Abstract
Background
To determine if an alternating regimen of the tyrosine kinase inhibitors imatinib and regorafenib improved outcomes in patients with advanced gastrointestinal stromal tumors.
Methods
ALTGIST (NCT02365441) was a randomized phase II study of standard treatment of imatinib (Arm A) compared with an experimental alternating regimen of imatinib and regorafenib (Arm B). Primary outcome was best objective tumor response (OTR) at nine months.
Results
Seventy-six eligible patients (Arm A 36, Arm B 40) enrolled were evaluable. Median follow-up was 46.0 months (range 6.5–64.6). Best responses and OTR were similar at 9 months. Eighteen (50.0%) Arm A patients and twelve (30.0%) Arm B patients discontinued treatment due to progressive disease. No Arm A patients stopped protocol therapy due to unacceptable toxicity, with 12 (30.0%) stopping in Arm B. Twelve (33.2%) Arm A patients and 12 (30.0%) Arm B patients experienced at least one serious adverse event, mostly grade 3. Secondary endpoints of PFS at 1 and OS at 1 year were not statistically different.
Conclusions
Alternation of imatinib and regorafenib did not impact on 9 months objective response nor on the secondary objectives of PFS and OS. Patients in the alternating arm experienced more toxicity and protocol discontinuations.
Clinical trial registration
NCT02365441.
Similar content being viewed by others
Background
While modern treatment of metastatic gastrointestinal stromal tumors (GIST) with imatinib (Glivec, Gleevec) is highly active, with clinical benefit rates of over 80%, most patients will ultimately progress. Two large pivotal randomized trials of 400 mg versus 800 mg of imatinib conducted in Europe/Australasia and North America respectively [1, 2], demonstrated that the median progression-free survival (PFS) was approximately two years. However, more recent data suggests that the median PFS is longer than this in patient populations with a smaller tumor burden at the time of imatinib initiation [3]. The overall median survival of patients is currently at least five years [4]. The risk of progression and death can be estimated by a prognostic nomogram that takes into account factors such as tumor genotype, primary mitotic count, size of metastasis, hemoglobin and neutrophil count at start of treatment [5].
Tumors may express varying mechanisms of resistance to imatinib [6] as well as second-generation tyrosine kinase inhibitors (TKI), due either to the acquisition of secondary mutations in KIT [7] or potentially the selection of pre-existing drug-resistant clones [8]. The emergence of resistance to imatinib occurs despite initial response rates of well over 80% in the more sensitive tumors carrying mutations in exon 11 of KIT. Imatinib treatment interruption and resumption at progression was not found to impact on resistance to imatinib in a randomized trial testing interruption at 1, 3, and 5 years [9].
Given the exquisite sensitivity of GIST cells to imatinib in vitro [10], and the dramatic clinical effect of imatinib administered to people in the first-line setting [11], particularly in tumors carrying mutations in KIT exon 11 (representing approximately 65% of all patients), it is unclear why complete responses are comparatively rare and the cure rate so low, with the vast majority of patients ultimately dying with advanced disease. Primary resistance to imatinib which is relatively uncommon may be due to the patients having KIT exon 9, PDGFR D842V mutations or no KIT/PDGFR mutations. The majority of GIST tumors progressing on imatinib therapy demonstrate the development of secondary KIT or PDGFR mutations which are resistant to the tyrosine kinase inhibitor. These commonly involve either the kinase activation loop (exons 17 and 18) or the ATP-binding pocket (exons 13 and 14) conferring imatinib resistance [12].
We hypothesized that by eliminating the selective pressure of continued pharmacologic therapy — which encourages the proliferation of resistant clones — by temporarily discontinuing treatment (a “washout” period), and using a second TKI such as regorafenib with established activity in GIST related in part to additional mechanisms of action beyond inhibition of KIT,13, response rates and survival times may be improved. Tumor cells harboring secondary mutations that expand predominantly because of this selective pressure might also regress during a brief period free of TKI exposure and become more susceptible to therapy.
Given the above rationale, it was hypothesized that with the addition of regorafenib (Stivarga), a second targeted agent with very substantial activity in the third-line setting in GIST [13], residual tumor cells may be eradicated and some partial responders might be converted into complete responders after a brief drug-free period. Regorafenib [14] was chosen as it represents a TKI with inhibitory activity against VEGFR1-3, FGFR, PDGFRA, Tie 2, BRAF, RET, RAF and p38 MAPK signaling pathways in addition to KIT. Secondary KIT mutations of exon 17 [15] and 18 [16] may also be sensitive to regorafenib. In a randomized, placebo controlled phase III trial in 199 patients who had failed at least two previous lines of therapy for GIST, there was a statistically significant reduction in the risk of progression in the regorafenib arm compared to the placebo arm (median PFS 4.8 vs. 0.9 months, HR 0.27) [17].
