Abstract
This clinical trial (NCT05347641) evaluated efficacy and safety of penpulimab combined with rituximab, high-dose methotrexate, and cytarabine (Pen-RMA) in patients with newly diagnosed (ND) primary central nervous system lymphoma (PCNSL). Patients received an induction treatment of six cycles of Pen-RMA every 3 weeks. Patients younger than 60 years who achieved at least partial remission (PR) underwent autologous stem cell transplantation (ASCT), followed by 8 cycles of penpulimab as maintenance therapy. For patients over 60 years or not eligible for ASCT who achieved complete remission (CR) after induction therapy received 8 cycles of penpulimab as maintenance treatment, and those who achieved PR were treated with whole brain radiotherapy (WBRT) combined with 8 cycles of penpulimab. Patients who achieved stable disease (SD) or progressive disease (PD) were withdrawn from this study. The primary endpoint was 2-year progression-free survival (PFS). Between April 2022 and December 2023, twenty-six ND-PCNSL patients were enrolled, and 23 patients were included in the intention-to-treat analysis. The median age was 65 (38‒74) yr. The overall response rate (ORR) and CR rate after the induction therapy were 95.7% and 91.3%, respectively. At a median follow-up of 29.4 months, the median PFS and overall survival (OS) were not reached. 2-year PFS and OS were 70.7% and 75.0%, respectively. The most frequent treatment-related adverse events were leukopenia, anemia, neutropenia, and thrombocytopenia. Grade 3 or worse treatment-related adverse events occurred in 7 (30.4%) of 23 patients. Cerebrospinal fluid (CSF) circulating-tumor DNA (ctDNA) monitoring was performed in 13 patients with imaging CR and 1 with PR after induction treatment. Among them, 8 had CSF ctDNA clearance while 6 were positive. Overall, Pen-RMA regimen demonstrated encouraging antitumor activity with a manageable toxicity in PCNSL.
Similar content being viewed by others
Introduction
Primary central nervous system lymphoma (PCNSL) is a B-cell non-Hodgkin lymphoma that is limited in the brain, spinal cord, cerebrospinal fluid (CSF) and/or the eye and without systemic lymphoma [1], and it was classified into large B-cell lymphomas (LBCL) of immune-privileged sites in the 5th edition of the World Health Organization Classification of Haematolymphoid Tumours (WHO-HAEM5) [2]. The prognosis of PCNSL patients is poor with the median survival only 1‒2 years [3, 4].
High-dose methotrexate (HD-MTX) (>3 g/m2 body surface) is the hallmark of induction therapy for PCNSL, mostly combined with other blood–brain barrier penetrating chemotherapeutic agents like cytarabine (Ara-C) or thiotepa. International Extranodal Lymphoma Study Group (IELSG) 32 study showed that MATRix regimen containing HD-MTX, Ara-C, thiotepa and rituximab yielded the best survival rates compared with group A (rituximab and HD-MTX) and group B (rituximab, HD-MTX and Ara-C) with the 2-year progression-free survival (PFS) was 61% and 2-year overall survival (OS) was 69%. However, MATRix group had a higher adverse events (AEs) rate [5]. In a larger phase III randomized controlled trial showed that the addition of rituximab to high-dose methotrexate, BCNU, teniposide, and prednisolone (R-MBVP/MBVP) in PCNSL was not efficacious, and the long-term results (median follow-up of 82.3 months) showed the 5-year OS for MBVP and R-MBVP was 49% (39‒59) and 53% (43‒63), respectively [6]. Autologous stem cell transplantation (ASCT) or whole-brain radiotherapy (WBRT) is recommended as consolidation treatment, significantly prolong the duration of the response [7]. Although highly responsive to first-line chemotherapy, recurrence is still common and most relapses occur within 2 years [1, 8]. Therefore, reducing the recurrence rate is an urgent issue that needs to be addressed. Thus, the development of therapeutic strategies that can achieve high response rates, low recurrence rates, and minimal toxicity remains a pressing issue in clinical practice.
Previous studies showed that amplification of 9p24.1 was frequently detected in PCNSL, and the expression of programmed death ligand 1 (PD-L1) was associated with an early mutation event in PCNSL [9, 10], and the activation of the programmed death (PD-1)/ PD-L1 axis was related to poor prognosis [11, 12]. Keytruda (pembrolizumab) and Opdivo (nivolumab) are two well-known PD-1 inhibitors that have shown significant efficacy in various cancers, including melanoma, non-small cell lung cancer, and renal cell carcinoma. Both agents have demonstrated promising results in relapsed or refractory (R/R) PCNSL. Graber et al. reported that pembrolizumab achieved a response rate of 40% in patients with R/R PCNSL [13]. Similarly, Nayak et al. showed that nivolumab had a response rate of 36% in a cohort of patients with R/R PCNSL [14]. These findings highlight the potential of PD-1 inhibitors in improving outcomes for patients with this challenging disease. Recently, Zhiyong Zeng et.al reported the results of a phase 2 study exploring the efficacy and safety of sintilimab (PD-1 inhibitor) combined with high-dose methotrexate, temozolomide, and rituximab (SMTR) in preliminary diagnosed PCNSL. The study showed that the overall response rate (ORR) was 96.3% and the 2-year PFS and OS rates were 57.2% (40.6‒80.8%) and 91.5% (80.7‒100%), respectively [15]. Therefore, the aforementioned studies showed a promising therapeutic effect of PD-1 inhibitor in PCNSL.
