Abstract
Background
Neoadjuvant combined immunotherapy has provided more treatment options for patients with gastric adenocarcinoma (GA). However, some GA patients, especially those with primitive enterocyte phenotype (GAPEP) show a poor response to immunotherapy, even with positive PD-L1 expression.
Method
We enrolled multiple cohorts from our center and utilized public data to identify the predictive factors and explore the immunosuppressive features of GAPEP by multi-omics methods.
Results
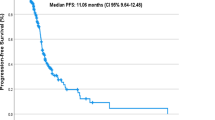
Forty-seven patients with neoadjuvant combined immunotherapy were enrolled. After testing, we found PD-L1 combined positive score (CPS) ≥ 50 in biopsy tissues was significantly associated with major pathological response (MPR) (P = 0.04). RNA testing and immunohistochemical staining highlighted the cytotoxicity-associated markers (GZMA and PRF1) as the predictors to better response. Notably, GAPEP patients demonstrated resistance to therapy and exhibited worse survival outcomes. Our own and public bulk/single-cell transcriptomic analyses identified PVR as a predictor of treatment resistance and as an important immune suppressor, especially in GAPEP. Cell interaction analyses, multiple staining, and cell experiments verified the association between GAPEP and PVR.
Conclusion
Cytotoxic markers, especially GZMA and PRF1, could predict the benefit of neoadjuvant combined immunotherapy in GA than PD-L1 CPS, while PVR is a negative predictor, particularly for GAPEP patients.

Highlights
-
IHC staining of cytotoxic markers such as GZMA and PRF1 could better predict ICI benefit in GA than PD-L1 CPS, while positive PVR IHC staining is a negative predictor.
-
GAPEP (GA with the primitive enterocyte phenotype) is more aggressive and more likely to resist immunotherapy.
-
Upregulation of PVR is prevalent in GAPEP, and PVR might be the key factor in promoting the immunosuppressive microenvironment of GAPEP.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Available. The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Zheng R, Zhang S, Zeng H, Wang S, Sun K, Chen R, et al. Cancer incidence and mortality in China, 2016. J Natl Cancer Cent. 2022;2:1–9.
Yang Z, Zhang C, Liu X, Che N, Feng Y, Xuan Y. SETD5 regulates glycolysis in breast cancer stem-like cells and fuels tumor growth. Am J Pathol. 2022;192:712–21.
Liu L, Woo Y, D’Apuzzo M, Melstrom L, Raoof M, Liang Y, et al. Immunotherapy-based neoadjuvant treatment of advanced microsatellite instability-high gastric cancer: a case series. J Natl Compr Canc Netw. 2022;20:857–65.
Dislich B, Mertz KD, Gloor B, Langer R, Interspatial distribution of tumor and immune cells in correlation with PD-L1 in molecular subtypes of gastric cancers. Cancers 2022, 14.
Goodman KA, Xu R-h, Chau I, Chen MH, Cho BC, Shah MA, et al. SKYSCRAPER-07: A phase III, randomized, double-blind, placebo-controlled study of atezolizumab with or without tiragolumab in patients with unresectable ESCC who have not progressed following definitive concurrent chemoradiotherapy. 2022, 40:TPS374-TPS374.
Fuchs CS, Ozguroglu M, Bang YJ, Di Bartolomeo M, Mandala M, Ryu MH, et al. Pembrolizumab versus paclitaxel for previously treated PD-L1-positive advanced gastric or gastroesophageal junction cancer: 2-year update of the randomized phase 3 KEYNOTE-061 trial. Gastric Cancer. 2022;25:197–206.
Tang X, Li M, Wu X, Guo T, Zhang L, Tang L, et al. Neoadjuvant PD-1 blockade plus chemotherapy induces a high pathological complete response rate and anti-tumor immune subsets in clinical stage III gastric cancer. Oncoimmunology. 2022;11:2135819.
Zhao Y, Bai Y, Shen M, Li Y. Therapeutic strategies for gastric cancer targeting immune cells: Future directions. Front Immunol. 2022;13:992762.
Xie T, Zhang Z, Zhang X, Qi C, Shen L, Peng Z. Appropriate PD-L1 cutoff value for gastric cancer immunotherapy: a systematic review and meta-analysis. Front Oncol. 2021;11:646355.
Kim TK, Vandsemb EN, Herbst RS, Chen L. Adaptive immune resistance at the tumour site: mechanisms and therapeutic opportunities. Nat Rev Drug Discov. 2022;21:529–40.