These strategies for improving response to therapy (treatment-free periods and addition of a second active agent) were incorporated in the design of this pragmatic trial, which was originally aimed to increase the OTR and PFS rates and ultimately increase the overall survival (OS) rate for advanced GIST.
Methods
Study design and participants
This phase II, randomized 1:1, open-label trial was conducted across 37 centers in 11 countries (Australia, Finland, France, Italy, Netherlands, Norway, Singapore, Slovakia, Spain, Sweden, United Kingdom). The trial sponsor was the Australasian Gastro-Intestinal Trials Group (AGITG) with collaborating groups being the AGITG, the European Organisation for Research and Treatment of Cancer (EORTC) and the Scandinavian Sarcoma Group (SSG). The study was coordinated by the National Health and Medical Research Council (NHMRC) Clinical Trials Centre, University of Sydney, Australia. See Supplemental Appendix for details of participating sites, investigators and contributing personnel. Patients aged 18 years or older, with a confirmed diagnosis of CD-117 positive, unresectable, metastatic GIST were eligible for this trial. Patients with CD-117 negative disease had to be DOG-1 positive or KIT/PDGFRA mutated. Patients with Eastern Cooperative Oncology Group performance status of 0 to 2 and adequate organ function were included. Patients were required to have measurable disease by Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. Patients must have had no prior history of taking a TKI for metastatic disease, with the exception of those patients who had up to and including 21 days of uninterrupted treatment on 400 mg daily of imatinib; imatinib therapy given as an adjuvant treatment and completed at least three months prior to entry into this trial was permitted. Patients who had progression of GIST while on adjuvant therapy were not eligible for this trial.
Key exclusion criteria included poorly controlled hypertension or persistent proteinuria of >3.5 g/24 h; inability to swallow or malabsorption; an arterial or venous thrombotic event within six months prior to randomization; known central nervous system metastases; active hepatitis B or C or HIV infection, and presence of known PDGFRA D842V mutation or other mutation known to cause imatinib resistance.
The trial was registered at ClinicalTrials.gov as NCT02365441.
Randomization
This was an open-label trial with central randomization by the NHMRC Clinical Trials Centre via a web-based system 1:1 by minimization to continuous imatinib (Arm A) or to alternating imatinib with regorafenib (Arm B). The randomization was stratified by site, receipt of previous adjuvant therapy (prior vs. none), and receipt of imatinib for metastatic disease for less than or equal to 21 days.
Procedures
Imatinib was administered orally at 400 mg once daily continuously on a 56-day cycle. In Arm B imatinib was administered at 400 mg orally daily for 21–25 days followed by a washout (drug-free) period of 3–7 days, then regorafenib 160 mg once daily orally for three weeks followed by a seven-day washout period. Treatment was continued until disease progression as per RECIST 1.1 or unacceptable adverse events.
Study treatment was delayed or stopped in case of occurrence of adverse events and resumed once protocol-defined criteria were met. See study protocols in Supplementary Appendix documents. Dose delays and reductions were permitted for imatinib and regorafenib for toxicities prespecified in the protocol.
Tumor assessment was by CT scan at baseline and every eight weeks for the first year on trial, and then every 12 weeks until disease progression or death, timed from the randomization date. PET scans were performed for Arm B patients enrolled into the PET substudy, patients were randomized to receive two PET scans either during the imatinib or regorafenib washout period one week apart, results yet to be reported. Hematology, biochemistry, and urine dipstick were performed before Day 1 of each cycle. Adverse events were recorded at each visit according to National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 4.03.
Blood for pharmacokinetic analysis were taken as follows: for imatinib levels, plasma samples were taken on Week 4, Day 1 of Cycles 1 and 2 in Arm A, and Week 3 (last day of imatinib administration) of Cycles 1 and 2 in Arm B; for regorafenib levels, plasma samples were taken on Week 7 (last day of regorafenib administration) of Cycles 1 and 2 in Arm B only. Serial blood samples were also collected at other timepoints for exploratory biomarker studies.
Outcomes
The primary outcome, objective tumor response (OTR) at 9 months, was measured, as defined by RECIST 1.1 (complete or partial response) and assessed by the investigator with central confirmation of response at or before nine months from the time of either (i) randomization or (ii) commencement of therapy (if patients were randomized during their first cycle of imatinib). Secondary outcomes were OS, PFS, treatment-related safety, toxicity, tolerability, clinical benefit rate (stable disease + partial response + complete response) following three cycles (24 weeks) of treatment, and time to treatment failure. OS was defined as the interval from either (i) randomization (if patients had not yet commenced treatment) or (ii) commencement of therapy (if patients were randomized during the first cycle of imatinib) to date of death from any cause, or the date of last known follow-up alive. PFS was the length of time from either (i) randomization (if patients had not yet commenced treatment) or (ii) commencement of therapy (if patients were randomized during the first cycle of imatinib) until disease progression defined according to RECIST 1.1 or death. Patients were censored at last known status if progressive disease (PD) had not yet occurred or censored if switched to non-protocol therapy; patients were still continued to be followed to PD if still on any part of protocol therapy (eg: imatinib alone); surgery for residual disease prior to PD was allowed as part of protocol therapy (so patients were still followed further for PD).
