Abstract
The treatment landscape of multiple myeloma (MM) has evolved significantly over four decades, driven by novel therapies and optimized supportive care. However, the attrition rate (AR), defined as the proportion of patients who die without advancing to the next line of therapy (LOT) after treatment failure, remains a major challenge. To assess how treatment patterns and outcomes have evolved, we analyzed 1,297 MM patients treated between 1980 and 2020, stratified by diagnosis period and age. ARs declined from 38–55% in the 1980s to 15–20% in 2010–2020, but remained high in older patients, with 46.9% of those over 80 unable to proceed beyond first LOT. While progression-free survival gains were primarily observed in the first LOT (15.8 to 24.1 months, p = 0.001), overall survival (OS) improved across all LOTs and age groups, likely due to more effective salvage therapies and supportive care. Achieving a complete response in first-line therapy was associated with a significant OS benefit (4.5 vs. 1.6 years, p < 0.001), underscoring its importance, as many patients, particularly older ones, are less likely to reach subsequent LOTs. Despite advances in MM treatment, patient loss to attrition remains a challenge, highlighting the need for more effective therapies early in the disease course.
Similar content being viewed by others
Introduction
Multiple myeloma (MM) is a clonal plasma cell proliferative disorder characterized by subsequent relapses and a need for successive lines of therapy (LOTs). While significant advancements have been made in the therapeutic landscape for MM, the disease remains incurable for most patients. In this setting, outcomes are limited by the progressive development of treatment resistance, lower response rates, and a shorter time to next treatment [1].
In a large cohort from three U.S. patient-level databases, more than one LOT was given to 79% of autologous transplant recipients, compared to only 43% of non-recipients [2]. This loss of patients not able to receive the next LOT after failing the previous one has been named the attrition rate (AR). Several studies have demonstrated that ARs tend to increase with each LOT [1, 3], with significant disparities based on factors such as patient’s age, performance status, and treatment regimen [4]. Moreover, the particularities of different health systems have an impact on treatment patterns and ARs. For instance, recent studies have reported ARs ranging from 21% to 57% across different healthcare settings [2, 3, 5, 6]. The AR can be particularly relevant as it reflects the increasing challenges posed by cumulative toxicity and treatment-related adverse effects, as well as disease progression [5].
The incorporation of novel agents, including proteasome inhibitors (PI), immunomodulatory drugs (IMiD), and monoclonal antibodies, has transformed the management of MM, improving quality of responses and, consequently, survival outcomes [2, 7, 8]. However, treatment attrition still remains a challenge. Since many patients may not reach later LOT, optimizing first-line therapy—rather than reserving the most effective options for subsequent lines—is critical, as achieving maximal progression-free survival (PFS) early has a direct impact on overall survival (OS) [9].
Given the evolving treatment landscape of MM, real-world studies with extended follow-up are crucial to understanding their impact on AR and survival. This study analyzes treatment patterns and long-term outcomes in a large cohort treated over 40 years.
Patients and methods
Study design and population
We conducted a retrospective review of clinical records of 1297 patients diagnosed and treated for MM at our institution (tertiary care center) from 1980 to 2020. This analysis excluded patients with smoldering MM. Data on demographic characteristics, clinical and laboratory features at diagnosis, treatment strategies, and follow-up were extracted from a prospectively maintained database, with the most recent update in December 2023.
Patients were stratified into four periods based on the decade of diagnosis: 1980–89 (n = 180), 1990–99 (n = 291), 2000–09 (n = 306), and 2010–20 (n = 520). Additionally, age at diagnosis was categorized into four groups, namely ≤60 years (n = 558), 61–70 years (n = 316), 71–80 years (n = 310), and >80 years (n = 113). The Durie–Salmon staging system was used instead of the International Staging System (ISS) in the 1980–89 period.
