Abstract
Multiple myeloma (MM), long viewed as incurable, is entering a transformative era marked by deeper remissions, prolonged survival, and the realistic prospect of cure. The greatest opportunity lies at diagnosis, before genomic heterogeneity and immune escape limit therapeutic impact. High-risk smoldering MM provides a preclinical window where early intervention with targeted antibodies or combinations can delay progression and, as in some trials, improve survival. Refining the threshold for treatment-entry requires integrated risk models that combine genomics, microenvironmental and immune profiling, and artificial intelligence. The therapeutic goal is sustained minimal residual disease (MRD) negativity at a sensitivity of 10⁻⁶, validated with functional imaging to exclude focal disease. Achieving two years of sustained MRD negativity in standard-risk and three years or longer in high-risk disease strongly predicts long-term progression-free survival; five years off therapy may approximate operational cure. Quadruplet induction, consolidation, and tailored maintenance regimens maximize depth and durability of response. To advance this vision, priorities include standardized MRD and imaging assays, MRD-adapted clinical trials, strategies balancing treatment toxicity with quality of life, and equitable global access. With decisive and risk-adapted strategies, achieving cure in MM within the next decade is no longer aspirational but increasingly within reach.
Similar content being viewed by others
Introduction
Multiple myeloma (MM), historically viewed as an incurable plasma cell malignancy, has in recent years seen dramatic improvements in outcomes: deeper remissions, longer survival, and increasingly, long-term disease-free intervals [1]. The time is ripe to set a bold goal: curing MM in a subset of patients, particularly if addressed aggressively at diagnosis. Achieving cure (operational or functional) for more patients within the next ten years is a realistic target, provided we sharpen three axes: early intervention, maximal depth and durability of response (especially sustained minimal residual disease [MRD] negativity), and balance these with safety and quality of life (QoL) [2]. Below we outline the rationale and propose key strategic imperatives, with current evidence to support them.
Why diagnosis is the best window for cure
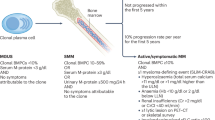
The concept of cure in MM hinges on eradicating or durably suppressing all malignant clone reservoirs before they adapt, diversify, or exploit microenvironmental niches. Once overt disease has evolved with organ damage, high disease burden, immune suppression, and genomic heterogeneity, it becomes exponentially harder to eliminate disease completely. Furthermore, asymptomatic disease states, particularly smoldering MM (SMM; especially high-risk SMM) offer a preclinical window. Emerging evidence shows that early intervention in high-risk SMM significantly delays progression to overt MM and, in some trials, improves overall survival. A recent meta-analysis of randomized trials in intermediate- or high-risk SMM reported a pooled hazard ratio of ~0.40 for disease progression or death in treated versus (vs.) observation arms [3]. The AQUILA trial showed that daratumumab monotherapy vs. observation in high-risk SMM significantly reduced risk of progression to MM and death [4]. The GEM-CESAR trial tested an intensive KRd-based strategy followed by, autologous hematopoietic cell transplant (AHCT), consolidation, and maintenance, achieving exceptionally high rates of MRD negativity at both 10⁻⁵ and 10⁻⁶ sensitivity, with a substantial proportion of patients maintaining deep, durable remissions [5]. Similarly, the ASCENT trial employed multiagent induction, AHCT, and consolidation, demonstrating that aggressive, myeloma-like treatment in high-risk SMM can convert a large fraction of patients to sustained MRD-negative status [6]. More recently, the ImmunoPRISM program has explored early multi-immunotherapy approaches, including combinations of monoclonal antibodies and immune-modulating backbones, aimed at inducing profound cytoreduction while the disease remains biologically less complex [7]. Early intervention, then, seems likely to shift the disease course, preventing the accrual of complications and perhaps enabling cure in more patients. The challenge is drawing the line: when to treat, in whom, how intensively.
