizes (e.g., ABCD) and incorporating more corticolimbic circuits (e.g., temporal-limbic circuitry) are needed.
Furthermore, there is growing evidence indicating sexually dimorphic responses to interpersonal or socioeconomic factors and ELA dimensions [12, 35, 55]; therefore, we additionally examined sex differences in the effects of ELA on rsFC development and in the mediating effects of rsFC on the associations between ELA and children’s behaviors. Previous studies found mixed results regarding performances in cognition among children who experienced ELA, including adaptation, maladaptation, and no differences. As such a deeper understanding of how multiple categories of ELA are related to the nuanced components of cognition (e.g., working memory, inhibitory control) and the underlying neural mechanisms is needed [56].
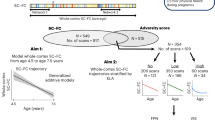
In summary, we had three major aims and hypotheses (Fig. 1). First, we examined the effects of six categories of ELA (i.e., interpersonal threat, socioeconomic threat, interpersonal unpredictability, socioeconomic unpredictability, interpersonal deprivation, and socioeconomic deprivation) on the changes in rsFC of the frontolimbic and temporal-limbic circuits (i.e., CON/RTN-amygdala/hippocampus) from baseline to 2-year follow-up. To examine the relative importance of different types of ELA, we controlled for concurrent ELA categories in a unified model. We hypothesized that CON-amygdala/hippocampus rsFC would decrease from baseline to 2-year follow-up, with greater exposures to ELA accelerating these changes based on the stress acceleration theory; however, as exploratory analyses, we did not make specific hypotheses regarding changes in RTN-amygdala/hippocampus rsFC and the effects of ELA on the changes due to limited prior research. Second, we examined the mediating effects of changes in rsFC and hypothesized that ELA would be associated with atypical development of CON/RTN-amygdala/hippocampus rsFC, which in turn would reduce adolescents’ concurrent cognitive abilities (crystallized cognition, fluid cognition, and specific task scores) and increase their subsequent behavioral problems at the 3-year follow-up, controlling for their baseline levels. We further tested sex differences in the two steps mentioned above but refrained from making specific hypotheses due to insufficient evidence regarding the complex effects of these ELA categories. Third, as exploratory analyses, we examined whether socioeconomic ELA dimensions indirectly predicted adolescent behavioral outcomes through serial mediation pathways involving interpersonal ELA dimensions and changes in rsFC.
The blue arrows represent the associations between interpersonal/socioeconomic early life adversity (ELA) dimensions andresting-state functional connectivity (RSFC) of the corticolimbic circuits (1.1). The purple arrows indicate the mediating role of RSFC in the link between ELA and adolescent behaviors (2.1). The red arrows represent the multiple mediation pathways through which interpersonal ELA and RSFC mediate the association between socioeconomic ELA andadolescent behaviors (3). Yellow indcates sex differences (1.2 & 2.2).
Materials and methods
Participants
Participants were from the ABCD study (https://abcdstudy.org/). The ABCD study is an ongoing longitudinal study that has recruited 11 868 children (9–10 years of age) from 21 study sites across the United States [57]. We used baseline, 1-year, 2-year, and 3-year follow-up data from the 5.0 release. After excluding participants who did not have resting-state functional MRI data (rs-fMRI), or whose rs-fMRI data were recommended for exclusion by the ABCD analytic core at baseline and 2-year follow-up, the final sample consisted of N = 5 885 participants (see Table 1 for demographic information).
All procedures were carried out in accordance with the ethical standards established by the 1964 Declaration of Helsinki and received approval from the central Institutional Review Board (IRB) at the University of California, San Diego, as well as from the local IRBs at each study site. All participants provided informed consent or assent, detailed ethics information can be found in previous study [58].
Environmental and behavioral data
Interpersonal and socioeconomic threat
We used the Conflict Subscale from the Family Environment Scale (FES) to measure interpersonal threat [59], which was reported by youth at baseline. This subscale consists of nine items assessing the amount of openly expressed conflict among family members. We used a sum score of these nine items. The higher the total score, the greater level of interpersonal threat the child experienced.
We used the Neighborhood Safety/Crime Survey to measure socioeconomic threat, which was reported by parents at baseline [60, 61]. This measure consists of three items assessing feelings about safety in the respondent’s neighborhood, which were rated on a 5-point Likert-type scale ranging from “1 = strongly disagree” to “5 = strongly agree”. We reversed the score and computed the mean score of these 3 items to measure socioeconomic threat. The higher the mean score, the greater level of socioeconomic threat the child experienced.
Interpersonal and socioeconomic unpredictability
We used a subset of items from the Life Events Scale (LES) to measure unpredictability [62,63,64], which was reported by youth at the 1-year follow-up. LES is a 25-item questionnaire measuring children’s stressful life events. We operationalized unpredictability from ancestral cue perspective by measuring classic cues (e.g., parental transition) that reliably indicated high unpredictability [65], which were similar to those employed in previous research. For example, Belsky and colleagues assessed unpredictability using three measures, containing paternal transitions, household moves, and parental employment transitions [66]; Mittal and colleagues measured unpredictability using three items from the Life Stress Inventory [67, 68]: changes in employment status during the prior year (e.g., periods of unemployment), changes in residence during the prior year (e.g., moving to a different house/apartment) and people moving in and out of the house during the prior year (e.g., mother’s romantic partners moving in or out; an immediate family member receiving a jail sentence).
We selected five items pertaining to the unpredictable experiences involving interactions among family members to assess interpersonal unpredictability: parents argued more than previously; one parent was away from home more often; parents separated or divorced; one of the parents/caregivers went to jail; got new stepmother or stepfather. We selected three items pertaining to the unpredictable experiences involving household socioeconomic changes to assess socioeconomic unpredictability: negative change in parent’s financial situation; mother/father figure lost job; parent/caregiver got a new job. Respondents reviewed the list of items and indicated which events occurred. We used the total number of events to reflect the level of interpersonal/socioeconomic unpredictability experienced by the children.
Interpersonal and socioeconomic deprivation
We used the Parental Monitoring Survey (PMS) to measure interpersonal deprivation [69, 70], which was reported by youth at baseline. The PMS consists of five items assessing parents’ active efforts to keep track of a child’s whereabouts and when they are at or not at home (e.g., who they are with; what they are doing). The scale was rated on a 5-point Likert-type scale ranging from “1 = never” to “5 = always or almost always”. We reversed the score and computed the mean score of these five items to measure interpersonal deprivation. The higher the score, the greater level of interpersonal deprivation the child experienced.
