Abstract
Recent genetic findings have highlighted the importance of metabolic factors in contributing to risk for anorexia nervosa. The treatment of anorexia nervosa, however, has largely not yet attempted to modulate metabolic pathways, given that knowledge and understanding of potential metabolic targets and treatment goals remain limited. In search of potential metabolic mechanisms at play in psychiatric disorders, we look at the brain and beyond, focusing on ketones, related molecules, and nutritional therapies, e.g. the ketogenic diet, highlighting their brain and behavioural effects. The aim of this review is therefore to summarize knowledge on the physiology of ketones and related molecules, as well as evidence on their role on energy balance, brain, and behaviour. We then review the role ketones might have on the pathophysiology of anorexia nervosa, and how the intake of exogenous ketones and ketone precursors might impact on brain and behaviour in anorexia nervosa. We conclude with an hypothesised model explaining the potential role of ketones in the pathophysiology of anorexia nervosa and propose that exogenous ketones and ketone precursors could be an alternative source of brain energy in this illness and deserve further investigation.
Similar content being viewed by others
Introduction
In the last years, a shift in the conceptualization of anorexia nervosa (AN) from being purely a brain-based disorder to a multi-factorial disease implicating a combination of psychiatric, metabolic, and anthropometric factors is emerging [1]. Genetic factors related to metabolism are important in AN [2], but their association with cognitions and behaviours characteristic of Eating Disorders (ED) in the general population is not clear yet [3]. Hence metabolic factors might be crucial for the transition from symptoms to a full-blown disorder, and might also be implicated in the maintenance of AN. Shifts in metabolism as precursors/catalysts of AN-like symptoms have been documented since Keys’ famous “Minnesota’ studies [4], however the role of metabolic factors in the pathophysiology of AN is not clear and complex to study.
Metabolism and metabolic markers are closely interconnected with nutrition. Substantial literature and evidence-based guidelines support medical nutrition therapy, i.e., refeeding, in the service of weight restoration and to offset the impact of starvation [5]. In medical settings, indeed, nutrition has traditionally been used as an adjunctive treatment to improve lipid or glucose metabolism and thereby aid disorders like hypercholesterolemia and diabetes [6]. In AN, although refeeding is an integral part of treatment, the potential role of nutrition in targeting cognitions and behaviours of AN, hence having a role beyond weight restoration per se, with the potential to change brain metabolism and function, has largely been overlooked.
Amongst nutritional ‘therapies’ ketogenic diets, in particular, have been shown to influence several biological processes, including mitochondrial energy metabolism, inflammatory processes, oxidative stress, monoaminergic activity in the brain, and slowing down the progression of neuro-degeneration [7, 8], and hence are considered ‘metabolic therapies’ in themselves [9,10,11]. In particular, recent evidence points to the ketogenic diet (KD) as a potential treatment for neurodegenerative and neuropsychiatric conditions [12, 13]. Initially used by clinicians in the 1920s as a treatment for epilepsy [14], this high-fat, moderate protein, low-carbohydrate diet facilitates the production of ketone bodies (KBs: D-β−hydroxybutyrate (D-BHB) and aceto-acetate (AcAc)) from fatty acids, which serve as an alternative fuel, diverting away from the use of glucose as main energy source of the brain. By adhering to a sustained KD, an individual achieves a level of nutritional ketosis [15], characterized by high levels of blood ketones. Ketones are actively transported to the brain via Mono-Carboxylate transporters, where they are used as energy sources preferentially to glucose, and where they can mitigate the energy gap during times of glucose deprivation or increased energy demands [16]. This is evident for example in fasting states, during chronic energy restriction (with a significant reduction in caloric intake), and following significant [17] carbohydrate restriction [18, 19].
Fasting ketosis has a sexual dimorphism, in that pre-menopausal women develop fasting ketosis faster than men. This has been suggested to associate with a divergence in metabolic profiles between men and women [20]. The metabolic response to calorie restriction also differs between women and men; a recent review highlighted increased lipid metabolism during calorie restriction in women [21]. Women seem to rely more on lipolysis to release substrates in circulation in the context of fasting and exercise compared to men (who rely more on carbohydrate metabolism [22]).
Ketosis and metabolic adaptation to starvation
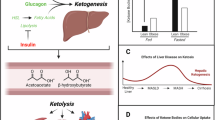
Ketogenesis is the biochemical process through which the body generates KBs in the liver from fatty acids as a source of energy [23]. Produced continuously in low concentrations under normal physiological conditions, the production of KBs rapidly increases in response to starvation, fasting, and prolonged exercise, or in dysregulated metabolic states, such as type 1 diabetes [24]. KBs are used by the brain, the heart, the kidney, and smooth muscles, while concurrently preserving vital glucose/gluconeogenic reserves [25]. Although the human brain is only 2% of the body volume, it consumes over 20% of its energy at rest, and accordingly the brain is particularly vulnerable to changes in metabolism [26]. While glucose is the brain’s default fuel, KBs provide 27% more free energy than glucose [27] while also causing less oxidative stress [28] and, therefore, they are highly effective energy substrates under conditions of low glucose. Indeed, KBs can provide up to 60% of the brain energy needs during fasting and reduction of glucose availability [29]. Notably, after 3 days of fasting a switch occurs and, instead of glucose, lipids and the resulting ketones are utilised as the primary source of fuel [29].
KBs and glucose metabolism are strongly linked to each other [30]. There is evidence that some neurons (e.g., glutamatergic neurons in the rat hippocampus) may preferentially use D-BHB for the generation of Acetyl-CoA, even if glucose is present [31]. Furthermore, KBs represent the main fuel of choice in humans in specific conditions (such as endurance exercise). Five separate studies of 39 high-performance athletes undergoing nutritional ketosis showed increased triacylglycerol oxidation in skeletal muscle and decreased glycolysis and lactate production [32]. This is important in this context, as increased lactate levels have been related to decreased motivation to exert effort [33]. The metabolic fuel shift in athletes occurred even in the presence of normal muscle glycogen, co-ingested carbohydrates, and elevated insulin [32]. Several mechanisms have been proposed to explain the reduction of the glycolytic flow by KBs, including an inhibitory feedback of glycolysis by a high acetyl-CoA/CoA or NADH/NAD+ (nicotinamide adenine dinucleotide) ratio and the involvement of uncoupling proteins (UCP2, UCP3, UCP4) [34]. In healthy resting conditions, the energy balance is maintained by a feedback loop mechanism at the level of the pyruvate dehydrogenase complex since the downstream metabolites such as ATP and Acetyl-CoA act as negative regulators. Hence, as more KBs are metabolized in the mitochondria to form Acetyl-CoA, less glucose is utilized [31, 35].
Other effects of ketone bodies and ketogenic diet on the brain
In addition to their role in body and brain metabolism, KBs have been (directly or indirectly via effects secondary to a ketogenic diet) shown to have other effects on the brain. In addition to brain metabolism, KBs affect regulation of cell signalling, neuronal firing, the release of glutamate, and gene expression modulation in the brain or neuronal cultures [36, 37]. There is also evidence of effects of exogenous ketones on circulating dopamine in humans and mice. [38, 39]. KBs act as signalling molecules that coordinate a complex responsive network in the brain [35].
KBs also modulate neuronal excitability through several mechanisms; for example, via a reduction in neuronal firing secondary to a direct or indirect regulation of the activity of several ion channels, particularly potassium ATP-sensitive (K-ATP) channels [40, 41]. The effect of ketones on neuronal firing is one of the main mechanisms through which ketogenic diets might have their beneficial effect on epilepsy [42] It has been shown that physiological concentrations of D-BHB and AcAc reduced the spontaneous firing rate of neurons [43], and restored energy status, calcium homeostasis, and M-channel activity [42] in slices from different rodent brain regions. A critical role of K-ATP channels as mediating the effect of KBs activity on neuronal firing has been emphasized in a mouse genetic model with enhanced KB metabolism [44, 45].
Further, KBs have also been shown to impair the release of the excitatory neurotransmitter glutamate [46, 47]. D-BHB directly inhibits the neuronal vesicular glutamate transporter (VGLUT) by binding to its allosteric site [48]. VGLUTs promote glutamate uptake in synaptic vesicles for subsequent release [49]. By limiting glutamate uptake into synaptic vesicles, KBs were shown to reduce the secretion of glutamate into the synaptic cleft, thus further blocking post-synaptic neuronal excitability [50].
KBs have also been found to promote long–term adaptive modifications through changes in gene expression. Notably, KBs play a role in upregulating the expression of the Brain-Derived Neurotrophic Factor (BDNF) in the central nervous system [51]. BDNF is crucial in the development of neuronal circuits regulating cognition, anxiety, and eating-related behaviours [52,53,54]. Effects of KBs on BDNF have been shown in exposure to a ketogenic diet in humans and rodents. For example, an up-regulation of BDNF expression has been shown in cerebral cortical neurons in mice through D-BHB action [17, 51]. Exploratory studies in human adults have found associations between D-BHB and plasma BDNF either secondary to a KD intervention or in the context of exogenous ketone ingestion [55,56,57].