Patients who had received at least one dose of the study drugs were included in evaluation of treatment safety.
The clinical benefit rate was calculated by summing the number of participants assessed as having a complete response, partial response, or stable disease within the first 24 weeks from randomization, and dividing this by the total number of participants evaluable for response (according to RECIST version 1.1) within the first 24 weeks. Time to treatment failure was defined as the time from either (i) randomization (if patients had not yet commenced treatment) or (ii) commencement of therapy (if patients were randomized during their first cycle of imatinib) to treatment discontinuation for any reason, including disease progression, treatment toxicity, patient preference, or death.
Statistical analysis
The study was originally planned to enroll 240 patients in a 1:1 randomization ratio. Analyses were to include all patients who were randomized. Treatment activity was to be assessed by the proportion, together with the 95% confidence interval, of patients not progressing at 24 months. In order to demonstrate a relative increase in progression free survival (PFS) at 24 months from the date of randomization from an expected 78% to 88%, with 80% power and 95% confidence based on A’Hern’s adjustment to Fleming’s design, approximately 110 evaluable participants were required in each arm. Thus 240 participants were proposed allowing for approximately a 10% drop-out rate. Eighty percent of participants were expected to achieve a clinical benefit at 24 months (CBR – rate of complete or partial response, or stable disease). A secondary outcome was to determine whether a minimum 25% relative increase of the CBR in the experimental cohort could be attained.
Due to logistical difficulties with opening centers to recruitment across multiple countries in Europe and Asia as well as slow enrollment, the funding available for this study was reduced, necessitating a revision of the recruitment target to 76 patients in a 1:1 randomization ratio in July 2017. The primary endpoint was altered to the OTR at or before nine months as it was uncertain at that date what amount of funding would be available to continue followup of patients enrolled and data collection. Time to treatment failure was altered to consider events in the first 12 months. Patients not experiencing treatment failure within 12 months would be censored at 12 months. PFS and overall survival at one year were new secondary objectives. These changes were made with the process being blinded to trial outcome data by randomized group. Based on a 95% one-sided confidence interval, for different levels of the true underlying response rate, the minimum number of responses required to be observed in the alternating group to classify the alternating regimen as active was as follows: 40%, ≥ 10 responses; 50%, ≥ 13 responses; 60%, ≥ 17 responses; 70%, ≥ 22 responses.
Role of funding source
The funder of the study had no role in the study design, study execution, data collection, data analyses, data interpretation or decision to submit results for publication.
Results
Between June 2015 and September 2018, 78 patients were screened, of whom two were excluded from randomization. We randomly assigned 76 patients, with 36 in the Arm A (imatinib only) and 40 in Arm B (imatinib alternating with regorafenib) (Table 1). Baseline disease characteristics and demographic characteristics are given in Table 1. The patients were predominately male in both arms. The mean age was 63.1 in Arm A and 59.3 in Arm B, with the stomach the most common primary site in both Arm A (38.9%) and Arm B (35.0%). Exon 11 and 9 KIT mutations were the most common alterations.
At data cut off, on 31 December 2020, median follow-up time was 46.0 months (range 6.5– 64.6 months) for all patients. Relative dose intensity was 95.0% for imatinib in Arm A and 98.9% for imatinib and 84.1% for regorafenib in Arm B. The best responses to Arm A and Arm B treatments were similar at nine months; 0 vs. 1 patients had complete response, 22 vs. 23 partial response, 14 vs. 15 stable disease (Table 2). The primary endpoint of OTR at or before nine months was 61.1% (95% CI, 44.9–75.2%) and 60.0% (95% CI, 44.6–73.7%), respectively, in Arm A and Arm B. Estimated clinical benefit rate (CR + PR + SD) at three cycles (24 weeks) was achieved in 97.2% patients in Arm A (95% CI 85.8–99.5%) and 87.5% in Arm B (95% CI 73.9–94.5%).
There were 8 (22.2%; 95% CI, 11.7–38.1%) and 6 (15.0%; 95% CI, 7.1–29.1%) patients respectively in Arm A and Arm B who underwent surgical resection of residual disease. Of these patients 7 had complete excision of disease and 6 incomplete excision. Two patients had disease progression prior to surgery and one had progression after surgery. Eight patients received non protocol systemic therapy after surgery.