We used the rate of poverty for participants’ valid primary addresses, geocoded at the census tract level, to measure socioeconomic deprivation. These data are from Vox.com (https://www.vox.com/a/lead-exposure-risk-map). The poverty rate reflects the proportion of individuals living in poverty (−125% of poverty level) in the neighborhood environment. The higher the proportion, the greater level of socioeconomic deprivation the child experienced.
Crystallized and fluid cognition
We used the NIH Cognition Toolbox to measure children’s cognitive abilities, which comprises seven tasks: Picture Vocabulary, Flanker Inhibitory Control & Attention, Picture Sequence Memory, Dimensional Change Card Sort, Pattern Comparison Processing Speed, Oral Reading Recognition, and List Sorting Working Memory [71]. The cognitive tasks were not administered at the 3-year follow-up; therefore, we used the measures at the 2-year follow-up. The Dimensional Change Card Sort and List Sorting Working Memory were not administered at the 2-year follow-up; therefore, we used the other five measures at baseline and the 2-year follow-up. We used the uncorrected scaled scores (recommended using for longitudinal analyses by the ABCD group) of three measures (i.e., Flanker Inhibitory Control & Attention, Picture Sequence Memory, and Pattern Comparison Processing Speed) to calculate a mean score of fluid cognition. We used the uncorrected scaled scores of the crystallized cognition composite (i.e., Picture Vocabulary and Oral Reading Recognition). We also included the five task-specific scores. The higher the scores, the greater level of cognitive abilities the children had.
Internalizing ang externalizing problems
We used the Child Behavior Checklist (CBCL) [72, 73], which measured children’s psychopathology and behaviors over the past 6 months. Parents reported at baseline and at the 3-year follow-up. Given we examined longitudinal changes in this study, we used raw scores rather than sex- and age-corrected T-scores, which was recommended in previous research [74], from the internalizing and externalizing symptom subscales, which were rated on a 3-point Likert-type scale ranging from “0 = not true” to “2 = very true”. The higher the scores, the greater level of internalizing or externalizing problems the children had.
Imaging data
Imaging procedures have been described in detail in previous study [57]. Participants underwent neuroimaging scans using standardized protocols across sites. They completed four or five 5-min resting-state scans with their eyes open, ensuring a minimum of 8 min of data with related low-motion [75]. In this study, we used resting-state functional MRI data at baseline and at the 2-year follow-up. The preprocessing of the data was conducted by the ABCD Data Analysis and Informatics Core using the standardized ABCD pipeline [75]. The participant’s time courses were pre-processed and then mapped onto the cortical surface. Average cortical time courses were derived based on the Gordon functional parcellation [46], and subcortical time-courses were also computed and labeled using atlas-based segmentation [76]. The rsFC between Gordon networks and subcortical regions were calculated (Pearson correlation) and then Fischer z transformed. We focused on the changes in rsFC between CON/RTN (Cingulo-opercular Network/Retrosplenial Temporal Network) and amygdala/hippocampus (left and right; aglh represents the left amygdala, agrh represents the right amygdala, hplh represents the left hippocampus, hprh represents the right hippocampus), therefore a total of 8 rsFC values.
Statistical analyses
First, to test the associations between interpersonal/socioeconomic ELA dimensions and changes in rsFC, we conducted models using cluster robust standard errors (CR-SEs; the highest level - site ID was modeled as clustering variable, TYPE = COMPLEX) [77] to account for the clustering structure (i.e., multiple children from the same family and site) in Mplus Version 7.4 [78], with rsFC values as outcomes separately and six ELA categories as predictors (i.e., interpersonal threat, socioeconomic threat, interpersonal unpredictability, socioeconomic unpredictability, interpersonal deprivation, and socioeconomic deprivation) in the unified models. Moreover, we included an interaction term of ELA and sex to examine sex difference in the effects of ELA on rsFC development. Second, we examined the mediating roles of rsFC in linking interpersonal/socioeconomic ELA dimensions and adolescents’ cognitive abilities as well as behavioral problems, using 95% bias-corrected confidence intervals (CIs) with 5 000 bootstrap samples in the mediation analysis. Moreover, we included an interaction term of ELA and sex to examine sex difference in the mediating effects of rsFC on the associations between ELA and children’s behaviors. Finally, we conducted the serial mediation models using the same methods to examine whether socioeconomic ELA dimensions indirectly predicted adolescent behavioral outcomes through serial mediation pathways involving interpersonal ELA dimensions and changes in rsFC.
In addition to controlling for the concurrent ELA categories in the unified models to examine their relative importance, we controlled for sex, age, scanner type, and mean framewise displacement when predicting rsFC at the 2-year follow-up. We also controlled for rsFC at baseline, therefore, the outcome was residualized to eliminate baseline connectivity effects, leaving only variances attributable to developmental change. Moreover, we included parental mental health, the number of peers, and community SES as covariates of predictors and brain variables. In models predicting cognitive abilities at the 2-year follow-up, we covaried sex, parents’ highest education level, family income, and cognitive abilities at baseline, as well as age at the 2-year follow-up. In models predicting behavioral problems at the 3-year follow-up, we also covaried sex, parents’ highest education level, family income, and internalizing/externalizing problems at baseline, as well as age at the 3-year follow-up. We used winsorization method to address outliers. Specifically, observations with absolute standard scores greater than four on any model variables were replaced with the nearest maximum or minimum value [79, 80]. In all models, residuals were examined to assess normality, and full information maximum likelihood (FIML) estimation was utilized to address missing data in the study variables. All statistical tests were two-sided, and we controlled for multiple comparisons using the false discovery rate (FDR) correction for the coefficients across the 8 tests of single interpersonal or socioeconomic ELA dimension.
Results
Correlations between study variables and changes in rsFC
Correlations between all study variables were shown in Supplementary Fig. 1. Pair-sample t-test indicated that rsFC of CON-aglh, CON-agrh, CON-hplh, CON-hprh, RTN-agrh, and RTN-hplh decreased, rsFC of RTN-aglh, and RTN-hprh increased from baseline to 2-year follow-up (Fig. 2).
rsFC resting-state functional connectivity, CON cingulo-opercular network, RTN retrosplenial temporal network, aglh left amygdala, agrh right amygdala, hplh left hippocampus, hprh right hippocampus. **** p < 0.0001.
Associations between interpersonal/socioeconomic ELA dimensions with changes in rsFC and sex differences
We found significant associations between interpersonal/socioeconomic ELA dimensions and accelerated development of rsFC. Specifically, interpersonal unpredictability was associated with a greater decrease in rsFC of CON-aglh, CON-agrh, CON-hprh, and RTN-hplh, as well as a greater increase in rsFC of RTN-hprh; socioeconomic deprivation was associated with a greater decrease in rsFC of CON-aglh, CON-agrh, CON-hprh, and RTN-hplh, as well as a greater increase in rsFC of RTN-aglh and RTN-hprh (Fig. 3 and Supplementary Table 1).
