Abstract
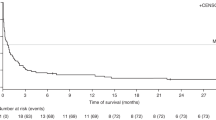
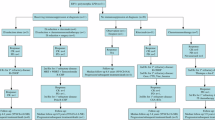
The aim of this study was to determine the current approach of EBV-driven post-transplant complications in context of monitoring, diagnosis, prevalence and treatment in EBMT transplant centers. Routine serology testing in patient and donor before HCT is performed in 95.5% centers. Pretransplant EBV-DNA is routinely tested in all patients in 32.7% centers. Monitoring for EBV infection is feasible in 98.2% centers: including 66.7% centers using standardized PCR. Post-HCT regular monitoring is performed in all patients in 80.5% centers. Anti-EBV prophylaxis with rituximab is used in 12.4% centers. Frequency of csEBV-DNA-emia was 7.4% (adults: 6.2%, children: 12.6%). The PCR threshold used to start preemptive treatment was differentiated among centers. Frequency of EBV-PTLD was 1.6% (adults: 1.3%; children: 3.5%). First-line therapy of EBV-driven complications was rituximab and reduction of immunosuppressive therapy. The rate of failure of first-line preemptive treatment was 12.0%. EBV-specific viral-specific T-lymphocytes were available in 46.0% centers. A number of new experimental therapies were given in 28 patients with resistant/refractory PTLD. In conclusion, the prevalence of EBV-DNA-emia and EBV-PTLD over the period 2020–2021 decreased in comparison to historical data. New trends (routine pretransplant screening for EBV-DNA, wider access to VST, new experimental therapies) are being observed in management of EBV infection after allo-HCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Data availability
Data represent the results of the survey. Only one answer was possible from each center. No personal data are included. With the survey design of the study, no ethical review board approval was necessary. Data set remains the property of EBMT. The results of the survey are available upon reasonable request to the corresponding author.
References
Styczynski J. Managing post-transplant lymphoproliferative disorder. Expert Opinion on Orphan Drugs. 2017;5:19–35.
Styczynski J, Tridello G, Gil L, Ljungman P, Hoek J, Iacobelli S, et al. Impact of donor Epstein-Barr virus serostatus on the incidence of graft-versus-host disease in patients with acute leukemia after hematopoietic stem-cell transplantation: a study from the Acute Leukemia and Infectious Diseases Working Parties of the European Society for Blood and Marrow Transplantation. J Clin Oncol. 2016;34:2212–20.
Styczynski J, Tridello G, Gil L, Ljungman P, Mikulska M, Ward KN, et al. Prognostic impact of EBV serostatus in patients with lymphomas or chronic malignancies undergoing allogeneic HCT. Bone Marrow Transplant. 2019;54:2060–71.
Styczynski J, Tridello G, Gil L, Ljungman P, Mikulska M, van der Werf S, et al. Prognostic impact of Epstein–Barr virus serostatus in patients with nonmalignant hematological disorders undergoing allogeneic hematopoietic cell transplantation: the study of Infectious Diseases Working Party of the European Society for Blood and Marrow Transplantation. Acta Haematol Pol. 2020;51:73–80.
Galazka P, Szafranska M, Jaremek K, Rutkowska D, Czyzewski K, Debski R, et al. Long-term follow-up of pediatric patients with EBV-related post-transplant lymphoproliferative disorder. Acta Haematol Pol. 2021;52:597–600.
Kolodziejczak M, Gil L, de la Camara R, Styczynski J. Impact of donor and recipient Epstein-Barr Virus serostatus on outcomes of allogeneic hematopoietic cell transplantation: a systematic review and meta-analysis. Ann Hematol. 2021;100:763–77.
Styczynski J, van der Velden W, Fox CP, Engelhard D, de la Camara R, Cordonnier C, et al. Management of Epstein-Barr virus infections and post-transplant lymphoproliferative disorders in patients after allogeneic hematopoietic stem cell transplantation: Sixth European Conference on Infections in Leukemia (ECIL-6) guidelines. Haematologica. 2016;101:803–11.
