Abstract
Background
K-homology-type splicing regulatory protein (KSRP) is an RNA-binding protein involved in mRNA decay and translational repression through recognition of adenine–uracil-rich elements. Although KSRP regulates approximately 16% of transcript expression, its role in cancer remains poorly defined.
Methods
KSRP expression was analysed using qPCR, Western blot, and immunohistochemistry. Its functional role in follicular thyroid cancer (FTC) was examined through in vitro and in vivo assays. Luciferase reporter and rescue experiments were performed to elucidate the underlying molecular mechanisms.
Results
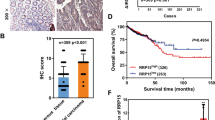
KSRP was significantly upregulated in FTC tissues and metastatic cell lines. Functional studies demonstrated that KSRP enhances the invasiveness and stemness of FTC cells. Mechanistically, KSRP promotes nuclear accumulation and transcriptional activity of β-catenin by downregulating the Wnt inhibitors DACT2 and SFRP2.
Conclusion
This study identifies KSRP as an oncogenic factor in FTC that activates Wnt/β-catenin signalling, suggesting its potential as a therapeutic target for FTC patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data supporting the findings of this study are available from the corresponding author upon reasonable request.
References
Gerstberger S, Hafner M, Tuschl T. A census of human RNA-binding proteins. Nat Rev Genet. 2014;15:829–45.
Hentze MW, Castello A, Schwarzl T, Preiss T. A brave new world of RNA-binding proteins. Nat Rev Mol Cell Biol. 2018;19:327–41.
Iadevaia V, Gerber AP. Combinatorial control of mRNA fates by RNA-binding proteins and non-coding RNAs. Biomolecules. 2015;5:2207–22.
Gherzi R, Lee KY, Briata P, Wegmuller D, Moroni C, Karin M, et al. A KH domain RNA binding protein, KSRP, promotes ARE-directed mRNA turnover by recruiting the degradation machinery. Mol Cell. 2004;14:571–83.
Briata P, Chen CY, Ramos A, Gherzi R. Functional and molecular insights into KSRP function in mRNA decay. Biochim Biophys Acta. 2013;1829:689–94.
Trabucchi M, Briata P, Garcia-Mayoral M, Haase AD, Filipowicz W, Ramos A, et al. The RNA-binding protein KSRP promotes the biogenesis of a subset of microRNAs. Nature. 2009;459:1010–4.
Garcia-Mayoral MF, Hollingworth D, Masino L, Diaz-Moreno I, Kelly G, Gherzi R, et al. The structure of the C-terminal KH domains of KSRP reveals a noncanonical motif important for mRNA degradation. Structure. 2007;15:485–98.
Palzer KA, Bolduan V, Kafer R, Kleinert H, Bros M. Pautz A. The role of KH-type splicing regulatory protein (KSRP) for immune functions and tumorigenesis. Cells. 2022;11:1482.
Gherzi R, Chen CY, Ramos A, Briata P. KSRP controls pleiotropic cellular functions. Semin Cell Dev Biol. 2014;34:2–8.
Chien MH, Lee WJ, Yang YC, Li YL, Chen BR, Cheng TY, et al. KSRP suppresses cell invasion and metastasis through miR-23a-mediated EGR3 mRNA degradation in non-small cell lung cancer. Biochim Biophys Acta Gene Regul Mech. 2017;1860:1013–24.
Yang YC, Lin YW, Lee WJ, Lai FR, Ho KH, Chu CY, et al. The RNA-binding protein KSRP aggravates malignant progression of clear cell renal cell carcinoma through transcriptional inhibition and post-transcriptional destabilization of the NEDD4L ubiquitin ligase. J Biomed Sci. 2023;30:68.
Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73:17–48.
Tronko M, Brenner AV, Bogdanova T, Shpak V, Oliynyk V, Cahoon EK, et al. Thyroid neoplasia risk is increased nearly 30 years after the Chernobyl accident. Int J Cancer. 2017;141:1585–8.
