Abstract
Introduction
Selective internal radiation therapy (SIRT) and sorafenib are used for patients with liver metastases from uveal melanoma. We conducted a phase I study to determine the optimal timing of administration of sorafenib in combination with radioembolisation.
Methods
Patients received radioembolisation plus sorafenib initiated 14, 11, or 3 days after or 7 days before radioembolisation. The primary endpoint was safety. Secondary endpoints included efficacy and evaluation of circulating angiogenic factors. Sorafenib plasma levels were quantified retrospectively.
Results
Ten patients received radioembolisation plus sorafenib. The most common grade 2–4 adverse events included rash, abdominal pain, fatigue and lymphocytopenia. Best response was partial response (30%) and stable disease (70%). Median progression-free and overall survival were 6.2 (4.8–not reached) and 11.7 months (9.4–not reached), respectively. Circulating angiogenic factors showed transient elevation at Day 10 post-SIRT when sorafenib was started 11–14 days after SIRT, but not when sorafenib was started before or just after SIRT. Most steady-state sorafenib plasma concentrations were in the upper percentiles relative to reference curves for single-agent sorafenib.
Conclusions
SIRT plus sorafenib was feasible, but appeared more toxic than sorafenib alone, presumably due to altered pharmacokinetics. Sorafenib may hinder the angiogenic response when initiated before or shortly after SIRT.
Clinical trial registration
Registry: ClinicalTrials.gov, NCT01893099.
KeyPoints
-
Phase I trial testing optimal timing of sorafenib with SIRT for patients with MUM.
-
Most common DLTs were grade 3/4 rash (60%) and grade 3 lymphopenia (60%).
-
Best response per RECIST was partial response in 30% and stable disease in 70%.
-
Initiation of sorafenib before or soon after SIRT blunted the angiogenic response.
-
Sorafenib plasma levels were higher vs historical data for single-agent sorafenib.
Similar content being viewed by others
Introduction
Uveal melanoma is the most common primary intraocular malignancy in adults and accounts for 3–5% of melanomas [1, 2]. Approximately 30–50% of patients ultimately develop metastases, with liver involvement in >90% of cases [3,4,5,6]. Liver metastases are an important cause of morbidity and mortality, leading to poor overall survival (OS) of <9 months in historical series [7].
The only approved systemic treatment for metastatic uveal melanoma (MUM) is tebentafusp, a bispecific T-cell engager that prolongs OS in patients with the HLA-A*02:01 genotype [1, 8]. Unlike cutaneous melanomas, most uveal melanomas are characterised by low tumour mutational burden and alterations in GNAQ/GNA11 [9]. Limited efficacy has been observed in MUM with established therapies for cutaneous melanoma, including BRAF/MEK inhibitors, immune checkpoint inhibitors, and chemotherapy [9]. Angiogenesis is involved in the pathophysiology of MUM and the development of liver metastases, especially those with a nodular growth pattern [10, 11].
Both angiogenesis and aberrant mitogen-activated protein kinase (MAPK) signalling are targeted by sorafenib, a small-molecule, multikinase inhibitor of VEGFRs, PDGFR-β, FLT3, c-KIT, CRAF and BRAF [12]. In a phase II trial in MUM, sorafenib provided disease control in 31% of patients at 24 weeks [13]. Sunitinib, another multitarget tyrosine kinase and angiogenesis inhibitor, also showed clinical benefit in patients with MUM [14].
Given the prevalence and clinical significance of liver metastases, patients with predominantly hepatic involvement often undergo liver-directed therapies, including surgery, ablative treatment, selective internal radiation therapy (SIRT; radioembolisation), chemoembolisation, immunoembolisation or intra-arterial hepatic chemotherapy [15, 16]. In a systematic review of retrospective and prospective studies in 268 patients, SIRT using 90Y-microspheres achieved a disease control rate of 67.5%, with a median PFS of 5.5 months and median OS of 12.3 months [17].