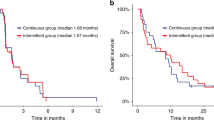
At data cut off, on 31 December 2020, 12 (33.2%) patients in Arm A and 9 (22.5%) patients in Arm B were still on study treatment; 18 patients (50.0%) in Arm A and 12 patients (30.0%) in Arm B had discontinued protocol therapy due to progressive disease. The secondary endpoint of PFS at one year was Arm A 83.3% (95% CI 66.6–92.1) and Arm B 87.5% (95% CI, 72.5–94.6), plogrank = 0.71. The median PFS was 37.0 months and 36.7 months in arm A and arm B, respectively. OS at one year was 97.2% (95% CI 81.9–99.6) and 97.5% (95% CI 84.5–99.6) respectively; plog rank = 0.30. The median overall survival was not reached in any arm. The Kaplan-Meier curves for PFS and OS are shown in Fig. 1.
Kaplan Meier survival curves for progression free Panel a and overall survival Panel b.
An exploratory subgroup analysis was performed with respect to CBR, PFS and OS according to the exon 11 KIT mutation status. In patients with exon 11 KIT mutation (n = 60, 27 Arm A and 33 Arm B) the CBR were 100% (95% CI 87.5–100) and 93.9% (95% CI 80.4-98.3) in Arm A (27/27 patients) and Arm B (31/33 patients), respectively (p = 0.6); in patient with non-exon 11 KIT mutation (n = 8), the CBR were 66.7% (95% CI 20.8–93.9) and 80.0% (95% CI 37.6–96.4) in Arms A (2/3 patients) and B (4/5 patients), respectively (p = 1.0). In patients with exon 11 KIT mutation, the estimated 12-month PFS was 85.2% (95% CI 65.2–94.2) in Arm A and 93.9% (95% CI 77.9–98.4) in Arm B, plogrank = 0.88. In patients with non-exon 11 KIT mutation, the estimated 12-months PFS was 33.3% (95% CI 0.9–77.4) in Arm A and 80.0% (95% CI 20.4–96.9) in Arm B, plogrank = 0.26. The OS at one year for patients with exon 11 KIT mutation was 96.3% (95% CI 76.5–99.5) in Arm A and 100% (95% CI not estimable) in Arm B, plogrank = 0.79. The OS at one year for patients with non-exon 11 KIT mutation was 100% (95% CI not estimable) in both Arm A and Arm B, plogrank = 0.14. The mutation analysis results of the cohort are shown in Table 1.
Kaplan-Meier estimate of treatment failure at 12 months was 83.3% (66.7–92.1) for Arm A and 75.0% (58.5–85.7) for Arm B, plogrank = 0.36 with a hazard ratio of 1.60 (0.58–4.39), alternating versus imatinib only.
As data collection was able to be continued beyond the data cutoff of December 2020, an updated analysis was carried out of PFS and OS as of the last available date of May 18, 2023. This found no difference in PFS or OS. At four years the PFS rates were Arm A: 40.0% (23.8-55.7) and Arm B 35.8% (20.8–0.51.0) plogrank = 0.68 and estimated OS rates were: 69.1% (51.2–81.6) and 74.1% (57.2–85.2) respectively, plogrank = 0.46. See Supplementary Fig. 1 for graph of PFS. In Arm A 14 of the 38 (37%) disease progression (PD) events were due to new lesion development compared to 12 out of 40 (30%) in Arm B.
Duration of response (defined as time from registration to disease progression or death in patients who achieve complete or partial response) was also explored up to the last available follow-up. This found a median duration of response of 57.5 months with Arm A (48.6– 66.8) and 40.2 months with arm B (38.8–49.9), p = 0.612. See Supplemental Fig. 2 for graph of PFS in responders.
Treatment-related adverse events of any grade occurred in 36 (100%) of 36 patients in Arm A and 40 (100%) of 40 patients in Arm B. Grade 3 or 4 adverse events occurred in 16 (44.4%) and 23 (57.5%) patients in Arm A and Arm B, respectively (Table 3). The most common grade 3 or 4 adverse events were anemia and diarrhea in Arm A. In Arm B, the most common grade 3 or 4 adverse events were palmar-plantar erythrodysesthesia syndrome and hypertension. Dose reductions occurred in 11 (30.5%) patients in Arm A and 25 (62.5%) patients in Arm B. Hematological toxicity led to dose delay in 2 patients in Arm A and one patient in Arm B due to regorafenib. Twelve patients (30.0%) in Arm B stopped protocol therapy due to unacceptable toxicity, and none in Arm A. Twelve (31.5%) patients in Arm A and 12 (30.0%) in Arm B experienced at least one serious adverse event. The most common serious treatment-related adverse event was diarrhea (3 [7.8%] of 38) in Arm A and abdominal pain (2 [5.0%] of 40) in Arm B.