All together these data indicate that KBs exhibit diverse effects on the brain that might be relevant to psychiatric disorders characterized by dysfunction in energy metabolism [58, 59].
In contrast, mixed findings are available on the effect of the KD on brain serotonin and dopamine levels, with evidence of an increase in dopamine but not serotonin [60] in rodents, and a study on children on a KD showing a decrease in serotonin and dopamine metabolites in CSF [61]. The KD seems to modulate serotonin metabolism [62] and down-regulate serotonin in the colon (whilst increasing brain BDNF) in an animal model of irritable bowel syndrome [63].
Evidence on the gut-brain axis has proliferated in recent years, with important insights into the crucial role of some gut cells (e.g. the enterochromaffin cells) in gut-brain signalling [64,65,66,67,68]. The enterochromaffin cells in fact are affected by diet, for example by ‘sensing’ diets high in fat and sugar content and releasing serotonin in response [69]. Serotonin release in the gut can modulate brain circuits and in turn affect emotion and behaviour [70].
An additional way in which KB and the KD can impact on the brain is via their effect on gut microbiome. There is increasing evidence of the impact of the KD on gut microbiome [65, 71], and many studies show a positive effect of this diet on gut dysbiosis [13, 72,73,74,75]. However, the breadth of evidence on this topic goes beyond the scope of the current review.
Exogenous ketone supplementation and ketogenic precursors
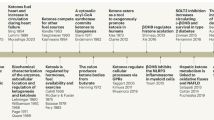
An alternative approach to obtain the putative benefits of KBs, without the dietary restrictions inherent to the KD, involves supplementation with “exogenous ketones” (EKs) [67, 76]. See Table 1 for an overview on EKs. EK precursors were developed to generate a dose-dependent transient increase of blood KBs regardless of dietary macronutrient intake. EK precursors can induce a rapid and controlled ketosis within minutes to hours in contrast to KDs, which require 3 to 5 days of strict dietary intervention [77]. They may represent an alternative tool to induce ketosis for individuals unwilling to adhere to a KD or when the KD is contraindicated [76]. EK precursors have been suggested as a therapeutic tool in chronic disease, with impact on metabolic, cognitive and immune function amongst others [78], but evidence is still scarce.
Several EK precursors are under investigation and include medium-chain triglycerides (MCTs) of various chain length, D-BHB salts, and R-1,3-Butandiol (R-1,3-BD). Synthetic Ketone Esters (KE) combining some of these precursors by ester bonds have also been developed. Upon oral intake, KEs are cleaved by gastric and blood esterases and their individual ketone precursor components released into systemic circulation where they are converted to KBs by the liver. Below, we briefly review the most well described EK precursors that have been tested in humans and highlight some of their reported cerebral metabolic effects.
Medium-chain triglycerides
Ketogenic Medium-chain triglycerides (MCTs) are made of saturated fats of six to ten carbons in chain length. They are rapidly hydrolysed by gastric lipase [79], and the resulting medium chain free fatty acid (MCFFA) rapidly absorbed, and directly oxidized by the liver, bypassing the carnitine regulatory pathway and leading to efficient ketone production. Bolus ingestion of emulsified MCTs provides a rapid increase in blood KBs. MCFFA chain length of 8 carbons (C8) provides the highest amount of blood KBs, while MCFFA of 10 carbons (C10) are less potent at producing ketones [80]. MCTs can specifically affect brain function and increasing research has shown improvements in neurobehavioral symptoms -including anxiety- and cognitive function with MCT intake [81, 82].
Intake of an MCT-based ketogenic drink for 6 months improved performance in several cognitive functions, such as memory, task switching, attention, in individuals with mild-cognitive impairment [83]. In addition, a positive effect on Alzheimer’s Disease symptoms has been shown following intake of a MCT C8 supplement [84, 85]. In healthy participants, MCTs can increase brain NAD redox ratio [86], which is known to be decreased in psychotic disorders [87]. Interestingly MCTs rich in C10 had a beneficial effect on the frequency of seizures or paroxysmal events in individuals with drug-resistant epilepsy, despite absent or very low levels of ketosis [88]. This suggests that C10 might have a direct mechanism of action perhaps unrelated to ketosis.
D-Beta-Hydroxybutyrate salt
Perhaps the most physiological way to raise blood ketones is via the oral intake of D-Beta-Hydroxybutyrate (D-BHB). Exogenous D-BHB is directly absorbed into the circulation, with some of it being converted to AcAc by the liver, and both ketones being distributed throughout the body. Oral D-BHB increases blood KBs to millimolar levels [89]. At the same dose, D-BHB is significantly more ketogenic and provides fewer calories than a racemic mixture of D and L-BHB or medium chain triglyceride. In a whole-body ketone positron emission tomography pilot study, it was observed that after D-BHB consumption, the ketone tracer 11C-acetoacetate is rapidly metabolized by the brain, but mostly by the heart and the kidneys. Beyond brain energy rescue, this opens additional opportunities for therapeutic exploration of D-BHB supplements as a “super fuel” in cardiac and chronic kidney diseases. While D-BHB, the natural and metabolically active ketone, is rapidly metabolized to energy, L-BHB is not and is slowly eliminated via the urine [89, 90].
R-1,3-Butanediol
Following ingestion, R-1,3-butanediol (R-1,3-BD) is rapidly absorbed in the gut and increases blood R-1,3-BD before undergoing rapid hepatic conversion to D-BHB and AcAc [91]. Recently, R-1,3-BD has been tested in human and show similar increase in blood KB compared to D-BHB [92]. In the context of endurance exercise, racemic R + S-1,3-Butanediol was also tested in human alone or in combination with a glucose supplement [93, 94].
Ketone ester (R)-3-hydroxybutyl (R)-3-hydroxybutyrate
(R)-3-hydroxybutyl (R)-3-hydroxybutyrate (HHB-KE) is a synthetic ketone ester where D-BHB and R-1,3-BD are held together by an ester bond. HHB-KE is cleaved by blood esterase to release its individual components and increases significantly blood KBs [90] following acute and chronic consumption [95]. HHB-KE has been tested in humans for its athletic improving and weight loss properties [32, 96], and more recently for the potential beneficial effects on cognition and executive functions in both healthy young men [97] and in cognitively intact adults [98]. In a brain Magnetic Resonance Spectroscopic (MRS) study in healthy participants, oral intake of HHB-KE was shown to reduce GABA and Glu in the anterior and posterior cortices, an effect specific to HHB-HE and not observed after administration of glucose [99]. HHB-HE was also shown to increase brain network stabilization [100], a measure correlated with brain activity and cognitive acuity, suggesting that dietary interventions resulting in ketone utilization increase available brain energy and thus may show potential in protecting the aging brain.
Ketone diester 1,3-butanediol acetoacetate
R,S-1,3-butanediol acetoacetate diester (BD-AcAc2) combine 2 molecules of acetoacetate (AcAc) and one molecule of racemic 1,3-BD linked by 2 ester bonds [101]. BD-AcAc2 has been shown to induce therapeutic ketosis in several pre-clinical models and in one clinical study performed in elite athletes, where millimolar of blood KBs were achieved [102].
Ketone diester bis hexanoyl R-1,3-butanediol
A novel ketone ester, bis hexanoyl R-1,3-butanediol (BH-BD) combines two MCFFA hexanoic acids esterified on a R-1,3-BD backbone. Pre-clinical results demonstrated that BH-BD is efficiently cleaved by esterase into its individual components, which can then be metabolized into BHB [103]. Acute and chronic studies in healthy adults demonstrate that BH-BD dose dependently increased blood ketone concentration to millimolar level independent of nutritional state [104, 105].
The potential role of ketones in the pathophysiology of Anorexia Nervosa
AN is principally characterized by severe restriction of food intake. This severe food restriction overrides physiological feeding requirements, with associated undernutrition, starvation, and a series of serious medical complications including: extremely low weight, cardiovascular instability, bone demineralisation, and cachexia [106]. In addition to its effects on the body, AN also has effects on the brain. Evidence from an international consortium showed that severely underweight individuals diagnosed with AN differed in cortical thickness, volume of subcortical regions, and (albeit to a lesser degree) of cortical areas compared to normal-weight controls and patients with partially recovered weight [68]. Moreover, larger effects were associated with a lower Body Mass Index (BMI), thus emphasizing the strong impact of undernutrition and starvation on the brain [68]. In a narrative review, Ayton & Ibrahim [107] highlight the role of KBs and the KD in AN in the context of broader metabolic influences and dietary intake in AN.