Chen Y, Jia K, Sun Y, Zhang C, Li Y, Zhang L, et al. Predicting response to immunotherapy in gastric cancer via multi-dimensional analyses of the tumour immune microenvironment. Nat Commun. 2022;13:4851.
Huang K, Hu M, Chen J, Wei J, Qin J, Lin S, et al. Multi-omics perspective reveals the different patterns of tumor immune microenvironment based on programmed death ligand 1 (PD-L1) expression and predictor of responses to immune checkpoint blockade across pan-cancer. Int J Mol Sci 2021, 22.
Haas M, Buttner M, Rau TT, Fietkau R, Grabenbauer GG, Distel LV. Inflammation in gastric adenocarcinoma of the cardia: how do EBV infection, Her2 amplification and cancer progression influence tumor-infiltrating lymphocytes? Virchows Arch. 2011;458:403–11.
Yamazawa S, Ushiku T, Shinozaki-Ushiku A, Hayashi A, Iwasaki A, Abe H, et al. Gastric cancer with primitive enterocyte phenotype: an aggressive subgroup of intestinal-type adenocarcinoma. Am J Surg Pathol. 2017;41:989–97.
Zhang Q, Kim DH, Xu Y, Wang W, Medeiros LJ. Clinicopathological features of syncytial variant nodular sclerosis Hodgkin lymphoma. Hum Pathol. 2022;119:105–13.
Akazawa Y, Saito T, Hayashi T, Yanai Y, Tsuyama S, Akaike K, et al. Next-generation sequencing analysis for gastric adenocarcinoma with enteroblastic differentiation: emphasis on the relationship with hepatoid adenocarcinoma. Hum Pathol. 2018;78:79–88.
Mei Y, Li M, Wen J, Kong X, Li J. Single-cell characteristics and malignancy regulation of alpha-fetoprotein-producing gastric cancer. Cancer Med. 2023;12:12018–33.
Kwon MJ, Byeon S, Kang SY, Kim KM. Gastric adenocarcinoma with enteroblastic differentiation should be differentiated from hepatoid adenocarcinoma: A study with emphasis on clear cells and clinicopathologic spectrum. Pathol Res Pr. 2019;215:152525.
Xia R, Zhou Y, Wang Y, Yuan J, Ma X. Hepatoid adenocarcinoma of the stomach: current perspectives and new developments. Front Oncol. 2021;11:633916.
Kang M, Ma X, Shi J, Chen G, Jin X, Wang J, et al. Distinct molecular phenotype and the potential prognostic value of immune prognostic index and tumor infiltrating lymphocytes in hepatoid adenocarcinoma of stomach. Transl Oncol. 2022;19:101380.
He F, Fu Y, Sun Q, Geng P, Zheng Z, Pu X, et al. Integrated clinicopathological and immunohistochemical analysis of gastric adenocarcinoma with hepatoid differentiation: an exploration of histogenesis, molecular characteristics, and prognostic markers. Hum Pathol. 2021;115:37–46.
Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, et al. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017;67:93–9.
He L, Xiao J, Zheng P, Zhong L, Peng Q. Lymph node regression grading of locally advanced rectal cancer treated with neoadjuvant chemoradiotherapy. World J Gastrointest Oncol. 2022;14:1429–45.
Thorsson V, Gibbs DL, Brown SD, Wolf D, Bortone DS, Ou Yang TH, et al. The immune landscape of cancer. Immunity. 2018;48:812–30.e814.
Yoshihara K, Shahmoradgoli M, Martinez E, Vegesna R, Kim H, Torres-Garcia W, et al. Inferring tumour purity and stromal and immune cell admixture from expression data. Nat Commun. 2013;4:2612.
Newman AM, Liu CL, Green MR, Gentles AJ, Feng W, Xu Y, et al. Robust enumeration of cell subsets from tissue expression profiles. Nat Methods. 2015;12:453–7.
Ma C, Storer CE, Chandran U, LaFramboise WA, Petrosko P, Frank M, et al. Crohn’s disease-associated ATG16L1 T300A genotype is associated with improved survival in gastric cancer. EBioMedicine. 2021;67:103347.
Oh SC, Sohn BH, Cheong JH, Kim SB, Lee JE, Park KC, et al. Clinical and genomic landscape of gastric cancer with a mesenchymal phenotype. Nat Commun. 2018;9:1777.
