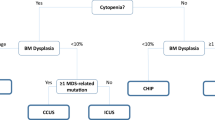
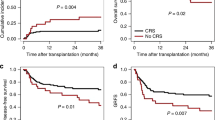
Abstract
Higher-risk myelodysplastic syndrome (HR-MDS) is a heterogeneous group of hematopoietic malignancies primarily affecting the elderly, characterized by ineffective hematopoiesis, cytopenias, and a risk of transformation to acute myeloblastic leukemia. This review outlines the current landscape of HR-MDS management, focusing on risk stratification, treatment options, and challenges. The International Prognostic Scoring Systems (IPSS-R and IPSS-M) classify patients into risk categories, integrating cytogenetics and molecular data to guide therapy. Hypomethylating agents remain the standard of care for non-transplant-eligible patients, though their efficacy varies, with median overall survival ranging from 13-19 months. Promising novel agents include anti-apoptotic drugs (e.g., venetoclax), mutation-targeted drugs (e.g., TP53, IDH1/2), signal transduction inhibitors, inflammation pathway inhibitors and immune checkpoint inhibitors. Combinations of hypomethylating agents and these novel agents have shown promise in early trials as initial or salvage therapy but have failed to improve survival in phase III studies. Allogeneic hematopoietic stem cell transplantation is the only potentially curative option, yet its applicability is limited by patient age, comorbidities, and donor availability. Post-transplant relapse monitoring via chimerism and measurable residual disease is critical, with preemptive donor lymphocyte infusion recommended for relapse prevention. Future research should focus on mutation-driven therapies and inclusive trial designs to optimize HR-MDS management.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Mina A, Komrokji R. How I treat higher-risk MDS. Blood. 2025;145:2002–11. https://doi.org/10.1182/blood.2024025271.
Shrestha A, Stewart E, Cress R. Are we undercounting MDS? J Registry Manag. 2021;48:168–73.
Khoury JD, Solary E, Abla O, Akkari Y, Alaggio R, Apperley JF, et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia. 2022;36:1703–19. https://doi.org/10.1038/s41375-022-01613-1.
Arber DA, Orazi A, Hasserjian RP, Borowitz MJ, Calvo KR, Kvasnicka HM, et al. International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: integrating morphologic, clinical, and genomic data. Blood. 2022;140:1200–28. https://doi.org/10.1182/blood.2022015850.
Greenberg PL, Tuechler H, Schanz J, Sanz G, Garcia-Manero G, Sole F, et al. Revised international prognostic scoring system for myelodysplastic syndromes. Blood. 2012;120:2454–65. https://doi.org/10.1182/blood-2012-03-420489.
Bernard E, Tuechler H, Greenberg PL, Hasserjian RP, Arango Ossa JE, Nannya Y, et al. Molecular International Prognostic Scoring System for Myelodysplastic Syndromes. NEJM Evid. 2022;1:EVIDoa2200008. https://doi.org/10.1056/EVIDoa2200008.
Kroger N. Treatment of high-risk myelodysplastic syndromes. Haematologica. 2025;110:339–49. https://doi.org/10.3324/haematol.2023.284946.
Pfeilstocker M, Tuechler H, Sanz G, Schanz J, Garcia-Manero G, Sole F, et al. Time-dependent changes in mortality and transformation risk in MDS. Blood. 2016;128:902–10. https://doi.org/10.1182/blood-2016-02-700054.
Baer C, Huber S, Hutter S, Meggendorfer M, Nadarajah N, Walter W, et al. Risk prediction in MDS: independent validation of the IPSS-M-ready for routine? Leukemia. 2023;37:938–41. https://doi.org/10.1038/s41375-023-01831-1.
Sanz GF. In MDS, is higher risk higher reward? Hematol Am Soc Hematol Educ Program. 2019;2019:381–90. https://doi.org/10.1182/hematology.2019000042.
Venugopal S, Sekeres MA. Drug development in higher-risk myelodysplastic syndromes. Blood Cancer J. 2024;14:187. https://doi.org/10.1038/s41408-024-01171-y.