Nevertheless, most PD-1 inhibitors are humanized IgG4 monoclonal antibodies whose Fc domains retain the structural signature of native IgG4, which may diminish anti-tumor efficacy [16,17,18,19]. Engagement of Fc-γ receptors by the IgG4 Fc fragment has been shown to initiate antibody-dependent cellular cytotoxicity (ADCC) and to precipitate the release of pro-inflammatory cytokines, thereby increasing the incidence and severity of immune-related adverse events (irAEs) [20]. More evidence further demonstrates that these antibodies bind FcγRI+ macrophages, trigger antibody-dependent phagocytosis (ADCP), deplete PD-1⁺ T cells, and amplify systemic cytokine release, collectively exacerbating irAE risk [17]. Penpulimab is a humanized IgG1 monoclonal antibody targeting PD-1, which removes ADCC, ADCP, complement dependent cytotoxicity (CDC) and reduces the irAEs by modifying Fc segment [21]. In August 2021, penpulimab was approved in China for the treatment of R/R classic Hodgkin's lymphoma [22].
Therefore, we performed the single-center, single-arm, phase 2 study to evaluate the efficacy and activity of penpulimab in combination with rituximab, high-dose methotrexate, and Ara-C (Pen-RMA) regimen in ND-PCNSL patients.
Methods
Patients and study design
Patients were required to meet the following key inclusion criteria
(a) age between 18 and 75 years; (b) histologic diagnosis of diffuse large B-cell lymphoma (DLBCL) confined to the central nervous system (CNS) according to the 2016 World Health Organization classification, the lesions were located in the brain, leptomeninges, spinal cord, CSF, and/or eyes; (c) Eastern Cooperative Oncology Group performance status (ECOG PS) score from 0 to 4;(d) at least one measurable lesion based on magnetic resonance (MR) imaging; (e) absolute neutrophil count ≥1.0 × 109/L; platelet count ≥50 × 109/L; levels of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) ≤ 2.5×upper limit of normal (ULN); serum total bilirubin ≤1.5 × ULN; creatinine clearance ≥90 mL/min.
Key exclusion criteria include
(a) previous immune checkpoint therapy, radiotherapy or chemotherapy for PCNSL; (b) history of allogeneic organ transplantation or allogeneic hematopoietic stem cell transplantation (allo-HSCT); (c) active, known or suspected autoimmune disease; (d) history of other malignancies, human immunodeficiency virus infection, active hepatitis B infection, and interstitial lung disease.
This study was approved by an institutional review board and ethics committee at Jiangsu Province Hospital in accordance with the Declaration of Helsinki (2022-SR-166). Informed consent was obtained from all subjects. The study is registered on clinicaltrials.gov (NCT05347641). The study protocol is available online. The last patient was enrolled in this study on December 28, 2023. The last follow-up date of this report was January 31, 2025. Follow-up is still ongoing.
Procedures
The Pen-RMA induction treatment included six cycles of rituximab 375 mg/m2 intravenously on Day 0; methotrexate 3.5 g/m2 intravenously over 3 h on Day 1; Ara-C 2 g/m2 intravenously every 12 h from Day 2 to Day 3 for patients younger than 60 years and ECOG PS ≤ 2, starting from the second cycle; for patients older than 60 years or ECOG PS > 2, the Ara-C dose was reduced to 1 g/m2 every 12 h on Day 2 from the second cycle; penpulimab 200 mg intravenously on Day 5. In the induction phase, patients received Pen-RMA every three weeks per cycle.
Disease assessment was conducted by positron emission tomography/computed tomography (PET/CT) and brain MR after three cycles and six cycles of treatment, respectively. Patients with partial response (PR) or complete response (CR) at mid-assessment continued with the treatment. After the end of treatment (EOT) assessment, patients younger than 60 years who achieved PR or CR underwent ASCT (conditioning regimen consisting of thiotepa 300 mg/m2, days -6, -5; busulfan 3.2 mg/kg, days -4, -3, -2), and then received 8 cycles of penpulimab as maintenance therapy. For patients over 60 years or not eligible for ASCT who achieved CR after induction therapy were treated with penpulimab as maintenance treatment, and those who reached PR received 36 Gy WBRT combined with 8 cycles of penpulimab. Patients with stable disease (SD) or progressive disease (PD) were withdrawn at the mid or EOT assessment.
Outcomes
The primary endpoint was 2-year PFS. Tumor assessments were conducted according to the International Primary Central Nervous System Lymphoma Collaborative Group (IPCG) criteria [23] after 3 cycles and 6 cycles during induction, every 3 months during maintenance, and every 6 months after completion of the planned treatment until disease progression or death. Safety was assessed throughout the treatment period, and adverse events were defined and graded according to National Cancer Institute-Common Terminology Criteria for Adverse Events, version 5.0. Secondary outcomes were ORR and 2-year OS rate. Other research objectives include the role of CSF circulating-tumor DNA (CSF-ctDNA) as a minimal residual disease (MRD) monitoring tool in predicting relapses in PCNSL.