A rationale for combining SIRT with sorafenib lies in the role of MAPK activation and angiogenesis in radiotherapy resistance. Additionally, the potential for vascular normalisation under antiangiogenic therapy may enhance radiosensitivity and improve local SIRT delivery [18,19,20]. Sorafenib has demonstrated a radiosensitising effect in preclinical models of various tumours, including melanoma, which was schedule-dependent [21,22,23]. No interventional trial has yet evaluated SIRT combined with sorafenib in patients with MUM, and the optimal schedule of administration is unknown.
This phase I trial was conducted to evaluate the safety and tolerability of different schedules of SIRT with 90Y-microspheres plus sorafenib in patients with MUM and liver metastases. We also assessed circulating angiogenic factors and the pharmacokinetics of sorafenib when combined with SIRT.
Methods
Trial design
This was a four-cohort, single-centre, non-randomised, single-arm, phase I trial of SIRT plus sorafenib (ClinicalTrials.gov, NCT01893099). Sorafenib was initiated 14 days after SIRT (Cohort 1), 11 days after SIRT (Cohort 2), 3 days after SIRT (Cohort 3), and 7 days before SIRT (Cohort 4) (Supplementary Fig. 1). The design was based on a traditional ‘3 + 3’ dose escalation scheme, with a planned enrolment of 3–6 patients per cohort. Among the first three patients per cohort, the third was enrolled once the first two had completed the dose-limiting toxicity (DLT) evaluation period. If fewer than two DLTs were observed in the first three patients, three more patients could be included in the cohort.
The trial was conducted in accordance with the Declaration of Helsinki, local regulations, and the International Council for Harmonisation’s Good Clinical Practice guidelines. The protocol was approved by the institutional ethics committee, and all patients provided written informed consent.
Patients
Eligible adults (>18 years) with histologically confirmed uveal melanoma and measurable liver metastases based on RECIST v1.1 were included if they had an Eastern Cooperative Oncology Group performance status 0–1, life expectancy ≥12 weeks, and adequate hepatic, renal, and bone marrow function. Concomitant extrahepatic metastases were permitted if not imminently life-threatening. Exclusion criteria included history of cardiac disease, uncontrolled hypertension, HIV infection, chronic hepatitis B or C, organ allograft, active serious infections, symptomatic brain or meningeal metastases, evidence or history of bleeding diathesis and renal dialysis. Prior antiangiogenic therapy was not allowed, and prior chemotherapy, immunotherapy, or major surgery had to be completed ≥4 weeks before starting study treatment.
Treatment
All patients received SIRT (90Y-loaded resin microspheres; SIR-Spheres®; Sirtex Medical, Sydney, Australia) plus sorafenib (Nexavar®; Bayer Healthcare Pharmaceuticals, Berlin, Germany). Before SIRT, patients underwent hepatic and visceral angiography, with 99mTc-macroaggregated albumin (MAA) whole-body scintigraphy and liver-centred single-photon emission computed tomography with computed tomography (SPECT/CT) for dosimetry planning and assessment of lung shunt fraction. Coil embolisation was performed before SIRT to prevent gastrointestinal leak. SIRT was delivered in a single session on Day 1 to one or both liver lobes via a femoral arterial approach using a catheter guided into the hepatic arteries. SIRT activity was calculated from body surface area or the partition model based on 99mTc-MAA SPECT/CT, and was optimised to deliver >100 Gy absorbed dose to the tumour and <20 Gy to normal perfused liver [24]. Post-SIRT dosimetry was computed using 90Y PET. Sorafenib 400 mg was administered twice daily (BID), orally, until disease progression or intolerance.
Assessments
Clinical assessments were performed at baseline, weekly until Day 30, and monthly thereafter. 18F-fluorodeoxyglucose (FDG)-PET/CT was conducted at baseline, at week 8, and then every 12 weeks until progression. The response was assessed according to RECIST and PERCIST [25, 26]. Adverse events (AEs) were evaluated according to the National Cancer Institute Common Terminology Criteria for Adverse Events Version 4.0 (May 2009). DLTs were defined as any grade 3/4 AEs occurring within the first 30 days after SIRT, except for procedure-related pain and grade 3 alanine aminotransferase or aspartate aminotransferase elevation.