Marjanska A, Styczynski J. Who is the patient at risk for EBV reactivation and disease: expert opinion focused on post-transplant lymphoproliferative disorders following hematopoietic stem cell transplantation. Expert Opin Biol Ther. 2023;23:539–52.
Styczynski J, Reusser P, Einsele H, de la Camara R, Cordonnier C, Ward KN, et al. Management of HSV, VZV and EBV infections in patients with hematological malignancies and after SCT: guidelines from the Second European Conference on Infections in Leukemia. Bone Marrow Transplant. 2009;43:757–70.
Styczynski J, Gil L, Tridello G, Ljungman P, Donnelly JP, van der Velden W, et al. Response to rituximab-based therapy and risk factor analysis in Epstein Barr Virus-related lymphoproliferative disorder after hematopoietic stem cell transplant in children and adults: a study from the Infectious Diseases Working Party of the European Group for Blood and Marrow Transplantation. Clin Infect Dis. 2013;57:794–802.
Williams-Aziz SL, Hartline CB, Harden EA, Daily SL, Prichard MN, Kushner NL, et al. Comparative activities of lipid esters of cidofovir and cyclic cidofovir against replication of herpesviruses in vitro. Antimicrob Agents Chemother. 2005;49:3724–33.
Wang FZ, Roy D, Gershburg E, Whitehurst CB, Dittmer DP, Pagano JS. Maribavir inhibits Epstein-Barr virus transcription in addition to viral DNA replication. J Virol. 2009;83:12108–17.
Whitehurst CB, Sanders MK, Law M, Wang FZ, Xiong J, Dittmer DP, et al. Maribavir inhibits Epstein-Barr virus transcription through the EBV protein kinase. J Virol. 2013;87:5311–5.
Camargo JF, Morris MI, Abbo LM, Simkins J, Saneeymehri S, Alencar MC, et al. The use of brincidofovir for the treatment of mixed dsDNA viral infection. J Clin Virol. 2016;83:1–4.
Kinzel M, Kalra A, Khanolkar RA, Williamson TS, Li N, Khan F, et al. Rituximab toxicity after preemptive or therapeutic administration for post-transplant lymphoproliferative disorder. Transplant Cell Ther. 2023;29:43.e1–43.e8.
Acknowledgements
The authors acknowledge the cooperation of the participating teams and their staff. They acknowledge the excellent work of the IDWP-EBMT Data Office in Leiden. The authors thank following centers participating in the survey: CIC141 (Domenico Russo, Unit of Blood Diseases and Bone Marrow Transplantation, Brescia University, Brescia, Italy); CIC145 (Martin Kaufmann, Robert_Bosch_Krankenhaus, Stuttgart, Germany); CIC 159 (Abdullah Alsharm, King Fahad Medical City, Riyadh, Saudi Arabia); CIC 161 (Ron Ram, Tel Aviv Sourasky Medical Center, Tel Aviv, Israel); CIC 169 (Zubeyde Nur Ozkurt, Gazi University Faculty of Medicine, Ankara, Turkey); CIC 187 (Zahit Bolaman, Adnan Menderes University Med. Faculty, Aydin, Turkey); CIC 202 (Jakob Passweg, University Hospital, Basel, Basel, Switzerland); CIC 203 (Peter von dem Borne, Leiden University Hospital, Leiden, The Netherlands); CIC 207 (Régis Peffault de Latour, Saint-Louis Hospital, BMT Unit, Paris, France); CIC 212 (Stephan Mielke, Karolinska University Hospital, Stockholm, Sweden); CIC 215 (Philippe Lewalle, Institut