Lei Z, Tan IB, Das K, Deng N, Zouridis H, Pattison S, et al. Identification of molecular subtypes of gastric cancer with different responses to PI3-kinase inhibitors and 5-fluorouracil. Gastroenterology. 2013;145:554–65.
Qian Z, Zhu G, Tang L, Wang M, Zhang L, Fu J, et al. Whole genome gene copy number profiling of gastric cancer identifies PAK1 and KRAS gene amplification as therapy targets. Genes Chromosomes Cancer. 2014;53:883–94.
Lou S, Wang Y, Zhang J, Yin X, Zhang Y, Wang Y, et al. Patient-level DNA damage repair pathway profiles and anti-tumor immunity for gastric cancer. Front Immunol. 2021;12:806324.
Pan X, Li X, Dong L, Liu T, Zhang M, Zhang L, et al. Tumour vasculature at single-cell resolution. Nature. 2024;632:429–36.
Ding J, Adiconis X, Simmons SK, Kowalczyk MS, Hession CC, Marjanovic ND, et al. Systematic comparison of single-cell and single-nucleus RNA-sequencing methods. Nat Biotechnol. 2020;38:737–46.
Chiang EY, Mellman I. TIGIT-CD226-PVR axis: advancing immune checkpoint blockade for cancer immunotherapy. J Immunother Cancer 2022:10.
O’Donnell JS, Madore J, Li XY, Smyth MJ. Tumor intrinsic and extrinsic immune functions of CD155. Semin Cancer Biol. 2020;65:189–96.
Molfetta R, Zitti B, Lecce M, Milito ND, Stabile H, Fionda C, et al. CD155: A multi-functional molecule in tumor progression. Int J Mol Sci 2020:21.
Bronte V. The expanding constellation of immune checkpoints: a DNAMic control by CD155. J Clin Invest. 2018;128:2199–201.
Lee BR, Chae S, Moon J, Kim MJ, Lee H, Ko HW, et al. Combination of PD-L1 and PVR determines sensitivity to PD-1 blockade. JCI Insight 2020, 5.
Wang JB, Li P, Liu XL, Zheng QL, Ma YB, Zhao YJ, et al. An immune checkpoint score system for prognostic evaluation and adjuvant chemotherapy selection in gastric cancer. Nat Commun. 2020;11:6352.
Gorvel L, Olive D. Targeting the “PVR-TIGIT axis” with immune checkpoint therapies. F1000Res 2020:9.
Rooney MS, Shukla SA, Wu CJ, Getz G, Hacohen N. Molecular and genetic properties of tumors associated with local immune cytolytic activity. Cell. 2015;160:48–61.
Cao X, Cai SF, Fehniger TA, Song J, Collins LI, Piwnica-Worms DR, et al. Granzyme B and perforin are important for regulatory T cell-mediated suppression of tumor clearance. Immunity. 2007;27:635–46.
Chen Q, Wang C, Lei X, Huang T, Zhou R, Lu Y. Immune cytolytic activity for comprehensive insights of the immune landscape in endometrial carcinoma. J Oncol. 2022;2022:9060243.
Gao Z, Tao Y, Lai Y, Wang Q, Li Z, Peng S, et al. Immune cytolytic activity as an indicator of immune checkpoint inhibitors treatment for prostate cancer. Front Bioeng Biotechnol. 2020;8:930.
Reis ES, Mastellos DC, Ricklin D, Mantovani A, Lambris JD. Complement in cancer: untangling an intricate relationship. Nat Rev Immunol. 2018;18:5–18.
Wu B, Zhong C, Lang Q, Liang Z, Zhang Y, Zhao X, et al. Poliovirus receptor (PVR)-like protein cosignaling network: new opportunities for cancer immunotherapy. J Exp Clin Cancer Res. 2021;40:267.
Kumar V, Ramnarayanan K, Sundar R, Padmanabhan N, Srivastava S, Koiwa M, et al. Single-cell atlas of lineage states, tumor microenvironment, and subtype-specific expression programs in gastric cancer. Cancer Discov. 2022;12:670–91.
Zhou KI, Peterson B, Serritella A, Thomas J, Reizine N, Moya S, et al. Spatial and Temporal Heterogeneity of PD-L1 expression and tumor mutational burden in gastroesophageal adenocarcinoma at baseline diagnosis and after chemotherapy. Clin Cancer Res. 2020;26:6453–63.
Ribeiro HSC, Menezes JN, da Costa WL Jr, F de Jesus VH, Diniz AL, et al. PD-L1 expression in gastric and gastroesophageal junction cancer patients treated with perioperative chemotherapy. J Surg Oncol. 2022;126:150–60.