Death while on treatment or follow-up occurred in 11 (30.6%) of 36 patients in Arm A and 12 (30.0%) of 40 patients in Arm B. Among these, 9/11 (81.8%) and 9/12 (75.0%) were related to disease progression in Arm A and Arm B, respectively. Death due to study drug toxicity was reported in one patient in Arm B (myocardial infarction) and no patients in Arm A.
Discussion
While our working hypothesis was that rotation of treatment would limit the emergence of clonal resistance, we observed no difference in the primary endpoint of objective tumor response at 9 months nor on secondary endpoints of PFS and OS in this prospective randomized study testing for the first time the rotation of TKIs as a strategy for first line treatment.
The endpoints were non-significantly different between the alternating therapy arm compared to the imatinib-only arm. In addition, there was no difference in subgroup analysis based on the presence or absence of an exon 11 KIT mutation, history of prior imatinib treatment (as adjuvant therapy or before randomization) and the tumor site. Although the observed DFS in Arm B was 80% while in Arm A it was 33.3% in the non-exon 11 patients, this non-significant trend was not accounted for by differences in the numbers of exon 9 and KIT/PDGFRA negative (wild type) patients between the two arms. These two genotypes are sensitive to regorafenib but are relatively less sensitive and resistant to imatinib respectively.
Mutations in the KIT and PDGFRA receptors are critical gain of function drivers in 90% of patients with GIST [18, 19]. Inhibition of these receptors with imatinib results in improved survival in the majority of patients with GIST; however after an initial clinical response, tumor progression occurs in most patients [2]. In around 90% of patients imatinib failure is due to secondary mutations in KIT in two regions of the kinase domain: the ATP binding pocket and the activation loop [8, 20]. A preclinical study in imatinib-resistant GIST using alternating regorafenib and sunitinib reported that regorafenib mainly inhibits KIT kinases with a mutation in the activation loop and sunitinib inhibits kinases with an ATP binding pocket mutation [21]. This study suggested that a combination of TKIs may prevent polyclonal imatinib resistance due to the development of secondary mutations. Serano et al. conducted a phase Ib study to investigate the feasibility and tolerability of continuous treatment with rapid alternation of sunitinib for 3 days followed by regorafenib for 4 days in patients with TKI-resistant GIST [22]. They reported stable disease in 31% patients (4/13 patients), a median PFS of 1.9 months (95% CI 1.4–3.6 months) and a median OS of 10.8 months (95% CI, 5.9–∞), and mainly grade 1–2 adverse events. Although this schedule was tolerable and feasible, low plasma drug concentrations of both sunitinib and regorafenib were found below threshold target inhibition concentrations suggesting that clinically meaningful drug exposure was not achieved. As sustained stable disease was seen in the study with clinical benefit it was thought by the authors that partial KIT inhibition was still achieved but not enough to suppress all the KIT resistant mutations. Our study reported good relative dose intensity in both arms (94% in Arm A; 100% for imatinib and 86% for regorafenib in Arm B) but pharmacokinetic analysis of the blood samples collected is awaited to see if drugs levels correlate with clinical endpoints in Arm B.
Treatment-related toxicity with imatinib and regorafenib was similar to that expected for either agent alone. The most common grade 3 or 4 toxicity was anemia and diarrhea in Arm A and palmar-plantar erythrodysesthesia syndrome and hypertension in Arm B. Toxicity was higher in the alternating arm compared to single-agent imatinib affecting the tolerability of this regimen with many dose reductions and discontinuation of drug administration due to side effects. There were no drug-related deaths in the imatinib-only arm and one death due to myocardial infarction in the alternating arm.
The treatment paradigm for GIST is continuously evolving. There are now three further options for GIST: avapritinib, ripretinib and pimitespib. Updated results from the phase I NAVIGATOR study reported 84% ORR in patients with the rare D842V and other exon 18 mutations and avapritinib is now considered first-line therapy in the uncommon patients with metastatic GIST with a PDGFRA D842V mutation [23]. Ripretinib was compared with placebo in the phase III INVICTUS trial in patients with advanced GIST who had received three or more lines of therapy [24]. They reported a PFS of 6.3 vs. 1.0 months and an OS of 15.1 vs. 6.6 months in the ripretinib group, respectively, compared to placebo, but ripretinib has not been evaluated as the first-line treatment for advanced GIST. Ripretinib has also been compared against sunitinib following imatinib failure in advanced GIST in the INTRIGUE study [25]. This did not show a meaningful difference in the median progression free survivals. The CHAPTER-GIST-301 phase III trial of the heat shock protein 90 (HSP90) inhibitor pimitespib demonstrated improved PFS of 2.8 months versus 1.4 months and cross over adjusted overall survival compared to placebo in fourth line therapy after imatinib, sunitinib and regorafenib [26].