Fenaux P, Mufti GJ, Hellstrom-Lindberg E, Santini V, Finelli C, Giagounidis A, et al. Efficacy of azacitidine compared with that of conventional care regimens in the treatment of higher-risk myelodysplastic syndromes: a randomised, open-label, phase III study. Lancet Oncol. 2009;10:223–32. https://doi.org/10.1016/S1470-2045(09)70003-8.
Bernal T, Martinez-Camblor P, Sanchez-Garcia J, de Paz R, Luno E, Nomdedeu B, et al. Effectiveness of azacitidine in unselected high-risk myelodysplastic syndromes: results from the Spanish registry. Leukemia. 2015;29:1875–81. https://doi.org/10.1038/leu.2015.115.
Garcia-Manero G, Montalban-Bravo G, Berdeja JG, Abaza Y, Jabbour E, Essell J, et al. Phase 2, randomized, double-blind study of pracinostat in combination with azacitidine in patients with untreated, higher-risk myelodysplastic syndromes. Cancer. 2017;123:994–1002. https://doi.org/10.1002/cncr.30533.
Garcia JS, Swords RT, Roboz GJ, Jacoby MA, Garcia-Manero G, Hong WJ, et al. A systematic review of higher-risk myelodysplastic syndromes clinical trials to determine the benchmark of azacitidine and explore alternative endpoints for overall survival. Leuk Res. 2021;104:106555. https://doi.org/10.1016/j.leukres.2021.106555.
Ades L, Girshova L, Doronin VA, Diez-Campelo M, Valcarcel D, Kambhampati S, et al. Pevonedistat plus azacitidine vs azacitidine alone in higher-risk MDS/chronic myelomonocytic leukemia or low-blast-percentage AML. Blood Adv. 2022;6:5132–45. https://doi.org/10.1182/bloodadvances.2022007334.
Sallman DA, Al Malki MM, Asch AS, Wang ES, Jurcic JG, Bradley TJ, et al. Magrolimab in combination with azacitidine in patients with higher-risk myelodysplastic syndromes: final results of a phase Ib study. J Clin Oncol. 2023;41:2815–26. https://doi.org/10.1200/JCO.22.01794.
Gilead Statement on Discontinuation of Phase 3 ENHANCE-3 Study in AML; https://www.gilead.com/company/company-statements/2024/gilead-statement-on-discontinuation-of-phase-3-enhance-3-study-in-aml.
Garcia JS, Platzbecker U, Odenike O, Fleming S, Fong CY, Borate U, et al. Efficacy and safety of venetoclax plus azacitidine for patients with treatment-naive high-risk myelodysplastic syndromes. Blood. 2025;145:1126–35. https://doi.org/10.1182/blood.2024025464.
Study Of Venetoclax Tablet With Intravenous or Subcutaneous Azacitidine to Assess Change in Disease Activity In Adult Participants With Newly Diagnosed Higher-Risk Myelodysplastic Syndrome (Verona). http://clinicaltrials.gov/study/NCT04401748. Accessed, 2025.
Sallman DA, DeZern AE, Garcia-Manero G, Steensma DP, Roboz GJ, Sekeres MA, et al. Eprenetapopt (APR-246) and azacitidine in TP53-mutant myelodysplastic syndromes. J Clin Oncol. 2021;39:1584–94. https://doi.org/10.1200/JCO.20.02341.
Cluzeau T, Sebert M, Rahme R, Cuzzubbo S, Lehmann-Che J, Madelaine I, et al. Eprenetapopt plus azacitidine in TP53-mutated myelodysplastic syndromes and acute myeloid leukemia: a phase II study by the groupe francophone des myelodysplasies (GFM). J Clin Oncol. 2021;39:1575–83. https://doi.org/10.1200/JCO.20.02342.
Aprea Therapeutics. Aprea Therapeutics announces results of primary endpoint from phase 3 trial of eprenetapopt in TP53 mutant myelodysplastic syndromes (MDS). https://ir.aprea.com/news-releases/news-release-details/aprea-therapeutics-announces-results-primary-endpoint-phase-3.
Sekeres MA, Watts J, Radinoff A, Sangerman MA, Cerrano M, Lopez PF, et al. Randomized phase 2 trial of pevonedistat plus azacitidine versus azacitidine for higher-risk MDS/CMML or low-blast AML. Leukemia. 2021;35:2119–24. https://doi.org/10.1038/s41375-021-01125-4.