Definitions and outcomes of interest
A LOT was defined as the administration of one or a combination of two or more anti-MM agents, continued until either discontinuation for at least 60 days or the introduction of a new agent. Maintenance after ASCT was considered part of the first-line therapy. The AR was defined as the proportion of patients who died without receiving the next LOT after failing the previous one. Time to next treatment or death (TTNTD) was calculated as the interval between treatment initiation and either the start of the subsequent LOT or death from any cause, whichever occurred first. Treatment responses were assessed using different response criteria based on the year of diagnosis [10,11,12,13]. The overall response rate (ORR) was defined as the proportion of patients achieving a partial response (PR) or better, defined by a ≥ 50% reduction in serum monoclonal (M) protein, a ≥ 90% reduction in 24-h urine M-protein, and ≥50% reduction in the size of soft tissue plasmacytomas if present at diagnosis. Complete response (CR) was defined by a negative serum and urine immunofixation, disappearance of any soft tissue plasmacytomas, and <5% bone marrow plasma cells. Time-to-event analyses began at the time of MM diagnosis or the initiation of a new relevant LOT.
Statistical analysis
Continuous variables were summarized as medians with interquartile ranges (IQR) and compared using Student’s t-test, ANOVA, or appropriate non-parametric methods. Categorical variables were presented as frequencies and percentages, with statistical comparisons made using the Cochran–Armitage test for trend in proportions. Median follow-up was calculated using the reverse Kaplan–Meier method. ARs were reported for all patients and across up to four LOTs.
Univariate and multivariate logistic regression models were employed to identify factors associated with treatment response. Time-to-event analyses began at the time of MM diagnosis for the first LOT or the initiation of a relevant LOT in subsequent ones. Cox proportional hazards regression with backward stepwise selection was utilized to identify factors associated with OS. All statistical tests were two-tailed, with significance set at an alpha level of 0.05. The statistical analyses were performed using Stata v.11, RStudio 2024.04.1, and GraphPad Prism 8.2.1. The code used to generate the results is available from the corresponding author upon reasonable request, due to the use of patient data subject to confidentiality restrictions.
Results
Patient characteristics
We analyzed data from 1297 patients treated between 1980 and 2020, with a median follow-up of 134 months (IQR 72–208). Patient’s characteristics are summarized in Table 1. The median age at diagnosis increased over time, from 64 years (IQR 56–72) between 1980–89 to 66 years (IQR 57–76) (p < 0.002) in the most recent period, mostly due to a significant rise in the percentage of patients older than 80 years (up to 13%) in the last period studied. Six hundred and seventy-one (51.7%) were males and 626 (48.3%), females. Although patients were diagnosed at various disease stages, a significantly higher proportion were diagnosed at an earlier stage in the most recent decade (2010–2020). Compared to the first period with ISS staging (1990–1999), more patients were classified as ISS Stage I (31.1% vs. 26.2%, p < 0.001). Additionally, compared to the earliest period studied (1980–1989), fewer patients were diagnosed with a Durie–Salmon Stage III score (40.7% vs. 61.5%, p < 0.001) or with renal insufficiency (Durie–Salmon Stage B) (13.6% vs. 27.4%, p < 0.001). The functional status of patients at diagnosis, measured by the Eastern Cooperative Oncology Group Performance Status (ECOG-PS), showed a notable improvement over time, with the percentage of patients having an ECOG-PS higher than 1 dropping from 73% in the 1980–89 period to 28% in the 2010–2020 period (p < 0.001). There was an increase in the proportion of patients who received an ASCT from 29.9% in the second, 49% in the third, and 46.7% in the fourth period.
Number of lines of treatment
The number of LOT received per patient increased in later periods. The proportion of patients that received at least 3 LOT increased from 26% between 1980–1989, to 56.2% between 2000–2009 (p < 0.001), while the patients who received at least 4 lines increased from 10.5% to 40.5% in this period (p < 0.001). The proportion of patients receiving more LOT in later periods is probably underestimated due to a shorter follow-up. These data are summarized in Table 2.
Treatment patterns
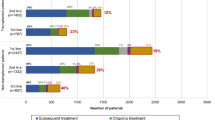
Treatment regimens evolved significantly over time with the introduction of novel agents. Until 1999, nearly all patients received chemotherapy across all treatment lines.