Starvation and energy for the brain in AN
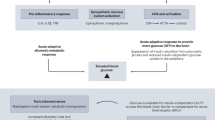
Behavioural characteristic of AN, such as caloric restriction and undereating, lead to low nutrient and fat intake, eventually leading to starvation (Fig. 1). In healthy individuals, once the starved state is established, mild ketosis is reached after 12–14 h of fasting [108] and ketones sharply increase after 24–28 h of water-only fasting [109]. At this stage, ketones will become the main source of energy for the brain, as discussed above, and will provide faster fuel (especially for the brain) than glucose. Although this has not been directly shown in AN, brain PET studies in AN show brain glucose hypometabolism, with lower brain glucose metabolism being ‘state’ rather than ‘trait’ dependent, and potentially due to low BMI and starvation [110]. However, to date there are no studies directly measuring brain KB utilization in AN [110], and few reporting ketosis in individuals with AN [111].
AN reduces nutrient and fat intake, leading to interconnected changes in peripheral and brain metabolism. These alterations contribute to decreased anxiety and neurocircuit dysfunction, which further drive behavioral dysfunction and reinforce the cycle.
The link between metabolism and behaviour in animal models of AN
So far, it has been difficult to model AN in mice, as the aetiology of the disorder likely involves complex interactions between genetic, environmental, social, and cultural factors [112]. Several approaches involving genetic, environmental and/or dietary manipulations have been used. The activity-based AN model (ABA) has been widely used in preclinical studies of AN. This well-established model resembles key aspects of AN, like self-starvation, excessive running, severe weight loss, loss of reproductive cycles, hypothermia, hyperghrelinemia and hypoleptinemia [113]. In the ABA paradigm, peripubertal mice are singly housed with ad libitum access to water and a running wheel. Mice are food-restricted and access to food is limited to few hours per day. During the experimental paradigm, mice typically engage in excessive running till exhaustion and prefer to run instead of eating even if food is available. Previous studies [114,115,116] have shown that providing a high fat and low carbohydrate diet interferes with the cascade leading to self-starvation and death in ABA. The fat content of the diet was the protective factor in both, preventing the initial self-starvation, and rescuing animals once the starvation began. A study [117, 118] found that a class of hypothalamic neurons called Agouti-related peptide neurons (AgRP) control compulsive exercise and survival in the ABA model. AgRP neurons residing in the arcuate nucleus of the hypothalamus promote adaptation during starvation by controlling nutrient partitioning and peripheral substrate preference [119, 120]. They have also been implicated in circuitry controlling non-feeding behaviour, including that associated with reward, anxiety and compulsive like behaviours [121,122,123]. Chronic disruption of the AgRP circuit led to a severe reduction of food intake and induced 100% lethality in the ABA model. Reduced levels of free fatty acids and low glucose levels paralleled this event. Further, chemogenetic activation of the AgRP neurons promoted the development of compulsive exercise, but not anxiety-like behaviours [117].
Importantly, and consistent with previous evidence, the death of animals with AgRP impaired circuity was prevented by an exposure to a high-fat diet. The elevated fat content in the diet also prevented the long-term behavioural impact of food-restricted mice with elevated exercise volume such as compulsive exercise. This study [117] identified a fundamental feeding circuit that couples metabolic peripheral changes with anxious and compulsive behaviours causing AN-like symptoms in mice. An additional study [124] validated the importance of the AgRP circuit in promoting maladaptive feeding behaviour. In line with these preclinical observations, individuals with AN have elevated circulating blood levels of AgRP compared to controls [125] and AgRP levels have been associated with cognitive rigidity in these individuals [126]. Altogether, this preclinical evidence supports a link between alterations in fat metabolism and AN-related behavioural changes.
Anorexia nervosa, anxiety, and ketone bodies
Anxiety is a common occurrence in the acute phases of AN and lifetime [127,128,129]. In a recent study, we have shown the longitudinal relationship between anxiety and ‘fasting’ (i.e., food restriction to control weight or weight gain, a behaviour typical of AN) in adolescence [130]. It has been hypothesized that in some individuals, particularly those that are anxious premorbidly (who are also at higher risk of developing AN), starvation has a beneficial anxiolytic effect [74]. However, traditional anxiolytic treatments (i.e., selective serotonin reuptake inhibitors –SSRIs) are not beneficial during the starved state in patients with AN. Among other reasons, this probably involves changes in the availability of pre-synaptic serotonin and glucose hypo-metabolism, which are characteristic of acute AN [131,132,133].
Importantly, substantial emerging work is pointing to metabolic and mitochondrial dysfunctions in the brain and periphery in the context of anxiety [134,135,136,137]. Emerging evidence also suggests the effectiveness of KD in exerting anxiolytic effects, possibly by improving transport and control of energy substrates in key brain regions involved in anxiety regulation, such as the medial prefrontal cortex [81]. Recent reviews have summarized the evidence supporting the impact of low carbohydrate/KD diets on anxiety and mood disorders [13, 138] although the small number of studies and their great heterogeneity may undermine the interpretation of the results, it would appear that the use of these diets has a beneficial effect in psychiatric conditions characterized among other things by anxiety dysregulation.
Given the evidence highlighted above, it is plausible that additionally to providing faster and essential (given the depletion of glucose) fuel for the brain, KBs will, through their membrane stabilization properties and anxiety-reducing effect [139] lead to a state of reduced anxiety, facilitating a vicious cycle that will persist due to these positive effects.
Starvation also promotes alterations in peripheral metabolism and in brain mitochondria, further resulting in behavioural and neurocircuit dysfunction (Fig. 1). Excessive exercise might also contribute to maintain this vicious cycle, given that physical activity induces the release of KBs (in particular D-BHB) into the bloodstream [139]. Accordingly, it is possible that maintenance of AN symptoms is partly due to the positive effect of ketosis on reducing anxiety in the starved state in vulnerable individuals, hence breaking the vicious cycle highlighted in Fig. 1 might be difficult during refeeding without replacing the anxiolytic effect of ketosis.
Potential benefits of exogenous ketones/ketogenic diet on psychopathology and brain functions related to AN
There is evidence that ketones and MCTs might improve anxiety in humans and animal models. Recent preclinical work has supported the effectiveness of an MCT diet to reduce anxiety-like behaviours and improve social competitiveness [81], a behavioural component that is deterred by high anxiety both in animal models and humans [75, 140, 141].
Mechanistically, the study by Hollis et al. showed that MCT treatment leads to changes in mitochondrial protein content and function in the medial prefrontal cortex (mPFC) and the nucleus accumbens (NAc), two brain regions critically implicated in executive function, motivation and rewards, among other functions [81]. Further, sub-chronic (7 days) oral administration of exogenous ketone supplements, such as KE, KS and KS-MCT evoked an anxiolytic effect in wild type and transgenic rat model on elevated plus maze in correlation with increased levels of BHB [142]. The KD might be also beneficial at the cognitive and behavioral levels, improving sleep, female sexual function, and non-compulsive physical activity, as shown in a previous study in female patients with obesity [18].
A potential role for exogenous ketones and related molecules in refeeding or treatment of AN
All the evidence reviewed above led us to put forward a model whereby exogenous ketones supplementation in AN could help both behaviour, comorbid psychopathology (e.g., anxiety), and dysfunctional metabolism through pleiotropic effects. These involve hormonal and metabolic regulation, increased mitochondrial energy metabolism, decreased inflammatory processes and oxidative stress, and modulation of neurotransmitter activity. Critically, for individuals with AN, KB supplementation could also promote skeletal muscle anabolism [143] (Fig. 2). We suggest that nutritional ketosis could protect individuals with AN from the detrimental effects of starvation while still contributing to the ‘positive’ metabolic effects of KBs on the brain and behaviour through synergistic mechanisms. Critically, nutritional/exogenous ketosis could provide additional energy without breaking the ketosis state and its positive anxiolytic effects.
KB supplementation may improve brain metabolism and behavior, promote skeletal muscle anabolism, reduce catabolism, and modulate hormonal, metabolic, inflammatory, and oxidative stress pathways in AN patients. Created in BioRender. Miletta, M. (2025) https://BioRender.com/kcdobbx.
So far, only two studies, one with a small number of patients (n = 5) and a case report, employed the KD in individuals with AN [66, 144]. In the study by Calabrese et al. [66] individuals with AN (weight-recovered but still symptomatic) were given a KD diet, with a macronutrient ratio of 70% fat, 20% protein and 10% carbohydrate, with the aim of inducing a stable degree of ketosis for at least 4 weeks. Ketamine injections were used in combination with the KD. Despite the small number of patients, individuals with AN showed some improvements as evidenced by both a decrease in the Eating Disorders Examination-Questionnaire scores (particularly on the global scale and eating restraint subscale) and a decrease in the clinical impairment assessment [66]. However, no changes were reported on acceptance of self and body, lack of symptomatic behaviour, social and emotional connection, and BMI. The study also showed no adverse effects, nor active suicidal ideation, secondary to the KD diet, hence suggesting a KD diet is feasible in individuals with AN who are weight-recovered.