The ALTGIST trial was a well conducted international randomized study which recruited patients from 11 countries thus reducing the risk of bias and improving generalizability of the outcomes. The study adopted RECIST 1.1 standardized criteria in tumor response measurement which were centrally corroborated to improve the validity of the data. The premature discontinuation of the study led to a smaller sample size than originally planned could mean that a moderate treatment effect could be missed. However there were no signals seen that would warrant further investigation of this strategy.
In summary there was no meaningful difference in the primary endpoint of OTR and in PFS between the groups in this final analysis of AGITG ALT-GIST. The difference in the Kaplan-Meier estimate of OS and time to treatment failure at 12 months were also not significant. Post-hoc analyses to the time of last available follow-up found no differences in duration of response or PFS and OS at four years between the study arms. No new or unexpected toxicity was observed, with increased toxicity seen, as expected, in the alternating arm. This alternating schedule does not warrant further study. We conclude that clinicians should continue the current standard of first-line single-agent continuous imatinib until disease progression on highest tolerable dose.
Data availability
De-identified individual participant data collected during the AGITG ALT-GIST trial will be shared after article publication with no end date. These data will be available to researchers who provide a methodologically sound proposal for the purposes of achieving specific aims outlined in that proposal. Proposals should be directed to the Clinical Trials Project Manager via email to altgist.study@sydney.edu.au and will be reviewed by the AGITG ALT-GIST study management committee. Requests to access data to undertake hypothesis-driven research will not be unreasonably withheld. To gain access, data requesters will need to sign a data access agreement and to confirm that data will only be used for the agreed purpose for which access was granted.
References
Verweij J, Casali PG, Zalcberg J, LeCesne A, Reichardt P, Blay JY, et al. Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: Randomised trial. Lancet. 2004;364:1127–34.
Blanke CD, Rankin C, Demetri GD, Ryan CW, Von Mehren M, Benjamin RS, et al. Phase III randomized, intergroup trial assessing imatinib mesylate at two dose levels in patients with unresectable or metastatic gastrointestinal stromal tumors expressing the kit receptor tyrosine kinase: S0033. J Clin Oncol. 2008;26:626–32.
Von Mehren M, Heinrich M, Joensuu H, Blanke C, Wehrle E, Demetri G. Follow-up results after 9 years of the ongoing, phase II B2222 trial of imatinib mesylate (IM) in patients (pts) with metastatic or unresectable KIT+ gastrointestinal stromal tumors (GIST). Journ. al Clin Oncol. 2011;29:10016.
Heinrich MC, Rankin C, Blanke CD, Demetri GD, Borden EC, Ryan CW, et al. Correlation of long-term results of imatinib in advanced gastrointestinal stromal tumors with next-generation sequencing results: Analysis of phase 3 SWOG Intergroup trial S0033. JAMA Oncol. 2017;3:944–52.
Lee CK, Goldstein D, Gibbs E, Joensuu H, Zalcberg J, Verweij J, et al. Development and validation of prognostic nomograms for metastatic gastrointestinal stromal tumour treated with imatinib. Eur J Cancer. 2015;51:852–60.
Seeber A, Perathoner A, Kocher F. Resistance in gastrointestinal stromal tumors. Memo Mag Eur Med Oncol. 2019;12:140–3.
Antonescu CR, Besmer P, Guo T, Arkun K, Hom G, Koryotowski B, et al. Acquired resistance to imatinib in gastrointestinal stromal tumor occurs through secondary gene mutation. Clin Cancer Res. 2005;11:4182–90.
Desai J, Shankar S, Heinrich MC, Fletcher JA, Fletcher CD, Manola J, et al. Clonal evolution of resistance to imatinib in patients with metastatic gastrointestinal stromal tumors. Clin Cancer Res. 2007;13:5398–405.
Patrikidou A, Chabaud S, Ray-Coquard I, Bui BN, Adenis A, Rios M, et al. Influence of imatinib interruption and rechallenge on the residual disease in patients with advanced GIST: results of the BFR14 prospective French Sarcoma Group randomised, phase III trial. Ann Oncol. 2013;24:1087–93.
Heinrich MC, Griffith DJ, Druker BJ, Wait CL, Ott KA, Zigler AJ. Inhibition of c-kit receptor tyrosine kinase activity by STI 571, a selective tyrosine kinase inhibitor. Blood. 2000;96:925–32.
Demetri GD, Von Mehren M, Blanke CD, Van den Abbeele AD, Eisenberg B, Roberts PJ, et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med. 2002;347:472–80.
Kee D, Zalcberg JR. Current and emerging strategies for the management of imatinib-refractory advanced gastrointestinal stromal tumors. Ther Adv Med Oncol. 2012;4:255–70.