Between 2000 and 2009, chemotherapy remained the predominant first-line therapy (75.2%), though the use of bortezomib (Bor) (10%) and thalidomide/lenalidomide (T/R) (11.1%) increased. In second-line therapy, Bor-based regimens (40%) were most common, followed by chemotherapy (36.9%) and T/R-based regimens (30%). The distribution in third-line therapy was more balanced, with chemotherapy (30%), Bor (24%), and T/R (24%), while in fourth-line therapy, chemotherapy remained dominant, though T/R and Bor continued to be used in a subset of patients.
By 2010–2020, treatment patterns shifted significantly, with Bor-T/R-based regimens becoming the most frequent first-line therapy (35.2%), followed by Bor-based regimens (31.3%). Chemotherapy use declined to 15.4%, while T/R-based regimens alone were used in 8.3% of patients. Anti-CD38 therapy was introduced in this period but remained uncommon in first-line therapy (2.3%). In second-line therapy, treatment distribution was more diverse, with chemotherapy, T/R-based regimens, and carfilzomib (K) each used in about 20% of patients, followed by Bor-based (18%) and CD38-based therapies (14%). Chemotherapy remained the most frequent third-line therapy (18%), while T/R-based regimens were used in 10% of patients, similar to Bor, K, pomalidomide, and CD38-based therapies. In fourth-line therapy, CD38-based regimens were the second most common (17%), while T/R-based regimens were used less frequently (Fig. 1).
LoT line of treatment, Chemo chemotherapy, Bor bortezomib, T thalidomide, R lenalidomide, B bortezomib, K carfilzomib, Pom pomalidomide.
Attrition rate
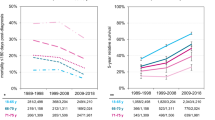
In our cohort, all patients received first-line treatment. The AR increased progressively with each LOT across all time periods. Moreover, the AR for every LOT decreased over the successive time periods. In the first time period (1980–1989), the AR was 37.8% after the first LOT, 55% after the second, and 59.6% and 63.2% after the third and fourth, respectively. In the 1990–1999 period, the ARs were 31.6%, 35.4%, 54.5%, and 43.4% after the first, second, third and fourth LOTs, respectively. During 2000–2009, the ARs were 17.9%, 20.6%, 23.8%, and 36.3% for the corresponding lines. Finally, in the most recent period studied (2010–2020), the ARs were lower; 15% for both the first and second LOT, 20.5% for the third, and 17.7% for the fourth LOT (Fig. 2A and Supplementary Table 1).
A Attrition rate by line of treatment and period. B Attrition rate by line of treatment and age.
Regarding the patient’s age at diagnosis and regardless of the time period, the ARs were higher in older patients. For those under 60 years of age, the AR was 19% after the first LOT, 17% after the second, and 26.2% after both the third and fourth. In contrast, for patients over 80, the ARs were 46.9%, 50%, 69.2%, and 66.7% for the first, second, third and fourth LOT, respectively (Fig. 2B and Supplementary Table 1).
When considering transplant status, the ARs were higher in patients who did not undergo an ASCT, being 29.2%, 36%, 42.8%, and 43.8% after the first, second, third and fourth LOT, respectively. In comparison, patients who received an ASCT had ARs of 11%, 10.7%, 18.8%, and 19.5%. These data are summarized in Supplementary Table 1.
Time to next treatment or death
The TTNTD was shorter after each LOT. We also observed that TTNTD was more prolonged in the last two periods studied (2000–2020), but only after the first and second LOT (p = 0.0003), while no differences were observed after subsequent lines, where TTNTD remained relatively stable across decades. After the first line, TTNTD increased from 15.4 months in the 1980s to 23.9 months in 2010–2020. In the second line, TTNTD was shorter in 2010–2020 (8.1 months) compared to 2000–2009 (12.1 months), possibly due to the earlier use of highly active agents in first-line therapy during the last decade, leading to more resistant disease at the time of second-line initiation. These results are summarized in Table 3.
Regarding patient age, TTNTD was significantly longer in younger patients after the first LOT (p < 0.0001), likely due to the use of more intensive treatment regimens. However, no significant differences were observed regarding age in subsequent lines. This may be explained by selection bias, as only patients who are fit enough to continue treatment reach later lines, potentially minimizing age-related differences. Among patients under 60 years, TTNTD progressively decreased from 26.8 months after the first line to 5.6 months after the fourth. In those aged 61–70 years, TTNTD ranged from 20.0 months in the first line to 5.8 months in the fourth. In older age groups, TTNTD was generally shorter, particularly in patients over 80, where it remained around 9 months across all lines. Detailed age-related data are presented in Table 3.