Short NJ, Muftuoglu M, Ong F, Nasr L, Macaron W, Montalban-Bravo G, et al. A phase 1/2 study of azacitidine, venetoclax and pevonedistat in newly diagnosed secondary AML and in MDS or CMML after failure of hypomethylating agents. J Hematol Oncol. 2023;16:73 https://doi.org/10.1186/s13045-023-01476-8.
Zeidan AM, Xiao Z, Sanz G, Giagounidis A, Sekeres M, Lao Z et al. Primary results of the phase III stimulus-Mds2 study of sabatolimab + azacitidine vs placebo + azacitidine as frontline therapy for patients with higher-risk Mds Or Cmml-2. EHA Library. 2024:S180.
Zeidan AM, Ando K, Rauzy O, Turgut M, Wang MC, Cairoli R, et al. Sabatolimab plus hypomethylating agents in previously untreated patients with higher-risk myelodysplastic syndromes (STIMULUS-MDS1): a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Haematol. 2024;11:e38–50. https://doi.org/10.1016/S2352-3026(23)00333-2.
Garcia-Manero G. Current status of phase 3 clinical trials in high-risk myelodysplastic syndromes: pitfalls and recommendations. Lancet Haematol. 2023;10:e71–e78. https://doi.org/10.1016/S2352-3026(22)00265-4.
Brunner AM, Platzbecker U, DeZern AE, Zeidan AM. Are we ready for “triplet” therapy in higher-risk MDS? Clin Hematol Int. 2023;5:88301. https://doi.org/10.46989/001c.88301.
Santini V. How I treat MDS after hypomethylating agent failure. Blood. 2019;133:521–9. https://doi.org/10.1182/blood-2018-03-785915.
Prebet T, Gore SD, Esterni B, Gardin C, Itzykson R, Thepot S, et al. Outcome of high-risk myelodysplastic syndrome after azacitidine treatment failure. J Clin Oncol. 2011;29:3322–7. https://doi.org/10.1200/JCO.2011.35.8135.
Gurnari C, Xie Z, Zeidan AM. How I manage transplant ineligible patients with myelodysplastic neoplasms. Clin Hematol Int. 2023;5:8–20. https://doi.org/10.1007/s44228-022-00024-4.
Sebert M, Renneville A, Bally C, Peterlin P, Beyne-Rauzy O, Legros L, et al. A phase II study of guadecitabine in higher-risk myelodysplastic syndrome and low blast count acute myeloid leukemia after azacitidine failure. Haematologica. 2019;104:1565–71. https://doi.org/10.3324/haematol.2018.207118.
Garcia-Manero G, McCloskey J, Griffiths EA, Yee KWL, Zeidan AM, Al-Kali A, et al. Oral decitabine-cedazuridine versus intravenous decitabine for myelodysplastic syndromes and chronic myelomonocytic leukaemia (ASCERTAIN): a registrational, randomised, crossover, pharmacokinetics, phase 3 study. Lancet Haematol. 2024;11:e15–26. https://doi.org/10.1016/S2352-3026(23)00338-1.
Rodriguez-Sevilla JJ, Adema V, Garcia-Manero G, Colla S. Emerging treatments for myelodysplastic syndromes: Biological rationales and clinical translation. Cell Rep Med. 2023;4:100940. https://doi.org/10.1016/j.xcrm.2023.100940.
Desikan SP, Montalban-Bravo G, Ohanian M, Daver NG, Kadia TM, Venugopal S et al. Results of a phase 1 trial of azacitidine with venetoclax in relapsed/refractory higher-risk myelodysplastic syndrome (MDS). J Clin Oncol. 2022. https://doi.org/10.1200/JCO.2022.40.16_suppl.e19068.
Zeidan AM, Borate U, Pollyea DA, Brunner AM, Roncolato F, Garcia JS, et al. A phase 1b study of venetoclax and azacitidine combination in patients with relapsed or refractory myelodysplastic syndromes. Am J Hematol. 2023;98:272–81. https://doi.org/10.1002/ajh.26771.
Stein EM, DiNardo CD, Pollyea DA, Fathi AT, Roboz GJ, Altman JK, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017;130:722–31. https://doi.org/10.1182/blood-2017-04-779405.