Precision in defining treatment-entry: SMM and active MM boundary
We must refine risk stratification not only to identify high-risk SMM patients who benefit from early therapy, but equally to avoid overtreating those with indolent disease who may remain stable for years without progression. Current tools include the SLiM-CRAB criteria, the 20/2/20 model, cytogenetics (e.g. del(17p), t(4;14), 1q21 gain), circulating tumor cell load, genomic signatures, and features of the immune microenvironment. But there is heterogeneity and imperfect predictive value [8]. Work in artificial intelligence and machine learning applied to MM genomics plus microenvironment and immune profiling promises to sharpen predictions of who will progress and when. In fact, the concept of genomic MM and genomic MGUS was recently introduced to differentiate the subsets of MGUS and SMM that are biologically malignant with genomic features indistinguishable from MM from the subset that is premalignant and unlikely to progress to malignancy [9]. Such modeling could allow us to trigger therapy just early enough to prevent irreversible damage or clonal escape, but avoid overtreating those who may never progress. Early trials in SMM also underscore that toxicity and QoL must not be neglected [3].
Maximizing depth of response: MRD negativity at high sensitivity (≥10−6) as a surrogate for cure
MRD has become the most powerful early prognostic marker in MM. Achieving MRD negativity predicts longer progression-free survival (PFS) and overall survival [10]. Achieving sustained MRD negativity (over time), particularly at high sensitivity (10⁻⁶), appears to correlate with outcomes approaching those expected of cure [11]. For instance, a recent observational study reported that patients with MRD negativity sustained for more than three years had a 10-year PFS of ~88% [12]. In the PERSEUS frontline phase 3 trial, the use of a quadruplet induction (daratumumab + bortezomib, lenalidomide, and dexamethasone [VRd]) plus daratumumab + lenalidomide maintenance yielded much higher rates of MRD negativity at 10⁻⁶, and significantly higher rates of sustained MRD negativity at 12 and 24 months, vs. VRd without daratumumab. In both standard- and high-risk cytogenetic subgroups, PFS at 48 months in patients achieving sustained MRD negativity exceeded 94% [13]. Furthermore, recent advances in MRD assessment are now pushing sensitivity thresholds beyond the conventional 10⁻⁶ level, with next-generation sequencing platforms and enhanced multiparametric flow cytometry capable of detecting residual disease down to 10⁻⁷ [14, 15]. These ultra-sensitive assays have the potential to refine our definitions of “deep” and “sustained” MRD negativity, particularly as we attempt to correlate depth with long-term relapse risk and the likelihood of operational cure.
Functional imaging (e.g. fludeoxyglucose-18-positron emission tomography/computed tomography [PET/CT], whole-body diffusion-weighted magnetic resonance imaging) is critical to complement bone marrow MRD assays, given that patches of disease may exist outside the marrow or in focal lesions that escape catchment. Studies show that combining imaging with bone marrow MRD results improves prognostic stratification [16]. Thus, frontline strategies should aim at induction + consolidation + maintenance such that: (i) depth of response reaches MRD negativity at sensitivity ≥10⁻⁶; (ii) duration of therapy is long enough to embed durability of MRD negativity; and (iii) functional imaging is also negative.
MGUS-like immune control
An additional pathway toward long-term disease control, independent of strict MRD negativity, is the emergence of a “MGUS-like” immune reconstitution phenotype in a subset of patients [17, 18]. These individuals do not achieve deep MRD negativity by conventional or next-generation assays, yet they maintain remarkably stable, low-level clonal populations that behave biologically more like monoclonal gammopathy of undetermined significance than active myeloma. This state appears to reflect effective immune surveillance and microenvironmental control rather than complete eradication of malignant cells, and is associated with prolonged remission and excellent quality of life. Recognizing this alternative route to functional cure is essential, as it broadens our understanding of durable disease control: cure may be achieved not only through maximal cytoreduction, but also through restored immune equilibrium capable of maintaining long-term stability without continuous therapy.
Treatment duration by risk levels: defining standards
To move from long remission toward cure, we must define treatment durations tailored by patient risk. In standard-risk patients, the aim is to achieve sustained MRD negativity (10⁻⁶) for ~2 years including negative functional imaging. In high-risk patients, the risk of relapse is higher. Thus, we should aim for at least ~3 years of sustained MRD negativity (10⁻⁶) including negative imaging. These durations are not arbitrary: in several studies, duration of sustained MRD negativity correlates with longer PFS and fewer relapses. For example, in the integrated next generation sequencing MRD study post-autologous stem cell transplant, patients with stable MRD negativity over multiple measurements ( ≥ 1 year apart) including PET/CT, showed significantly longer time to next treatment, even in those with high-risk cytogenetics [16]. Moreover, sustained MRD negativity off therapy for ≥5 years might serve as an operational definition of cure i.e. clinical remission without treatment, no progression, no disease symptoms, and QoL close to normal. Evidence for this kind of long-term treatment-free remission is emerging, though still limited [8].