Responses
Treatment responses improved significantly across all LOT over the decades. For first-line therapy, ORR increased from 46.7% in 1980–89 to 75.5% in 2011–20 (p < 0.001), while second-line therapy resulted in an ORR rise from 19.3% to 53.1% over the same periods (p < 0.001). Similar improvements were observed after third- and fourth-line therapies, with ORRs increasing from 15.6% and 5.9% in 1980–89 to 41.2% and 37.8% in 2011–20, respectively. CR rates also improved significantly in recent decades. Among patients diagnosed between 1980 and 1989, only one achieved CR, and it occurred in third-line therapy. In contrast, in the last period studied (2011–20), CR rates reached 16.4% in the first line, 10.4% in the second, 8.8% in the third, and 5.5% in the fourth (Fig. 3A).
A Overall response rate by line of treatment and period. B Overall response rate by line of treatment and age. NR not reached.
Regarding age, the responses declined with increasing patient’s age and successive LOT. However, the differences regarding the age of the patient in the ORR are only statistically significant in the first LOT (p < 0.001), probably due to the small number of older patients treated in later lines. Younger patients ( ≤ 60 years) demonstrated the most favorable outcomes, with an ORR of 67.9% in the first line, including a 13% CR rate (p = 0.001), and an ORR of 32% in the fourth line, with a CR rate of only 5.8%. Among patients aged 61–70 years, the ORR dropped from 64.9% (CR 12%) in the first line to 25.5% (CR 4.1%) in the fourth. For those aged 71–80 years, the decline was more pronounced, with ORRs falling from 53.9% (CR 6.1%) to 24.6% (CR 1.8%). The oldest group ( > 80 years) achieved only a 43.4% ORR and 3.5% CR in the first line, and an ORR of 16.7% with no CR by the fourth line (Fig. 3B).
Progression-free survival and overall survival
In the earlier periods, PFS was more limited, consistent with the use of conventional chemotherapy and the absence of novel agents. PFS improved significantly for first-line therapy across the studied periods, increasing from a median of 15.8 months (95% CI, 13.3–20.5) in 1980–89 to 24.1 months (95% CI, 20.9–28.1) in 2010–20 (p = 0.001). In contrast, over the different time periods, no significant improvement in PFS was observed after second-line or subsequent treatments (Fig. 4A and Supplementary Table 2).
A Progression-free survival by line of treatment and period. B Overall survival by line of treatment and period. NR not reached.
Age at diagnosis only showed a significant effect in the first LOT. Patients aged ≤60 years exhibited the longest PFS in the first line, with a median of 27.6 months (95% CI, 24.3–32.4), compared to 20.9 months (95% CI, 17.3–24.3) for patients aged 61–70 years, 16.4 months (95% CI, 14.7–18.5) for those aged 71–80 years, and 10.3 months (95% CI 8.1–17.5) for patients aged >80 years (p < 0.0001). However, in subsequent lines, no differences in PFS were found across age groups, with median PFS of approximately 8, 6, and 4 months in the second, third, and fourth lines, respectively (Supplementary Table 3 and Supplementary Fig. 1).
OS has also significantly improved over time. Median survival increased progressively from 26.5 months (95% CI, 22.4–34.5) in 1980–89 to 70.7 months (95% CI, 61.5–82.7) in 2010–20 for first-line therapy (p < 0.001), from 11.0 months (95% CI, 7.2–15.7) to 42.6 months (95% CI, 32.8–51.5) for second-line (p < 0.001), from 10.2 months (95% CI, 7.4–22.6) to 24.7 months (95% CI, 21.2–33.2) for third-line (p = 0.002), and from 14.2 months (95% CI, 5.52–53.5) to 18.0 months (95% CI, 14.6–26.4) for fourth-line therapy (p = 0.76; Fig. 4B and Supplementary Table 2). OS decreased progressively with age across all therapy lines. For first-line therapy, younger patients (under 60 years) had the highest OS (70.5 months), while those over 80 had the lowest (24.4 months) (p < 0.001). In second-line therapy, OS ranged from 36.4 months for patients under 60 to 19.3 months for those over 80 (p = 0.012). For third- and fourth-line therapies, OS differences by age were less pronounced and not statistically significant (p = 0.48 and p = 0.83, respectively). (Supplementary Table 3 and Supplementary Fig. 1).
