Abstract
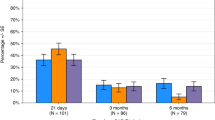
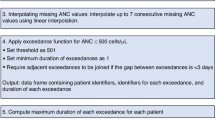
Late Immune Effector Cell-Associated HematoToxicity (ICAHT) is a recognized complication following CAR T therapy, yet their incidence and clinical consequences remain poorly defined. We assessed early and late hematotoxicity in 290 patients with large B-cell lymphoma receiving CAR T therapy. Early ICAHT ( ≤ 30 days post-infusion) occurred in 78% of patients (grade 1: 22%, grade 2: 26%, grade 3: 22%, grade 4: 8.1%). Cumulative incidence of late ICAHT ( > 30 days post-infusion) by day 100 was 45% (95% CI 39–51) for any grade, 40% (95% CI 34-46) for grade≥2, 22% (95% CI 17–27) for grade ≥3, and 7.2% (95% CI 4.4–11) for grade 4. Cumulative incidences of late moderate-severe thrombocytopenia ( < 50 × 103/μL) and anemia ( < 8 g/dL) at day 100 were 20% (95% CI 15–25) and 14% (95% CI 10-19), respectively. Early severe ICAHT (grade ≥3) was independently associated with late severe ICAHT (p < 0.001), late severe thrombocytopenia (p < 0.001), and late moderate-severe anemia (p = 0.037). Late severe ICAHT was associated with an increased hazard of late infections (HR 2.85 [95% CI 1.18–6.88], p = 0.032). These findings highlight late hematologic toxicity as a frequent and clinically relevant complication of CAR T therapy. Incorporating ICAHT grading into post-infusion monitoring may inform preventive strategies to mitigate long-term complications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The data are available from the corresponding author upon reasonable request.
References
Abramson, Palomba JS, Gordon LI ML, Lunning MA, Wang M, Arnason J, et al. Lisocabtagene maraleucel for patients with relapsed or refractory large B-cell lymphomas (TRANSCEND NHL 001): a multicentre seamless design study. Lancet. 2020;396:839–52.
Locke FL, Ghobadi A, Jacobson CA, Miklos DB, Lekakis LJ, Oluwole OO, et al. Long-term safety and activity of axicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): a single-arm, multicentre, phase 1-2 trial. Lancet Oncol. 2019;20:31–42.
Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med. 2017;377:2531–44.
Schuster SJ, Bishop MR, Tam CS, Waller EK, Borchmann P, McGuirk JP, et al. Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N Engl J Med. 2019;380:45–56.
Hirayama AV, Turtle CJ. Toxicities of CD19 CAR-T cell immunotherapy. Am J Hematol. 2019;94:S42–S9.
Brudno JN, Kochenderfer JN. Recent advances in CAR T-cell toxicity: mechanisms, manifestations and management. Blood Rev. 2019;34:45–55.
Karschnia P, Jordan JT, Forst DA, Arrillaga-Romany IC, Batchelor TT, Baehring JM, et al. Clinical presentation, management, and biomarkers of neurotoxicity after adoptive immunotherapy with CAR T cells. Blood. 2019;133:2212–21.
Sandler RD, Tattersall RS, Schoemans H, Greco R, Badoglio M, Labopin M, et al. Diagnosis and Management of Secondary HLH/MAS Following HSCT and CAR-T Cell Therapy in Adults; A Review of the Literature and a Survey of Practice Within EBMT Centres on Behalf of the Autoimmune Diseases Working Party (ADWP) and Transplant Complications Working Party (TCWP). Front Immunol. 2020;11:524.
Hill JA, Li D, Hay KA, Green ML, Cherian S, Chen X, et al. Infectious complications of CD19-targeted chimeric antigen receptor-modified T-cell immunotherapy. Blood. 2018;131:121–30.
Morris EC, Neelapu SS, Giavridis T, Sadelain M. Cytokine release syndrome and associated neurotoxicity in cancer immunotherapy. Nat Rev Immunol. 2022;22:85–96.