Sorafenib plasma levels were quantified using a validated high-performance liquid chromatography-tandem mass spectrometry method [27], with lower limits of quantification and detection of 10 and 1 ng/mL, respectively.
Translational research
Plasma cytokines measurements were done in duplicate at the Center for Integrative Genomics, Faculty of Biology and Medicine, University of Lausanne, using commercially available kit from Sigma Aldrich Human angiopoietin (RAB0015-1KT), Merck Millipore Thrombospondin1 Human Sepsis Immune Response (HSP3MAG-63K), Human Angiogenesis Panel 2, sVEGFR1, sVEGFR2, Angiostatin, Thrombospondin-2 (HANG2MAG-12K-04), Biorad - Bioplex Pro Human Cancer1: FGF-basic (171BC502M), sTIE-2 (171BC514M), PDGF-AB BB (171BC511M), Pro Human Cancer2: Angiopoietin-2 (171BC601M),VEGF-A (171BC616M). Eleven angiogenic factors were evaluated in plasma samples collected at baseline and on Days 2, 3, 7, 10, 14, 17, 24, 31, and 60. These were categorised as proangiogenic (FGF-basic, VEGF-A, PDGF-AB/BB, angiopoetin-2), antiangiogenic (angiostatin, thrombospondin [TSP]-1/2), and other factors (angiopoetin-1, soluble VEGF receptor [sVEGFR]1/2 and soluble TIE-2).
Statistics
The primary endpoint was safety. Secondary endpoints included PFS and OS, analysed by Kaplan–Meier methods using the R library ‘survival’ (https://doi.org/10.1002/sim.956). Safety and efficacy outcomes were summarised descriptively without formal hypothesis testing. Associations between steady state (ss) sorafenib plasma concentrations and safety and efficacy were retrospectively evaluated.
Given limited cohort size and similarity of results between cohorts, Cohorts 1 and 2 were analysed together as the ‘delayed sorafenib introduction cohorts’ (DIC), and Cohorts 3 and 4 as the ‘early sorafenib introduction cohorts’ (EIC) for selected endpoints. This approach allowed us to better assess differences according to variations in sorafenib administration schedules. Angiogenic factors were analysed individually or grouped as proangiogenic, antiangiogenic, or other factors. To cluster time-series data, we first utilised the dynamic time warping algorithm implemented in the R package ‘TSdist’ (https://doi.org/10.32614/RJ-2016-058), which measures the similarity between two temporal sequences by calculating the distances between different time-series. These computed distances were then clustered using the average hierarchical clustering algorithm.
Results
Patients and treatment
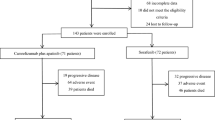
Of 11 patients screened between 2013 and 2016, one was excluded due to hepatic arterial anatomy inappropriate for SIRT. Ten patients were enrolled, with three patients each in Cohorts 1 and 3, and two patients each in Cohorts 2 and 4. The sample size reflects the premature closure of all cohorts due to DLTs.
Patient characteristics are detailed in Table 1. The median time to metastases was 25 months (range 3–133). The extent of liver involvement was <25% in all patients and two patients had unilobar disease. Three patients (30.0%) had minimal extrahepatic disease. Prior treatments included adjuvant fotemustine and percutaneous ablation of three liver metastases, each in one patient (10.0%).
All patients received SIRT and sorafenib. The median activity of SIRT administered was 1.4 GBq (range, 1.0–2.8). SIRT administration was bilobar in six patients (60.0%) and unilobar in four patients (40.0%). Among the patients with unilobar administration, one patient had unilateral lobe involvement, and two had minor tumor infiltration in the untreated lobe. The fourth patient had bilobar involvement, but treatment of the less-involved left lobe was postponed by 8 months due to pain during the initial procedure. Post 90Y SIRT dosimetry showed a median (range) estimated absorbed dose of 149.4 Gy (47.3–356) and 40.3 Gy (25.8–49) for tumour and non-tumour perfused liver tissue, respectively. The median duration of sorafenib treatment was 71 days (range, 11–780). Sorafenib was discontinued due to AEs in six patients (60.0%) and disease progression in four patients (40.0%).