Progression-free survival and overall survival according to treatment regimen
While the global analysis of PFS showed significant improvement only in first-line therapy, specific novel regimens introduced during 2000–2009 and 2010–2020 contributed to meaningful PFS and OS gains in later lines.
Between 2000 and 2009, Bor-T/R-based regimens in first-line therapy achieved a PFS of 51.1 months, compared to 27.4 months for Bor-based therapies (p = 0.086), translating into OS gains of 96.3 and 79.0 months, respectively (p = 0.04). In second-line therapy, no significant PFS differences were observed, but OS was notably prolonged with K-based (68.6 months), Bor-based (38.1 months), and T/R-based therapies (41.9 months), compared to chemotherapy (17.3 months, p < 0.0001). However, sample sizes were small, with only 2 patients receiving CD38-based and Bor-T/R-based regimens, both of whom had not reached PFS and OS. In third-line therapy, K-based regimens showed the highest PFS (35.2 months, p = 0.0011), followed by CD38-based regimens (32.1 months), though patient numbers were low (4 and 5, respectively). OS varied significantly by treatment, with OS not reached in CD38-based and K-treated patients (p < 0.0001). In fourth-line therapy, no significant PFS differences were found, and the use of K-based, CD38-based, and triplet regimens was minimal (Supplementary Fig. 2).
Between 2010 and 2020, first-line therapy further improved, with Bor-T/R-based regimens achieving a PFS of 44.5 months, CD38-based combinations reaching 35.5 months, and K-based therapies achieving 30.3 months (p < 0.0001). OS was not reached in the CD38 group, whereas it reached 106.0 months in Bor-T/R-treated patients, compared to 31.7 months with chemotherapy (p < 0.0001). In second-line therapy, K-based regimens had a PFS of 19.8 months, and CD38-based therapies reached 11.9 months, both surpassing chemotherapy (6.2 months, p = 0.0022). OS in this setting was significantly prolonged, with survival not reached in pomalidomide-based, CD38-based, and K-based regimens, compared to 21.6 months with chemotherapy (p < 0.0001). The use of Bor-T/R regimens in this line was minimal (Fig. 5). In third-line therapy, PFS differences were not statistically significant, and OS was at the limit of significance (p = 0.19 and p = 0.05, respectively). In fourth-line therapy, PFS was longer in the Bor-T/R and T/R-based groups (8.3 months each) than in the chemotherapy group (2.7 months), though OS differences between treatment groups were not significant (Supplementary Fig. 3).
A Progression-free survival after first and second line of treatment by treatment regimen between 2010–20. B Overall survival after first and second line of treatment by treatment regimen between 2010–20. QMT Chemotherapy, Bor bortezomib, T thalidomide, R lenalidomide, B bortezomib, K carfilzomib, Pom pomalidomide.
Survival impact of CR achieved after 1st line of therapy
Achieving a CR after the first LOT has emerged as an important factor in improving long-term outcomes in MM. Therefore, we evaluated the impact on survival of attaining CR in first-line therapy. Survival was measured from the date CR was achieved to avoid immortal time bias to ensure that survival outcomes accurately reflected the effect of CR, independent of time already survived before response.
Our results indicate that patients who achieved CR had a median OS of 4.5 years (95% CI 3.9–4.9), compared to 1.6 years (95% CI 1.35–2.11) for those who did not achieve CR (p < 0.001) (Supplementary Fig. 3). This association remained statistically significant after adjusting for age, sex, Durie–Salmon stage, renal function, performance status, and the treatment period, as assessed through multivariate analysis (Supplementary Table 4). These adjustments confirm that achieving CR is independently associated with longer OS across various patient subgroups.
Discussion
In the current study, we analyzed a series of 1297 MM patients treated over a 40-year period, with a median follow-up of 11 years. This long-term follow-up offers a unique opportunity to examine real-world treatment patterns, ARs, and survival outcomes across four decades of evolving MM management.