George S, Wang Q, Heinrich MC, Corless CL, Zhu M, Butrynski JE, et al. Efficacy and safety of regorafenib in patients with metastatic and/or unresectable GI stromal tumor after failure of imatinib and sunitinib: a multicenter phase II trial. J Clin Oncol. 2012;30:2401.
Wilhelm SM, Dumas J, Adnane L, Lynch M, Carter CA, Schütz G, et al. Regorafenib (BAY 73‐4506): A new oral multikinase inhibitor of angiogenic, stromal and oncogenic receptor tyrosine kinases with potent preclinical antitumor activity. Int J Cancer. 2011;129:245–55.
Yeh CN, Chen MH, Chen YY, Yang CY, Yen CC, Tzen CY, et al. A phase II trial of regorafenib in patients with metastatic and/or a unresectable gastrointestinal stromal tumor harboring secondary mutations of exon 17. Oncotarget. 2017;8:44121–30.
Grellety T, Kind M, Coindre J-M, Italiano A. Clinical activity of regorafenib in PDGFRA-mutated gastrointestinal stromal tumor. Future Science OA 2015; 1;1:FSO33.
Demetri GD, Reichardt P, Kang Y-K, Blay J-Y, Rutkowski P, Gelderblom H, et al. Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): An international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381:295–302.
Heinrich MC, Corless CL, Duensing A, McGreevey L, Chen C-J, Joseph N, et al. PDGFRA activating mutations in gastrointestinal stromal tumors. Science. 2003;299:708–10.
Hirota S, Isozaki K, Moriyama Y, Hashimoto K, Nishida T, Ishiguro S, et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998;279:577–80.
Heinrich MC, Corless CL, Blanke CD, Demetri GD, Joensuu H, Roberts PJ, et al. Molecular correlates of imatinib resistance in gastrointestinal stromal tumors. J Clin Oncol. 2006;24:4764–74.
Serrano C, Mariño-Enríquez A, Tao DL, Ketzer J, Eilers G, Zhu M, et al. Complementary activity of tyrosine kinase inhibitors against secondary kit mutations in imatinib-resistant gastrointestinal stromal tumours. Br J Cancer. 2019;120:612–20.
Serrano C, Leal A, Kuang Y, Morgan JA, Barysauskas CM, Phallen J, et al. Phase I study of rapid alternation of sunitinib and regorafenib for the treatment of tyrosine kinase inhibitor refractory gastrointestinal stromal tumors. Clin Cancer Res. 2019;25:7287–93.
Jones RL, Serrano C, von Mehren M, George S, Heinrich MC, Kang YK, et al. Avapritinib in unresectable or metastatic PDGFRA D842V-mutant gastrointestinal stromal tumours: Long-term efficacy and safety data from the NAVIGATOR phase I trial. Eur J Cancer. 2021;145:132–42.
Blay JY, Serrano C, Heinrich MC, Zalcberg J, Bauer S, Gelderblom H, et al. Ripretinib in patients with advanced gastrointestinal stromal tumours (INVICTUS): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2020;21:923–34.
Bauer S, Jones RL, Blay J-Y, Gelderblom H, George S, Schöffski P, et al. Ripretinib versus sunitinib in patients with advanced gastrointestinal stromal tumor after treatment with imatinib (INTRIGUE): a randomized, open-label, Phase III Trial. J Clin Oncol. 2022;40:3918–28.
Kurokawa Y, Honma Y, Sawaki A, Naito Y, Iwagami S, Komatsu Y, et al. Pimitespib in patients with advanced gastrointestinal stromal tumor (CHAPTER-GIST-301): a randomized, double-blind, placebo-controlled phase III trial. Ann Oncol. 2022;33:959–67.
Funding
Funding for this study was provided by Bayer Healthcare. The Australasian Gastro-Intestinal Trials Group (AGITG) was the sponsor of this study and this was a collaboration with the European Organisation for Research and Treatment of Cancer and the Scandinavian Sarcoma Group. We thank the patients, families and caregivers for participation in this study as well as the investigators and site study personnel. AGITG ALT-GIST was supported by an unrestricted research grant from Bayer HealthCare Pharmaceuticals to the Australasian Gastro-Intestinal Trials Group (AGITG) to conduct the study independently. The preliminary results of this study was presented at the American Society of Clinical Oncology Annual Scientific Meeting on 1st of June 2019 in Chicago. Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
DY, JZ, VG, JS and HJ conceived and designed this study. DY, JZ, JB, ME, AI, NS, CU, HG and HJ acquired data. TP, SM, JS were involved in operational aspects of the study. DE, VG and JS were involved in data analysis and interpretation. DY drafted the manuscript. All authors revised the manuscript and approved the final version. DY confirms he had full access to the data in the study and assumes full responsibility for the decision to submit for publication.
