Abstract
Background
Fusobacterium nucleatum inhabits the oral cavity and affects the progression of gastrointestinal cancer. Our prior findings link F. nucleatum to poor prognosis in oesophageal squamous cell carcinoma via NF-κB pathway. However, its role in oesophagogastric junction and gastric adenocarcinoma remains unexplored. We investigated whether F. nucleatum influences these cancers, highlighting its potential impact.
Methods
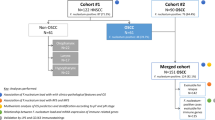
Two cohorts of EGJ and gastric adenocarcinoma patients (438 from Japan, 380 from the USA) were studied. F. nucleatum presence was confirmed by qPCR, FISH, and staining. Patient overall survival (OS) was assessed based on F. nucleatum positivity. EGJ and gastric adenocarcinoma cell lines were exposed to F. nucleatum to study molecular and phenotypic effects, validated in xenograft mouse model.
Results
In both cohorts, F. nucleatum-positive EGJ or gastric adenocarcinoma patients had notably shorter OS. F. nucleatum positivity decreased in more acidic tumour environments. Cancer cell lines with F. nucleatum showed enhanced proliferation and NF-κB activation. The xenograft model indicated increased tumour growth and NF-κB activation in F. nucleatum-treated cells. Interestingly, co-occurrence of F. nucleatum and Helicobacter pylori, a known risk factor, was rare.
Conclusions
F. nucleatum can induce the NF-κB pathway in EGJ and gastric adenocarcinomas, leading to tumour progression and poor prognosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ferlay J, Ervik M, Lam F, Colombet M, Mery L, Piñeros M, et al. Global Cancer Observatory: Cancer Today. [(accessed on 1 Feb 2023)]. Available online: https://gco.iarc.fr/today
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209–49.
Spechler SJ, Souza RF. Barrett’s esophagus. N. Engl J Med. 2014;371:836–45.
Quante M, Graham TA, Jansen M. Insights Into the Pathophysiology of Esophageal Adenocarcinoma. Gastroenterology. 2018;154:406–20.
Doorakkers E, Lagergren J, Santoni G, Engstrand L, Brusselaers N. Helicobacter pylori eradication treatment and the risk of Barrett’s esophagus and esophageal adenocarcinoma. Helicobacter. 2020;25:e12688.
Coleman HG, Xie SH, Lagergren J. The Epidemiology of Esophageal Adenocarcinoma. Gastroenterology. 2018;154:390–405.
Reynolds JV, Preston SR, O’Neill B, Lowery MA, Baeksgaard L, Crosby T, et al. Trimodality therapy versus perioperative chemotherapy in the management of locally advanced adenocarcinoma of the oesophagus and oesophagogastric junction (Neo-AEGIS): an open-label, randomised, phase 3 trial. Lancet Gastroenterol Hepatol. 2023;8:1015–27.
Imamura Y, Watanabe M, Oki E, Morita M, Baba H. Esophagogastric junction adenocarcinoma shares characteristics with gastric adenocarcinoma: Literature review and retrospective multicenter cohort study. Ann Gastroenterol Surg. 2021;5:46–59.
Kato K, Doki Y, Ogata T, Motoyama S, Kawakami H, Ueno M, et al. First-line nivolumab plus ipilimumab or chemotherapy versus chemotherapy alone in advanced esophageal squamous cell carcinoma: a Japanese subgroup analysis of open-label, phase 3 trial (CheckMate 648/ONO-4538-50). Esophagus. 2023;20:291–301.
Janjigian YY, Shitara K, Moehler M, Garrido M, Salman P, Shen L, et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet. 2021;398:27–40.
Integrative HMP (iHMP) Research Network Consortium. The Integrative Human Microbiome Project. Nature. 2019;569:641–8.
Galeano Nino JL, Wu H, LaCourse KD, Kempchinsky AG, Baryiames A, Barber B, et al. Effect of the intratumoral microbiota on spatial and cellular heterogeneity in cancer. Nature. 2022;611:810–7.
Fan Y, Pedersen O. Gut microbiota in human metabolic health and disease. Nat Rev Microbiol. 2021;19:55–71.
Sepich-Poore GD, Zitvogel L, Straussman R, Hasty J, Wargo JA, Knight R. The microbiome and human cancer. Science. 2021;371:eabc4552.
Holt RA, Cochrane K. Tumor Potentiating Mechanisms of Fusobacterium nucleatum, A Multifaceted Microbe. Gastroenterology. 2017;152:694–6.
Brennan CA, Garrett WS. Fusobacterium nucleatum - symbiont, opportunist and oncobacterium. Nat Rev Microbiol. 2019;17:156–66.
Ugai T, Shimizu T, Kawamura H, Ugai S, Takashima Y, Usui G, et al. Inverse relationship between Fusobacterium nucleatum amount and tumor CD274 (PD-L1) expression in colorectal carcinoma. Clin Transl Immunol. 2023;12:e1453.