Lei M, Siemers NO, Pandya D, Chang H, Sanchez T, Harbison C, et al. Analyses of PD-L1 and Inflammatory Gene Expression Association with Efficacy of Nivolumab +/- Ipilimumab in Gastric Cancer/Gastroesophageal Junction Cancer. Clin Cancer Res. 2021;27:3926–35.
Kawazoe A, Shitara K, Kuboki Y, Bando H, Kojima T, Yoshino T, et al. Clinicopathological features of 22C3 PD-L1 expression with mismatch repair, Epstein-Barr virus status, and cancer genome alterations in metastatic gastric cancer. Gastric Cancer. 2019;22:69–76.
Kim JY, Kim WG, Kwon CH, Park DY. Differences in immune contextures among different molecular subtypes of gastric cancer and their prognostic impact. Gastric Cancer. 2019;22:1164–75.
Oh S, Kim E, Lee H. Comparative Impact of PD-1 and PD-L1 inhibitors on advanced esophageal or gastric/gastroesophageal junction cancer treatment: a systematic review and meta-analysis. J Clin Med 2021:10.
van den Ende T, van den Boorn HG, Hoonhout NM, van Etten-Jamaludin FS, Meijer SL, Derks S, et al. Priming the tumor immune microenvironment with chemo(radio)therapy: A systematic review across tumor types. Biochim Biophys Acta Rev Cancer. 2020;1874:188386.
Weigelin B, Friedl P. T cell-mediated additive cytotoxicity - death by multiple bullets. Trends Cancer. 2022;8:980–7.
Finn OJ. A Believer’s Overview of Cancer Immunosurveillance and Immunotherapy. J Immunol. 2018;200:385–91.
Hu Q, Nonaka K, Wakiyama H, Miyashita Y, Fujimoto Y, Jogo T, et al. Cytolytic activity score as a biomarker for antitumor immunity and clinical outcome in patients with gastric cancer. Cancer Med. 2021;10:3129–38.
Zhao R, Li H, Ge W, Zhu X, Zhu L, Wan X, et al. Comprehensive analysis of genomic alterations in hepatoid adenocarcinoma of the stomach and identification of clinically actionable alterations. Cancers 2022:14.
Wang B, Du C, Li L, Xie Y, Hu C, Li Z, et al. New substituted molecular classifications of advanced gastric adenocarcinoma: characteristics and probable treatment strategies. J Natl Cancer Cent. 2022;2:50–9.
Gao J, Zheng Q, Xin N, Wang W, Zhao C. CD155, an onco-immunologic molecule in human tumors. Cancer Sci. 2017;108:1934–8.
Lozano E, Joller N, Cao Y, Kuchroo VK, Hafler DA. The CD226/CD155 interaction regulates the proinflammatory (Th1/Th17)/anti-inflammatory (Th2) balance in humans. J Immunol. 2013;191:3673–80.
Braun M, Aguilera AR, Sundarrajan A, Corvino D, Stannard K, Krumeich S, et al. CD155 on Tumor Cells Drives Resistance to Immunotherapy by Inducing the Degradation of the Activating Receptor CD226 in CD8(+) T Cells. Immunity. 2020;53:805–23.e815.
Chan CJ, Martinet L, Gilfillan S, Souza-Fonseca-Guimaraes F, Chow MT, Town L, et al. The receptors CD96 and CD226 oppose each other in the regulation of natural killer cell functions. Nat Immunol. 2014;15:431–8.
Rodriguez-Abreu D, Johnson ML, Hussein MA, Cobo M, Patel AJ, Secen NM, et al. Primary analysis of a randomized, double-blind, phase II study of the anti-TIGIT antibody tiragolumab (tira) plus atezolizumab (atezo) versus placebo plus atezo as first-line (1L) treatment in patients with PD-L1-selected NSCLC (CITYSCAPE). 2020;38:9503.
van der Ploeg AH, Kumpf O, Seelow E, Almanza LC, Schlag PM, Schumann RR, et al. The course of gastric cancer following surgery is associated with genetic variations of the interleukin-1 receptor antagonist and interleukin-1beta. Gastric Cancer. 2015;18:77–83.
Frentzas S, Meniawy T, Kao SC-H, Wang R, Zuo Y, Zheng H, et al. AdvanTIG-105: Phase 1 dose-escalation study of anti-TIGIT monoclonal antibody ociperlimab (BGB-A1217) in combination with tislelizumab in patients with advanced solid tumors. 2021;39:2583.