Safety
DLTs were reported in all patients with no apparent difference between cohorts. The most frequent clinical DLT was rash, which was reported in six patients (60.0%) (grade 3, five patients [50.0%]; grade 4, one patient [10.0%]). The most common laboratory DLT was lymphocyte count decreased, also reported in six patients (60.0%, all grade 3). Although rash was easily managed with dose interruption or reduction, and the lymphocyte count decreases were not considered clinically relevant, these DLTs prevented cohort expansion.
Across the entire study period, the most common grade 2–4 AEs were abdominal pain (nine patients; 90.0%), rash (six patients; 60.0%), fatigue (five patients; 50.0%), and weight loss (four patients; 40.0%). The most frequent laboratory abnormalities were lymphocyte count decreased (six patients; 60.0%) and gamma-glutamyl transferase increased (four patients; 40.0%). Table 2 shows the total number of grade 2–4 AE episodes reported during the entire study period.
Early SIRT-related toxicity included transient procedure-associated abdominal pain in six patients (60.0%). Delayed toxicities causing pain and considered likely related to SIRT included two episodes of gastric ulcers and one of duodenitis, where microspheres were found in subsequent biopsies.
The sorafenib dose was reduced in six patients (60.0%), all due to AEs occurring during the DLT evaluation period. Six patients (60.0%) discontinued sorafenib due to skin toxicity (four patients; 40.0%), fatigue and anorexia (one patient; 10.0%), and epigastric pain (one patient; 10.0%). No treatment-related deaths occurred.
Response and survival
Best responses per RECIST were partial response in three patients (30.0%; one each in Cohorts 1–3) and stable disease in seven patients (70.0%; Suppl. Table 1). Two patients were not evaluable per PERCIST due to absence of FDG uptake by liver metastases. Among the remaining eight patients, the best response was a complete metabolic response in one patient (12.5%; Cohort 1; Suppl. Figure 2) and partial metabolic response in three patients (37.5%; one each in Cohorts 1–3) for a metabolic response rate of 50.0%. Stable metabolic disease was reported in four patients (50.0%; Suppl. Table 1). Among the patients with a metabolic response, two had partial response and two had stable disease according to RECIST criteria.
The first sites of progression were hepatic-only in five patients (50.0%), hepatic plus extrahepatic in four patients (40.0%; bone in three patients, lung in one patient), and extrahepatic-only in one patient (10.0%; lymph node).
Median (95% confidence interval [CI]) PFS was 6.2 months (4.8–not reached [nr]) in the overall population, 8.2 months (5.9–nr) in the EIC (n = 5), and 5.3 months (4.8–nr) in the DIC (n = 5) (Fig. 1A). Median OS (95% CI) was 11.7 months (9.4–nr), 20 months (11.0–nr), and 10.8 months (7.9–nr), respectively (Fig. 1B).
a PFS; b OS. PFS progression-free survival, OS overall survival, EIC early sorafenib introduction cohort, DIC delayed sorafenib introduction cohort.
Angiogenic factors
In the DIC, most angiogenic factors showed a biphasic pattern, with a 46% increase from baseline at Day 10 after SIRT across all factors (log2 fold-change [log2FC] 0.55; p = 0.045), followed by a decrease to baseline 7–10 days after sorafenib introduction (Day 24: −16%, log2FC 0.24, p = 0.25; Day 60: 2%, log2FC 0.03, p = 1) (Fig. 2A). PDGF-AB/BB and TSP-1 showed the most pronounced changes, with increases of 4.0-fold (log2FC 2.0; p = 0.33) and 5.3-fold (log2FC 2.4; p = 0.3), respectively, at Day 10. Unlike most other factors, TSP-2 increased soon after SIRT and remained elevated, while angiostatin remained unchanged (Fig. 3A).