ARs in our study reflect a consistent pattern seen in similar studies in other countries. Our results show an improvement in AR in recent periods, probably reflecting earlier diagnosis, access to newer therapeutic options, and optimized supportive care practices in recent decades, such as more effective antibiotic treatments and transfusion protocols.
Moreover, earlier diagnosis allows initiating therapy in less advanced disease stages, with more preserved renal function and better performance status, which allows using more intensive treatments. For instance, our ARs after the first and second LOT in the 2010–2020 period were both 15%, compared to 38% and 55%, respectively, in the 1980–1989 period. In later LOT, dynamics may differ. As options narrow and disease worsens, decisions are driven more by urgency and overall care goals than by response depth. This may explain the stable attrition beyond the third line despite advances. Still, our data show clear progress: ARs after third and fourth lines remained at 18–20% recently, down from nearly 60% in the earliest period.
Since no similar studies include data prior to 2008, direct comparisons can only be made for the most recent period. When focusing on this period, our ARs are consistent with reports from the Austrian Myeloma Registry, which reported ARs of 19% after first-line therapy and around 22% for subsequent lines and the Italian study, which showed ARs ranging from 26% to 37% across first to fifth lines [5, 6]. In contrast, higher ARs have been observed in U.S. datasets, with up to 50% in patients not undergoing ASCT and 21–37% in ASCT recipients. These differences could be attributed to differences in patient populations, healthcare systems, access to novel therapies, and the definition of the AR [2].
Differences in AR definitions make direct comparisons between studies difficult. Some include only patients who die or progress, while others also count those lost to follow-up or still in remission. U.S. databases often use a broader definition, considering any patient not receiving a next LOT, which may overestimate attrition. In contrast, the Canadian Myeloma Research Group defined AR more narrowly, including only those who failed to continue due to progression or death, and excluding patients in remission off-treatment [14]. To illustrate the variability in how attrition is reported, we summarized key data from major studies in MM (Supplementary Table 5), underscoring the need for standardized definitions.
Despite significant improvements in OS across all therapy lines, PFS only improved significantly in the first line. This aligns with other studies showing declining response rates and treatment durations with successive lines due to increasing treatment resistance and disease progression [15, 16]. The relatively modest PFS observed during the earlier decades likely reflects the absence of novel agents, particularly PI, IMiDs, and anti-CD38 monoclonal antibodies, which only became widely available in the later periods. This evolution in therapeutic strategy has had a clear impact on disease control and attrition across all LOTs. Nonetheless, these benefits are diluted in global analyses due to high AR and treatment heterogeneity. The increased OS despite limited PFS gains reflects the impact of effective salvage therapies and the higher proportion of patients receiving multiple LOT in recent years. While PFS improvements remain concentrated in first-line treatment, introducing triplets and novel agents has extended disease control and OS across successive lines. Other contributing factors could be improved supportive care, as well as earlier diagnosis and better patient fitness in the most recent decades [15, 16].
Beyond pharmacological and supportive advances, behavioral factors may also explain recent declines in AR. Patients achieving deep responses like CR or sustained minimal residual disease (MRD)-negativity may be more willing to continue therapy despite toxicity, and physicians are more likely to support it when efficacy is clearly demonstrated. In the phase 3 PERSEUS trial, D-VRd showed lower discontinuation due to toxicity (9.1% vs. 22.5%) and higher CR (87.9%) and MRD-negativity (75.2%) than VRd, despite its greater intensity, highlighting how perceived benefit can reduce dropout [17].
Although the prognostic value of CR is well established, our results provide robust confirmation of its impact on survival in real-world clinical practice. This reinforces the rationale for response-adapted treatment strategies and further supports the growing interest in minimal residual disease (MRD) as a potential surrogate endpoint, especially as deeper responses become increasingly achievable in early LOT.