Targeted next-generation sequencing (TGS) was performed at a Clinical Laboratory Improvement Amendments and College of American Pathologists-accredited testing laboratory (Nanjing Geneseeq Technology Inc, Nanjing, China) [24]. Sequencing libraries were constructed using the KAPA Hyper Prep Kit (KAPA Biosystems) and sequenced on Illumina HiSeq 4000 using a panel of 475 leukemia- and lymphoma-related genes. The criteria of genotyping were according to the LymphGen algorithm [25]. We conducted TGS on formalin-fixed paraffin-embedded (FFPE) tissue or aqueous humor, peripheral blood (PB) specimens and CSF before treatment, and collected CSF at the end of induction treatment or at the time of PD and upon completion of maintenance treatment.
Statistical analysis
The sample size was calculated on the basis of data from studies of the RMA regimen in patients with newly diagnosed (ND) PCNSL in real-world in our center [26]. The null hypothesis assumed a 2-year PFS rate of 39% for RMA regimen and an estimated 2-year PFS rate of 65% for patients treated with Pen-RMA. Therefore, based on an exact binomial test with a two-side α of 0.05, β of 0.2, and a 20% dropout tare, a sample size of 23 patients were required.
The Wilson method was used to estimate the 95% confidence intervals (CIs) of a binomial distribution. The Kaplan–Meier method was applied to estimate PFS and OS, and their corresponding 95% CIs. The Cox proportional hazards regression model was employed to estimate hazard ratios (HRs) and 95% CIs. Univariable Cox regression analysis was performed to evaluate the association between clinicopathological characteristics and PFS (calculated from the date of first treatment until the date of disease progression or death from any cause), OS (calculated from the date of first treatment to the date of death from any cause). Fisher exact test and Pearson χ² test were used to determine the associations between two categorical variables. Statistical analyses were performed using SPSS software (version 22.0) and R (version 3.6.0). We used two-sided P values for all tests, and a significant level of less than 0.05 was considered statistically significant.
Results
Characteristics of patients
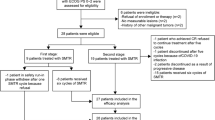
Between April 2022 and December 2023, 30 ND-PCNSL patients were assessed for eligibility. Ultimately, 26 patients were recruited. One patient discontinued treatment after 1 cycle and two discontinued after 2 cycles due to personal reasons. A total of 23 patients were included in the intention-to-treat analysis (Fig. 1). The patients’ characteristics at enrollment are shown in Table 1. The median age was 65 years (range 38‒74). Patients older than 60 years accounted for 60.9% (14/23). The distribution of patients based on the IELSG risk score was 5 (21.7%) in the low-risk group (0–1 socre), 7 (30.4%) in the intermediate-risk group (2–3 scores), and 11 (47.8%) in the high-risk group (4–5 scores). Seventeen (73.9%) patients displayed an ECOG PS ≥ 2 (five with 2 scores, four with 3 scores, and eight with 4 scores). Three (13.0%) patients were diagnosed with germinal center B-cell (GCB) and 14 (60.9%) were non-GCB. All patients had parenchymal brain involvement, except for one with additional vitreous involvement; none had meningeal involvement. Lumbar puncture was performed for testing of CSF and not for administering intrathecal therapy.
PCNSL primary central nervous system lymphoma, CR complete response, PR partial response, SD stable disease, ASCT autologous stem cell transplantation, WBRT whole brain radiotherapy.
Response and survival
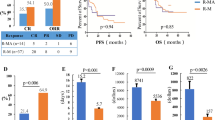
All 23 patients were assessed for efficacy (Fig. 2). One patient was evaluated as SD after 3 cycles of treatment. At the end of induction therapy, 21 patients were assessed as CR, and 1 patient was PR. The ORR was 95.7% (22/23) [95% confidence interval (CI): 83.2‒99.8%] and the CR rate (CRR) was 91.3% (21/23) [95% CI: 81.2‒99.9%] after completing the induction phase. After evaluation by the investigators, 7 patients underwent ASCT and subsequently received maintenance treatment with penpulimab. A 67-year-old patient was assessed as PR at the end of induction therapy, followed by WBRT and received penpulimab maintenance treatment (Fig. 1). Fourteen patients proceeded directly to penpulimab maintenance therapy. Three patients experienced disease progression during the maintenance therapy phase. Two patients died of pneumonia induced by COVID-19 infection during the maintenance therapy, although they were assessed as PR and CR respectively after induction therapy and had no disease progression before death.
The PFS (A) and OS (B) of the 23 PCNSL patients. Patients who received penpulimab maintenance therapy after ASCT had a better PFS and OS than penpulimab maintenance group, but there were no statistically significant differences in PFS (P = 0.16, HR 4.45, 95% CI 0.55–36.13) and OS (P = 0.20, HR 4.15, 95% CI 0.48–35.80) between the two groups (C, D). Swimmer plot of responses to and duration of treatment in our study. Each bar corresponds to one patient (E). PCNSL primary central nervous system lymphoma, PFS progression-free survival, OS overall survival, CR complete response, PR partial response, SD stable disease, PD progressive disease, CSF Cerebrospinal fluid, ctDNA circulating-tumor DNA, ASCT autologous stem cell transplantation, WBRT whole brain radiotherapy.