Another study, the only one of its kind, administered MCT to patients with AN, with the aim of investigating changes in gut hormones in response to MCTs [145]. This study used a high ( > 6 g/day), medium (1–6 g/day) and low (0 g/day) MCT supplement for 6 weeks as an add-on to treatment as usual (cognitive behavioural therapy with nutrition treatment) for AN and showed an increase in acylated ghrelin [145]. Tolerability of MCT supplementation was shown and no adverse effects were reported.
Although recent studies have primarily examined ketogenic interventions in weight-restored patients [66], there is a strong theoretical basis for considering the role of KBs or KD in refeeding of AN individuals. Moreover, although KBs might be promising in the treatment of AN, several considerations are required, including: 1. the need for a balanced nutritional/refeeding protocol that meets all nutritional requirements in individuals with acute AN; and 2. the need for a multi-pronged approach which will require a combination of psychotherapy, nutritional and other approaches, as well as ketone-based interventions. Therefore KBs or KD are unlikely to be a stand-alone treatment for AN, but might offer benefits in integration with other treatment approaches. Given the cognitive and behavioural effects of KBs highlighted above, and the need to promote balanced nutritional intake and not reinforce restrictive eating, we suggest that they might complement existing psychotherapy treatments.
Additionally, rigorous clinical trials are needed to establish the safety, feasibility, and efficacy of any ketone-based approach.
Summary and future directions
Evidence that alterations in metabolism might play a role in the development of AN is increasing, however the role of nutrition as a treatment per se contributing to recovery in AN has not been adequately studied. Shifts in metabolism during fasting states give a prominent role to ketones, which are a more effective source of energy, particularly for the brain, as compared to glucose. Ketones have multiple effects on the brain and behaviour; hence, exogenous ways of supplementing ketones have been increasingly studied in relation to psychiatric disorders. The anxiolytic effect of EK as well as the sexual dimorphism of fasting ketosis give rise to interesting speculations about the role starvation-induced ketosis might have in AN. Metabolic changes involving ketones that occur following fasting and related physiological and brain changes are likely to be relevant to the pathophysiology of AN. Links between ketosis, fat metabolism, and behaviour relevant to AN have also been identified in animal models. Currently, there are no effective pharmacological/nutritional treatments for AN, either to treat the acute phase or to prevent relapse. However, early evidence of positive effects of exogenous ketones, including and going beyond the KD, on symptoms of AN warrant further investigation. We present here a model postulating that by addressing underlying aberrant metabolic characteristics, nutritional ketosis (exogenously induced) might prove to be a novel and useful adjunctive therapeutic approach to treat AN. Future studies in rodents, as well as parallel studies in individuals with AN, are warranted in which either a KD or specific ketone supplementation are given as potential treatments, both as a proof of principle in support of human clinical studies and as the means to achieve a mechanistic understanding of their effects at the cellular, molecular and circuit levels. Further investigation of ketosis and the effects of exogenous ketones in AN might benefit from a longitudinal perspective, across stages of illness, and using an integrated approach -including studying other metabolic and hormonal factors. This approach would allow a broader understanding of metabolic changes occurring over time, and in a wider context.
Lastly, a more integrated view of pathophysiological changes that are triggered by starvation and that occur in AN, that goes beyond a focus on behaviour as purely brain-mediated, but integrates how behaviour is modulated and impacted upon by metabolism is likely to shed light on our understanding and therapeutic approaches to this highly lethal disorder.
References
Bulik CM, Carroll IM, Mehler P. Reframing anorexia nervosa as a metabo-psychiatric disorder. Trends Endocrinol Metab. 2021;32:752–61. https://doi.org/10.1016/j.tem.2021.07.010.
Watson HJ, Yilmaz Z, Thornton LM, Hubel C, Coleman JRI, Gaspar HA, et al. Genome-wide association study identifies eight risk loci and implicates metabo-psychiatric origins for anorexia nervosa. Nat Genet. 2019;51:1207–14. https://doi.org/10.1038/s41588-019-0439-2.
Abdulkadir M, Hubel C, Herle M, Loos RJF, Breen G, Bulik CM, Micali N. Eating disorder symptoms and their associations with anthropometric and psychiatric polygenic scores. Eur Eat Disord Rev. 2022;30:221–36. https://doi.org/10.1002/erv.2889.
Ancel Keys JB, Henschel A, Mickelsen O, Taylor HL. The biology of human starvation. Nature. 1950;2:765–1386. https://doi.org/10.1038/170177A0.
Mehler PS, Winkelman AB, Andersen DM, Gaudiani JL. Nutritional rehabilitation: practical guidelines for refeeding the anorectic patient. J Nutr Metab. 2010;2010:625782 https://doi.org/10.1155/2010/625782.
Evert AB, Dennison M, Gardner CD, Garvey WT, Lau KHK, MacLeod J, et al. Nutrition therapy for adults with diabetes or prediabetes: a consensus report. Diabetes Care. 2019;42:731–54. https://doi.org/10.2337/dci19-0014.
Jiang Z, Yin X, Wang M, Chen T, Wang Y, Gao Z, Wang Z. Effects of ketogenic diet on neuroinflammation in neurodegenerative diseases. Aging Dis. 2022;13:1146–65. https://doi.org/10.14336/AD.2021.1217.
Grochowska K, Przeliorz A. The effect of the ketogenic diet on the therapy of neurodegenerative diseases and its impact on improving cognitive functions. Dement Geriatr Cogn Dis Extra. 2022;12:100–6. https://doi.org/10.1159/000524331.
Altayyar M, Nasser JA, Thomopoulos D, Bruneau M Jr. The implication of physiological ketosis on the cognitive brain: a narrative review. Nutrients. 2022;14:513 https://doi.org/10.3390/nu14030513.
Achanta LB, Rae CD. beta-Hydroxybutyrate in the brain: one molecule, multiple mechanisms. Neurochem Res. 2017;42:35–49. https://doi.org/10.1007/s11064-016-2099-2.
Garner S, Davies E, Barkus E, Kraeuter AK. Ketogenic diet has a positive association with mental and emotional well-being in the general population. Nutrition. 2024;124:112420 https://doi.org/10.1016/j.nut.2024.112420.
Kraeuter AK, Phillips R, Sarnyai Z. Ketogenic therapy in neurodegenerative and psychiatric disorders: From mice to men. Prog Neuropsychopharmacol Biol Psychiatry. 2020;101:109913 https://doi.org/10.1016/j.pnpbp.2020.109913.
Dietch DM, Kerr-Gaffney J, Hockey M, Marx W, Ruusunen A, Young AH, et al. Efficacy of low carbohydrate and ketogenic diets in treating mood and anxiety disorders: systematic review and implications for clinical practice. BJPsych Open. 2023;9:e70 https://doi.org/10.1192/bjo.2023.36.
Kossoff E, Cervenka M. Ketogenic dietary therapy controversies for its second century. Epilepsy Curr. 2020;20:125–9. https://doi.org/10.1177/1535759719890337.
Gershuni VM, Yan SL, Medici V. Nutritional ketosis for weight management and reversal of metabolic syndrome. Curr Nutr Rep. 2018;7:97–106. https://doi.org/10.1007/s13668-018-0235-0.
Cunnane SC, Trushina E, Morland C, Prigione A, Casadesus G, Andrews ZB, et al. Brain energy rescue: an emerging therapeutic concept for neurodegenerative disorders of ageing. Nat Rev Drug Discov. 2020;19:609–33. https://doi.org/10.1038/s41573-020-0072-x.
Marosi K, Kim SW, Moehl K, Scheibye-Knudsen M, Cheng A, Cutler R, et al. 3-Hydroxybutyrate regulates energy metabolism and induces BDNF expression in cerebral cortical neurons. J Neurochem. 2016;139:769–81. https://doi.org/10.1111/jnc.13868.
Castro AI, Gomez-Arbelaez D, Crujeiras AB, Granero R, Aguera Z, Jimenez-Murcia S, et al. Effect of a very low-calorie ketogenic diet on food and alcohol cravings, physical and sexual activity, sleep disturbances, and quality of life in obese patients. Nutrients. 2018;10:1348. https://doi.org/10.3390/nu10101348.
Watanabe M, Risi R, Masi D, Caputi A, Balena A, Rossini G, et al. Current evidence to propose different food supplements for weight loss: a comprehensive review. Nutrients. 2020;12:2873. https://doi.org/10.3390/nu12092873.
Marinou K, Adiels M, Hodson L, Frayn KN, Karpe F, Fielding BA. Young women partition fatty acids towards ketone body production rather than VLDL-TAG synthesis, compared with young men. Br J Nutr. 2011;105:857–65. https://doi.org/10.1017/S0007114510004472.