Mima K, Sukawa Y, Nishihara R, Qian ZR, Yamauchi M, Inamura K, et al. Fusobacterium nucleatum and T Cells in Colorectal Carcinoma. JAMA Oncol. 2015;1:653–61.
Mima K, Cao Y, Chan AT, Qian ZR, Nowak JA, Masugi Y, et al. Fusobacterium nucleatum in Colorectal Carcinoma Tissue According to Tumor Location. Clin Transl Gastroenterol. 2016;7:e200.
Mima K, Nishihara R, Qian ZR, Cao Y, Sukawa Y, Nowak JA, et al. Fusobacterium nucleatum in colorectal carcinoma tissue and patient prognosis. Gut. 2016;65:1973–80.
Wang N, Fang JY. Fusobacterium nucleatum, a key pathogenic factor and microbial biomarker for colorectal cancer. Trends Microbiol. 2023;31:159–72.
Yang Y, Weng W, Peng J, Hong L, Yang L, Toiyama Y, et al. Fusobacterium nucleatum Increases Proliferation of Colorectal Cancer Cells and Tumor Development in Mice by Activating Toll-Like Receptor 4 Signaling to Nuclear Factor-kappaB, and Up-regulating Expression of MicroRNA-21. Gastroenterology. 2017;152:851–66.e824.
Zhang Y, Zhang L, Zheng S, Li M, Xu C, Jia D, et al. Fusobacterium nucleatum promotes colorectal cancer cells adhesion to endothelial cells and facilitates extravasation and metastasis by inducing ALPK1/NF-kappaB/ICAM1 axis. Gut Microbes. 2022;14:2038852.
Castellarin M, Warren RL, Freeman JD, Dreolini L, Krzywinski M, Strauss J, et al. Fusobacterium nucleatum infection is prevalent in human colorectal carcinoma. Genome Res. 2012;22:299–306.
Kostic AD, Gevers D, Pedamallu CS, Michaud M, Duke F, Earl AM, et al. Genomic analysis identifies association of Fusobacterium with colorectal carcinoma. Genome Res. 2012;22:292–8.
Lee DW, Han SW, Kang JK, Bae JM, Kim HP, Won JK, et al. Association Between Fusobacterium nucleatum, Pathway Mutation, and Patient Prognosis in Colorectal Cancer. Ann Surg Oncol. 2018;25:3389–95.
Lee JB, Kim KA, Cho HY, Kim D, Kim WK, Yong D, et al. Association between Fusobacterium nucleatum and patient prognosis in metastatic colon cancer. Sci Rep. 2021;11:20263.
Yamamura K, Baba Y, Nakagawa S, Mima K, Miyake K, Nakamura K, et al. Human Microbiome Fusobacterium Nucleatum in Esophageal Cancer Tissue Is Associated with Prognosis. Clin Cancer Res. 2016;22:5574–81.
Yamamura K, Baba Y, Miyake K, Nakamura K, Shigaki H, Mima K, et al. Fusobacterium nucleatum in gastroenterological cancer: Evaluation of measurement methods using quantitative polymerase chain reaction and a literature review. Oncol Lett. 2017;14:6373–8.
Nomoto D, Baba Y, Liu Y, Tsutsuki H, Okadome K, Harada K, et al. Fusobacterium nucleatum promotes esophageal squamous cell carcinoma progression via the NOD1/RIPK2/NF-kappaB pathway. Cancer Lett. 2022;530:59–67.
Di Pilato V, Freschi G, Ringressi MN, Pallecchi L, Rossolini GM, Bechi P. The esophageal microbiota in health and disease. Ann N. Y Acad Sci. 2016;1381:21–33.
Guo Y, Cao XS, Zhou MG, Yu B. Gastric microbiota in gastric cancer: Different roles of Helicobacter pylori and other microbes. Front Cell Infect Microbiol. 2022;12:1105811.
Guccione C, Yadlapati R, Shah S, Knight R, Curtius K. Challenges in Determining the Role of Microbiome Evolution in Barrett’s Esophagus and Progression to Esophageal Adenocarcinoma. Microorganisms. 2021;9:2003.
Brusilovsky M, Bao R, Rochman M, Kemter AM, Nagler CR, Rothenberg ME. Host-Microbiota Interactions in the Esophagus During Homeostasis and Allergic Inflammation. Gastroenterology. 2022;162:521–34.e528.
Yang L, Lu X, Nossa CW, Francois F, Peek RM, Pei Z. Inflammation and intestinal metaplasia of the distal esophagus are associated with alterations in the microbiome. Gastroenterology. 2009;137:588–97.
Muszynski D, Kudra A, Sobocki BK, Folwarski M, Vitale E, Filetti V, et al. Esophageal cancer and bacterial part of gut microbiota - A multidisciplinary point of view. Front Cell Infect Microbiol. 2022;12:1057668.