While our study provides valuable insights, it is not without limitations. The retrospective design and single-center nature may introduce bias and limit generalizability. However, the long-term, prospectively maintained database ensures consistent data, detailed clinical information, and complete follow-up. As a major referral center for MM in Catalonia, our institution covers a broad patient population, enhancing the real-world relevance of these findings. Still, future multi-center or registry-based studies would help validate and expand these results. Additionally, variations in treatment protocols and patient characteristics across regions and studies complicate direct comparisons. Nevertheless, these differences are themselves valuable, reflecting the diversity of real-world practice and the influence of healthcare systems on treatment access and outcomes. Unlike larger registry-based studies, such as Fonseca et al., our continuously updated long-term database allows for a more detailed exploration of patient characteristics and treatment responses, enabling a deeper understanding of treatment patterns and outcomes. Finally, we are aware that stratification reduced sample sizes in some subgroups, possibly affecting statistical power. However, the robustness of the data and long-term follow-up support the validity of our findings.
It is important to note that anti-CD38 monoclonal antibodies and quadruplet regimens have been introduced more recently as part of first-line therapy, leading to their underrepresentation in our study. With extended follow-up, we will be able to assess their real-world impact, along with the effects of emerging therapies such as bispecific antibodies and CAR-T cell therapies in this clinical setting.
In summary, our study demonstrates a significant decrease in the AR over the years, with 15–20% of patients being lost after each LOT in the most recent period. A significant proportion of patients received three or more LOT in recent decades. There is a significant prolongation of PFS with the first line of treatment, while no differences were observed in subsequent lines when analyzed globally. However, when stratified by treatment regimens, PFS improvements become evident, particularly with the incorporation of novel agents such as anti-CD38 monoclonal antibodies and carfilzomib, which have contributed to prolonged disease control in later lines. A prolongation of OS is also observed over time, likely related to the greater availability of salvage treatments, earlier diagnosis, and improved supportive care. Further therapeutic advancements in MM treatment and wider access to emerging therapies are essential to continue improving outcomes, both in the first line and particularly in subsequent treatment lines, and also in older patients, where attrition remains high.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Yong K, Delforge M, Driessen C, Fink L, Flinois A, Gonzalez-McQuire S, et al. Multiple myeloma: patient outcomes in real‐world practice. Br J Haematol. 2016;175:252.
Fonseca R, Usmani SZ, Mehra M, Slavcev M, He J, Cote S, et al. Frontline treatment patterns and attrition rates by subsequent lines of therapy in patients with newly diagnosed multiple myeloma. BMC Cancer. 2020;20:1–11.
Tang W, Yang J, Li Y, Zhang L, Li H, Wang J, et al. Treatment attrition rates and relevant risk factors in multiple myeloma: a real-world study in China. Front Pharmacol. 2023;14:979111.
Terpos E, Suzan F, Goldschmidt H. Going the distance: Are we losing patients along the multiple myeloma treatment pathway? Crit Rev Oncol Hematol. 2018;126:19–23.
Benda MA, Ulmer H, Weger R, Reimann P, Lang T, Pichler P, et al. Attrition RAtes in Multiple Myeloma Treatment under Real World Conditions—an Analysis from the Austrian Myeloma Registry (AMR). Cancers (Basel). 2023;15:962.
Morè S, Corvatta L, Manieri MV, Olivieri A, Offidani M. Real-world assessment of treatment patterns and outcomes in patients with relapsed-refractory multiple myeloma in an Italian haematological tertiary care centre. Br J Haematol. 2023;201:432–42.
Kumar SK, Rajkumar SV, Dispenzieri A, Lacy MQ, Hayman SR, Buadi FK, et al. Improved survival in multiple myeloma and the impact of novel therapies. Blood. 2008;111:2516–20.
Kaddoura M, Moritz B, Dingli D, Buadi FK, Lacy MQ, Gertz MA, et al. Impact of achieving a complete response to initial therapy of multiple myeloma and predictors of subsequent outcome. Am J Hematol 2022; 97:267–73.
Goldman-Mazur S, Visram A, Rajkumar SV, Kapoor P, Dispenzieri A, Lacy MQ, et al. Predictors and impact of timing of disease progression following primary therapy in multiple myeloma. Clin Lymphoma Myeloma Leuk. 2024;24:104–11.
Durie BGM, Harousseau JL, Miguel JS, Bladé J, Barlogie B, Anderson K, et al. International uniform response criteria for multiple myeloma. Leukemia. 2006;20:1467–73.