At a median follow-up of 29.4 months [95% CI: 16.9‒36.6 months], the 1-year PFS rate and OS rate were 87.0% [95% CI: 72.5‒97.1%] and 87.0% [95% CI: 80.7‒97.6%], respectively. The 2-year PFS rate was 70.7% [95% CI: 50.1‒91.3%], thus meeting the primary endpoint of the study, and the 2-year OS rate was 75.0% [95% CI: 50.9‒90.1%]. The median PFS, OS and duration of response (DOR) were not reached. Patients who received penpulimab maintenance therapy after ASCT (7 patients) had a 2-year PFS rate and OS rate of 100%. Patients in the penpulimab maintenance group (14 patients) had a 2-year PFS rate of 68.2% [95% CI: 24.1‒73.7%], and an OS rate of 75.7% [95% CI: 25.0‒80.6%]. However, there were no statistically significant differences in PFS (P = 0.16, HR 4.45, 95% CI 0.55–36.13) and OS (P = 0.20, HR 4.15, 95% CI 0.48–35.80) between the two groups (Fig. 2).
Safety
AEs are detailed in Table 2. All 23 patients experienced at least one treatment-related adverse event (TRAE) during the safety assessment period. The most common TRAEs across all grades were hematological toxicities (17, 73.9%), including leukopenia neutropenia (11, 47.8%), neutropenia (9, 39.1%), anemia (10, 43.5%) and thrombocytopenia (5, 21.7%), other non-hematological toxicities were increased levels of ALT (8,34.8%) and AST (7, 30.4%), pneumonia (8,34.8%) and renal dysfunction (6, 26.1%). Four patients (17.4%) experienced immune-related AEs. Grade 3 or worse TRAEs occurred in 7 (30.4%) of 23 patients, including 1 with neutropenia, 1 with neutropenia and renal dysfunction, 1 with renal dysfunction, 2 with pneumonia (8.7%), 1 with rash and 1 with hypothyroidism.
During the induction treatment, two patients discontinued induction therapy after four cycles due to grade 3 renal dysfunction, and two patients had dose reductions because of hematological toxicities. Three patients discontinued penpulimab maintenance due to a treatment-related immune adverse event during maintenance treatment (1 had severe rash and 2 had hypophysitis), which occurred during the third, fifth, and sixth maintenance treatments, respectively.
Genomic profiling
At diagnosis, tumor tissue samples were collected from eight patients, as well as CSF samples from 17 patients and blood samples from 19 patients for TGS testing. The detection of at least one pathogenic mutation or copy number variation (CNV) was 100% (8/8), 76.5% (13/17) and 68.4% (13/19) in tumor tissue, CSF, and plasma at baseline, respectively.
Paired tissue and CSF specimens from four patients harbored 74 somatic mutations, 55.4 % (41/74) of which were concomitantly detected in CSF. Analysis of seven tissue—plasma paired specimens revealed 165 variants, yet only 13.3 % (22/165) were traceable in plasma. Among sixteen CSF—plasma paired specimens, 273 mutations were cataloged; merely 14.6 % (40/273) of the CSF-identified alterations were recovered from the corresponding plasma. The most common gene mutations in tumor tissue were MYD88 (75%), PIM1 (75%), CDKN2A (75%), ETV6 (62.5%) and KMT2D (62.5%). The top 5 gene mutations in CSF were MYD88 (52.9%), PIM1 (52.9%), CD79B (52.9%), BTG2 (47.1%) and KMT2D (41.2%), and the top 5 gene mutations in plasma were MYD88 (21.1%), PIM1 (21.1%), DNMT3A (15.8%), FAT1 (15.8%), and KMT2D (15.8%) (Fig. 3).
A CSF (B), and plasma (C) at baseline, and consistency results of paired samples (D–F). CR complete response, PR partial response, SD stable disease, PD progressive disease, CSF cerebrospinal fluid, ctDNA circulating-tumor DNA, EOT end of treatment.
Somatic mutations from 22 patients were subjected to LymphGen classification. Of the 8 tissue samples, 5 (62.5%) were classified as the MCD subtype, 1 (12.5%) as BN2, 1 (12.5%) as ST2, and 1 (12.5%) as a composite MCD/ST2 subtype. Of the 17 CSF samples, mutations were detected in 13, of which 8 (61.5%) were classified as MCD and 5 (38.5%) as other subtypes. Of the 19 plasma samples, mutations were detectable in 13, with only 2 (15.4%) classified as MCD and the remaining as other subtypes.
Fourteen patients underwent dynamic CSF-ctDNA testing, while three patients declined dynamic monitoring. Results showed that eight patients (8/14, 57.1%) had negative CSF-ctDNA after induction therapy, with imaging assessments of CR. Among the six patients with positive CSF-ctDNA, imaging assessments showed that five achieved CR and one achieved PR. The latter patient died from COVID-19 during maintenance therapy. Following maintenance therapy, CSF-ctDNA became undetectable in two of the five patients; two others developed radiological progression after two cycles, while the remaining patient maintained CR despite a concurrent rise in CSF-ctDNA.