DiNardo CD, Stein EM, de Botton S, Roboz GJ, Altman JK, Mims AS, et al. Durable remissions with ivosidenib in IDH1-mutated relapsed or refractory AML. N Engl J Med. 2018;378:2386–98. https://doi.org/10.1056/NEJMoa1716984.
Woods AC, Pulte ED, Wang X, Vallejo J, Chadda R, Zheng N, et al. FDA approval summary: ivosidenib for treatment of adult patients with relapsed/refractory myelodysplastic syndrome with an IDH1 mutation. Clin Cancer Res. 2025. https://doi.org/10.1158/1078-0432.CCR-25-1005.
Sebert M, Cluzeau T, Beyne Rauzy O, Stamatoulas Bastard A, Dimicoli-Salazar S, Thepot S, et al. Ivosidenib monotherapy is effective in patients with IDH1 mutated myelodysplastic syndrome (MDS): the idiome phase 2 study by the GFM group. Blood. 2021;138:62. https://doi.org/10.1182/blood-2021-146932.
Stein EM, Fathi AT, DiNardo CD, Pollyea DA, Roboz GJ, Collins R, et al. Enasidenib in patients with mutant IDH2 myelodysplastic syndromes: a phase 1 subgroup analysis of the multicentre, AG221-C-001 trial. Lancet Haematol. 2020;7:e309–19. https://doi.org/10.1016/S2352-3026(19)30284-4.
DiNardo CD, Venugopal S, Lachowiez C, Takahashi K, Loghavi S, Montalban-Bravo G, et al. Targeted therapy with the mutant IDH2 inhibitor enasidenib for high-risk IDH2-mutant myelodysplastic syndrome. Blood Adv. 2023;7:2378–87. https://doi.org/10.1182/bloodadvances.2022008378.
Sebert M, Chevret S, Dimicoli-Salazar S, Cluzeau T, Rauzy O, Stamatoulas Bastard A, et al. Enasidenib (ENA) Monotherapy in Patients with IDH2 mutated Myelodysplastic Syndrome (MDS), the ideal phase 2 study by the GFM and Emsco groups. Blood. 2024;144:1839. https://doi.org/10.1182/blood-2024-201317.
Cortiana V, Vallabhaneni H, Ghazal J, Itodo K, Kassim Dohadwala T, Park C, et al. Beyond hypomethylating agents: novel therapies and targeted approaches. Clin Hematol Int. 2025;7:24–35. https://doi.org/10.46989/001c.142956.
Moyo TK, Watts JM, Skikne BS, Mendler JH, Klimek VM, Chen S-C, et al. Preliminary results from a phase II study of the combination of pevonedistat and azacitidine in the treatment of MDS and MDS/MPN after failure of DNA methyltransferase inhibition. Blood. 2019;134:4236. https://doi.org/10.1182/blood-2019-130003.
Garcia-Manero G, Winer ES, DeAngelo DJ, Tarantolo SR, Sallman DA, Dugan J, et al. Phase 1/2a study of the IRAK4 inhibitor CA-4948 as monotherapy or in combination with azacitidine or venetoclax in patients with relapsed/refractory (R/R) acute myeloid leukemia or lyelodysplastic syndrome. J Clin Oncol. 2022;40:7016. https://doi.org/10.1200/JCO.2022.40.16_suppl.7016.
Garcia-Manero G, Fenaux P, Al-Kali A, Baer MR, Sekeres MA, Roboz GJ, et al. Rigosertib versus best supportive care for patients with high-risk myelodysplastic syndromes after failure of hypomethylating drugs (ONTIME): a randomised, controlled, phase 3 trial. Lancet Oncol. 2016;17:496–508. https://doi.org/10.1016/S1470-2045(16)00009-7.
Garcia-Manero G, Ribrag V, Zhang Y, Farooqui M, Marinello P, Smith BD. Pembrolizumab for myelodysplastic syndromes after failure of hypomethylating agents in the phase 1b KEYNOTE-013 study. Leuk Lymphoma. 2022;63:1660–8. https://doi.org/10.1080/10428194.2022.2034155.