Rejeski K, Greco R, Onida F, Sanchez-Ortega I, Bonini C, Sureda A, et al. An International Survey on Grading, Diagnosis, and Management of Immune Effector Cell-Associated Hematotoxicity (ICAHT) Following CAR T-cell Therapy on Behalf of the EBMT and EHA. Hemasphere. 2023;7:e889.
Wudhikarn K, Tomas AA, Flynn JR, Devlin SM, Brower J, Bachanova V, et al. Low toxicity and excellent outcomes in patients with DLBCL without residual lymphoma at the time of CD19 CAR T-cell therapy. Blood Adv. 2023;7:3192–8.
Jain T, Knezevic A, Pennisi M, Chen Y, Ruiz JD, Purdon TJ, et al. Hematopoietic recovery in patients receiving chimeric antigen receptor T-cell therapy for hematologic malignancies. Blood Adv. 2020;4:3776–87.
Rejeski K, Perez A, Sesques P, Hoster E, Berger C, Jentzsch L, et al. CAR-HEMATOTOX: a model for CAR T-cell-related hematologic toxicity in relapsed/refractory large B-cell lymphoma. Blood. 2021;138:2499–513.
Beyar-Katz O, Rejeski K, Shouval R. Immune effector cell-associated hematotoxicity: mechanisms, clinical manifestations, and management strategies. Haematologica. 2025;110:1254–68.
Rejeski K, Wang Y, Hansen DK, Iacoboni G, Bachy E, Bansal R, et al. Applying the EHA/EBMT grading for ICAHT after CAR-T: comparative incidence and association with infections and mortality. Blood Adv. 2024;8:1857–68.
Harris PA, Taylor R, Minor BL, Elliott V, Fernandez M, O’Neal L, et al. The REDCap consortium: Building an international community of software platform partners. J Biomed Inf. 2019;95:103208.
Rejeski K, Subklewe M, Aljurf M, Bachy E, Balduzzi A, Barba P, et al. Immune effector cell-associated hematotoxicity: EHA/EBMT consensus grading and best practice recommendations. Blood. 2023;142:865–77.
Liang EC, Rejeski K, Fei T, Albittar A, Huang JJ, Portuguese AJ, et al. Development and validation of an automated computational approach to grade immune effector cell-associated hematotoxicity. Bone Marrow Transplant. 2024;59:910–7.
Lee DW, Santomasso BD, Locke FL, Ghobadi A, Turtle CJ, Brudno JN, et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol Blood Marrow Transpl. 2019;25:625–38.
Luft T, Benner A, Jodele S, Dandoy CE, Storb R, Gooley T, et al. EASIX in patients with acute graft-versus-host disease: a retrospective cohort analysis. Lancet Haematol. 2017;4:e414–e23.
Pennisi M, Sanchez-Escamilla M, Flynn JR, Shouval R, Alarcon Tomas A, Silverberg ML, et al. Modified EASIX predicts severe cytokine release syndrome and neurotoxicity after chimeric antigen receptor T cells. Blood Adv. 2021;5:3397–406.
Cheson BD, Fisher RI, Barrington SF, Cavalli F, Schwartz LH, Zucca E, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol. 2014;32:3059–68.
Rejeski K, Sanz J, Fei T, Nair MS, Hashmi H, Avigdor A, et al. T-ICAHT: grading and prognostic impact of thrombocytopenia after CAR T-cell therapy. Blood. 2025.
Frenking JH, Zhou X, Wagner V, Hielscher T, Kauer J, Mai EK, et al. EASIX-guided risk stratification for complications and outcome after CAR T-cell therapy with ide-cel in relapsed/refractory multiple myeloma. J Immunother Cancer. 2024;12.
Rejeski K, Perez A, Iacoboni G, Penack O, Bucklein V, Jentzsch L, et al. The CAR-HEMATOTOX risk-stratifies patients for severe infections and disease progression after CD19 CAR-T in R/R LBCL. J Immunother Cancer. 2022;10.