Jules Bordet, Brussels, Belgium); CIC 219 (Samppa Ryhänen, University of Helsinki, Helsinki, Finland); CIC 227 (Werner Rabitsch, Medizinische Universitaet Wien, Vienna, Austria); CIC 234 (Xavier Poiré, Cliniques Universitaires St. Luc, Brussels, Belgium); CIC 237 (Nicole Blijlevens, Nijmegen Medical Centre, Nijmegen, The Netherlands); CIC 239 (Jürgen Kuball, University Medical Centre, Utrecht, The Netherlands); CIC 242 (Arancha Bermúdez Rodríguez, Hospital U. Marqués de Valdecilla, Santander, Spain); CIC 246 (A.E.C. Broers, Erasmus MC Cancer Institute, Rotterdam, The Netherlands); CIC 248 (Stella Santarone, Ospedale Civile, Pescara, Italy); CIC 258 (Polina Stepensky, Hadassah University Hospital, Jerusalem, Israel); CIC 259 (Thomas Schroeder, University Hospital, Essen, Duesseldorf, Germany); CIC 260 (Rodrigo Martino, Hospital Santa Creu i Sant Pau, Barcelona, Spain); CIC 261 (Yves Chalandon, Département d’Oncologie, Service d’Hématologie, Geneva, Switzerland); CIC 264 (Xavier Leleu, Hopital La Miletrie, Poitiers, France); CIC 265 (Francesco Onida, Fondazione IRCCS – Granda, Milano, Italy); CIC 267 (Edouard Forcade, CHU Bordeaux, Pessac, France); CIC 274 (Maura Faraci, HSCT Unit, IRCCS Institute G. Gaslini, Genova, Italy); CIC 283 (Stig Lenhoff, Skanes University Hospital, Lund, Sweden); CIC 285 (Alessandra Biffi, Clinica di Oncoematologia Pediatrica, Padova, Italy); CIC 304 (Riccardo Saccardi, Azienda Ospedaliera Universitaria Careggi, Firenze, Italy); CIC 307 (Simona Sica, Universita Cattolica S. Cuore, Rome, Italy); CIC 331 (Corrado Tarella, European Institute of Oncology, Milano, Italy); CIC 334 (Tayfun Güngör, University Children’s Hospital, Zürich, Switzerland); CIC 341 (Francesco Paolo Tambaro, Azienda Ospedaliera di Rilievo Nazionale, Napoli, Italy); CIC 352 (Marc Bierings, Princess Maxima Center, University Hospital for Children (WKZ), Utrecht, The Netherlands); CIC 401 (Huang He, First Affiliated Hospital of Zhejiang University School of Medicine, Hangzhou, China); CIC 434 (Aloysius Ho, Singapore General Hospital, Singapore, Singapore); CIC 443 (Xiao-jun Huang, Peking University People’s Hospital, Institute of Haematology, Beijing, China); CIC 444 (Mohsen Al Zahrani, King Abdul - Aziz Medical City, Riyadh, Saudi Arabia); CIC 452 (Petr Sedlacek, University Hospital Motol, Prague, Czech Republic; CIC 464 (Gaurav Kharya, Indraprastha Apollo Hospitals, New Delhi, India); CIC 465 (Rajat Argawal, BMJH – SANKALP, Bangalore, India); CIC 468 (Siret Ratip, Altunizade Acibadem Hospital, Istanbul, Turkey); CIC 505 (Claudia Rössig, Universitaetsklinikum Muenster, Muenster, Germany); CIC 507 (Jolanta Gozdzik, University Children’s Hospital in Krakow, Krakow, Poland); CIC 512 (Estelle Verburgh, University of Cape Town Faculty of Health Sciences, Cape Town, South Africa); CIC 515 (Urpu Salmenniemi, HUCH Comprehensive Cancer Center, Helsinki, Finland); CIC 520 (Denis A. Fedorenko, Clinic of Hematology and Cellular Therapy, Moscow, Russian Federation); CIC 521 (Robert F. Wynn, Royal Manchester Children’s