Shao D, Vogtmann E, Liu A, Qin J, Chen W, Abnet CC, et al. Microbial characterization of esophageal squamous cell carcinoma and gastric cardia adenocarcinoma from a high-risk region of China. Cancer. 2019;125:3993–4002.
Liou JM, Chen CC, Chang CM, Fang YJ, Bair MJ, Chen PY, et al. Long-term changes of gut microbiota, antibiotic resistance, and metabolic parameters after Helicobacter pylori eradication: a multicentre, open-label, randomised trial. Lancet Infect Dis. 2019;19:1109–20.
Freedberg DE, Kim LS, Yang YX. The Risks and Benefits of Long-term Use of Proton Pump Inhibitors: Expert Review and Best Practice Advice From the American Gastroenterological Association. Gastroenterology. 2017;152:706–15.
Amieva M, Peek RM Jr. Pathobiology of Helicobacter pylori-Induced Gastric Cancer. Gastroenterology. 2016;150:64–78.
Schulz C, Schutte K, Koch N, Vilchez-Vargas R, Wos-Oxley ML, Oxley APA, et al. The active bacterial assemblages of the upper GI tract in individuals with and without Helicobacter infection. Gut. 2018;67:216–25.
Boehm ET, Thon C, Kupcinskas J, Steponaitiene R, Skieceviciene J, Canbay A, et al. Fusobacterium nucleatum is associated with worse prognosis in Lauren’s diffuse type gastric cancer patients. Sci Rep. 2020;10:16240.
Lagergren J, Lagergren P. Recent developments in esophageal adenocarcinoma. CA Cancer J Clin. 2013;63:232–48.
Cossentino MJ, Wong RK. Barrett’s esophagus and risk of esophageal adenocarcinoma. Semin Gastrointest Dis. 2003;14:128–35.
Pohl H, Wrobel K, Bojarski C, Voderholzer W, Sonnenberg A, Rosch T, et al. Risk factors in the development of esophageal adenocarcinoma. Am J Gastroenterol. 2013;108:200–7.
Gao P, Cai N, Yang X, Yuan Z, Zhang T, Lu M, et al. Association of Helicobacter pylori and gastric atrophy with adenocarcinoma of the esophagogastric junction in Taixing, China. Int J Cancer. 2022;150:243–52.
Johnson DA, Fennerty MB. Heartburn severity underestimates erosive esophagitis severity in elderly patients with gastroesophageal reflux disease. Gastroenterology. 2004;126:660–4.
Rubinstein MR, Wang X, Liu W, Hao Y, Cai G, Han YW. Fusobacterium nucleatum promotes colorectal carcinogenesis by modulating E-cadherin/beta-catenin signaling via its FadA adhesin. Cell Host Microbe. 2013;14:195–206.
Wu B, Crampton SP, Hughes CC. Wnt signaling induces matrix metalloproteinase expression and regulates T cell transmigration. Immunity. 2007;26:227–39.
Shi S, Liu Y, Wang Z, Jin X, Yan W, Guo X, et al. Fusobacterium nucleatum induces colon anastomosis leak by activating epithelial cells to express MMP9. Front Microbiol. 2022;13:1031882.
Acknowledgements
We thank J. Iacona, Ph.D., from Edanz (https://jp.edanz.com/ac) for editing this manuscript.
Funding
This work was supported in part by Grants-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (grant numbers 17H04273, 17K19702, and 17KK0195 to Y.B.).
Author information
Authors and Affiliations
Contributions
YH conducted experiments and contributed to manuscript writing. YB provided guidance and supervision throughout the project. EO, KH, KY, TT, KK, MI, YM, and QG, REW were involved in data collection and analysis. HT and TS provided guidance on microbial experimental techniques. YK assisted in the confirmation of pathological specimens. JAA and HB supervised the project. All authors contributed to the conception, design, and interpretation of the study, and critically reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Informed consent was obtained from all the participants. This study was conducted in accordance with the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board of Kumamoto University (#1365).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hara, Y., Baba, Y., Oda, E. et al. Presence of Fusobacterium nucleatum in relation to patient survival and an acidic environment in oesophagogastric junction and gastric cancers. Br J Cancer 131, 797–807 (2024). https://doi.org/10.1038/s41416-024-02753-0
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-024-02753-0
This article is cited by
-
Fusobacterium nucleatum: ecology, pathogenesis and clinical implications
Nature Reviews Microbiology (2026)
-
Fusobacterium nucleatum drives gastric cancer metastasis via Gbp-CypA-NF-κB-mediated CXCL8 crosstalk between tumor cells and mast cells
Cell Communication and Signaling (2025)
-
Oral microbes and gastrointestinal cancers: new strategies and insights
Clinical and Translational Oncology (2025)