Blue lines indicated the associations between interpersonal unpredictability and changes in rsFC; red lines indicated the associations between socioeconomic deprivation and changes in rsFC. Solid lines represented positive effects; dashed lines represented negative effects. rsFC resting-state functional connectivity, CON cingulo-opercular network, RTN retrosplenial temporal network, aglh left amygdala, agrh right amygdala, hplh left hippocampus, hprh right hippocampus.
Moreover, we found that sex significantly moderated the associations between interpersonal threat and rsFC of CON-aglh, CON-agrh, CON-hprh, and RTN-agrh, as well as the associations between interpersonal deprivation and rsFC of RTN-hprh (Supplementary Table 2). Specifically, for boys, interpersonal threat was associated with a greater decrease in rsFC of CON-aglh, CON-agrh, and CON-hprh, but with a smaller decrease in rsFC of RTN-agrh. For girls, none of these effects were significant. Additionally, the association between interpersonal deprivation and changes in rsFC of RTN-hprh was not significant for either boys or girls (Supplementary Table 3).
Mediating roles of changes in rsFC between interpersonal/socioeconomic ELA dimensions with adolescents’ cognitive abilities/behavioral problems and sex differences
Based on the significant results of the associations between interpersonal/socioeconomic ELA dimensions and rsFC between CON/RTN and amygdala/hippocampus (left and right), we conducted further analyses to test if rsFC mediated the associations of interpersonal unpredictability and socioeconomic deprivation with cognitive abilities as well as behavioral problems, respectively. The results showed that rsFC of CON-aglh, CON-agrh, and CON-hprh mediated the associations between interpersonal unpredictability and crystallized cognition. Specifically, greater interpersonal unpredictability at the 1-year follow-up was associated with a greater decrease in rsFC of CON-aglh, CON-agrh, and CON-hprh, which was associated with lower crystallized cognition at the 2-year follow-up. Moreover, rsFC of CON-aglh, CON-agrh, CON-hprh, and RTN-hplh mediated the associations between socioeconomic deprivation and crystallized cognition, rsFC of RTN-hplh mediated the associations between socioeconomic deprivation and fluid cognition, and rsFC of CON-hprh mediated the associations between socioeconomic deprivation and internalizing problems. Specifically, greater socioeconomic deprivation at baseline was associated with a greater decrease in rsFC of CON-aglh, CON-agrh, CON-hprh, and RTN-hplh, which was associated with lower crystallized cognition at the 2-year follow-up. Greater socioeconomic deprivation at baseline was associated with a greater decrease in rsFC of RTN-hplh, which was associated with lower fluid cognition at the 2-year follow-up. Additionally, greater socioeconomic deprivation at baseline was associated with a greater decrease in rsFC of CON-hprh, which was associated with lower internalizing problems at the 3-year follow-up (Fig. 4 and Supplementary Table 4).
The two ends of the lines were the lower and upper limits of the confidence interval, and the middle points were the estimates of the mediating effects. The y-axis on the left side of the lines corresponded to the independent variables and mediators, while the colors represented the dependent variables in the legend. rsFC resting-state functional connectivity, IU interpersonal unpredictability, SD socioeconomic deprivation, CON cingulo-opercular network, RTN retrosplenial temporal network, aglh left amygdala, agrh right amygdala, hplh left hippocampus, hprh right hippocampus. +p < 0.1, * p < 0.05, ** p < 0.01.
Moreover, we found that rsFC of CON-agrh mediated the association between interpersonal unpredictability and performance of Picture Sequence Memory, CON-aglh and CON-agrh mediated the association between interpersonal unpredictability and performance of Picture Vocabulary. Specifically, greater interpersonal unpredictability was associated with a greater decrease in rsFC of CON-agrh, which was associated with lower scores of Picture Sequence Memory. Greater interpersonal unpredictability was associated with a greater decrease in rsFC of CON-aglh and CON-agrh, which were associated with lower scores of Picture Vocabulary. Moreover, rsFC of CON-hprh and RTN-hplh mediated the association between socioeconomic deprivation and the performance of Flanker Inhibitory Control & Attention, rsFC of CON-agrh mediated the association between socioeconomic deprivation and the performance of Pattern Comparison Processing Speed, rsFC of CON-aglh and CON-agrh mediated the association between socioeconomic deprivation and the performance of Picture Sequence Memory, and rsFC of CON-aglh, CON-agrh, and RTN-hplh mediated the association between socioeconomic deprivation and the performance of Picture Vocabulary. Specifically, greater socioeconomic deprivation was associated with a greater decrease in rsFC of CON-hprh and RTN-hplh, which were associated with lower scores of Flanker Inhibitory Control & Attention. Greater socioeconomic deprivation was associated with a greater decrease in rsFC of CON-aglh and CON-agrh, which were associated with lower scores of Picture Sequence Memory. Greater socioeconomic deprivation was associated with a greater decrease in rsFC of CON-aglh, CON-agrh, and RTN-hplh, which were associated with lower scores of Picture Vocabulary. Greater socioeconomic deprivation was associated with a greater decrease in rsFC of CON-agrh, which was associated with higher scores of Pattern Comparison Processing Speed (Supplementary Table 4).
As for sex differences, we found no sex differences in the mediating effects of rsFC on the associations between interpersonal/socioeconomic ELA dimensions and adolescents’ cognitive abilities/behavioral problems (Supplementary Table 5).
Serial mediation models of interpersonal unpredictability and changes in rsFC
Based on the significant results of the simple mediating effects, we conducted further analyses to test if interpersonal unpredictability and rsFC mediated the associations between socioeconomic deprivation and cognitive abilities as well as behavioral problems, respectively. The results showed that greater socioeconomic deprivation was associated with decreased crystallized cognition through greater interpersonal unpredictability and a greater decrease in rsFC of CON-aglh/CON-agrh/CON-hprh (Fig. 5 and Supplementary Table 6); greater socioeconomic deprivation was associated with worse performance of Picture Sequence Memory through greater interpersonal unpredictability and a greater decrease in rsFC of CON-agrh; greater socioeconomic deprivation was associated with worse performance of Picture Vocabulary through greater interpersonal unpredictability and a greater decrease in rsFC of CON-aglh and CON-agrh (Supplementary Table 6).
A Multiple mediation pathways through which interpersonal unpredictability and CON-aglh rsFC mediate the association between socioeconomic deprivation andadolescent crystallized cognition. B Multiple mediation pathways through which interpersonal unpredictabilityand CON-agrh rsFC mediate the association between socioeconomic deprivation andadolescent crystallized cognition. C Multiple mediation pathways through which interpersonal unpredictability and CON-hprh rsFC mediate the association between socioeconomic deprivation and adolescent crystallized cognition.