Suchacki KJ, Thomas BJ, Ikushima YM, Chen KC, Fyfe C, Tavares AAS, et al. The effects of caloric restriction on adipose tissue and metabolic health are sex- and age-dependent. eLife. 2023;12:e88080 https://doi.org/10.7554/eLife.88080.
Hedrington MS, Davis SN. Sexual dimorphism in glucose and lipid metabolism during fasting, hypoglycemia, and exercise. Front Endocrinol (Lausanne). 2015;6:61. https://doi.org/10.3389/fendo.2015.00061.
Dhillon KK, Gupta S Biochemistry, Ketogenesis. In: StatPearls. Treasure Island (FL); 2022.
Robinson AM, Williamson DH. Physiological roles of ketone bodies as substrates and signals in mammalian tissues. Physiol Rev. 1980;60:143–87. https://doi.org/10.1152/physrev.1980.60.1.143.
Cahill GF Jr. Starvation in man. N Engl J Med. 1970;282:668–75. https://doi.org/10.1056/NEJM197003192821209.
Rolfe DF, Brown GC. Cellular energy utilization and molecular origin of standard metabolic rate in mammals. Physiol Rev. 1997;77:731–58. https://doi.org/10.1152/physrev.1997.77.3.731.
Prince A, Zhang Y, Croniger C, Puchowicz M. Oxidative metabolism: glucose versus ketones. Adv Exp Med Biol. 2013;789:323–8. https://doi.org/10.1007/978-1-4614-7411-1_43.
Prins ML, Matsumoto JH. The collective therapeutic potential of cerebral ketone metabolism in traumatic brain injury. J Lipid Res. 2014;55:2450–7. https://doi.org/10.1194/jlr.R046706.
Owen OE, Morgan AP, Kemp HG, Sullivan JM, Herrera MG, Cahill GF Jr. Brain metabolism during fasting. J Clin Invest. 1967;46:1589–95. https://doi.org/10.1172/JCI105650.
Smith RL, Soeters MR, Wust RCI, Houtkooper RH. Metabolic flexibility as an adaptation to energy resources and requirements in health and disease. Endocr Rev. 2018;39:489–517. https://doi.org/10.1210/er.2017-00211.
Valente-Silva P, Lemos C, Kofalvi A, Cunha RA, Jones JG. Ketone bodies effectively compete with glucose for neuronal acetyl-CoA generation in rat hippocampal slices. NMR Biomed. 2015;28:1111–6. https://doi.org/10.1002/nbm.3355.
Cox PJ, Kirk T, Ashmore T, Willerton K, Evans R, Smith A, et al. Nutritional ketosis alters fuel preference and thereby endurance performance in athletes. Cell Metab. 2016;24:256–68. https://doi.org/10.1016/j.cmet.2016.07.010.
Clairis N, Barakat A, Brochard J, Xin L, Sandi C. A neurometabolic mechanism involving dmPFC/dACC lactate in physical effort-based decision-making. Mol Psychiatry. 2024. https://doi.org/10.1038/s41380-024-02726-y.
Srivastava S, Kashiwaya Y, King MT, Baxa U, Tam J, Niu G, et al. Mitochondrial biogenesis and increased uncoupling protein 1 in brown adipose tissue of mice fed a ketone ester diet. FASEB J. 2012;26:2351–62. https://doi.org/10.1096/fj.11-200410.
Garcia-Rodriguez D, Gimenez-Cassina A. Ketone bodies in the brain beyond fuel metabolism: from excitability to gene expression and cell signaling. Front Mol Neurosci. 2021;14:732120. https://doi.org/10.3389/fnmol.2021.732120.
Mattson MP, Moehl K, Ghena N, Schmaedick M, Cheng A. Intermittent metabolic switching, neuroplasticity and brain health. Nat Rev Neurosci. 2018;19:63–80. https://doi.org/10.1038/nrn.2017.156.
Jensen NJ, Wodschow HZ, Nilsson M, Rungby J. Effects of ketone bodies on brain metabolism and function in neurodegenerative diseases. Int J Mol Sci. 2020;21:8767 https://doi.org/10.3390/ijms21228767.
Poffe C, Robberechts R, Stalmans M, Vanderroost J, Bogaerts S, Hespel P. Exogenous ketosis increases circulating dopamine concentration and maintains mental alertness in ultra-endurance exercise. J Appl Physiol (1985). 2023;134:1456–69. https://doi.org/10.1152/japplphysiol.00791.2022.
Mahajan VR, Nadel JA, King MT, Pawlosky RJ, Davis MI, Veech RL, et al. Ketone ester-enriched diet ameliorates motor and dopamine release deficits in MitoPark mice. Eur J Neurosci. 2024;60:6875–90. https://doi.org/10.1111/ejn.16601.
Yellen G. Ketone bodies, glycolysis, and KATP channels in the mechanism of the ketogenic diet. Epilepsia. 2008;49:80–82. https://doi.org/10.1111/j.1528-1167.2008.01843.x. Suppl 8.
Martinez-Francois JR, Fernandez-Aguera MC, Nathwani N, Lahmann C, Burnham VL, Danial NN, Yellen G. BAD and K(ATP) channels regulate neuron excitability and epileptiform activity. eLife. 2018;7:e32721. https://doi.org/10.7554/eLife.32721.
De La Rossa A, Laporte MH, Astori S, Marissal T, Montessuit S, Sheshadri P, et al. Paradoxical neuronal hyperexcitability in a mouse model of mitochondrial pyruvate import deficiency. eLife. 2022;11:e72595 https://doi.org/10.7554/eLife.72595.
Ma W, Berg J, Yellen G. Ketogenic diet metabolites reduce firing in central neurons by opening K(ATP) channels. J Neurosci. 2007;27:3618–25. https://doi.org/10.1523/JNEUROSCI.0132-07.2007.
Gimenez-Cassina A, Martinez-Francois JR, Fisher JK, Szlyk B, Polak K, Wiwczar J, et al. BAD-dependent regulation of fuel metabolism and K(ATP) channel activity confers resistance to epileptic seizures. Neuron. 2012;74:719–30. https://doi.org/10.1016/j.neuron.2012.03.032.
Foley J, Burnham V, Tedoldi M, Danial NN, Yellen G. BAD knockout provides metabolic seizure resistance in a genetic model of epilepsy with sudden unexplained death in epilepsy. Epilepsia. 2018;59:e1–e4. https://doi.org/10.1111/epi.13960.
Calderon N, Betancourt L, Hernandez L, Rada P. A ketogenic diet modifies glutamate, gamma-aminobutyric acid and agmatine levels in the hippocampus of rats: A microdialysis study. Neurosci Lett. 2017;642:158–62. https://doi.org/10.1016/j.neulet.2017.02.014.
Yudkoff M, Daikhin Y, Horyn O, Nissim I, Nissim I. Ketosis and brain handling of glutamate, glutamine, and GABA. Epilepsia. 2008;49:73–75. https://doi.org/10.1111/j.1528-1167.2008.01841.x. Suppl 8.
Naito S, Ueda T. Characterization of glutamate uptake into synaptic vesicles. J Neurochem. 1985;44:99–109. https://doi.org/10.1111/j.1471-4159.1985.tb07118.x.
Eriksen J, Li F, Edwards RH. The mechanism and regulation of vesicular glutamate transport: Coordination with the synaptic vesicle cycle. Biochim Biophys Acta Biomembr. 2020;1862:183259. https://doi.org/10.1016/j.bbamem.2020.183259.
Juge N, Gray JA, Omote H, Miyaji T, Inoue T, Hara C, et al. Metabolic control of vesicular glutamate transport and release. Neuron. 2010;68:99–112. https://doi.org/10.1016/j.neuron.2010.09.002.
Sleiman SF, Henry J, Al-Haddad R, El Hayek L, Abou Haidar E, Stringer T, et al. Exercise promotes the expression of brain derived neurotrophic factor (BDNF) through the action of the ketone body beta-hydroxybutyrate. eLife. 2016;5:e15092. https://doi.org/10.7554/eLife.15092.
Kernie SG, Liebl DJ, Parada LF. BDNF regulates eating behavior and locomotor activity in mice. EMBO J. 2000;19:1290–1300. https://doi.org/10.1093/emboj/19.6.1290.
Egan MF, Kojima M, Callicott JH, Goldberg TE, Kolachana BS, Bertolino A, et al. The BDNF val66met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function. Cell. 2003;112:257–69. https://doi.org/10.1016/s0092-8674(03)00035-7.
Chen ZY, Jing D, Bath KG, Ieraci A, Khan T, Siao CJ, et al. Genetic variant BDNF (Val66Met) polymorphism alters anxiety-related behavior. Science. 2006;314:140–3. https://doi.org/10.1126/science.1129663.