Kitahara CM, K Rmendiné Farkas D, Jørgensen JOL, Cronin-Fenton D, Sørensen HT. Benign thyroid diseases and risk of thyroid cancer: a nationwide cohort study. J Clin Endocrinol Metab. 2018;103:2216–24.
Carling T, Udelsman R. Thyroid cancer. Annu Rev Med. 2014;65:125–37.
Nikiforov YE, Nikiforova MN. Molecular genetics and diagnosis of thyroid cancer. Nat Rev Endocrinol. 2011;7:569–80.
Fagin JA, Krishnamoorthy GP, Landa I. Pathogenesis of cancers derived from thyroid follicular cells. Nat Rev Cancer. 2023;23:631–50.
Gou Q, Gao L, Nie X, Pu W, Zhu J, Wang Y, et al. Long noncoding RNA AB074169 inhibits cell proliferation via modulation of KHSRP-mediated CDKN1a expression in papillary thyroid carcinoma. Cancer Res. 2018;78:4163–74.
Antonello ZA, Nucera C. Orthotopic mouse models for the preclinical and translational study of targeted therapies against metastatic human thyroid carcinoma with BRAF(V600E) or wild-type BRAF. Oncogene. 2014;33:5397–404.
Nucera C, Nehs MA, Mekel M, Zhang X, Hodin R, Lawler J, et al. A novel orthotopic mouse model of human anaplastic thyroid carcinoma. Thyroid. 2009;19:1077–84.
Goretzki PE, Frilling A, Simon D, Roeher HD. Growth regulation of normal thyroids and thyroid tumors in man. Recent Results Cancer Res. 1990;118:48–63.
Yeh MW, Rougier JP, Park JW, Duh QY, Wong M, Werb Z, et al. Differentiated thyroid cancer cell invasion is regulated through epidermal growth factor receptor-dependent activation of matrix metalloproteinase (MMP)-2/gelatinase A. Endocr Relat Cancer. 2006;13:1173–83.
Paget S. The distribution of secondary growths in cancer of the breast. 1889. Cancer Metastasis Rev. 1989;8:98–101.
Miller HE, Bishop AJR. Correlation AnalyzeR: functional predictions from gene co-expression correlations. BMC Bioinform. 2021;22:206.
Zhang M, Weng W, Zhang Q, Wu Y, Ni S, Tan C, et al. The lncRNA NEAT1 activates Wnt/beta-catenin signaling and promotes colorectal cancer progression via interacting with DDX5. J Hematol Oncol. 2018;11:113.
Zheng H, Chen C, Luo Y, Yu M, He W, An M, et al. Tumor-derived exosomal BCYRN1 activates WNT5A/VEGF-C/VEGFR3 feedforward loop to drive lymphatic metastasis of bladder cancer. Clin Transl Med. 2021;11:e497.
Wang X, Hu H, Liu H. RNA binding protein Lin28B confers gastric cancer cells stemness via directly binding to NRP-1. Biomed Pharmacother. 2018;104:383–9.
Cao Y, Geng J, Wang X, Meng Q, Xu S, Lang Y, et al. RNA-binding motif protein 10 represses tumor progression through the Wnt/beta- catenin pathway in lung adenocarcinoma. Int J Biol Sci. 2022;18:124–39.
Riesco-Eizaguirre G, Santisteban P. Molecular biology of thyroid cancer initiation. Clin Transl Oncol. 2007;9:686–93.
Xing M. Molecular pathogenesis and mechanisms of thyroid cancer. Nat Rev Cancer. 2013;13:184–99.
Garcia-Rostan G, Tallini G, Herrero A, D’Aquila TG, Carcangiu ML, Rimm DL. Frequent mutation and nuclear localization of beta-catenin in anaplastic thyroid carcinoma. Cancer Res. 1999;59:1811–5.