Discussion
This is the first study to integrate two classification models/perspectives of ELA – the dimensional models and the proximity (interpersonal and socioeconomic) of ELA, and to examine their associations with adolescent altered neural development, including changes in rsFC of frontolimbic and temporal-limbic circuits (i.e., CON/RTN-amygdala/hippocampus) implicated in cognition and behavioral problems. We found that the frontolimbic circuits segregated with development, which was in line with previous research [43]. Limited studies, however, had identified the developmental trajectory of the temporal-limbic circuitry. This study provided initial evidence for the typical developmental patterns of the temporal-limbic circuits: the rsFC between RTN and left amygdala and right hippocampus increased, whereas the rsFC between RTN and right amygdala and left hippocampus decreased. Controlling for concurrent ELA categories, interpersonal unpredictability and socioeconomic deprivation were associated with accelerated development of frontolimbic and temporal-limbic circuits. These findings were consistent with the stress acceleration hypothesis [25] and extended this theory by containing more widespread cortical cortex (i.e., temporal cortex).
These findings supported and refined the dimensional models by identifying distinct effects of ELA dimensions (i.e., deprivation and unpredictability, rather than threat) on the development of corticolimbic circuits. Previous research has indicated that deprivation, but not threat, was associated with accelerated development of task-based functional connectivity between emotion circuits [10]. Exposed to deprivation led to reduced proliferation or accelerated pruning of synaptic connections, which may underlie the accelerating effect [81]. Moreover, unpredictable caregiving accelerating maturation of the corticolimbic circuitry was embedded through early activation of the neuroendocrine and stress response dysregulation of autonomic nervous systems [52, 82]. Threat was not associated with the development of corticolimbic circuits in the unified models; however, threat may be associated with other regions that were not measured in this study (e.g., frontoparietal network, default mode network (DMN), SN, precuneus, insula, and putamen) [14, 27].
The findings highlighted the significant impact of the proximity of ELA, whether interpersonal or socioeconomic, on children’s neural and behavioral development, aligning with the integrated model of dimensions that incorporates both proximal and distal cues to detect harshness and unpredictability [19]. When controlling for concurrent ELA categories, interpersonal unpredictability and socioeconomic deprivation emerged as the most robust factors influencing individuals’ brain and behaviors. The findings indicated that different proximities (i.e., socioeconomic or interpersonal) within a single ELA dimension were distinctively associated with the development of the frontolimbic circuits, which might help explain the inconsistent associations between ELA dimensions and the frontolimbic circuits in previous research [9, 10, 16,17,18]. Moreover, the significant role of socioeconomic deprivation in adolescents’ development, which highlighted the myriad challenges faced by individuals exposed to impoverished environment, could be attributed to its association with various risky environmental factors, including exposure to toxins and stressors, adverse dietary factors, and lack of educational opportunities [83, 84]. Prior research showed that family financial difficulty (socioeconomic deprivation) had a stronger association with functional brain organization compared to other forms of ELA, such as parental neglect (interpersonal deprivation) [32]. This study extended the measure of socioeconomic deprivation from household SES to the neighborhood level, identifying its widespread effects on the maturation of corticolimbic circuits. The finding was consistent with previous research showing that neighborhood socioeconomic disadvantages were associated with structure and rsFC alterations of the brain regions implicated in affective processing (e.g., the insula, ventromedial prefrontal cortex, amygdala, and hippocampus) [85, 86].
Interestingly, interpersonal unpredictability exerted a similarly significant effect on the development of the frontolimbic and temporal-limbic circuits, highlighting the importance of parental caregiving during humans’ extended period of development [31]. Although limited studies have distinguished between interpersonal and socioeconomic unpredictability, prior studies identified significant effects of caregiving unpredictability on adolescents’ neural and behavioral development. For example, prenatal exposure to maternal mood unpredictability (interpersonal unpredictability) was associated with a weakened and inflexible SN in adolescence [15]. Moreover, the long-lasting implications of interpersonal unpredictability were demonstrated through extensive rodent studies conducted by Baram and colleagues, who identified that unpredictable, fragmented parental signals (interpersonal unpredictability) dysregulate key physiological systems, including the hypothalamic-pituitary-adrenal (HPA) axis, gut-brain axis, and metabolism, induce aberrant synaptic connectivity, and disrupted the maturation of cortical and limbic circuits, which underlie later memory impairments and anhedonia-like behaviors [87,88,89]. Compared with socioeconomic unpredictability, interpersonal unpredictability was more proximal to children, violated children’s basic safety needs, heightened their sensitivity to parent–child negative signals, and had a stronger impact on children’s frontolimbic circuitry involved in their survival and emotional processing [90, 91].
Moreover, we found that interpersonal threat was associated with boys’ rsFC in the corticolimbic circuits but showed no such association in girls. This finding was inconsistent with previous research indicating girls may exhibit heightened emotional responses following family conflicts [92]. However, in terms of brain development, particularly the maturation of corticolimbic circuitry as examined in this study, whether boys or girls appear to be more vulnerable remains inconsistent [93, 94] and requires further detailed investigation in future research.
In analyses of the simple mediation models, we found that rsFC of corticolimbic circuits mediated the association between interpersonal unpredictability/socioeconomic deprivation and decreased crystallized cognition, as well as decreased internalizing problems. The findings indicated that adolescents who experienced ELA might exhibit both adaptive and maladaptive behaviors with the “stress accelerated” corticolimbic development. This accelerating effect might lead to side effects—imbalanced maturation of the hippocampal cingulum and uncinate fasciculus—which impairs cognition [93]; crystallized cognition was more likely to be affected due to its reliance on experiential accumulation [95]. However, ELA or life stress events might have an ontogenetic adaptation by reducing internalizing problems in the short term [25, 43, 52]. Notably, repeated exposure to ELA might impose an allostatic load, accumulating burdens on individual’s body [40]; the long-term effects of ELA are unclear and need further study. In this study, ELA was not associated with adolescents’ externalizing problems via the corticolimbic circuits. Externalizing problems, however, may be associated with other brain metrics that were not measured in this study (e.g., rsFC between CON and DMN) [96]. The coexistence of adaptive and maladaptive behaviors underscored the need for future research to examine the long-term effects of interpersonal and socioeconomic ELA dimensions on brain and behaviors as well as the underlying neural mechanisms.