Corresponding author
Ethics declarations
Competing interests
DY reports leadership or fudiciary roles in other board, society, committee or advocacy group for Bayer and Specialised Therapeutics. JRZ reports grants or contracts from Bristol Myers Squibb, AstraZeneca, Pfizer, IQvia, Mylan, Ipsen, Eisai, Medtronic, MSD Oncology, Servier; consulting fees from Merck, Sharp & Dohme, Specialised Therapeutics, CEND, Deciphera, Revolution Medicine, FivePHusion, Genorbio, 1Global, Novotech, Alloplex Biotheapeutics Inc, NOUS Consulting and Oncology Republic; support for attending meetings and/or travel from MSD Oncology, ICON Group, PRAXIS; Leadership or fiduciary role in other board, society, committee or advocacy group, paid or unpaid from ICON group, Lipotex and PRAXIS; Stock or stock options from Biomarin, Ophthea, Amarin, Concert Pharmaceuticals, Frequency Therapeutics, Gilead, Madrigal Pharmaceuticals, UniQure, Zogenix, Orphazyme, Moderna Therapeutics, TWST, Novavax, Teladoc, Gilead Sciences, MSD Oncology and Viatris. JYB reports support for the present manuscript from InterSARC grant, NETSARC and LYRICAN + °; Consulting fees from Bayer and Novartis. ME reports Grants or contracts from institutional support from Novartis for the adjuvant GIST trial SSG XXII; Consulting fees from Consultant for Blueprint Medicines until 2021; Participation on a Data Safety Monitoring Board or Advisory Board as Chairman of the Data Management Board for IMPRESS Norway; Leadership or fiduciary role in other board, society, committee or advocacy group, paid or unpaid as Chairman of the Ethical Advisory Board for SELNET. AI reports Grants or contracts from BAYER, ROCHE, ASTRAZENECA, MERCK, MSD and BMS; consulting fees from BAYER, ROCHE, ASTRAZENECA, MERCK, MSD and BMS. NS provided consultation or attended advisory boards for Boehringer Ingelheim, Cogent Biosciences, Ellipses Pharma, Incyte, Luszana; received research grants from Abbvie, Actuate Therapeutics, Amgen, Array, Ascendis Pharma, AstraZeneca, Bayer, Blueprint Medicines, Boehringer Ingelheim, BridgeBio, Bristol-Myers Squibb, Cantargia, CellCentric, Cogent Biosciences, Cresecendo Biologics, Cytovation, Deciphera, Dragonfly, Eli Lilly, Exelixis, Genentech, GlaxoSmithKline, IDRx, Immunocore, Incyte, InteRNA, Janssen, Kinnate Biopharma, Kling Biotherapeutics, Luszana, Merck, Merck Sharp & Dohme, Merus, Molecular Partners, Navire Pharma, Novartis, Numab Therapeutics, Pfizer, Relay Pharmaceuticals, Revolution Medicin, Roche, Sanofi, Seattle Genetics, Taiho, Takeda (all outside the submitted work, all payment to the Netherlands Cancer Institute). KB reports payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from Novartis; participation on a Data Safety Monitoring Board or Advisory Board for GSK, Bayer, NEC Oncoimmunity, Incyte. CG reports grants or contracts from study sponsor for costs of study conduct. JS reports support for the present manuscript from Bayer; grants or contracts from Roche, BMS, AZ, MSD, Pfizer, National Health and Medical Research Council, Australia; consulting fees from Detsamma Investments Pty Ltd as FivepHusion. HJ reports support for the present manuscript from Scandinavian Sarcoma Group; consulting fees from Orion Pharma andNeutron Therapeutics; payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from Deciphera Pharmaceuticals; patents planned, issued or pending from Sartar Therapeutics; participation on a Data Safety Monitoring Board or Advisory Board for Neutron Therapeutics, Orion Pharma and Maud Kuistila Foundation; leadership or fiduciary role in other board, society, committee or advocacy group, paid or unpaid for Orion Pharma; stock or stock options from Orion Pharma and Sartar Therapeutics. DE, SM, TP, VG, HG do not report any conflicts of interest.
Ethics approval and consent to participate
All participants provided written informed consent according to International Conference on Harmonization and Good Clinical Practices guidelines and national or local regulations. Lead ethics approval was granted by the Sydney Local Health District Human Research Ethics Committee (Protocol No X14‐0218 & HREC/14/RPAH/286) for the AGITG sites. The study was approved by the human research ethics committee of each participating country or institution. The study was performed in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yip, D., Zalcberg, J., Blay, JY. et al. Imatinib alternating with regorafenib compared to imatinib alone for the first-line treatment of advanced gastrointestinal stromal tumor: The AGITG ALT-GIST intergroup randomized phase II trial. Br J Cancer 132, 897–904 (2025). https://doi.org/10.1038/s41416-025-02983-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-025-02983-w