Log2 fold-change in plasma levels of antiangiogenic, proangiogenic and other factors in the DIC (a) and EIC (b). DIC delayed sorafenib introduction cohort, EIC early sorafenib introduction cohort, SIRT selective internal radiation therapy.
Log2 fold-change in plasma levels in the DIC (a) and EIC (b). DIC delayed sorafenib introduction cohort, EIC early sorafenib introduction cohort, SIRT selective internal radiation therapy.
Compared with the DIC, the angiogenic response after SIRT in the EIC appeared blunted, with no biphasic pattern or increase in angiogenic factors observed (Fig. 2B). Angiogenic factors showed a small decrease of 15% at Day 10 (log2FC −0.23, p = 0.33) and 39% at Day 24 (log2FC −0.71; p = 0.032) across all factors. The two factors most affected were PDGF-AB/BB and TSP-1, which decreased at Day 10 (PDGF-AB/BB: log2FC −0.38; p = 0.45; TSP-1: log2FC −0.36; p = 0.4) and Day 24 (PDGF-AB/BB: log2FC −1.65; p = 0.01; TSP-1: log2FC −1.78; p = 0.009). Of note, one value for both factors was affected by a preanalytical or transcription error at Day 30. As in the DIC, TSP-2 increased soon after SIRT and remained elevated (Fig. 3B).
Based on clustering analysis, within each cohort, the proangiogenic factors FGF-basic, VEGF-A, and angiopoietin-2 showed a similar response profile, whereas the response of antiangiogenic factors diverged. In both cohorts, close clustering of TSP-1 and PDGF-AB/BB was observed, supporting their direct interaction, whereas TSP-2 was separated from the other factors (Supplementary Fig. 3).
Sorafenib plasma levels
Given that sorafenib is metabolised by the liver and that SIRT can cause transient liver function impairment, we explored whether an increase in sorafenib plasma concentrations post-SIRT could account for the high rate of DLTs and skin toxicity. Of 41 plasma samples collected during the DLT evaluation period, eight were not collected at steady-state (achieved after 7 days at a constant sorafenib dose) and were excluded from analyses, although the concentrations for the eight samples were within the range of steady-state level observed for the respective patient.
In the 33 remaining steady-state plasma samples (median per patient, 2.5; range, 1–8), the median sorafenib concentration was 6963 ng/mL (range, 2517–19’829). Most values were in the upper range of, or higher than, pharmacokinetic curves for sorafenib reported in the literature. Specifically, >50% of patients had concentrations above the 75th percentile, as reported by Jain et al. (Fig. 4) [28], and 20% above the 90th percentile, as reported by Inaba et al. [29]. Although sorafenib concentrations showed considerable inter-individual and inter-cohort variation, no statistically significant difference was observed between the DIC and EIC. Patients in Cohort 4, who initiated sorafenib 7 days before SIRT, had values in the range of the reference literature, with a median of 4741 ng/mL (range, 2517–7744). In contrast, patients who initiated sorafenib after SIRT had higher values, with median (range) concentrations of 9907 ng/mL (3508–19,829) overall, 8315 ng/mL (4307–12,859) in Cohort 1, 5720 ng/mL (4208–11,311) in Cohort 2, and 11,485 ng/mL (3508–19,829) in Cohort 3.
Plasma concentrations from patients treated with SIRT plus sorafenib are represented by circles (where the time after dose is known) or diamonds (where the time after dose in not known, arbitrarily shown mid-dosing interval). For graphical purposes, two samples with high levels of sorafenib ( > 19,000 ng/mL) are not shown. Pharmacokinetic curves were retrieved and adapted from Jain et al. for five patients receiving sorafenib 400 mg BID (blue) or 200 mg BID (red), with solid lines indicating the median and broken lines indicating the 25th and 75th percentiles. Data points shown at 12 h correspond to any trough levels measured at 12 or 24 h. BID twice daily, SIRT selective internal radiation therapy.