In predicting the performance of specific cognitive tasks, socioeconomic deprivation impacted a wide range of tasks, including Flanker Inhibitory Control & Attention, Pattern Comparison Processing Speed, Picture Sequence Memory, and Picture Vocabulary, while interpersonal unpredictability was only associated with picture-related tasks, through rsFC of the corticolimbic circuits. These findings were aligned with the definition of deprivation - limited or reduced social and cognitive inputs from the environment [6, 7] - thereby restricting children’s general cognitive development. Interestingly, socioeconomic deprivation improved the performance of Pattern Comparison Processing Speed (information processing). This phenomenon can be explained by the Hidden Talent Hypothesis, as ELA might has an ontogenetic adaptation to enhance cognitive flexibility [97]. The results highlighted the importance of examining the impact of ELA on specific cognitive aspects, as there may be nuanced difference among them. Interpersonal unpredictability impaired specifically picture-related vocabulary knowledge and memory, which may be due to children’s biased attention to parent-child negative signals as they exhibited greater ventral attention network activation during sad or emotional scenes [91].
Moreover, we found that interpersonal unpredictability and corticolimbic rsFC mediated the association between socioeconomic deprivation and decreased crystallized cognition. The finding demonstrated the possibility that caregiving unpredictability may be a process through which other forms of ELA (e.g., poverty) influence development [35]. Socioeconomic deprivation occurred alongside various types of ELA and exerted a notable influence on the immediate environment, including disrupted family functioning, affecting communication, and parental roles [98, 99]; therefore, economic strain, which was responsible for cognitive development (e.g., language processing and executive functions) [28,29,30] may operate through a more unpredictable family environment and inconsistent parental behaviors.
The serial mediation models helped us understand the complex mechanisms underlying the effects of ELA on brain and behaviors, allowing us to identify the root causes and pinpoint intervention targets - neighborhood poverty, to promote stable family environments and children’s optimal neural and behavioral development. This conclusion was aligned with the United Nations’ 2030 Sustainable Development Goal 1—end poverty in all its forms everywhere (https://sdgs.un.org/goals/goal1). This study underscored the critical roles of family stability and socioeconomic conditions in shaping child developmental outcomes. Policymakers and educators could use these insights to design targeted interventions, such as expanding access to affordable childcare, offering direct financial support to low-income families, and implementing parental training programs to reduce threat and unpredictability in family environments. Furthermore, longitudinal studies, like the ABCD Study, can be leveraged to track policy effectiveness over time, providing empirical evidence for refining interventions to support vulnerable children.
Limitations
This study has some limitations. First, we only tested how interpersonal/socioeconomic ELA dimensions associated with neural and behavioral development over a three-year period. With the continuation of ABCD study, we hope to examine the prolonged effects of ELA as the participants enter middle and late adolescence. Additionally, we did not specifically assess ELA in infancy or early childhood, which represent critical periods warranting further exploration. Second, we assessed the rsFC of corticolimbic circuits and cognitive abilities both at the 2-year follow-up due to the absence of cognitive assessments at the 3-year follow-up in the ABCD dataset. Future research can further track changes in cognitive ability with the continuation of the ABCD study. Third, we measured unpredictability from an ancestral cues perspective, which made it difficult to clearly distinguish between threat/deprivation and unpredictability. Future research can combine the perspectives of statistical learning and ancestral cues [65]. Finally, we utilized generalized scales rather than those specifically designed to measure ELA, which was insufficient to fully capture the different dimensions of ELA and might affect the generalizability of the conclusions. Future research should employ more targeted measurements to validate these findings. Particularly, although informed by previous studies [66, 67], we assessed unpredictability using items drawn from another measure rather than a validated instrument specifically designed for unpredictability. Although our findings suggested potential differential effects of interpersonal vs. socioeconomic ELA on children’s neural and behavioral development, the results should be interpreted with caution. The lack of a validated measure of unpredictability may account for these findings. Future studies should utilize validated tools, such as the Questionnaire of Unpredictability in Childhood (QUIC) [100], to replicate our findings.
Conclusions
This study found distinct associations between multiple ELA categories and adolescents’ development. More importantly, this study highlighted the roles of interpersonal unpredictability and socioeconomic deprivation as pivotal environmental factors to influence the development of the corticolimbic circuits implicated in cognitive function and psychopathology. It is crucial for parents to provide stable and predictable family environments and for neighborhoods to provide more enriched environment to benefit the normative neurodevelopment of children and adolescents, thereby to promote adaptive cognitive and emotional development.
Data availability
Neuroimaging and behavioral data from ABCD dataset are obtained from https://nda.nih.gov/abcd with the approval of the ABCD consortium.
Code availability
The code used for the analyses is available upon reasonable request from the corresponding author.
References
Sacks V, Murphey D. The prevalence of adverse childhood experiences, nationally, by state, and by race or ethnicity. Child Trends. 2018;20:2018.
Yeo G, Lansford JE, Hirshberg MJ, Tong EMW. Associations of childhood adversity with emotional well-being and educational achievement: a review and meta-analysis. J Affect Disord. 2024;347:387–98.
Ireton R, Hughes A, Klabunde M. A functional magnetic resonance imaging meta-analysis of childhood trauma. Biol Psychiatry Cogn Neurosci Neuroimaging. 2024;9:561–70.
Tottenham N, Galván A. Stress and the adolescent brain: amygdala-prefrontal cortex circuitry and ventral striatum as developmental targets. Neurosci Biobehav Rev. 2016;70:217–27.
Ellis BJ, Figueredo AJ, Brumbach BH, Schlomer GL. Fundamental dimensions of environmental risk : the impact of harsh versus unpredictable environments on the evolution and development of life history strategies. Hum Nat. 2009;20:204–68.
McLaughlin KA, Sheridan MA, Lambert HK. Childhood adversity and neural development: deprivation and threat as distinct dimensions of early experience. Neurosci Biobehav Rev. 2014;47:578–91.
Sheridan MA, McLaughlin KA. Dimensions of early experience and neural development: deprivation and threat. Trends Cogn Sci. 2014;18:580–5.
Liu S, Fisher PA. Early experience unpredictability in child development as a model for understanding the impact of the COVID-19 pandemic: a translational neuroscience perspective. Dev Cogn Neurosci. 2022;54:101091.
Colich NL, Rosen ML, Williams ES, McLaughlin KA. Biological aging in childhood and adolescence following experiences of threat and deprivation: a systematic review and meta-analysis. Psychol Bull. 2020;146:721–64.
McLaughlin KA, Weissman D, Bitrán D. Childhood adversity and neural development: a systematic review. Annu Rev Dev Psychol. 2019;1:277–312.
Goetschius LG, Hein TC, McLanahan SS, Brooks-Gunn J, McLoyd VC, Dotterer HL, et al. Association of childhood violence exposure with adolescent neural network density. JAMA Netw Open. 2020;3:e2017850.
Rakesh D, Allen NB, Whittle S. Longitudinal changes in within-salience network functional connectivity mediate the relationship between childhood abuse and neglect, and mental health during adolescence. Psychol Med. 2023;53:1552–64.