Correlation of biomarkers with clinical outcomes
Univariate analysis showed that among traditional prognostic factors, elevated CSF protein levels at diagnosis were associated with poor PFS (P = 0.03) and OS (P = 0.03), while IELSG score, lactate Dehydrogenase (LDH), ECOG score, deep-brain lesions, cell of origin (COO) classification and age were not associated with prognosis. Our results also showed that the persistence of CSF-ctDNA at the end of induction treatment was associated with poor PFS (P = 0.04) and OS (P = 0.04) (Fig. 4).
Progression-free survival (a) and overall survival (b). IELSG international extranodal lymphoma study group, ECOG eastern cooperative oncology group, COO cell of origin, LDH lactate dehydrogenase, CSF cerebrospinal fluid, ctDNA circulating-tumor DNA, EOT end of treatment.
Discussion
HD-MTX is the hallmark of induction therapy for PCNSL, and MATRix regimen showed favorable survival outcomes, but with a high incidence of AEs. Previous studies had indicated that amplification of 9p24.1 was commonly observed in PCNSL, and the upregulation of the PD-1/PD-L1 axis was associated with unfavorable prognosis [10,11,12], and PD-1 antibody has been reported to have encouraging efficacy in PCNSL patients [13,14,15, 27]. PD-1 inhibitors can re-engage cytotoxic T cell-mediated antitumor activity by blocking the interaction between PD-1 on T cells and PD-L1 on tumor cells, thereby thwarting tumor immune evasion. Zhou et al. found that T cells residing in the deep cervical lymph nodes have the capacity to infiltrate the intracranial space and CSF via the bloodstream. Conversely, T cells and antigens originating from the CNS can migrate into the peripheral lymphatic system, where they can stimulate the activation of T cells within the lymphoid tissues [28]. Our study of Pen-RMA regimen represents a novel therapeutic approach for ND-PCNSL. This combination aims to enhance both chemotherapeutic efficacy and immune response against PCNSL with manageable AEs.
Although preliminary results indicate that BTK inhibitors have reached high response rates in R/R PCNSL patients [29, 30], and recent reports also showed that BTK inhibitors combined with chemotherapy significantly improved the ORR and CRR in ND- PCNSL patients at the end of induction therapy [31, 32], the long-term relapse rate remains relatively high. Gao et al. reported the efficacy of high-dose methotrexate plus ibrutinib and temozolomide (MIT) in the treatment of ND-PCNSL. The regimen induced a high ORR of 93.9%, with 72.7% of patients achieving CR; nevertheless, 2-year PFS and OS rates remained 57.6 % (95% CI 49.0–66.2) and 84.8%, respectively [32].
Our study achieved the primary endpoint, with a 2-year PFS rate of 70.7% (95% CI 50.1–91.3%) in the intention-to-treat population, surpassing the 39% rate assumed under the null hypothesis, which was derived from our real-world experience with RMA without PD-1 inhibitor [26]. However, the historical controls were not collected prospectively nor reviewed under the same imaging schedule. Therefore, cross-cohort comparisons should be considered hypothesis-generating rather than definitive evidence of superiority. When placed alongside the only available randomized evidence (the IELSG32 phase II trial), our 2-year PFS rate lies between the 80% (95% CI 70–90%) observed with the consolidative WBRT group and the 69% (95% CI 59–79%) reported with ASCT group [5, 7].
ASCT or WBRT is particularly important in the consolidation treatment of PCNSL, but how to further improve long-term survival remains to be explored. The Mayo Clinic recently conducted a retrospective analysis of 148 PCNSL patients who received HD-MTX induction therapy at the Mayo Clinic between October 2010 and June 2022. Patients subsequently underwent ASCT consolidation therapy (n = 70) or HD-MTX maintenance therapy (n = 37). In the ASCT cohort, after a median follow-up of 4.5 years, the median PFS was 8.3 years and median OS had not yet been reached. At 5-years postinduction treatment initiation, the PFS and OS rates from start of postinduction therapy in the ASCT cohort were 74.6% and 76.0%, respectively, and in the HD-MTX maintenance cohort were 72.6% and 82.4%, respectively [33]. Notably, patients treated with MATRix achieved 7-year OS rates of 71% with subsequent ASCT (n = 25) and 70% with WBRT (n = 31) [34]. Although our results showed that patients who underwent ASCT followed by penpulimab maintenance therapy remained CR during follow-up, with both 2-year PFS and OS rates at 100%, this subgroup is smaller than the ASCT cohorts described in IELSG-32 or the Mayo Clinic series. Therefore, whether penpulimab maintenance truly adds incremental benefit after ASCT requires validation in larger, preferably randomized cohorts.