Garcia-Manero G, Sasaki K, Montalban-Bravo G, Daver NG, Jabbour EJ, Alvarado Y, et al. A phase II study of nivolumab or ipilimumab with or without azacitidine for patients with myelodysplastic syndrome (MDS). Blood. 2018;132:465. https://doi.org/10.1182/blood-2018-99-119424.
Gerds AT, Scott BL, Greenberg P, Lin TL, Pollyea DA, Verma A, et al. Atezolizumab alone or in combination did not demonstrate a favorable risk-benefit profile in myelodysplastic syndrome. Blood Adv. 2022;6:1152–61. https://doi.org/10.1182/bloodadvances.2021005240.
Patnaik MM, Tefferi A. Luspatercept use for lower risk myelodysplastic syndromes: active but not enough. Am J Hematol. 2023;98:1171–5. https://doi.org/10.1002/ajh.27003.
Bejar R, Lord A, Stevenson K, Bar-Natan M, Perez-Ladaga A, Zaneveld J, et al. TET2 mutations predict response to hypomethylating agents in myelodysplastic syndrome patients. Blood. 2014;124:2705–12. https://doi.org/10.1182/blood-2014-06-582809.
Traina F, Visconte V, Elson P, Tabarroki A, Jankowska AM, Hasrouni E, et al. Impact of molecular mutations on treatment response to DNMT inhibitors in myelodysplasia and related neoplasms. Leukemia. 2014;28:78–87. https://doi.org/10.1038/leu.2013.269.
Seiler M, Yoshimi A, Darman R, Chan B, Keaney G, Thomas M, et al. H3B-8800, an orally available small-molecule splicing modulator, induces lethality in spliceosome-mutant cancers. Nat Med. 2018;24:497–504. https://doi.org/10.1038/nm.4493.
Steensma DP, Wermke M, Klimek VM, Greenberg PL, Font P, Komrokji RS, et al. Phase I first-in-human dose escalation study of the oral SF3B1 modulator H3B-8800 in myeloid neoplasms. Leukemia. 2021;35:3542–50. https://doi.org/10.1038/s41375-021-01328-9.
Zampini M, Riva E, Lanino L, Sauta E, Antunes Dos Reis R, Ejarque RMA, et al. Characterization and clinical implications of p53 dysfunction in patients with myelodysplastic syndromes. J Clin Oncol. 2025;43:2069–83. https://doi.org/10.1200/JCO-24-02394.
Schimmer RR, Kovtonyuk LV, Klemm N, Fullin J, Stolz SM, Mueller J, et al. TP53 mutations confer resistance to hypomethylating agents and BCL-2 inhibition in myeloid neoplasms. Blood Adv. 2022;6:3201–6. https://doi.org/10.1182/bloodadvances.2021005859.
Jabbour E, Short NJ, Montalban-Bravo G, Huang X, Bueso-Ramos C, Qiao W, et al. Randomized phase 2 study of low-dose decitabine vs low-dose azacitidine in lower-risk MDS and MDS/MPN. Blood. 2017;130:1514–22. https://doi.org/10.1182/blood-2017-06-788497.
Sasaki K, Jabbour E, Montalban-Bravo G, Darbaniyan F, Do K-A, Class C, et al. Low-dose decitabine versus low-dose azacitidine in lower-risk MDS. NEJM Evid. 2022;1. https://doi.org/10.1056/EVIDoa2200034.
Platzbecker U, Schetelig J, Finke J, Trenschel R, Scott BL, Kobbe G, et al. Allogeneic hematopoietic cell transplantation in patients age 60-70 years with de novo high-risk myelodysplastic syndrome or secondary acute myelogenous leukemia: comparison with patients lacking donors who received azacitidine. Biol Blood Marrow Transpl. 2012;18:1415–21. https://doi.org/10.1016/j.bbmt.2012.05.003.
Robin M, Porcher R, Ades L, Raffoux E, Michallet M, Francois S, et al. HLA-matched allogeneic stem cell transplantation improves outcome of higher risk myelodysplastic syndrome A prospective study on behalf of SFGM-TC and GFM. Leukemia. 2015;29:1496–1501. https://doi.org/10.1038/leu.2015.37.