Yang Y, Kong T, Ji F, Liu R, Luo L. Associations among environmental unpredictability, changes in resting-state functional connectivity, and adolescent psychopathology in the ABCD study. Psychol Med. 2024;54:4119–28.
Chahal R, Miller JG, Yuan JP, Buthmann J, Gotlib I. An exploration of dimensions of early adversity and the development of functional brain network connectivity during adolescence: implications for trajectories of internalizing symptoms. Dev Psychopathol. 2022;34:557–71.
Jirsaraie RJ, Palma AM, Small SL, Sandman CA, Davis EP, Baram TZ, et al. Prenatal exposure to maternal mood entropy is associated with a weakened and inflexible salience network in adolescence. Biol Psychiatry Cogn Neurosci Neuroimaging. 2024;9:207–16.
Cisler JM, Scott Steele J, Smitherman S, Lenow JK, Kilts CD. Neural processing correlates of assaultive violence exposure and PTSD symptoms during implicit threat processing: a network-level analysis among adolescent girls. Psychiatry Res. 2013;214:238–46.
Thomason ME, Marusak HA, Tocco MA, Vila AM, McGarragle O, Rosenberg DR. Altered amygdala connectivity in urban youth exposed to trauma. Soc Cogn Affect Neurosci. 2015;10:1460–8.
Saxbe D, Khoddam H, Piero LD, Stoycos SA, Gimbel SI, Margolin G, et al. Community violence exposure in early adolescence: longitudinal associations with hippocampal and amygdala volume and resting state connectivity. Dev Sci. 2018;21:e12686.
Ellis BJ, Sheridan MA, Belsky J, McLaughlin KA. Why and how does early adversity influence development? Toward an integrated model of dimensions of environmental experience. Dev Psychopathol. 2022;34:447–71.
Bronfenbrenner, U (eds) The ecology of human development: experiments by nature and design. Cambridge:Harvard University Press; 1979.
Martin P, Martin M. Proximal and distal influences on development: the model of developmental adaptation. Dev Rev. 2002;22:78–96.
Vannucci A, Fields A, Bloom PA, Camacho NL, Choy T, Durazi A et al. Probing the content of affective semantic memory following caregiving‐related early adversity. Dev Sci. 2024. https://doi.org/10.1111/desc.13518.
Vannucci A, Fields A, Hansen E, Katz A, Kerwin J, Tachida A, et al. Interpersonal early adversity demonstrates dissimilarity from early socioeconomic disadvantage in the course of human brain development: a meta-analysis. Neurosci Biobehav Rev. 2023;150:105210.
Bick J, Nelson CA. Early adverse experiences and the developing brain. Neuropsychopharmacology. 2016;41:177–96.
Callaghan BL, Tottenham N. The stress acceleration hypothesis: effects of early-life adversity on emotion circuits and behavior. Curr Opin Behav Sci. 2016;7:76–81.
Thijssen S, Collins PF, Luciana M. Does pubertal stage mediate the association between family environment and structure and function of the amygdala-mPFC circuit? A replication study of the longitudinal ABCD cohort. Dev Cogn Neurosci. 2022;56:101120.
van der Werff SJA, Pannekoek JN, Veer IM, van Tol M-J, Aleman A, Veltman DJ, et al. Resting-state functional connectivity in adults with childhood emotional maltreatment. Psychol Med. 2013;43:1825–36.
Brito NH, Noble KG. Socioeconomic status and structural brain development. Front Neurosci. 2014;8:276.
Michael C, Tillem S, Sripada CS, Burt SA, Klump KL, Hyde LW. Neighborhood poverty during childhood prospectively predicts adolescent functional brain network architecture. Dev Cogn Neurosci. 2023;64:101316.
Sripada C, Gard AM, Angstadt M, Taxali A, Greathouse T, McCurry K, et al. Socioeconomic resources are associated with distributed alterations of the brain’s intrinsic functional architecture in youth. Dev Cogn Neurosci. 2022;58:101164.
Barnett W, Hansen CL, Bailes LG, Humphreys KL. Caregiver–child proximity as a dimension of early experience. Dev Psychopathol. 2022;34:647–65.
Vedechkina M, Astle DE, Holmes J. Dimensions of early life adversity and their associations with functional brain organisation. Imaging Neurosci. 2024;2:1–25.
Dennison MJ, Rosen ML, Sambrook KA, Jenness JL, Sheridan MA, McLaughlin KA. Differential associations of distinct forms of childhood adversity with neurobehavioral measures of reward processing: a developmental pathway to depression. Child Dev. 2019;90:e96–e113.
Mrug S, Loosier PS, Windle M. Violence exposure across multiple contexts: individual and joint effects on adjustment. Am J Orthopsychiatry. 2008;78:70–84.
Davis EP, Glynn LM. Annual research review: the power of predictability–patterns of signals in early life shape neurodevelopment and mental health trajectories. J Child Psychol Psychiatry. 2024;65:508–34.
Sosu EM, Schmidt P. Changes in cognitive outcomes in early childhood: the role of family income and volatility. Front Psychol. 2022;13:758082.
Li Z, Liu S, Hartman S, Belsky J. Interactive effects of early-life income harshness and unpredictability on children’s socioemotional and academic functioning in kindergarten and adolescence. Dev psychol. 2018;54:2101.
Pilarz AR, Hill HD. Unstable and multiple child care arrangements and young children’s behavior. Early Child Res Q. 2014;29:471–83.
McLaughlin KA, Sheridan MA. Beyond cumulative risk: a dimensional approach to childhood adversity. Curr Dir Psychol Sci. 2016;25:239–45.
Vaidya N, Marquand AF, Nees F, Siehl S, Schumann G. The impact of psychosocial adversity on brain and behaviour: an overview of existing knowledge and directions for future research. Mol Psychiatry. 2024. https://doi.org/10.1038/s41380-024-02556-y.
Ho TC, King LS. Mechanisms of neuroplasticity linking early adversity to depression: developmental considerations. Transl Psychiatry. 2021;11:517.
Daliri MR, Behroozi M. Advantages and disadvantages of resting state functional connectivity magnetic resonance imaging for clinical applications. OMICS J Radiol. 2013;3:1–2.
Brieant AE, Sisk LM, Gee DG. Associations among negative life events, changes in cortico-limbic connectivity, and psychopathology in the ABCD Study. Dev Cogn Neurosci. 2021;52:101022.
Kaiser RH, Clegg R, Goer F, Pechtel P, Beltzer M, Vitaliano G, et al. Childhood stress, grown-up brain networks: corticolimbic correlates of threat-related early life stress and adult stress response. Psychol Med. 2018;48:1157–66.