Hospital, Manchester, United Kingdom); CIC 528 (Wolfgang Holter, Department of Pediatrics and Adolescent Medicine, St. Anna Children’s Hospital, Medical University of Vienna, Vienna, Austria); CIC 538 (Jaroslaw Dybko, Department of Hematology and Transplantology of Lower Silesian Center of Oncology, Wroclaw, Poland); CIC 543 (Angela Cuoghi, Azienda Ospedaliero Universitaria di Modena Policlinico, Modena, Italy); CIC 561 (Ioanna Sakellari, George Papanicolaou General Hospital, Thessaloniki, Greece); CIC 565 (Gwendolyn Van Gorkom, University Hospital Maastricht, Maastricht, The Netherlands); CIC 574 (Ludek Raida, Olomouc University Hospital, Olomouc, Czech Republic); CIC 576 (Maria Jesús Pascual Cascon, Hospital Regional de Málaga, Malaga, Spain); CIC 587 (Massimo Martino, Grande Ospedale Metropolitano Bianchi Melacrino Morelli – Centro Unico Trapianti A. Neri, Reggio C, Italy); CIC 599 (Peter Hauser, Borsod-Abaúj – Zemplén County Central Hospital, Miskolc, Hungary); CIC 604 (Panagiotis Tsirigotis, Attikon University General Hospital, Athens, Greece); CIC 613 (Josep Maria Ribera Santasusana, ICO-Hospital Universitari Germans Trias i Pujol, Badalona, Spain); CIC 614 (Nicolaus Kröger, University Hospital Eppendorf, Hamburg, Germany); CIC 623 (Fabio Benedetti, Policlinico G.B. Rossi, Verona, Italy); CIC 627 (Ali Unal, Erciyes Medical School, Kayseri, Turkey); CIC 630 (Rik Schots, Universitair Ziekenhuis Brussel, Brussels, Belgium); CIC 642 (Soledad González Muñiz, Hospital Universitario Central de Asturias, Oviedo, Spain); CIC 654 (Jessica Höll, Martin-Luther-Univ. Halle-Wittenberg, Halle, Germany); CIC 656 (Jan Vydra, Institute of Hematology and Blood Transfusion, Prague, Czech Republic); CIC 658 (Alessandro Rambaldi, ASST Papa Giovanni XXIII, Bergamo, Italy); CIC 670 (Ronit Elhasid, Tel Aviv Sourasky Medical Center, Tel Aviv, Israel); CIC 671 (Hélène Labussière-Wallet, Centre Hospitalier Lyon Sud, Lyon, France); CIC 677 (Grzegorz Helbig, Silesian Medical Academy, Katowice, Poland); CIC 684 (Ivana Bodova, Pediatric University Teaching Hospital, Bratislava, Slovak Republic); CIC 685 (Levent Undar, Akdeniz University School of Medicine, Antalya, Turkey); CIC 692 (Maurizio Musso, Ospedale La Maddalena - Dpt. Oncologico, Palermo, Italy); CIC 693 (Kazimierz Halaburda, Institute of Hematology and Transfusion Medicine, Warsaw, Poland); CIC 704 (Deborah Richardson, Southampton General Hospital, Southampton, United Kingdom); CIC 711 (Melissa Gabriel, The Children’s Hospital at Westmead, Sydney, Australia); CIC 717 (Jenny Byrne, Nottingham University, Nottingham, United Kingdom); CIC 718 (Pavel Jindra, Charles University Hospital, Pilsen, Czech Republic); CIC 725 (Aleksandr Kulagin, RM Gorbacheva Research Institute, Pavlov University, St. Petersburg, Russian Federation); CIC 730 (Lidia Gil, Poznan University of Medical Sciences, Poznan, Poland); CIC 732 (Marta Gonzalez Vicent, Niño Jesus Children’s Hospital, Madrid, Spain); CIC 737 (Jose Rifón, Clínica Universitaria de Navarra, Pamplona, Spain); CIC 750 (Bernd Gruhn, University of