Cordas Dos Santos DM, Tix T, Shouval R, Gafter-Gvili A, Alberge JB, Cliff ERS, et al. A systematic review and meta-analysis of nonrelapse mortality after CAR T cell therapy. Nat Med. 2024;30:2667–78.
Raj SS, Fei T, Fried S, Ip A, Fein JA, Leslie LA, et al. An inflammatory biomarker signature of response to CAR-T cell therapy in non-Hodgkin lymphoma. Nat Med. 2025;31:1183–94.
Rejeski K, Jain MD, Shah NN, Perales MA, Subklewe M. Immune effector cell-associated haematotoxicity after CAR T-cell therapy: from mechanism to management. Lancet Haematol. 2024;11:e459–e70.
Locke FL, Rossi JM, Neelapu SS, Jacobson CA, Miklos DB, Ghobadi A, et al. Tumor burden, inflammation, and product attributes determine outcomes of axicabtagene ciloleucel in large B-cell lymphoma. Blood Adv. 2020;4:4898–911.
de Boer JW, Keijzer K, Pennings ERA, van Doesum JA, Spanjaart AM, Jak M, et al. Population-based external validation of the EASIX scores to predict CAR T-cell-related toxicities. Cancers. 2023;15.
Korell F, Penack O, Mattie M, Schreck N, Benner A, Krzykalla J, et al. EASIX and severe endothelial complications after CD19-directed CAR-T cell therapy-a cohort study. Front Immunol. 2022;13:877477.
Acosta-Medina AA, Johnson IM, Bansal R, Hathcock M, Kenderian SJ, Durani U, et al. Pre-lymphodepletion & infusion endothelial activation and stress index as predictors of clinical outcomes in CAR-T therapy for B-cell lymphoma. Blood Cancer J. 2023;13:7.
Stella F, Pennisi M, Chiappella A, Casadei B, Bramanti S, Ljevar S, et al. Prospective validation of CAR-HEMATOTOX and a simplified version predict survival in patients with large B-cell lymphoma treated with anti-CD19 CAR T-cells: data from CART-SIE study. Transpl Cell Ther. 2025;31:240.e1–e9.
Vic S, Thibert JB, Bachy E, Cartron G, Gastinne T, Morschhauser F, et al. Transfusion needs after CAR T-cell therapy for large B-cell lymphoma: predictive factors and outcome (a DESCAR-T study). Blood Adv. 2024;8:1573–85.
Porter TJ, Lazarevic A, Ziggas JE, Fuchs E, Kim K, Byrnes H, et al. Hyperinflammatory syndrome resembling haemophagocytic lymphohistiocytosis following axicabtagene ciloleucel and brexucabtagene autoleucel. Br J Haematol. 2022;199:720–7.
Lichtenstein DA, Schischlik F, Shao L, Steinberg SM, Yates B, Wang HW, et al. Characterization of HLH-like manifestations as a CRS variant in patients receiving CD22 CAR T cells. Blood. 2021;138:2469–84.
Hines MR, Keenan C, Maron Alfaro G, Cheng C, Zhou Y, Sharma A, et al. Hemophagocytic lymphohistiocytosis-like toxicity (carHLH) after CD19-specific CAR T-cell therapy. Br J Haematol. 2021;194:701–7.
Rejeski K, Burchert A, Iacoboni G, Sesques P, Fransecky L, Bucklein V, et al. Safety and feasibility of stem cell boost as a salvage therapy for severe hematotoxicity after CD19 CAR T-cell therapy. Blood Adv. 2022;6:4719–25.
Gagelmann N, Wulf GG, Duell J, Glass B, van Heteren P, von Tresckow B, et al. Hematopoietic stem cell boost for persistent neutropenia after CAR T-cell therapy: a GLA/DRST study. Blood Adv. 2023;7:555–9.
Wesson W, Ahmed N, Rashid A, Tabak C, Logan E, Marchena-Burgos J, et al. Safety and efficacy of eltrombopag in patients with post-CAR T cytopenias. Eur J Haematol. 2024;112:538–46.