Ishigaki K, Namba H, Nakashima M, Nakayama T, Mitsutake N, Hayashi T, et al. Aberrant localization of beta-catenin correlates with overexpression of its target gene in human papillary thyroid cancer. J Clin Endocrinol Metab. 2002;87:3433–40.
Sastre-Perona A, Riesco-Eizaguirre G, Zaballos MA, Santisteban P. beta-catenin signaling is required for RAS-driven thyroid cancer through PI3K activation. Oncotarget. 2016;7:49435–49.
Cross DA, Alessi DR, Cohen P, Andjelkovich M, Hemmings BA. Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature. 1995;378:785–9.
Lee G, Goretsky T, Managlia E, Dirisina R, Singh AP, Brown JB, et al. Phosphoinositide 3-kinase signaling mediates beta-catenin activation in intestinal epithelial stem and progenitor cells in colitis. Gastroenterology. 2010;139:869–81: 881.e861-869.
Sastre-Perona A, Santisteban P. Wnt-independent role of beta-catenin in thyroid cell proliferation and differentiation. Mol Endocrinol. 2014;28:681–95.
Bikkavilli RK, Malbon CC. Dishevelled-KSRP complex regulates Wnt signaling through post-transcriptional stabilization of beta-catenin mRNA. J Cell Sci. 2010;123:1352–62.
Bae JA, Bae WK, Kim SJ, Ko YS, Kim KY, Park SY, et al. A new KSRP-binding compound suppresses distant metastasis of colorectal cancer by targeting the oncogenic KITENIN complex. Mol Cancer. 2021;20:78.
Gherzi R, Trabucchi M, Ponassi M, Ruggiero T, Corte G, Moroni C, et al. The RNA-binding protein KSRP promotes decay of beta-catenin mRNA and is inactivated by PI3K-AKT signaling. PLoS Biol. 2006;5:e5.
Nicastro G, Garcia-Mayoral MF, Hollingworth D, Kelly G, Martin SR, Briata P, et al. Noncanonical G recognition mediates KSRP regulation of let-7 biogenesis. Nat Struct Mol Biol. 2012;19:1282–6.
Michlewski G, Caceres JF. Antagonistic role of hnRNP A1 and KSRP in the regulation of let-7a biogenesis. Nat Struct Mol Biol. 2010;17:1011–8.
Acknowledgements
We thank the National RNAi Core Facility at Academia Sinica, Taiwan, for providing shRNA reagents and relevant services.
Funding
This study was supported by the National Science and Technology Council, Taiwan (grant number: 111-2628-B-002-023-MY3), Wan Fang Hospital (grant number: 112-wf-eva-04, 113-wf-swf-06), and Far Eastern Memorial Hospital (grant number: 114-FTN0014).
Author information
Authors and Affiliations
Contributions
Ke-Fan Pan: Study design and Writing - Original draft preparation. Han-Lin Chou: Methodology and Investigation. Wei-Li Wang: Investigation. Bo-Rong Chen: Investigation. Michael Hsiao: Resources. Kuo-Tai Hua: Writing – Review & editing, Supervision, and Project administration. Ming-Hsun Wu: Supervision and Project administration. All authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
This study was approved by the Institutional Review Board of National Taiwan University Hospital (institutional approval number: 201402068RINA), and written informed consent was obtained from all study participants enrolled, and all participants consented to participate in this work. Ethics approval for the animal studies was obtained from the Institutional Animal Care and Use Committee of National Taiwan University’s College of Medicine and College of Public Health (approval number: 20150359), and all procedures were performed in accordance with established guidelines.
Consent for publication
All authors were aware of the submission of the present study, and patients signed informed consent regarding publishing their data.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pan, KF., Chou, HL., Wang, WL. et al. KSRP-mediated Wnt/β-catenin activation promotes follicular thyroid cancer progression and stemness. Br J Cancer 133, 1111–1121 (2025). https://doi.org/10.1038/s41416-025-03142-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-025-03142-x