Intensifying frontline strategies: induction, consolidation, maintenance
To deliver the depth and durability we envision, each phase of initial therapy must be optimized. Induction should rely on quadruplet regimens in transplant-eligible patients, combining proteasome inhibitors, immunomodulatory drugs, monoclonal antibodies (CD38-directed or other antigens), and dexamethasone. The goal is to drive rapid, deep responses, minimizing residual disease and suppressing aggressive clones early. The PERSEUS trial, for example, compared DVRd (daratumumab + VRd) induction/consolidation plus DR maintenance vs. VRd + lenalidomide. DVRd achieved substantially higher MRD negativity rates (10⁻⁶) and sustained MRD over 12 or 24 months [9]. In the MIDAS trial, at the end of 4 cycle of induction with Isa-KRD (isatuximab, carfilzomib + Rd) 63% of the patients in the intention-to-treat population were MRD negative at 10 − 5 sensitivity [19]. At present, evidence clearly supports the addition of an anti-CD38 antibody to standard triplets for induction. Similarly, in transplant ineligible patients in the IMROZ trial, IsaVRd achieved significantly higher MRD negativity rates (10⁻⁶) compare to VRd, translating in an estimated PFS at 60 months of 63.2% in the IsaVRd (Isatuximab + VRD) group, as compared with 45.2% in the VRd group (P < 0.001) [20]. Similarly, in the BENEFIT trial, the 18-month MRD negativity rates at 10-5 were reported in 35 patients (26%, 95% confidence interval (CI) 19-34) in IsaRd versus 71 (53%, 95% CI 44-61) in Isa-VRd (odds ratio for MRD negativity 3.16, 95% CI 1.89-5.28, P < 0.0001) [21]. Looking ahead, the incorporation of antibodies against alternative targets or the use of bispecific antibodies may further increase the proportion of patients achieving MRD negativity at 10⁻⁶. Nevertheless, despite quadruplet induction regimens have delivered unprecedented rates of MRD negativity, including at sensitivities of 10⁻⁶, emerging long-term follow-up data make it clear that these approaches alone are likely not curative for many patients. Even with deep responses after induction, relapse eventually occurs, underscoring that depth without durability is insufficient to achieve cure. True long-term disease control therefore requires additional, strategically sequenced components, including effective consolidation and maintenance.
Following induction, AHCT remains the standard backbone for eligible patients, being both effective and cost-efficient. Nevertheless, recent artificial intelligence-based models integrating genomics, clinical variables, and microenvironmental features have been used to identify patients for whom AHCT is particularly effective versus patients for whom the impact is limited, supporting the development of more precise, risk-adapted cure strategies [9].
Consolidation therapy thereafter, whether with additional cycles of antibody-based combinations or novel agents, aims to deepen responses and convert residual disease into MRD negativity. For transplant-ineligible patients, consolidation may not be part of the treatment algorithm, but response intensification strategies tailored to patient fitness should still be pursued to maintain suppression of emergent clones.
Finally, for maintenance, duration, and intensity, agent choice should be risk adapted. Treatment should continue long enough to secure sustained MRD negativity, with strategies to stop safely in those who reach durable deep remission, off therapy. However, stopping prematurely risks relapse. For standard-risk disease, maintenance might be for ~2 years after attaining MRD negativity, with monitoring; for high-risk disease, perhaps three or more years and potentially longer, or continuous until evidence of durable MRD negativity off therapy is established. An important unresolved question is the optimal composition of maintenance. Lenalidomide remains the standard for most patients, but combinations (e.g. with a CD38 antibody) may be warranted for those with high-risk cytogenetics. Future strategies may also incorporate newer immunotherapies such as bispecific antibodies, CAR-T cells or CELMoDs, especially in patients at highest risk of relapse, to further eliminate resistant clones. Together, these elements may transform the remarkable early responses achieved with quadruplets into durable remissions compatible with operational cure.