Although the MATRix regimen achieved favorable survival outcomes in the IELSG32 study [34], it was associated with a high incidence of AEs, particularly hematological toxicity, especially in elderly patients [5]. In a retrospective cohort of 51 transplant-ineligible patients with ND-PCNSL (median age 71 years), Kim et al. employed a dose-modified etoposide and Ara-C consolidation protocol, reducing Ara-C to 1 g/m2 every 12 h on days 1–4 (total 8 g/m2). Despite the adjustment, grade 4 neutropenia and thrombocytopenia occurred in all patients, with 94% developing febrile neutropenia and a treatment-related mortality of 7.8% [35]. Zhao et al. reported a modified regimen of rituximab, idarubicin, dexamethasone, Ara-C and MTX, using Ara-C at 1 g/m2 on days 2–3 in three patients with ND-PCNSL. All patients achieved CR after two cycles, with manageable toxicity and no treatment-related mortality [36]. Sun et al. compared a reduced Ara-C dose (0.5–1 g/m2 on day 2) with historical controls using higher doses (2 g/m2 every 12 h for four doses), showing that the rituximab, MTX, Ara-C and dexamethasone combination chemotherapy achieved a CR rate of 66.7% with no treatment-related deaths, supporting dose de-escalation to enhance tolerability and compliance [37]. These data indicated that dose de-escalation enhances tolerability and treatment compliance, potentially contributing to improved outcomes. Thus, in this study, we implemented a risk-adapted dosing strategy for Ara-C, prospectively de-escalating the dose to 1 g/m2 twice daily on day 2 for patients older than 60 years or with an ECOG PS > 2. Penpulimab, a PD-1 monoclonal antibody engineered to reduce Fc-mediated irAEs, was administered on day 5 of each cycle. This timing allows for optimal immune activation after the tumor microenvironment is disrupted by chemotherapy, potentially enhancing T-cell reactivity against residual lymphoma cells [38, 39]. Our results showed that all patients experienced TRAEs, but the majority were Grade 1–2. Only two patients died of COVID-19 pneumonia, while the other AEs were manageable. The incidence of irAEs was low and controllable. Given that a relatively large number of patients had an ECOG performance status of more than 2, with 8 patients having an ECOG score of 4, and the vast majority being elderly patients, the overall safety and tolerability were assessed to be good.
In this study, clinical indicators such as intermediate-to-high-risk IELSG scores, age >60 years, ECOG performance status, and involvement of deep brain tissue, which are considered high-risk factors, showed no statistically significant differences in patients' PFS and OS. Only elevated CSF protein levels at diagnosis and positive CSF-ctDNA at the end of induction treatment were associated with poor prognosis. Within the constraints of the present sample size, we were unable to demonstrate the expected prognostic impact of traditional high-risk features. This absence of statistical significance most likely reflects limited power rather than definitive evidence that Pen-RMA has overcome these risk factors. Whether the regimen truly mitigates high-risk biology awaits validation in larger, preferably randomized, cohorts.
The TGS results of this study showed that, compared to plasma, CSF had higher concordance of ctDNA with tissue. The LymphGen classification results indicated that both tissue and CSF were predominantly characterized by the MCD subtype, at 62.5% and 61.5%, respectively, and MYD88, PIM1, CDKN2A, ETV6, CD79B, BTG2, and KMT2D mutations were identified as the top mutated genes in tissue and CSF, which is consistent with the literature reports [32, 40].
Although the study had some limitations, Pen-RMA demonstrated preliminary evidence of both activity and safety in patients with ND-PCNSL, with durable remissions and low toxicity. Maintenance treatment with penpulimab after ASCT or WBRT was feasible and appeared to prolong disease control. These promising signals now need to be confirmed in a larger, adequately powered randomized controlled study that we are planning to formally evaluate the efficacy and long-term safety of Pen-RMA.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Grommes C, DeAngelis LM. Primary CNS lymphoma. J Clin Oncol. 2017;35:2410–8.
Alaggio R, Amador C, Anagnostopoulos I, Attygalle AD, Araujo IBO, Berti E, et al. The 5th edition of the World Health Organization classification of haematolymphoid tumours: lymphoid neoplasms. Leukemia. 2022;36:1720–48.
Fallah J, Qunaj L, Olszewski AJ. Therapy and outcomes of primary central nervous system lymphoma in the United States: analysis of the National Cancer Database. Blood Adv. 2016;1:112–21.
Norden AD, Drappatz J, Wen PY, Claus EB. Survival among patients with primary central nervous system lymphoma, 1973-2004. J Neurooncol. 2011;101:487–93.
Ferreri AJ, Cwynarski K, Pulczynski E, Ponzoni M, Deckert M, Politi LS, et al. Chemoimmunotherapy with methotrexate, cytarabine, thiotepa, and rituximab (MATRix regimen) in patients with primary CNS lymphoma: results of the first randomisation of the International Extranodal Lymphoma Study Group-32 (IELSG32) phase 2 trial. Lancet Haematol. 2016;3:e217–27.
Bromberg JEC, Issa S, van der Holt B, van der Meulen M, Dirven L, Minnema MC, et al. Survival, neurocognitive function, and health-related quality of life outcomes after rituximab-methotrexate, BCNU, teniposide, and prednisolone for primary CNS lymphoma: final results of the HOVON 105/ALLG NHL 24 study. Neuro Oncol. 2024;26:724–34.
Ferreri AJM, Cwynarski K, Pulczynski E, Fox CP, Schorb E, La Rosee P, et al. Whole-brain radiotherapy or autologous stem-cell transplantation as consolidation strategies after high-dose methotrexate-based chemoimmunotherapy in patients with primary CNS lymphoma: results of the second randomisation of the International Extranodal Lymphoma Study Group-32 phase 2 trial. Lancet Haematol. 2017;4:e510–e23.
Schaff LR, Grommes C. Primary central nervous system lymphoma. Blood. 2022;140:971–9.