Norgren J, Daniilidou M, Kareholt I, Sindi S, Akenine U, Nordin K, et al. Serum proBDNF is associated With changes in the Ketone body beta-Hydroxybutyrate and shows superior repeatability over mature BDNF: secondary outcomes from a cross-over trial in healthy older adults. Front Aging Neurosci. 2021;13:716594 https://doi.org/10.3389/fnagi.2021.716594.
Walsh JJ, Myette-Cote E, Little JP. The effect of exogenous ketone monoester ingestion on plasma BDNF during an oral glucose tolerance test. Front Physiol. 2020;11:1094 https://doi.org/10.3389/fphys.2020.01094.
Mohorko N, Cernelic-Bizjak M, Poklar-Vatovec T, Grom G, Kenig S, Petelin A, Jenko-Praznikar Z. Weight loss, improved physical performance, cognitive function, eating behavior, and metabolic profile in a 12-week ketogenic diet in obese adults. Nutr Res. 2019;62:64–77. https://doi.org/10.1016/j.nutres.2018.11.007.
Penninx B, Lange SMM. Metabolic syndrome in psychiatric patients: overview, mechanisms, and implications. Dialogues Clin Neurosci. 2018;20:63–73. https://doi.org/10.31887/DCNS.2018.20.1/bpenninx.
Ho C, Zhang M, Mak A, Ho R. Metabolic syndrome in psychiatry: Advances in understanding and management. Advances Psychiatr Treat. 2014;20:101–12. https://doi.org/10.1192/apt.bp.113.011619.
Church WH, Adams RE, Wyss LS. Ketogenic diet alters dopaminergic activity in the mouse cortex. Neurosci Lett. 2014;571:1–4. https://doi.org/10.1016/j.neulet.2014.04.016.
Dahlin M, Mansson JE, Amark P. CSF levels of dopamine and serotonin, but not norepinephrine, metabolites are influenced by the ketogenic diet in children with epilepsy. Epilepsy Res. 2012;99:132–8. https://doi.org/10.1016/j.eplepsyres.2011.11.003.
Operto FF, Matricardi S, Pastorino GMG, Verrotti A, Coppola G. The Ketogenic diet for the treatment of mood disorders in comorbidity with epilepsy in children and adolescents. Front Pharm. 2020;11:578396. https://doi.org/10.3389/fphar.2020.578396.
Orlando A, Chimienti G, Notarnicola M, Russo F. The Ketogenic diet improves gut-brain axis in a rat model of irritable bowel syndrome: impact on 5-HT and BDNF systems. Int J Mol Sci. 2022;23:1098. https://doi.org/10.3390/ijms23031098.
Wei L, Singh R, Ghoshal UC. Enterochromaffin Cells-Gut microbiota crosstalk: underpinning the symptoms, pathogenesis, and pharmacotherapy in disorders of gut-brain interaction. J Neurogastroenterol Motil. 2022;28:357–75. https://doi.org/10.5056/jnm22008.
Attaye I, van Oppenraaij S, Warmbrunn MV, Nieuwdorp M. The role of the gut microbiota on the beneficial effects of ketogenic diets. Nutrients. 2021;14:191. https://doi.org/10.3390/nu14010191.
Calabrese L, Scolnick B, Zupec-Kania B, Beckwith C, Costello K, Frank GKW. Ketogenic diet and ketamine infusion treatment to target chronic persistent eating disorder psychopathology in anorexia nervosa: a pilot study. Eat Weight Disord. 2022;27:3751–7. https://doi.org/10.1007/s40519-022-01455-x.
Evans M, McClure TS, Koutnik AP, Egan B. Exogenous ketone supplements in athletic contexts: past, present, and future. Sports Med. 2022;52:25–67. https://doi.org/10.1007/s40279-022-01756-2.
Walton E, Bernardoni F, Batury VL, Bahnsen K, Lariviere S, Abbate-Daga G, et al. Brain structure in acutely underweight and partially weight-restored individuals with anorexia nervosa: a coordinated analysis by the ENIGMA Eating Disorders Working Group. Biol Psychiatry. 2022;92:730–8. https://doi.org/10.1016/j.biopsych.2022.04.022.
Lumsden AL, Martin AM, Sun EW, Schober G, Isaacs NJ, Pezos N, et al. Sugar responses of human enterochromaffin cells depend on gut region, sex, and body mass. Nutrients. 2019;11:234 https://doi.org/10.3390/nu11020234.
Dong TS, Mayer E. Advances in brain-gut-microbiome interactions: a comprehensive update on signaling mechanisms, disorders, and therapeutic implications. Cell Mol Gastroenterol Hepatol. 2024;18:1–13. https://doi.org/10.1016/j.jcmgh.2024.01.024.
Rew L, Harris MD, Goldie J. The ketogenic diet: its impact on human gut microbiota and potential consequent health outcomes: a systematic literature review. Gastroenterol Hepatol Bed Bench. 2022;15:326–42. https://doi.org/10.22037/ghfbb.v15i4.2600.
Cabrera-Mulero A, Tinahones A, Bandera B, Moreno-Indias I, Macias-Gonzalez M, Tinahones FJ. Keto microbiota: a powerful contributor to host disease recovery. Rev Endocr Metab Disord. 2019;20:415–25. https://doi.org/10.1007/s11154-019-09518-8.
Xie G, Zhou Q, Qiu CZ, Dai WK, Wang HP, Li YH, et al. Ketogenic diet poses a significant effect on imbalanced gut microbiota in infants with refractory epilepsy. World J Gastroenterol. 2017;23:6164–71. https://doi.org/10.3748/wjg.v23.i33.6164.
Lloyd EC, Frampton I, Verplanken B, Haase AM. How extreme dieting becomes compulsive: A novel hypothesis for the role of anxiety in the development and maintenance of anorexia nervosa. Med Hypotheses. 2017;108:144–50. https://doi.org/10.1016/j.mehy.2017.09.001.
van der Kooij MA, Hollis F, Lozano L, Zalachoras I, Abad S, Zanoletti O, et al. Diazepam actions in the VTA enhance social dominance and mitochondrial function in the nucleus accumbens by activation of dopamine D1 receptors. Mol Psychiatry. 2018;23:569–78. https://doi.org/10.1038/mp.2017.135.
Poff AM, Koutnik AP, Egan B. Nutritional ketosis with ketogenic diets or exogenous ketones: features, convergence, and divergence. Curr Sports Med Rep. 2020;19:251–9. https://doi.org/10.1249/JSR.0000000000000732.
Omori NE, Woo GH, Mansor LS. Exogenous ketones and lactate as a potential therapeutic intervention for brain injury and neurodegenerative conditions. Front Hum Neurosci. 2022;16:846183 https://doi.org/10.3389/fnhum.2022.846183.
Falkenhain K, Islam H, Little JP. Exogenous ketone supplementation: an emerging tool for physiologists with potential as a metabolic therapy. Exp Physiol. 2023;108:177–87. https://doi.org/10.1113/EP090430.
Acquistapace S, Patel L, Patin A, Forbes-Blom E, Cuenoud B, Wooster TJ. Effects of interesterified lipid design on the short/medium chain fatty acid hydrolysis rate and extent (in vitro). Food Funct. 2019;10:4166–76. https://doi.org/10.1039/c9fo00671k.
St-Pierre V, Vandenberghe C, Lowry CM, Fortier M, Castellano CA, Wagner R, Cunnane SC. Plasma ketone and medium chain fatty acid response in humans consuming different medium chain triglycerides during a metabolic study day. Front Nutr. 2019;6:46 https://doi.org/10.3389/fnut.2019.00046.
Hollis F, Mitchell ES, Canto C, Wang D, Sandi C. Medium chain triglyceride diet reduces anxiety-like behaviors and enhances social competitiveness in rats. Neuropharmacology. 2018;138:245–56. https://doi.org/10.1016/j.neuropharm.2018.06.017.
Wang D, Mitchell ES. Cognition and synaptic-plasticity related changes in aged rats supplemented with 8- and 10-Carbon medium chain triglycerides. PLoS ONE. 2016;11:e0160159 https://doi.org/10.1371/journal.pone.0160159.
Fortier M, Castellano CA, St-Pierre V, Myette-Cote E, Langlois F, Roy M, et al. A ketogenic drink improves cognition in mild cognitive impairment: Results of a 6-month RCT. Alzheimers Dement. 2021;17:543–52. https://doi.org/10.1002/alz.12206.
Reger MA, Henderson ST, Hale C, Cholerton B, Baker LD, Watson GS, et al. Effects of beta-hydroxybutyrate on cognition in memory-impaired adults. Neurobiol Aging. 2004;25:311–4. https://doi.org/10.1016/S0197-4580(03)00087-3.
Newport MT, VanItallie TB, Kashiwaya Y, King MT, Veech RL. A new way to produce hyperketonemia: use of ketone ester in a case of Alzheimer’s disease. Alzheimers Dement. 2015;11:99–103. https://doi.org/10.1016/j.jalz.2014.01.006.