Safety, quality of life and fitness considerations
Aggressive therapy risks greater toxicity. Cure must not come at unacceptable cost, whether in terms of toxicity burden for patients or financial sustainability for healthcare systems. The key to implementation will be patient selection where fitness, frailty, and comorbidities must be rigorously assessed. Elderly or frail patients may not tolerate maximal therapy; in such cases, alternative regimens or less intensive but still depth-oriented therapy should be used. Monitoring and management of adverse events is of utmost importance. In trials of quadruplet regimens (e.g. those adding a CD38-directed antibody such as daratumumab), safety data have been favorable despite increased rates of neutropenia and thrombocytopenia, but manageable when carefully monitored. QoL has in many cases been preserved. In early intervention for SMM (e.g. daratumumab in AQUILA), safety and QoL metrics did not show major detriment [4].
Duration of therapy is another crucial parameter (fixed-duration therapy vs. continuous therapy). For some patients, stopping therapy after sufficiently durable MRD negativity (including imaging) may allow off-therapy periods with maintenance only, or no therapy. The trade-off between relapse risk and long-term toxicity together with patient preferences will matter. Finally, we need to leverage functional imaging and comprehensive MRD assessments. To avoid underestimating disease or missing focal lesions, imaging (PET/CT, whole body magnetic resonance imaging) should accompany marrow MRD assays. The combined MRD results with negative imaging status carries superior prognostic value [10].
Challenges and research gaps
To make this vision real, several challenges must be addressed [22].
We believe that standardization of MRD assays is essential. Sensitivity thresholds (10⁻⁵ vs. 10⁻⁶), sample handling, assays (next-generation sequencing vs. flow cytometry), marrow vs. peripheral blood, reproducible imaging criteria. Inter-institutional standardization is also essential. More powerful artificial intelligence/machine learning methods that integrate genomics, immune profiling, microenvironment features, and clinical risk to stratify patients early (e.g. risk of progression from SMM, risk of relapse even with MRD negativity) are needed. Current models are improving but still have error.
Cellular therapies may also contribute to a true curative fraction in multiple myeloma as suggested by a growing body of evidence. The most compelling data come from the CARTITUDE-1 study, whose 5-year update shows that approximately one-third of heavily pretreated patients remain treatment-free and progression-free, a landmark outcome that raises the possibility of long-term functional cure in a subset of individuals [23]. Biologically, these results likely reflect the combination of profound tumor debulking and immune reconstitution, with CAR-T cells reshaping host immunity in a manner difficult to achieve with conventional agents. Building on this foundation, earlier-line trials such as CARTITUDE-5 (evaluating cilta-cel at first line without AHCT, NCT04923893) and CARTITUDE-6 (testing CAR-T as frontline consolidation in place of or compared with AHCT, NCT05257083) aim to determine whether moving CAR-T therapy earlier in the disease course can further expand the pool of patients achieving durable, treatment-free remission. Collectively, these studies signal a pivotal shift: the integration of cellular therapy, particularly when applied earlier or in combination with MRD-directed strategies, may become a cornerstone of cure-oriented treatment paradigms in myeloma.
Finally, clinical trials with MRD-guided therapy decisions should be developed widely [19]. These trials should use MRD status (including imaging) to escalate, de-escalate, stop, or continue therapy. Such trials will test duration of therapy required for cure (e.g. how long is “sustained MRD negativity”). With respect to operational definitions of cure, data are still limited on true operational cure, as in prolonged off-therapy disease-free periods ( ≥ 5 years), normal life expectancy, preserved QoL. Studies involving more patients and longer follow-up are needed. Finally, ensuring access and tolerability in diverse patient populations based on age, fitness, health systems, cost, and patient preferences has to become a priority. Treatment must be feasible globally, not just in highly resourced centers or countries.