Nakamura R, Saber W, Martens MJ, Ramirez A, Scott B, Oran B, et al. Biologic assignment trial of reduced-intensity hematopoietic cell transplantation based on donor availability in patients 50-75 years of age with advanced myelodysplastic syndrome. J Clin Oncol. 2021;39:3328–39. https://doi.org/10.1200/JCO.20.03380.
Kroger N, Sockel K, Wolschke C, Bethge W, Schlenk RF, Wolf D, et al. Comparison between 5-azacytidine treatment and allogeneic stem-cell transplantation in elderly patients with advanced MDS according to donor availability (VidazaAllo Study). J Clin Oncol. 2021;39:3318–27. https://doi.org/10.1200/JCO.20.02724.
Gagelmann N, Eikema DJ, Stelljes M, Beelen D, de Wreede L, Mufti G, et al. Optimized EBMT transplant-specific risk score in myelodysplastic syndromes after allogeneic stem-cell transplantation. Haematologica. 2019;104:929–36. https://doi.org/10.3324/haematol.2018.200808.
Cutler CS, Lee SJ, Greenberg P, Deeg HJ, Perez WS, Anasetti C, et al. A decision analysis of allogeneic bone marrow transplantation for the myelodysplastic syndromes: delayed transplantation for low-risk myelodysplasia is associated with improved outcome. Blood. 2004;104:579–85. https://doi.org/10.1182/blood-2004-01-0338.
Tentori CA, Gregorio C, Robin M, Gagelmann N, Gurnari C, Ball S, et al. Clinical and genomic-based decision support system to define the optimal timing of allogeneic hematopoietic stem-cell transplantation in patients with myelodysplastic syndromes. J Clin Oncol. 2024;42:2873–86. https://doi.org/10.1200/JCO.23.02175.
Greenberg PL, Stone RM, Abaza Y, Al-Kali A, Anand S, Ball B, et al. NCCN Guidelines(R) Insights: Myelodysplastic Syndromes, Version 2.2025. J Natl Compr Canc Netw 2025;23:66–75.
Scheid C, Eikema DJ, van Gelder M, Salmenniemi U, Maertens J, Passweg J, et al. Does IPSS-R downstaging before transplantation improve the prognosis of patients with myelodysplastic neoplasms? Blood. 2024;144:445–56. https://doi.org/10.1182/blood.2023022273.
Grunwald MR, Zhang MJ, Elmariah H, Johnson MH, St Martin A, Bashey A, et al. Alternative donor transplantation for myelodysplastic syndromes: haploidentical relative and matched unrelated donors. Blood Adv. 2021;5:975–83. https://doi.org/10.1182/bloodadvances.2020003654.
Nakaya Y, Koh H, Konuma T, Shimomura Y, Ishiyama K, Itonaga H, et al. HLA-haploidentical peripheral blood stem cell transplantation with post-transplantation cyclophosphamide versus HLA-matched unrelated donor transplantation for myelodysplastic syndrome. Transpl Cell Ther. 2024;30:316 e311–316 e312. https://doi.org/10.1016/j.jtct.2023.10.021.
Beelen DW, Trenschel R, Stelljes M, Groth C, Masszi T, Remenyi P, et al. Treosulfan or busulfan plus fludarabine as conditioning treatment before allogeneic haemopoietic stem cell transplantation for older patients with acute myeloid leukaemia or myelodysplastic syndrome (MC-FludT.14/L): a randomised, non-inferiority, phase 3 trial. Lancet Haematol. 2020;7:e28–39. https://doi.org/10.1016/S2352-3026(19)30157-7.
Oran B, Ahn KW, Fretham C, Beitinjaneh A, Bashey A, Pawarode A, et al. Fludarabine and melphalan compared with reduced doses of busulfan and fludarabine improve transplantation outcomes in older patients with myelodysplastic syndromes. Transpl Cell Ther. 2021;27:921 e921–921 e910. https://doi.org/10.1016/j.jtct.2021.08.007.
Hou C, Zhou L, Yang M, Jiang S, Shen H, Zhu M, et al. The prognostic value of early detection of minimal residual disease as defined by flow cytometry and gene mutation clearance for myelodysplastic syndrome patients after myeloablative allogeneic hematopoietic stem-cell transplantation. Front Oncol. 2021;11:700234. https://doi.org/10.3389/fonc.2021.700234.