Rajkumar SV, Harousseau JL, Durie B, Anderson KC, Dimopoulos M, Kyle R, et al. Consensus recommendations for the uniform reporting of clinical trials: report of the International Myeloma Workshop Consensus Panel 1. Blood. 2011;117:4691.
Kumar S, Paiva B, Anderson KC, Durie B, Landgren O, Moreau P, et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol. 2016;17:e328–46.
Blade J, Samson D, Reece D, Apperley J, Bjorkstrand B, Gahrton G, et al. Criteria for evaluating disease response and progression in patients with multiple myeloma treated by high-dose therapy and haemopoietic stem cell transplantation. Myeloma Subcommittee of the EBMT. European Group for Blood and Marrow Transplant. Br J Haematol. 1998;102:1115–23.
McCurdy A, Mian H, LeBlanc R, Jimenez-Zepeda VH, Su J, Masih-Khan E, et al. Redefining attrition in multiple myeloma (MM): a Canadian Myeloma Research Group (CMRG) analysis. Blood Cancer Journal. 2023;13:1–6.
Verelst SGR, Blommestein HM, De Groot S, Gonzalez-McQuire S, Decosta L, De Raad JB. et al. Long-term outcomes in patients with multiple myeloma: a retrospective analysis of the Dutch Population-based Haematological Registry for Observational Studies (PHAROS). Hemasphere. 2018;2:e45.
Yong K, Delforge M, Driessen C, Fink L, Flinois A, Gonzalez-McQuire S, et al. Multiple myeloma: patient outcomes in real-world practice. Br J Haematol. 2016;175:252–64.
Sonneveld P, Dimopoulos MA, Boccadoro M, Quach H, Ho PJ, Beksac M, et al. Daratumumab, bortezomib, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2024;390:301–13.
Acknowledgements
We thank Esther Bladé for her technical support in this research.
Funding
This work has been supported in part by grants from the Instituto de Salud Carlos III (ISCIII) and co-founded by the European Union (FIS PI22/00647 and ICI19/00025), 2021SGR01292 (AGAUR; Generalitat de Catalunya), and Asociación Española Contra el Cancer (AECC) LABAE21971FERN. LGRL has received funding from the CRIS Cancer Foundation (project number: TRANSPHYSTALENT_23_6).
Author information
Authors and Affiliations
Contributions
LGRL, AD, AP, NT, JB, and LR designed the study. LGRL, AD, and AP analyzed the data, performed statistical analysis, and wrote and reviewed the manuscript. LGRL, AD, CFL, NT, MTC, DFM, JMM, NL, EC, DM, JB, and LR treated patients and reviewed the manuscript. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
LGRL: Honoraria and travel grants from Janssen, Amgen, GSK, BMS, Sanofi, and Menarini Stemline; AD: Honoraria from Janssen, travel grants from Sanofi, Janssen, Binding Side; DFM: Honoraria and travel grants from Janssen; MTC: Honoraria from Janssen, Sanofi, GSA, Amgen, and Pfizer, LR: Honoraria from BMS, Celgene, Amgen, Takeda, Sanofi, Janssen, GSK; JB: Honoraria from Janssen, Celgene, BMS, Amgen, Takeda, Oncopeptides; CFL: Consultancy: BeiGene, Sanofi, GSK, BMS, Janssen; Honoraria: BeiGene, Pfizer, Sanofi, GSK, Takeda, BMS, Janssen; Research Funding: Takeda, Amgen, BMS, Janssen. The rest of the authors declare no competing interests.
Ethics approval and consent to participate
All methods were performed in accordance with the relevant guidelines and regulations, including the Declaration of Helsinki. The study was approved by the Ethics Committee of the Hospital Clínic de Barcelona. Informed consent was obtained from all participants, authorizing the use of their anonymized clinical data for research purposes.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Rodríguez-Lobato, L.G., de Daniel, A., Pereira, A. et al. Attrition rates and treatment outcomes in multiple myeloma: real-world data over a 40-year period. Blood Cancer J. 15, 103 (2025). https://doi.org/10.1038/s41408-025-01311-y
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41408-025-01311-y