Nayyar N, White MD, Gill CM, Lastrapes M, Bertalan M, Kaplan A, et al. MYD88 L265P mutation and CDKN2A loss are early mutational events in primary central nervous system diffuse large B-cell lymphomas. Blood Adv. 2019;3:375–83.
Gravelle P, Burroni B, Pericart S, Rossi C, Bezombes C, Tosolini M, et al. Mechanisms of PD-1/PD-L1 expression and prognostic relevance in non-Hodgkin lymphoma: a summary of immunohistochemical studies. Oncotarget. 2017;8:44960–75.
Chapuy B, Roemer MG, Stewart C, Tan Y, Abo RP, Zhang L, et al. Targetable genetic features of primary testicular and primary central nervous system lymphomas. Blood. 2016;127:869–81.
Four M, Cacheux V, Tempier A, Platero D, Fabbro M, Marin G, et al. PD1 and PDL1 expression in primary central nervous system diffuse large B-cell lymphoma are frequent and expression of PD1 predicts poor survival. Hematol Oncol. 2017;35:487–96.
Graber JJ, Plato B, Mawad R, Moore DJ. Pembrolizumab immunotherapy for relapsed CNS Lymphoma. Leuk Lymphoma. 2020;61:1766–8.
Nayak L, Iwamoto FM, LaCasce A, Mukundan S, Roemer MGM, Chapuy B, et al. PD-1 blockade with nivolumab in relapsed/refractory primary central nervous system and testicular lymphoma. Blood. 2017;129:3071–3.
Zeng Z, Yang A, Yang J, Zhang S, Xing Z, Wang X, et al. Sintilimab (anti-PD-1 antibody) combined with high-dose methotrexate, temozolomide, and rituximab (anti-CD20 antibody) in primary central nervous system lymphoma: a phase 2 study. Signal Transduct Target Ther. 2024;9:229.
Shen L, Guo J, Zhang Q, Pan H, Yuan Y, Bai Y, et al. Tislelizumab in Chinese patients with advanced solid tumors: an open-label, non-comparative, phase 1/2 study. J Immunother Cancer. 2020;8:1.
Zhang T, Song X, Xu L, Ma J, Zhang Y, Gong W, et al. The binding of an anti-PD-1 antibody to FcgammaRIota has a profound impact on its biological functions. Cancer Immunol Immunother. 2018;67:1079–90.
Fang W, Yang Y, Ma Y, Hong S, Lin L, He X, et al. Camrelizumab (SHR-1210) alone or in combination with gemcitabine plus cisplatin for nasopharyngeal carcinoma: results from two single-arm, phase 1 trials. Lancet Oncol. 2018;19:1338–50.
Bruhns P. Properties of mouse and human IgG receptors and their contribution to disease models. Blood. 2012;119:5640–9.
Kinder M, Greenplate AR, Strohl WR, Jordan RE, Brezski RJ. An Fc engineering approach that modulates antibody-dependent cytokine release without altering cell-killing functions. MAbs. 2015;7:494–504.
Dahan R, Sega E, Engelhardt J, Selby M, Korman AJ, Ravetch JV. FcgammaRs modulate the anti-tumor activity of antibodies targeting the PD-1/PD-L1 Axis. Cancer Cell. 2015;28:285–95.
Dhillon S. Penpulimab: first approval. Drugs. 2021;81:2159–66.
Abrey LE, Batchelor TT, Ferreri AJ, Gospodarowicz M, Pulczynski EJ, Zucca E, et al. Report of an international workshop to standardize baseline evaluation and response criteria for primary CNS lymphoma. J Clin Oncol. 2005;23:5034–43.
Liang JH, Wu YF, Shen HR, Li Y, Liang JH, Gao R, et al. Clinical implications of CSF-ctDNA positivity in newly diagnosed diffuse large B cell lymphoma. Leukemia. 2024;38:1541–52.
Wright GW, Huang DW, Phelan JD, Coulibaly ZA, Roulland S, Young RM, et al. A probabilistic classification tool for genetic subtypes of diffuse large B cell lymphoma with therapeutic implications. Cancer Cell. 2020;37:551–68.
Du KX, Shen HR, Pan BH, Luthuli S, Wang L, Liang JH, et al. Prognostic value of POD18 combined with improved IELSG in primary central nervous system lymphoma. Clin Transl Oncol. 2024;26:720–31.
Qiu Y, Li Z, Pouzoulet F, Vishnu P, Copland JA 3rd, Knutson KL, et al. Immune checkpoint inhibition by anti-PDCD1 (anti-PD1) monoclonal antibody has significant therapeutic activity against central nervous system lymphoma in an immunocompetent preclinical model. Br J Haematol. 2018;183:674–8.
Zhou S, Xie J, Huang Z, Deng L, Wu L, Yu J, et al. Anti-PD-(L)1 immunotherapy for brain metastases in non-small cell lung cancer: mechanisms, advances, and challenges. Cancer Lett. 2021;502:166–79.
Grommes C, Pastore A, Palaskas N, Tang SS, Campos C, Schartz D, et al. Ibrutinib unmasks critical role of Bruton tyrosine kinase in primary CNS lymphoma. Cancer Discov. 2017;7:1018–29.