Conclusion
Multiple myeloma is no longer a uniformly fatal disease. With modern therapeutics, including proteasome inhibitors, immunomodulatory drugs, monoclonal antibodies, cellular therapies, combined with refined diagnostics, imaging, and MRD assessment, we are entering an era where functional cure is not just aspirational, but achievable for an increasing number of patients (Table 1). In fact, Long-term follow-up studies of intensively treated NDMM have already documented cases of ‘functional cure,’ characterized by treatment-free remission extending beyond a decade and near-normal life expectancy [24, 25]. If we act decisively at diagnosis, draw clearer lines for when to treat smoldering disease, optimize induction/consolidation/maintenance with the goal of sustained MRD negativity (10⁻⁶) including imaging, and preserve QoL, then it is entirely plausible to see cure rates rise significantly within the next 5-10 years. An important next step will be to refine the definition of which patients may already be “cured” with current quadruplet-based approaches. Are we curing primarily standard-risk, transplant-eligible patients, or could some transplant-ineligible patients also achieve operational cure with optimized frontline regimens? Equally critical is clarifying whether high-risk patients (defined by adverse cytogenetics or molecular features) can reach sustained MRD negativity with long-term benefit, and if so, which subgroups are most likely to derive such durable remissions. Beyond this, the pressing challenge is how to extend cure to a broader proportion of patients. Novel immunotherapies, in particular bispecific antibodies and emerging constructs, may play a pivotal role by further deepening responses or rescuing patients at risk of relapse. At the same time, the experience in high-risk smoldering MM reminds us of the limitations of early intervention, as even intensive strategies show that MRD negativity does not always translate into long-lasting benefit. Thus, the field must reconcile the promise of aggressive upfront treatment with the reality that true operational cure requires not just depth but also durability of response, and that this balance may differ between disease states and risk categories. We must commit to rigorous trials, to investing in predictive tools, and to treating the disease early but carefully. Cure is no longer a mirage but a horizon toward which MM research and clinical practice must stride with courage and precision.
References
Malard F, Neri P, Bahlis NJ, Terpos E, Moukalled N, Hungria VTM, et al. Multiple myeloma. Nat Rev Dis Prim. 2024;10:45.
Mohty M, Avet-Loiseau H, Harousseau J-L. Requirements for operational cure in multiple myeloma. Blood. 2021;138:1406–11.
Ntanasis-Stathopoulos I, Filippatos C, Malandrakis P, Kastritis E, Terpos E, Dimopoulos M-A, et al. Observation or treatment for smoldering multiple myeloma? A systematic review and meta-analysis of randomized controlled studies. Blood Cancer J. 2025;15:104.
Dimopoulos MA, Voorhees PM, Schjesvold F, Cohen YC, Hungria V, Sandhu I, et al. Daratumumab or active monitoring for high-risk smoldering multiple myeloma. N Engl J Med. 2025;392:1777–88.
Puig N, Agulló C, Contreras T, Pérez JJ, Aires I, Calasanz MJ, et al. Single-point and kinetics of peripheral residual disease by mass spectrometry to predict outcome in patients with high-risk smoldering multiple myeloma included in the GEM-CESAR trial. Haematologica. 2024;109:4056–66.
Kumar SK, Alsina M, Laplant B, Badros AZ, Abdallah A-O, Abonour R, et al. Fixed Duration Therapy with Daratumumab, Carfilzomib, Lenalidomide and Dexamethasone for High Risk Smoldering Multiple Myeloma-Results of the Ascent Trial. Blood. 2022;140:1830–2.
Nadeem O, Magidson S, Midha S, O’Donnell EK, Hartley-Brown MA, Sperling AS, et al. Immuno-PRISM: a randomized phase II platform study of bispecific antibodies in high-risk smoldering myeloma. Blood. 2023;142:206.
Engelhardt M, Kortüm KM, Goldschmidt H, Merz M. Functional cure and long-term survival in multiple myeloma: how to challenge the previously impossible. Haematologica. 2024;109:2420–35.
Maura F, Rajanna AR, Ziccheddu B, Poos AM, Derkach A, Maclachlan K, et al. Genomic classification and individualized prognosis in multiple myeloma. J Clin Oncol: Off J Am Soc Clin Oncol. 2024;42:1229–40.
Mohty M, Avet-Loiseau H, Malard F, Harousseau JL. Potential future direction of measurable residual disease evaluation in multiple myeloma. Blood. 2023;142:1509–17.
San-Miguel J, Avet-Loiseau H, Paiva B, Kumar S, Dimopoulos MA, Facon T, et al. Sustained minimal residual disease negativity in newly diagnosed multiple myeloma and the impact of daratumumab in MAIA and ALCYONE. Blood. 2022;139:492–501.
Chen LY, Thibaud S, Bodnar S, Chari A, Richter J, Cho HJ, et al. MRD-negative duration following latest line of therapy predicts long-term PFS in real-world patients with multiple myeloma. Blood Adv. 2025;9:176–9.
Sonneveld P, Dimopoulos MA, Boccadoro M, Quach H, Ho PJ, Beksac M, et al. Daratumumab, Bortezomib, Lenalidomide, and Dexamethasone for Multiple Myeloma. N Engl J Med. 2024;390:301–13.