Jena, Jena, Germany); CIC 756 (Raffaella Cerretti, “Tor Vergata” University of Rome, Rome, Italy); CIC 764 (Department of Pediatric Hematology and Oncology; Collegium Medicum Nicolaus Copernicus University Torun; Bydgoszcz, Poland); CIC 786 (Eva Maria Wagner-Drouet, University Medical Center Mainz, Mainz, Germany); CIC 788 (Paola Coccia, Azienda Ospedali Riuniti di Ancona, Ancona, Italy); CIC 790 (Arcangelo Prete, Azienda Ospedaliero Universitaria, Bologna, Italy); CIC 795 (Mario Petrini, Azienda Ospedaliero Universitaria Pisana, Pisa, Italy); CIC 796 (Franco Locatelli, IRRCS Ospedale Pediatrico Bambino Gesù, Rome, Italy); CIC 799 (Jan Zaucha, Medical University of Gdansk, Gdansk, Poland); CIC 808 (Martin Bornhäuser, Universitaetsklinikum Dresden, Dresden, Germany); CIC 810 (Jürgen Finke, University of Freiburg, Freiburg, Germany); CIC 817 (Krzysztof Kalwak, Medical University, Wroclaw, Poland); CIC 823 (Patrick Medd, University Hospitals Plymouth NHS Trust, Plymouth, United Kingdom); CIC 824 (Gergely Kriván, Central Hospital of Southern Pest, Budapest, Hungary); CIC 825 (Marco Ladetto, H SS. Antonio e Biagio, Alessandria, Italy); CIC 856 (Amir Ali Hamidieh, Children’s Medical Centre/Tehran University of Medical Sciences, Teheran, Iran); CIC 868 (Nicola Di Renzo, Unita Operativa di Ematologia e Trapianto di cellule staminali, Lecce, Italy); CIC 911 (Akif Yesilipek, Medical Park Antalya Hospital, Antalya, Turkey); CIC 926 (Anne Sirvent, CHU Lapeyronie, Montpellier, France); CIC 931 (Depei Wu, First Affiliated Hospital of Soochow University, Suzhou, China); CIC 933 (Katharine Patrick, Sheffield Childrens NHS Foundation Trust, Sheffield, United Kingdom); CIC 935 (Anca Colita, Fundeni Clinical Institute, Bucharest, Romania); CIC 977 (Pascal Turlure, CHRU Limoges, Limoges, France); CIC 985 (Claudia Sossa, FOSCAL-UNAB, Floridablanca, Colombia); CIC 998 (Angela Maria Trujillo, Instituto de Cancerologia S.A, Medellin, Colombia); CIC 1006 (Marco Zecca, Fondazione IRCCS Policlinico San Matteo, Pavia, Italy); CIC 1008 (Erlie Jiang, Institute of Hematology, Chinese Academy of Medical Sciences, Tianjin, China); CIC 1029 (Zhao Wang, Beijing Friendship Hospital, Capital Medical University, Beijing, China); CIC 1032 (Baodong Ye, First Affiliated Hospital of Zhejiang Chinese Medical University, Hangzhou, China).
Author information
Authors and Affiliations
Contributions
JS designed the study and drafted the manuscript. GT conducted the statistical analysis. LW and NK were responsible for the data collection and assembly. SC, LG, PL, MM, DA and RdlC were responsible for the integrity of the data and gave scientific input. All authors have seen and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Styczynski, J., Tridello, G., Wendel, L. et al. Prevalence, management, and new treatment modalities of EBV-DNA-emia and EBV-PTLD after allo-HCT: survey of Infectious Diseases Working Party EBMT. Bone Marrow Transplant 59, 59–65 (2024). https://doi.org/10.1038/s41409-023-02129-7
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-023-02129-7