Yang C, Cui Y, Ren X, Li M, Yu K, Shen S, et al. Orelabrutinib combined with lenalidomide and immunochemotherapy for relapsed/refractory primary central nervous system lymphoma: a retrospective analysis of case series. Front Oncol. 2022;12:901797.
Xie Y, Qu S, Liao LS, Zheng ZH, Lin Y, Chen WM, et al. Evaluation of the short-term efficacy and safety of orelabrutinib combined with high-dose methotrexate in the first-line treatment of elderly patients with high risk primary central nervous system lymphoma. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2023;31:1714–9.
Gao Y, Ping L, Shan C, Huang H, Li Z, Zhou H, et al. High-dose methotrexate, ibrutinib, and temozolomide in the treatment of newly diagnosed primary CNS lymphoma: a multicenter, prospective phase-II study. Blood Cancer Discov. 2025;6:191–202.
Hwang SR, Godby RC, Negaard BJ, Mwangi R, Nedved AN, Barreto JN, et al. Comparison of outcomes in postinduction strategies for primary central nervous system lymphoma: a Mayo Clinic experience. Blood Adv. 2025;9:924–32.
Ferreri AJM, Cwynarski K, Pulczynski E, Fox CP, Schorb E, Celico C, et al. Long-term efficacy, safety and neurotolerability of MATRix regimen followed by autologous transplant in primary CNS lymphoma: 7-year results of the IELSG32 randomized trial. Leukemia. 2022;36:1870–8.
Kim YR, Cho H, Kim SJ, Chung H, Kook HW, Jang JE, et al. Clinical outcomes of etoposide and cytarabine as consolidation in elderly patients with primary CNS lymphoma. Oncologist. 2024;29:e796–e802.
Zhao D, Qian L, Shen J, Liu X, Mei K, Cen J, et al. Combined treatment of rituximab, idarubicin, dexamethasone, cytarabine, methotrexate with radiotherapy for primary central nervous system lymphoma. J Cell Mol Med. 2014;18:1081–6.
Sun X, Liu J, Wang Y, Bai X, Chen Y, Qian J, et al. Methotrexate-cytarabine-dexamethasone combination chemotherapy with or without rituximab in patients with primary central nervous system lymphoma. Oncotarget. 2017;8:49156–64.
Zitvogel L, Kepp O, Senovilla L, Menger L, Chaput N, Kroemer G. Immunogenic tumor cell death for optimal anticancer therapy: the calreticulin exposure pathway. Clin Cancer Res. 2010;16:3100–4.
Zhao Z, Liu S, Sun R, Zhu W, Zhang Y, Liu T, et al. The combination of oxaliplatin and anti-PD-1 inhibitor promotes immune cells infiltration and enhances anti-tumor effect of PD-1 blockade in bladder cancer. Front Immunol. 2023;14:1085476.
Bonzheim I, Sander P, Salmeron-Villalobos J, Susskind D, Szurman P, Gekeler F, et al. The molecular hallmarks of primary and secondary vitreoretinal lymphoma. Blood Adv. 2022;6:1598–607.
Funding
This research was funded by the National Natural Science Foundation of China (grant number 82200887 and 82370194), Jiangsu Science and Technology Department (grant numbers BK20220716 and BE2023780), China Postdoctoral Science Foundation (grant numbers 2022M7114034 and 2023M741463), Nanjing Municipal Returned Overseas Scholars Foundation (grant number BSHNJ2023011), Jiangsu Province Hospital Talent Specialized Foundation (grant number MXJL202107), Jiangsu Province "333" Project (grant number BRA2018085), Jiangsu Province Hospital Clinical Diagnosis and Treatment Technologies Innovation Project Foundation (grant number JBGS202405). Jiangsu Province Hospital (the First Affiliated Hospital with Nanjing Medical University) Clinical Capacity Enhancement Project (JSPH-MC-2023-10).
Author information
Authors and Affiliations
Contributions
Conception and design: HRS and WX. Collection of study materials or patients’ data: HRS, JZW, HY, KXD, LS, YFW, WH, YL, YLK, XYZ, RG, JYL, LW, JHL and WX. Assembly of data and data analysis: HRS, JZW, and HY. Manuscript writing and editing: HRS, JZW, YH, JHL, LW and WX. Final approval of manuscript: HRS, JZW, HY, KXD, LS, YFW, WH, YL, YLK, XYZ, RG, JYL, LW, JHL and WX. Accountable for all aspects of the work: HRS, JZW, HY, KXD, LS, YFW, WH, YL, YLK, XYZ, RG, JYL, LW, JHL and WX.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This study was approved by an institutional review board and ethics committee at Jiangsu Province Hospital in accordance with the Declaration of Helsinki (2022-SR-166). All patients gave written informed consent before enrollment. The study is registered on clinicaltrials.gov (NCT05347641). The study protocol is available online.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shen, HR., Wu, JZ., Yin, H. et al. Penpulimab combined with rituximab, high-dose methotrexate, and cytarabine (Pen-RMA) in newly diagnosed primary central nervous system lymphoma (PCNSL): a phase 2 trial. Blood Cancer J. 16, 23 (2026). https://doi.org/10.1038/s41408-026-01450-w
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41408-026-01450-w