Xin L, Ipek O, Beaumont M, Shevlyakova M, Christinat N, Masoodi M, et al. Nutritional Ketosis increases NAD(+)/NADH ratio in healthy human brain: an in vivo study by (31)P-MRS. Front Nutr. 2018;5:62 https://doi.org/10.3389/fnut.2018.00062.
Dwir D, Khadimallah I, Xin L, Rahman M, Du F, Ongur D, Do KQ. Redox and immune signaling in schizophrenia: new therapeutic potential. Int J Neuropsychopharmacol. 2023;26:309–21. https://doi.org/10.1093/ijnp/pyad012.
Schoeler NE, Orford M, Vivekananda U, Simpson Z, Van de Bor B, Smith H, et al. K.Vita: a feasibility study of a blend of medium chain triglycerides to manage drug-resistant epilepsy. Brain Commun. 2021;3:fcab160 https://doi.org/10.1093/braincomms/fcab160.
Cuenoud B, Hartweg M, Godin JP, Croteau E, Maltais M, Castellano CA, et al. Metabolism of exogenous D-Beta-Hydroxybutyrate, an energy substrate avidly consumed by the heart and kidney. Front Nutr. 2020;7:13 https://doi.org/10.3389/fnut.2020.00013.
Stubbs BJ, Cox PJ, Evans RD, Santer P, Miller JJ, Faull OK, et al. On the metabolism of exogenous ketones in humans. Front Physiol. 2017;8:848 https://doi.org/10.3389/fphys.2017.00848.
Desrochers S, David F, Garneau M, Jette M, Brunengraber H. Metabolism of R- and S-1,3-butanediol in perfused livers from meal-fed and starved rats. Biochem J. 1992;285:647–53. https://doi.org/10.1042/bj2850647.
Falkenhain K, Daraei A, Little JP. The effect of novel exogenous ketone supplements on blood beta-hydroxybutyrate and glucose. J Diet Suppl. 2023:1–15, https://doi.org/10.1080/19390211.2023.2179152.
Scott BE, Laursen PB, James LJ, Boxer B, Chandler Z, Lam E, et al. The effect of 1,3-butanediol and carbohydrate supplementation on running performance. J Sci Med Sport. 2019;22:702–6. https://doi.org/10.1016/j.jsams.2018.11.027.
Shaw DM, Merien F, Braakhuis A, Plews D, Laursen P, Dulson DK. The effect of 1,3-Butanediol on cycling time-trial performance. Int J Sport Nutr Exerc Metab. 2019;29:466–73. https://doi.org/10.1123/ijsnem.2018-0284.
Soto-Mota A, Vansant H, Evans RD, Clarke K. Safety and tolerability of sustained exogenous ketosis using ketone monoester drinks for 28 days in healthy adults. Regul Toxicol Pharm. 2019;109:104506. https://doi.org/10.1016/j.yrtph.2019.104506.
Stubbs BJ, Cox PJ, Evans RD, Cyranka M, Clarke K, de Wet H. A ketone ester drink lowers human ghrelin and appetite. Obesity (Silver Spring). 2018;26:269–73. https://doi.org/10.1002/oby.22051.
Evans M, Egan B. Intermittent running and cognitive performance after ketone ester ingestion. Med Sci Sports Exerc. 2018;50:2330–8. https://doi.org/10.1249/MSS.0000000000001700.
Avgerinos KI, Mullins RJ, Egan JM, Kapogiannis D. Ketone ester effects on biomarkers of brain metabolism and cognitive performance in cognitively intact adults >/= 55 years old. A study protocol for a double-blinded randomized controlled clinical Trial. J Prev Alzheimers Dis. 2022;9:54–66. https://doi.org/10.14283/jpad.2022.3.
Hone-Blanchet A, Antal B, McMahon L, Lithen A, Smith NA, Stufflebeam S, et al. Acute administration of ketone beta-hydroxybutyrate downregulates 7T proton magnetic resonance spectroscopy-derived levels of anterior and posterior cingulate GABA and glutamate in healthy adults. Neuropsychopharmacology. 2023;48:797–805. https://doi.org/10.1038/s41386-022-01364-8.
Mujica-Parodi LR, Amgalan A, Sultan SF, Antal B, Sun X, Skiena S, et al. Diet modulates brain network stability, a biomarker for brain aging, in young adults. Proc Natl Acad Sci USA. 2020;117:6170–7. https://doi.org/10.1073/pnas.1913042117.
D’Agostino DP, Pilla R, Held HE, Landon CS, Puchowicz M, Brunengraber H, et al. Therapeutic ketosis with ketone ester delays central nervous system oxygen toxicity seizures in rats. Am J Physiol Regul Integr Comp Physiol. 2013;304:R829–836. https://doi.org/10.1152/ajpregu.00506.2012.
Leckey JJ, Ross ML, Quod M, Hawley JA, Burke LM. Ketone diester ingestion impairs time-trial performance in professional cyclists. Front Physiol. 2017;8:806 https://doi.org/10.3389/fphys.2017.00806.
Stubbs BJ, Blade T, Mills S, Thomas J, Yufei X, Nelson FR, et al. In vitro stability and in vivo pharmacokinetics of the novel ketogenic ester, bis hexanoyl (R)-1,3-butanediol. Food Chem Toxicol. 2021;147:111859 https://doi.org/10.1016/j.fct.2020.111859.
Chen O, Blonquist TM, Mah E, Sanoshy K, Beckman D, Nieman KM, et al. Tolerability and safety of a novel ketogenic ester, Bis-Hexanoyl (R)-1,3-Butanediol: a randomized controlled trial in healthy adults. Nutrients. 2021;13:2066 https://doi.org/10.3390/nu13062066.
Crabtree CD, Blade T, Hyde PN, Buga A, Kackley ML, Sapper TN, et al. Bis Hexanoyl (R)-1,3-Butanediol, a novel ketogenic ester, acutely increases circulating r- and s-ss-Hydroxybutyrate concentrations in healthy adults. J Am Nutr Assoc. 2023;42:169–77. https://doi.org/10.1080/07315724.2021.2015476.
Mitchell JE, Peterson CB. Anorexia nervosa. N Engl J Med. 2020;382:1343–51. https://doi.org/10.1056/NEJMcp1803175.
Ayton A, Ibrahim A. The Western diet: a blind spot of eating disorder research?-a narrative review and recommendations for treatment and research. Nutr Rev. 2020;78:579–96. https://doi.org/10.1093/nutrit/nuz089.
Owen OE, Reichard GA Jr. Human forearm metabolism during progressive starvation. J Clin Invest. 1971;50:1536–45. https://doi.org/10.1172/JCI106639.
Pietzner M, Uluvar B, Kolnes KJ, Jeppesen PB, Frivold SV, Skattebo O, et al. Systemic proteome adaptions to 7-day complete caloric restriction in humans. Nat Metab. 2024;6:764–77. https://doi.org/10.1038/s42255-024-01008-9.
van Waarde A, Audenaert K, Busatto Filho G, Buchpiguel CA, Dierckx R SPECT and PET in Eating Disorders. In: PET and SPECT in Psychiatry. 2nd edition. Springer Nature; 2021. pp. 741–72. ed.pp.
Bae EH, Lho H. Severe ketoacidosis in a patient with an eating disorder. Chonnam Med J. 2016;52:141–2. https://doi.org/10.4068/cmj.2016.52.2.141.
Siegfried Z, Berry EM, Hao S, Avraham Y. Animal models in the investigation of anorexia. Physiol Behav. 2003;79:39–45. https://doi.org/10.1016/s0031-9384(03)00103-3.
Klenotich SJ, Dulawa SC. The activity-based anorexia mouse model. Methods Mol Biol. 2012;829:377–93. https://doi.org/10.1007/978-1-61779-458-2_25.
Brown AJ, Avena NM, Hoebel BG. A high-fat diet prevents and reverses the development of activity-based anorexia in rats. Int J Eat Disord. 2008;41:383–9. https://doi.org/10.1002/eat.20510.
Barboriak JJ, Wilson AS. Effect of diet on self-starvation in the rat. J Nutr. 1972;102:1543–6. https://doi.org/10.1093/jn/102.11.1543.
Dong Y, Lin Y, Khatri L, Chao M, Aoki C. Ketogenic food ameliorates activity-based anorexia of adult female mice. Int J Eat Disord. 2025;58:317–35. https://doi.org/10.1002/eat.24323.
Miletta MC, Iyilikci O, Shanabrough M, Sestan-Pesa M, Cammisa A, Zeiss CJ, et al. AgRP neurons control compulsive exercise and survival in an activity-based anorexia model. Nat Metab. 2020;2:1204–11. https://doi.org/10.1038/s42255-020-00300-8.