Tobiasson M, Pandzic T, Illman J, Nilsson L, Westrom S, Ejerblad E, et al. Patient-specific measurable residual disease markers predict outcome in patients with myelodysplastic syndrome and related diseases after hematopoietic stem-cell transplantation. J Clin Oncol. 2024;42:1378–90. https://doi.org/10.1200/JCO.23.01159.
Chen W, Huang J, Zhao Y, Huang L, Yuan Z, Gu M, et al. Measurable residual disease monitoring by ddPCR in the early posttransplant period complements the traditional MFC method to predict relapse after HSCT in AML/MDS: a multicenter retrospective study. J Transl Med. 2024;22:410. https://doi.org/10.1186/s12967-024-05114-w.
Loke J, McCarthy N, Jackson A, Siddique S, Hodgkinson A, Mason J, et al. Posttransplant MRD and T-cell chimerism status predict outcomes in patients who received allografts for AML/MDS. Blood Adv. 2023;7:3666–76. https://doi.org/10.1182/bloodadvances.2022009493.
Guieze R, Damaj G, Pereira B, Robin M, Chevallier P, Michallet M, et al. Management of myelodysplastic syndrome relapsing after allogeneic hematopoietic stem cell transplantation: a study by the French society of bone marrow transplantation and cell therapies. Biol Blood Marrow Transpl. 2016;22:240–7. https://doi.org/10.1016/j.bbmt.2015.07.037.
Guisnel C, Schirmer L, Morisset S, Robin M, Labussiere-Wallet H, Dulery R, et al. On behalf of the SFGM-TC: prophylactic donor lymphocyte infusion in patients treated with allogeneic stem-cell transplantation for high-risk myelodysplastic syndrome and acute myeloid leukemia. Acta Haematol. 2023;146:230–9. https://doi.org/10.1159/000528184.
Rujkijyanont P, Morris C, Kang G, Gan K, Hartford C, Triplett B, et al. Risk-adapted donor lymphocyte infusion based on chimerism and donor source in pediatric leukemia. Blood Cancer J. 2013;3:e137. https://doi.org/10.1038/bcj.2013.39.
Kungwankiattichai S, Ponvilawan B, Roy C, Tunsing P, Kuchenbauer F, Owattanapanich W. Maintenance with hypomethylating agents after allogeneic stem cell transplantation in acute myeloid leukemia and myelodysplastic syndrome: a systematic review and meta-analysis. Front Med (Lausanne). 2022;9:801632. https://doi.org/10.3389/fmed.2022.801632.
Wattebled KJ, Drumez E, Coiteux V, Magro L, Srour M, Chauvet P, et al. Single-agent 5-azacytidine as post-transplant maintenance in high-risk myeloid malignancies undergoing allogeneic hematopoietic cell transplantation. Ann Hematol. 2022;101:1321–31. https://doi.org/10.1007/s00277-022-04821-y.
Wei Y, Xiong X, Li X, Lu W, He X, Jin X, et al. Low-dose decitabine plus venetoclax is safe and effective as post-transplant maintenance therapy for high-risk acute myeloid leukemia and myelodysplastic syndrome. Cancer Sci. 2021;112:3636–44.
Mishra A, Tamari R, DeZern AE, Byrne MT, Gooptu M, Chen YB, et al. Eprenetapopt plus azacitidine after allogeneic hematopoietic stem-cell transplantation for TP53-mutant acute myeloid leukemia and myelodysplastic syndromes. J Clin Oncol. 2022;40:3985–993. https://doi.org/10.1200/JCO.22.00181.
Acknowledgements
This work was supported by the National Key Research and Development Program of China (Key Program for International S&T Cooperation Projects of China) (2025YFE0151000) and the Fundamental Research Funds for the Zhejiang Provincial Universities (226-2025-00213).
Author information
Authors and Affiliations
Contributions
YY and MM conceptualized the work and drafted the initial manuscript. MM provided critical revision of the manuscript. EB and AB co-drafted sections of the manuscript, and provided key expert opinions and critical feedback.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ye, Y., Brissot, E., Bazarbachi, A. et al. Therapy of high-risk myelodysplastic syndromes. Bone Marrow Transplant (2026). https://doi.org/10.1038/s41409-026-02821-4
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41409-026-02821-4