Dosenbach NU, Fair DA, Miezin FM, Cohen AL, Wenger KK, Dosenbach RA, et al. Distinct brain networks for adaptive and stable task control in humans. Proc Natl Acad Sci. 2007;104:11073–8.
Gordon EM, Laumann TO, Adeyemo B, Huckins JF, Kelley WM, Petersen SE. Generation and evaluation of a cortical area parcellation from resting-state correlations. Cereb Cortex. 2016;26:288–303.
Safar K, Vandewouw MM, Taylor MJ. Atypical development of emotional face processing networks in autism spectrum disorder from childhood through to adulthood. Dev Cogn Neurosci. 2021;51:101003.
Vann SD, Aggleton JP, Maguire EA. What does the retrosplenial cortex do? Nat Rev Neurosci. 2009;10:792–802.
Mitchell AS, Czajkowski R, Zhang N, Jeffery K, Nelson AJD. Retrosplenial cortex and its role in spatial cognition. Brain Neurosci Adv. 2018;2:2398212818757098.
Grayson DS, Fair DA. Development of large-scale functional networks from birth to adulthood: a guide to the neuroimaging literature. Neuroimage. 2017;160:15–31.
Xiao Y, Friederici AD, Margulies DS, Brauer J. Longitudinal changes in resting-state fMRI from age 5 to age 6years covary with language development. Neuroimage. 2016;128:116–24.
Gee DG, Gabard-Durnam LJ, Flannery J, Goff B, Humphreys KL, Telzer EH, et al. Early developmental emergence of human amygdala-prefrontal connectivity after maternal deprivation. Proc Natl Acad Sci USA. 2013;110:15638–43.
Rakesh D, Kelly C, Vijayakumar N, Zalesky A, Allen NB, Whittle S. Unraveling the consequences of childhood maltreatment: deviations from typical functional neurodevelopment mediate the relationship between maltreatment history and depressive symptoms. Biol Psychiatry Cogn Neurosci Neuroimaging. 2021;6:329–42.
Dennis EL, Thompson PM. Typical and atypical brain development: a review of neuroimaging studies. Dialogues Clin Neurosci. 2013;15:359–84.
van Polanen M, Colonnesi C, Fukkink RG, Tavecchio LW. Is caregiver gender important for boys and girls? Gender-specific child–caregiver interactions and attachment relationships. Early Educ Dev. 2017;28:559–71.
Young ES, Vermeent S, Frankenhuis WE, Nivison MD, Simpson JA, Roisman GI. How does adversity relate to performance across different abilities within individuals? Dev Psychopathol. 2024;23:1–18. https://doi.org/10.1017/S0954579424001433.
Casey BJ, Cannonier T, Conley MI, Cohen AO, Barch DM, Heitzeg MM, et al. The Adolescent Brain Cognitive Development (ABCD) study: Imaging acquisition across 21 sites. Dev Cogn Neurosci. 2018;32:43–54.
Clark DB, Fisher CB, Bookheimer S, Brown SA, Evans JH, Hopfer C, et al. Biomedical ethics and clinical oversight in multisite observational neuroimaging studies with children and adolescents: the ABCD experience. Dev Cogn Neurosci. 2018;32:143–54.
Moos, RH & Moos, BS (eds) Family environment scale manual: development,applications,research. Palo Alto:Consulting Psychologists Press; 2002.
Echeverria SE, Diez-Roux AV, Link BG. Reliability of self-reported neighborhood characteristics. J Urban Health. 2004;81:682–701.
Mujahid MS, Diez Roux AV, Morenoff JD, Raghunathan T. Assessing the measurement properties of neighborhood scales: from psychometrics to ecometrics. Am J Epidemiol. 2007;165:858–67.
Grant KE, Compas BE, Thurm AE, McMahon SD, Gipson PY. Stressors and child and adolescent psychopathology: measurement issues and prospective effects. J Clin Child Adolesc Psychol. 2004;33:412–25.
Hoffman EA, Clark DB, Orendain N, Hudziak J, Squeglia LM, Dowling GJ. Stress exposures, neurodevelopment and health measures in the ABCD study. Neurobiol Stress. 2019;10:100157.
Tiet QQ, Bird HR, Davies M, Hoven C, Cohen P, Jensen PS, et al. Adverse life events and resilience. J Am Acad Child Adolesc Psychiatry. 1998;37:1191–1200.
Young ES, Frankenhuis WE, Ellis BJ. Theory and measurement of environmental unpredictability. Evol Hum Behav. 2020;41:550–6.
Belsky J, Schlomer GL, Ellis BJ. Beyond cumulative risk: distinguishing harshness and unpredictability as determinants of parenting and early life history strategy. Dev Psychol. 2012;48:662–73.
Mittal C, Griskevicius V, Simpson JA, Sung S, Young ES. Cognitive adaptations to stressful environments: when childhood adversity enhances adult executive function. J Pers Soc Psychol. 2015;109:604–21.
Cochrane R, Robertson A. The life events inventory: a measure of the relative severity of psycho-social stressors. J Psychosom Res. 1973;17:135–40.
Karoly HC, Callahan T, Schmiege SJ, Ewing SWF. Evaluating the hispanic paradox in the context of adolescent risky sexual behavior: the role of parent monitoring. J Pediatr Psychol. 2016;41:429–40.
Stattin H, Kerr M. Parental monitoring: a reinterpretation. Child Dev. 2000;71:1072–85.
McDonald S. Special series on the cognition battery of the NIH toolbox. J Int Neuropsychol Soc. 2014;20:487–651.
Achenbach, TM (eds) The achenbach system of empirically based assessment (ASEBA): development, findings, theory, and applications. Burlington:University of Vermont, Research Center for Children, Youth, & Families; 2009.
Karcher NR, Barch DM. The ABCD study: understanding the development of risk for mental and physical health outcomes. Neuropsychopharmacology. 2021;46:131–42.
Zhi D, Jiang R, Pearlson G, Fu Z, Qi S, Yan W, et al. Triple interactions between the environment, brain, and behavior in children: an ABCD study. Biol Psychiatry. 2024;95:828–38.
Hagler DJ Jr, Hatton S, Cornejo MD, Makowski C, Fair DA, Dick AS, et al. Image processing and analysis methods for the adolescent brain cognitive development study. Neuroimage. 2019;202:116091.
Fischl B, Salat DH, Busa E, Albert M, Dieterich M. Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron. 2002;33:341–55.
McNeish D, Stapleton LM, Silverman RD. On the unnecessary ubiquity of hierarchical linear modeling. Psychol Methods. 2017;22:114–40.
Muthén, LK & Muthén, BO (eds) The comprehensive modeling program for applied researchers user guide. Los Angeles: Muthén & Muthén; 2003.
Dixon WJ, Yuen KK. Trimming and winsorization: a review. Statistische Hefte. 1974;15:157–70.