Derman BA, Major A, Cooperrider J, Jiang K, Ramsland A, Karrison T, et al. Discontinuation of maintenance therapy in multiple myeloma guided by multimodal measurable residual disease negativity (MRD2STOP). Blood Cancer J. 2024;14:170.
Takamatsu H, Takezako N, Zheng J, Moorhead M, Carlton VEH, Kong KA, et al. Prognostic value of sequencing-based minimal residual disease detection in patients with multiple myeloma who underwent autologous stem-cell transplantation. Ann Oncol. 2017;28:2503–10.
Fonseca R, Arribas M, Wiedmeier-Nutor JE, Kusne YN, González Vélez M, Kosiorek HE, et al. Integrated analysis of next generation sequencing minimal residual disease (MRD) and PET scan in transplant eligible myeloma patients. Blood Cancer J. 2023;13:32.
Burgos L, Tamariz-Amador LE, Puig N, Cedena MT, Guerrero C, Jelínek T, et al. Definition and Clinical Significance of the Monoclonal Gammopathy of Undetermined Significance-Like Phenotype in Patients With Monoclonal Gammopathies. J Clin Oncol : Off J Am Soc Clin Oncol. 2023;41:3019–31.
Paiva B, Vídriales MB, Rosiñol L, Martínez-López J, Mateos MV, Ocio EM, et al. A multiparameter flow cytometry immunophenotypic algorithm for the identification of newly diagnosed symptomatic myeloma with an MGUS-like signature and long-term disease control. Leukemia. 2013;27:2056–61.
Perrot A, Lambert J, Hulin C, Pieragostini A, Karlin L, Arnulf B, et al. Measurable residual disease-guided therapy in newly diagnosed myeloma. N Engl J Med. 2025;393:425–37.
Facon T, Dimopoulos MA, Leleu XP, Beksac M, Pour L, Hájek R, et al. Isatuximab, bortezomib, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2024;391:1597–609.
Leleu X, Hulin C, Lambert J, Bobin A, Perrot A, Karlin L, et al. Isatuximab, lenalidomide, dexamethasone and bortezomib in transplant-ineligible multiple myeloma: the randomized phase 3 BENEFIT trial. Nat Med. 2024;30:2235–41.
Mohty M, Facon T, Malard F, Harousseau J-L. A roadmap towards improving outcomes in multiple myeloma. Blood Cancer J. 2024;14:135.
Jagannath S, Martin TG, Lin Y, Cohen AD, Raje N, Htut M, et al. Long-Term (≥5-Year) remission and survival after treatment with ciltacabtagene autoleucel in CARTITUDE-1 patients with relapsed/refractory multiple myeloma. J Clin Oncol : Off J Am Soc Clin Oncol. 2025;43:2766–71.
Usmani SZ, Crowley J, Hoering A, Mitchell A, Waheed S, Nair B, et al. Improvement in long-term outcomes with successive Total Therapy trials for multiple myeloma: are patients now being cured? Leukemia. 2013;27:226–32.
Usmani SZ, Hoering A, Cavo M, Miguel JS, Goldschimdt H, Hajek R, et al. Clinical predictors of long-term survival in newly diagnosed transplant eligible multiple myeloma - an IMWG Research Project. Blood Cancer J. 2018;8:123.
Acknowledgements
The authors would like to acknowledge the outstanding contribution, efforts and dedication of all stakeholders involved in the myeloma field.
Author information
Authors and Affiliations
Contributions
MM, FM, TF, and JLH conceived and designed the work, interpreted data and drafted the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
MM reports grants and lecture honoraria from Janssen, Sanofi, Maat Pharma and JAZZ Pharmaceuticals, lecture honoraria from Celgene, Amgen, BMS, Takeda, and Pfizer, grants from Roche, all outside the submitted work. FM reports lecture honoraria from Sanofi, Novartis, BMS, Astrazeneca, Therakos, Priothera, MSD and Jazz Pharmaceutical all outside the submitted work. The other authors declare no competing financial interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mohty, M., Malard, F., Facon, T. et al. Toward a cure for multiple myeloma within a decade. Blood Cancer J. 16, 33 (2026). https://doi.org/10.1038/s41408-026-01461-7
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41408-026-01461-7