Miletta MC, Horvath TL. Construction of activity-based anorexia mouse models. Bio Protoc. 2023;13:e4730. https://doi.org/10.21769/BioProtoc.4730.
Takahashi KA, Cone RD. Fasting induces a large, leptin-dependent increase in the intrinsic action potential frequency of orexigenic arcuate nucleus neuropeptide Y/Agouti-related protein neurons. Endocrinology. 2005;146:1043–7. https://doi.org/10.1210/en.2004-1397.
Cavalcanti-de-Albuquerque JP, Bober J, Zimmer MR, Dietrich MO. Regulation of substrate utilization and adiposity by Agrp neurons. Nat Commun. 2019;10:311. https://doi.org/10.1038/s41467-018-08239-x.
Dietrich MO, Zimmer MR, Bober J, Horvath TL. Hypothalamic Agrp neurons drive stereotypic behaviors beyond feeding. Cell. 2015;160:1222–32. https://doi.org/10.1016/j.cell.2015.02.024.
Dietrich MO, Bober J, Ferreira JG, Tellez LA, Mineur YS, Souza DO, et al. AgRP neurons regulate development of dopamine neuronal plasticity and nonfood-associated behaviors. Nat Neurosci. 2012;15:1108–10. https://doi.org/10.1038/nn.3147.
Li C, Hou Y, Zhang J, Sui G, Du X, Licinio J, et al. AGRP neurons modulate fasting-induced anxiolytic effects. Transl Psychiatry. 2019;9:111. https://doi.org/10.1038/s41398-019-0438-1.
Sweeney P, Bedenbaugh MN, Maldonado J, Pan P, Fowler K, Williams SY, et al. The melanocortin-3 receptor is a pharmacological target for the regulation of anorexia. Sci Transl Med. 2021;13:eabd6434. https://doi.org/10.1126/scitranslmed.abd6434.
Moriya J, Takimoto Y, Yoshiuchi K, Shimosawa T, Akabayashi A. Plasma agouti-related protein levels in women with anorexia nervosa. Psychoneuroendocrinology. 2006;31:1057–61. https://doi.org/10.1016/j.psyneuen.2006.06.006.
Sarrar L, Ehrlich S, Merle JV, Pfeiffer E, Lehmkuhl U, Schneider N. Cognitive flexibility and Agouti-related protein in adolescent patients with anorexia nervosa. Psychoneuroendocrinology. 2011;36:1396–406. https://doi.org/10.1016/j.psyneuen.2011.03.014.
Pallister E, Waller G. Anxiety in the eating disorders: understanding the overlap. Clin Psychol Rev. 2008;28:366–86. https://doi.org/10.1016/j.cpr.2007.07.001.
Swinbourne J, Hunt C, Abbott M, Russell J, St Clare T, Touyz SJA. The comorbidity between eating disorders and anxiety disorders: Prevalence in an eating disorder sample and anxiety disorder sample. Psychiatry, N. Z J o. 2012;46:118–31.
Godart N, Flament M, Curt F, Perdereau F, Lang F, Venisse J, et al. Anxiety disorders in subjects seeking treatment for eating disorders: a DSM-IV controlled study. Psychiatry Res. 2003;117:245–58.
Lloyd EC, Haase AM, Zerwas S, Micali N. Anxiety disorders predict fasting to control weight: A longitudinal large cohort study of adolescents. Eur Eat Disord Rev. 2020;28:269–81. https://doi.org/10.1002/erv.2714.
Scolnick B. Ketogenic diet and anorexia nervosa. Med Hypotheses. 2017;109:150–2. https://doi.org/10.1016/j.mehy.2017.10.011.
Frank GKW, Scolnick B. Therapeutic ketogenic diet as treatment for anorexia nervosa. Front Nutr. 2024;11:1392135. https://doi.org/10.3389/fnut.2024.1392135.
Marvanova M, Gramith K. Role of antidepressants in the treatment of adults with anorexia nervosa. Ment Health Clin. 2018;8:127–37. https://doi.org/10.9740/mhc.2018.05.127.
Filiou MD, Sandi C. Anxiety and Brain Mitochondria: A Bidirectional Crosstalk. Trends Neurosci. 2019;42:573–88. https://doi.org/10.1016/j.tins.2019.07.002.
Gebara E, Zanoletti O, Ghosal S, Grosse J, Schneider BL, Knott G, et al. Mitofusin-2 in the nucleus accumbens regulates anxiety and depression-like behaviors through mitochondrial and neuronal actions. Biol Psychiatry. 2021;89:1033–44. https://doi.org/10.1016/j.biopsych.2020.12.003.
Trumpff C, Monzel AS, Sandi C, Menon V, Klein HU, Fujita M, et al. Psychosocial experiences are associated with human brain mitochondrial biology. Proc Natl Acad Sci USA. 2024;121:e2317673121. https://doi.org/10.1073/pnas.2317673121.
Rosenberg AM, Saggar M, Monzel AS, Devine J, Rogu P, Limoges A, et al. Brain mitochondrial diversity and network organization predict anxiety-like behavior in male mice. Nat Commun. 2023;14:4726. https://doi.org/10.1038/s41467-023-39941-0.
Freyberg Z, Andreazza AC, McClung CA, Phillips ML. Linking mitochondrial dysfunction, neurotransmitter, neural network abnormalities and mania: Elucidating neurobiological mechanisms of the therapeutic effect of the ketogenic diet in Bipolar Disorder. Biol Psychiatry Cogn Neurosci Neuroimaging. 2024;10:267–77. https://doi.org/10.1016/j.bpsc.2024.07.011.
Nay K, Smiles WJ, Kaiser J, McAloon LM, Loh K, Galic S, et al. Molecular mechanisms underlying the beneficial effects of exercise on brain function and neurological disorders. Int J Mol Sci. 2021;22:4052. https://doi.org/10.3390/ijms22084052.
Hollis F, van der Kooij MA, Zanoletti O, Lozano L, Canto C, Sandi C. Mitochondrial function in the brain links anxiety with social subordination. Proc Natl Acad Sci USA. 2015;112:15486–91. https://doi.org/10.1073/pnas.1512653112.
Goette L, Bendahan S, Thoresen J, Hollis F, Sandi C. Stress pulls us apart: anxiety leads to differences in competitive confidence under stress. Psychoneuroendocrinology. 2015;54:115–23. https://doi.org/10.1016/j.psyneuen.2015.01.019.
Ari C, Kovacs Z, Juhasz G, Murdun C, Goldhagen CR, Koutnik AP, et al. Exogenous ketone supplements reduce anxiety-related behavior in sprague-dawley and wistar albino Glaxo/Rijswijk rats. Front Mol Neurosci. 2016;9:137 https://doi.org/10.3389/fnmol.2016.00137.
Chen J, Li Z, Zhang Y, Zhang X, Zhang S, Liu Z, et al. Mechanism of reduced muscle atrophy via ketone body (D)-3-hydroxybutyrate. Cell Biosci. 2022;12:94. https://doi.org/10.1186/s13578-022-00826-2.
Scolnick B, Zupec-Kania B, Calabrese L, Aoki C, Hildebrandt T. Remission from chronic anorexia nervosa with ketogenic diet and ketamine: case report. Front Psychiatry. 2020;11:763. https://doi.org/10.3389/fpsyt.2020.00763.
Kawai K, Nakashima M, Kojima M, Yamashita S, Takakura S, Shimizu M, et al. Ghrelin activation and neuropeptide Y elevation in response to medium chain triglyceride administration in anorexia nervosa patients. Clin Nutr ESPEN. 2017;17:100–4. https://doi.org/10.1016/j.clnesp.2016.10.001.
Acknowledgements
Dr. Nadia Micali is the recipient of a Laureate award by the Novo Nordisk Foundation (Grant No: NNF22OC0071010), and an investigator initiated study funded by Nestlé. Dr. Maria Consolata Miletta is the recipient of a Young Independent Investigator Research Grant by the Swiss Society of Endocrinology and Diabetology.
Author information
Authors and Affiliations
Contributions
N.M. and M. C.M. contributed equally. N.M. and M. C.M. conceived the idea and wrote the manuscript. E.P. contributed to literature review, figure preparation, and critical editing. CC, FL, CS, BC contributed intellectually and provided substantive revisions. CS, BC supervised the project and contributed to the content. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
Dr. Bernard Cuenoud is an employee of Nestlé Health Science. Dr. Carmen Sandi is a member of the SAB of Amazentis S.A. and Advisor for Vandria S.A. Dr. Christoffer Clemmensen is a co-founder of Ousia Pharma, a biotech company developing therapeutics for the treatment of obesity.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Micali, N., Miletta, M.C., Clemmensen, C. et al. Providing alternative fuel for the brain in anorexia nervosa: a review of the literature on ketones and their effects on metabolism and the brain. Transl Psychiatry 15, 412 (2025). https://doi.org/10.1038/s41398-025-03591-1
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41398-025-03591-1