Hair, JF, Black, WC, Babin, BJ, & Anderson, RE (eds) Multivariate data analysis. Upper Saddle River:Pearson Education; 2014.
McLaughlin KA, Sheridan MA, Nelson CA. Neglect as a violation of species-expectant experience: neurodevelopmental consequences. Biol Psychiatry. 2017;82:462–71.
Gee DG, Cohodes EM. Caregiving influences on development: a sensitive period for biological embedding of predictability and safety cues. Curr Dir Psychol Sci. 2021;30:376–83.
Jensen SKG, Berens AE, Nelson CA 3rd. Effects of poverty on interacting biological systems underlying child development. Lancet Child Adolesc Health. 2017;1:225–39.
Raphael D. Poverty in childhood and adverse health outcomes in adulthood. Maturitas. 2011;69:22–26.
Ip KI, Sisk LM, Horien C, Conley MI, Rapuano KM, Rosenberg MD, et al. Associations among household and neighborhood socioeconomic disadvantages, resting-state frontoamygdala connectivity, and internalizing symptoms in youth. J Cogn Neurosci. 2022;34:1810–41.
Webb EK, Weis CN, Huggins AA, Fitzgerald JM, Bennett K, Bird CM, et al. Neural impact of neighborhood socioeconomic disadvantage in traumatically injured adults. Neurobiol Stress. 2021;15:100385.
Baram TZ, Davis EP, Obenaus A, Sandman CA, Small SL, Solodkin A, et al. Fragmentation and unpredictability of early-life experience in mental disorders. Am J Psychiatry. 2012;169:907–15.
Walker CD, Bath KG, Joels M, Korosi A, Larauche M, Lucassen PJ, et al. Chronic early life stress induced by limited bedding and nesting (LBN) material in rodents: critical considerations of methodology, outcomes and translational potential. Stress. 2017;20:421–48.
Glynn LM, Baram TZ. The influence of unpredictable, fragmented parental signals on the developing brain. Front neuroendocrinol. 2019;53:100736.
Smith KE, Pollak SD. Rethinking concepts and categories for understanding the neurodevelopmental effects of childhood adversity. Perspect Psychol Sci. 2021;16:67–93.
Furtado EJ, Camacho MC, Chin JH, Barch DM. Complex emotion processing and early life adversity in the healthy brain network sample. Dev Cogn Neurosci. 2024;70:101469.
Landsheer JA, van Dijkum C. Male and female delinquency trajectories from pre through middle adolescence and their continuation in late adolescence. Adolescence. 2005;40:729–48.
Granger SJ, Glynn LM, Sandman CA, Small SL, Obenaus A, Keator DB, et al. Aberrant maturation of the uncinate fasciculus follows exposure to unpredictable patterns of maternal signals. J Neurosci. 2021;41:1242–50.
De Bellis MD, Hooper SR, Chen SD, Provenzale JM, Boyd BD, Glessner CE, et al. Posterior structural brain volumes differ in maltreated youth with and without chronic posttraumatic stress disorder. Dev psychopathol. 2015;27:1555–76.
Cattell RB Abilities: their structure, growth, and action. Oxford:Houghton Mifflin; 1971.
Chen Z, Guo Y, Suo T, Feng T. Coupling and segregation of large-scale brain networks predict individual differences in delay discounting. Biol Psychiatry. 2018;133:63–71.
Ellis BJ, Abrams LS, Masten AS, Sternberg RJ, Tottenham N, Frankenhuis WE. Hidden talents in harsh environments. Dev psychopathol. 2022;34:95–113.
Banovcinova A, Levicka J, Veres M. The impact of poverty on the family system functioning. Procedia - Soc Behav Sci. 2014;132:148–53.
Yob Z, Shaari MS, Esquivias MA, Nangle B, Muhamad WZAW. The impacts of poverty, unemployment, and divorce on child abuse in Malaysia: ARDL approach. Econ Soc. 2022;10:291.
Glynn LM, Stern HS, Howland MA, Risbrough VB, Baker DG, Nievergelt CM, et al. Measuring novel antecedents of mental illness: the Questionnaire of Unpredictability in Childhood. Neuropsychopharmacology. 2019;44:876–82.
Acknowledgements
Data used in the preparation of this article were obtained from the Adolescent Brain Cognitive DevelopmentSM (ABCD) Study (https://abcdstudy.org), held in the NIMH Data Archive (NDA). This is a multisite, longitudinal study designed to recruit more than 10 000 children age 9–10 and follow them over 10 years into early adulthood. The ABCD Study® is supported by the National Institutes of Health and additional federal partners under award numbers U01DA041048, U01DA050989, U01DA051016, U01DA041022, U01DA051018, U01DA051037, U01DA050987, U01DA041174, U01DA041106, U01DA041117, U01DA041028, U01DA041134, U01DA050988, U01DA051039, U01DA041156, U01DA041025, U01DA041120, U01DA051038, U01DA041148, U01DA041093, U01DA041089, U24DA041123, U24DA041147. A full list of supporters is available at https://abcdstudy.org/federal-partners.html. A listing of participating sites and a complete listing of the study investigators can be found at https://abcdstudy.org/consortium_members/. ABCD consortium investigators designed and implemented the study and/or provided data but did not necessarily participate in the analysis or writing of this report. This manuscript reflects the views of the authors and may not reflect the opinions or views of the NIH or ABCD consortium investigators. The ABCD data repository grows and changes over time. The ABCD data used in this report came from https://doi.org/10.15154/wcfv-q195. DOIs can be found at https://doi.org/10.15154/wcfv-q195. Additional support for this work was made possible from NIEHS R01-ES032295 and R01-ES031074.
Funding
This work was supported by the Humanities and Social Science Fund of Ministry of Education of China (RL, grant number 23YJC190015), the STI 2030-Major Projects (LL, grant number 2021ZD0200500), the Fundamental Research Funds for the Central Universities (LL, grant number 01900310400209543), the National Natural Science Foundation of China (RL, grant number 32400890), and the Fundamental Research Funds for the Central Universities of Beijing Normal University, Scientific Research Starting Project for Young Scholars (RL, grant number 2023NTSS21).
Author information
Authors and Affiliations
Contributions
Yumeng Yang proposed the study, analyzed the data, and wrote the manuscript. Tianjiao Kong assisted with data organization and formatting proofreading. Ran Liu and Liang Luo provided guidance on how to analyze the data and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yang, Y., Kong, T., Liu, R. et al. Associations of interpersonal and socioeconomic early life adversity dimensions with adolescents’ corticolimbic circuits, cognition, and mental health. Transl Psychiatry 15, 168 (2025). https://doi.org/10.1038/s41398-025-03384-6
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41398-025-03384-6