Drillet G, Lhomme F, De Guibert S, Manson G, Houot R. Prolonged thrombocytopenia after CAR T-cell therapy: the role of thrombopoietin receptor agonists. Blood Adv. 2023;7:537–40.
Corona M, Shouval R, Alarcon A, Flynn J, Devlin S, Batlevi C, et al. Management of prolonged cytopenia following CAR T-cell therapy. Bone Marrow Transplant. 2022;57:1839–41.
Alvarado LJ, Huntsman HD, Cheng H, Townsley DM, Winkler T, Feng X, et al. Eltrombopag maintains human hematopoietic stem and progenitor cells under inflammatory conditions mediated by IFN-gamma. Blood. 2019;133:2043–55.
Baden LR, Swaminathan S, Almyroudis NG, Angarone M, Baluch A, Barros N, et al. Prevention and treatment of cancer-related infections, version 3.2024, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Cancer Netw. 2024;22:617–44.
Los-Arcos I, Iacoboni G, Aguilar-Guisado M, Alsina-Manrique L, Diaz de Heredia C, Fortuny-Guasch C, et al. Recommendations for screening, monitoring, prevention, and prophylaxis of infections in adult and pediatric patients receiving CAR T-cell therapy: a position paper. Infection. 2021;49:215–31.
Acknowledgements
The reported research was supported in part by the National Institutes of Health/National Cancer Institute (NIH/NCI) Memorial Sloan Kettering Cancer Center Support Grant (P30 CA008748). Dr. Magdalena Corona’s work was supported by a grant from the Alfonso Martin Escudero Foundation, and Dr. Alejandro Luna’s work was supported by an FEHH/Gilead grant. Dr. Roni Shouval reports grant support from NIH-NCI K08-CA282987, Comedy vs. Cancer, and an award from the Long Island Sound Chapter, Swim Across America.
Author information
Authors and Affiliations
Contributions
M.C., P.B.D., and R.S. designed the study; M.C. and P.B.D. wrote the manuscript; M.C., K.R., R.S., A.L.A., collected and analyzed the data; S.B., J.F., and S.D. performed the statistical planning and analysis; R.J.L., M.S., G.L.S., M.L.P., A.B., L.F., J.L., G.S., J.P., S.A.G., MA.P., R.S., P.B.D. treated patients; all authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
P.B.D. has financial interest in Atara Biotherapeutics and has served on Advisory Board for Pierre Fabre Pharmaceuticals. Dr. Perales reports honoraria from Adicet, Allogene, Caribou Biosciences, Celgene, Bristol-Myers Squibb, Equilium, Exevir, ImmPACT Bio, Incyte, Kite/Gilead, Merck, Miltenyi Biotec, MorphoSys, Nektar Therapeutics, Novartis, Omeros, OrcaBio, Pierre Fabre, Sanofi, Syncopation, Takeda, VectivBio AG, and Vor Biopharma. He serves on DSMBs for Cidara Therapeutics and Sellas Life Sciences. He has ownership interests in Omeros and OrcaBio. He has received institutional research support for clinical trials from Allogene, Genmab, Incyte, Kite/Gilead, Miltenyi Biotec, Nektar Therapeutics, and Novartis. R.S. reports speaker honoraria from Sanofi, Incyte, and MSD. K.R. Kite/Gilead: Research Funding, Consultancy, Honoraria and travel support; Novartis: Honoraria; BMS/Celgene: Consultancy, Honoraria; Pierre-Fabre: travel support. CSL Behring: Consultancy. AB reports consultancy with BMS and AbbVie.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Corona, M., Brown, S., Rejeski, K. et al. Late hematologic toxicity after CAR T-cell therapy in large B-cell lymphoma: incidence, risk factors, and clinical impact. Bone Marrow Transplant 61, 142–148 (2026). https://doi.org/10.1038/s41409-025-02745-5
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-025-02745-5