Wang Y, Sun L, Li Z, Gao J, Ge S, Zhang C, et al. Hepatoid adenocarcinoma of the stomach: a unique subgroup with distinct clinicopathological and molecular features. Gastric Cancer. 2019;22:1183–92.
Liu Z, Wang A, Pu Y, Li Z, Xue R, Zhang C, et al. Genomic and transcriptomic profiling of hepatoid adenocarcinoma of the stomach. Oncogene. 2021;40:5705–17.
Seto M, Ohta M, Ikenoue T, Sugimoto T, Asaoka Y, Tada M, et al. Reduced expression of RAS protein activator like-1 in gastric cancer. Int J Cancer. 2011;128:1293–302.
Ubertini V, Norelli G, D’Arcangelo D, Gurtner A, Cesareo E, Baldari S, et al. Mutant p53 gains new function in promoting inflammatory signals by repression of the secreted interleukin-1 receptor antagonist. Oncogene. 2015;34:2493–504.
Refae S, Gal J, Ebran N, Otto J, Borchiellini D, Peyrade F, et al. Germinal Immunogenetics predict treatment outcome for PD-1/PD-L1 checkpoint inhibitors. Invest N Drugs. 2020;38:160–71.
Yang J, Hawkins OE, Barham W, Gilchuk P, Boothby M, Ayers GD, et al. Myeloid IKKbeta promotes antitumor immunity by modulating CCL11 and the innate immune response. Cancer Res. 2014;74:7274–84.
Schmidt-Supprian M, Tian J, Ji H, Terhorst C, Bhan AK, Grant EP, et al. I kappa B kinase 2 deficiency in T cells leads to defects in priming, B cell help, germinal center reactions, and homeostatic expansion. J Immunol. 2004;173:1612–9.
Pasparakis M, Schmidt-Supprian M, Rajewsky K. IkappaB kinase signaling is essential for maintenance of mature B cells. J Exp Med. 2002;196:743–52.
Thaker YR, Raab M, Strebhardt K, Rudd CE. GTPase-activating protein Rasal1 associates with ZAP-70 of the TCR and negatively regulates T-cell tumor immunity. Nat Commun. 2019;10:4804.
Wang B, Zhu Y, Wang S, Li Z, Wang L, Rao W, et al. Gastric tubular adenocarcinoma with diffuse neutrophils infiltrating: characteristics and probable treatment strategy. Gastric Cancer. 2024;27:86–101.
Chen Y, Jia K, Chong X, Xie Y, Jiang L, Peng H, et al. Implications of PD-L1 expression on the immune microenvironment in HER2-positive gastric cancer. Mol Cancer. 2024;23:169.
Groen-van Schooten TS, Franco Fernandez R, van Grieken NCT, Bos EN, Seidel J, Saris J, et al. Mapping the complexity and diversity of tertiary lymphoid structures in primary and peritoneal metastatic gastric cancer. J Immunother Cancer 2024:12.
Peng H, Jiang L, Yuan J, Wu X, Chen N, Liu D, et al. Single-cell characterization of differentiation trajectories and drug resistance features in gastric cancer with peritoneal metastasis. Clin Transl Med. 2024;14:e70054.
Funding
This work was supported by the Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences 2023-I2M-2-004 and 2021-I2M-1-067, the Beijing Hope Run Special Fund (No. LC2021A20) and Beijing Xisike Clinical Oncology Research Foundation (Y-MSDZD2022-0245).
Author information
Authors and Affiliations
Contributions
LX and JY formulated the study concept and design; BW and YW developed the methodology and performed the writing, review and revision of the paper; XS and YT provided the clinical data; HZ, YZ and NC analyzed and interpretated the data and performed statistical analysis; LG, JM, SW and LW provided technical and material support. All authors read and approved the final paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that there are no conflicts of interest regarding the publication of this paper.
Ethics approval and consent to participate
Ethical approval was granted by the CICAMS ethics committee prior to commencing this study (No21/105-2776). All methods were carried out in accordance with relevant guidelines and regulations, and informed consent was obtained from all participants.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, B., Wang, Y., Zhu, Y. et al. Predictive factors for neoadjuvant combined immunotherapy in gastric adenocarcinoma: Focusing on the primitive enterocyte phenotype and PVR. Br J Cancer 133, 255–269 (2025). https://doi.org/10.1038/s41416-025-03031-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-025-03031-3