Geometric mean steady-state concentrations were significantly higher in patients with hand-foot skin reaction versus those without hand–foot skin reaction (HFSR; all grade 1; p = 0.017; Fig. 5), but were not associated with other AEs (DLTs, or grade ≥2 rash or dermatologic events) (Supplementary Fig. 4), or PFS (data not shown). In this small cohort, we did not detect any hepatic toxicity attributable to either SIRT or sorafenib.
*p = 0.0167 by the Mann–Whitney test. Red diamond represent one patient with Grade 1 progressing to Grade 3 HFSR. HFSR hand-foot skin reaction. ss steady state.
In this exploratory analysis, mean sorafenib plasma steady-state levels were not correlated with the absolute level or change in levels of angiogenic factors in the DIC or EIC, even for factors presumably affected by sorafenib, like PDGF-AB/BB and VEGF-A (data not shown).
Discussion
To our knowledge, we report the first trial of SIRT plus sorafenib in patients with MUM and liver metastases. Administration of this combination was generally feasible, although we observed high rates of DLTs and grade 3/4 AEs, and 60.0% of patients had AEs leading to sorafenib discontinuation. Toxicity was more frequent and severe than reported with SIRT or sorafenib alone in recent studies in MUM [13, 17]. In a prior trial of sorafenib alone, 34.4% patients had grade 3/4 AEs and 41.4% required dose modification. The toxicity in the present study aligns with the distinct safety profiles of the respective modalities, and the significant increase in grade 3/4 AEs observed with SIRT plus sorafenib, compared with sorafenib alone, in a randomized trial in hepatocellular carcinoma (HCC) [30]. The most common grade 3/4 AEs were abdominal pain related to SIRT, along with rash, fatigue and lymphocyte count decrease, which are known AEs of sorafenib [31, 32], but seemed more severe in this setting.
The study provides preliminary evidence for the efficacy of SIRT plus sorafenib, with a response rate of 30.0% by RECIST and 50.0% by PERCIST. The disease control rate was 100% at the first evaluation at 2 months and 60.0% at the second evaluation at 5 months, which compares favourably with reports for SIRT or sorafenib alone in MUM [13, 17]. PFS was similar to that observed with first-line SIRT alone [17, 18], and appeared greater than that obtained with sorafenib monotherapy in prior study that reported a 24-week PFS rate of 31.2% [13].
We observed a median sorafenib plasma level of 6963 ng/mL (range, 2517–19,829). Although not statistically significant, patients who initiated sorafenib after SIRT had higher values than those starting before SIRT. Although no therapeutic window has been defined for sorafenib, >50% of patients had sorafenib plasma levels above the 75th percentile and 20% above the 90th percentile compared with existing population pharmacokinetic curves for 400 mg BID [28, 29], suggesting a modification of sorafenib pharmacokinetics by SIRT. As sorafenib is partially metabolised by the liver (notably by CYP3A4 and glucuronidation by UGT1A9 and UGT1A1 [33]), transient effects of radioembolisation on liver function may explain the higher levels of sorafenib observed within the 30 days post-SIRT. In the SORAMIC trial, sorafenib (200 mg BID for one week followed by 400 mg BID) was initiated three days after SIRT for patients with HCC with preserved liver function (Child-Pugh <7). Blood samples were drawn after a median of 25 weeks, and the mean trough level (Ctrough) of sorafenib was 3217 ng/mL [34].
Elevations in sorafenib plasma concentrations may have exacerbated toxicity in the present study. In the literature, grade 3 toxicity has been correlated with sorafenib Ctrough > 7700 ± 3600 ng/mL [35], and sorafenib plasma levels of >5780 ng/mL and >4780 ng/mL predicted grade 2 HFSR and hypertension, respectively [33]. Correlation between sorafenib plasma levels and HFSR was also shown by Inaba et al. [29]. In our study, most AEs occurred within 8 weeks of treatment initiation, with six patients with grade >1 rash, six patients with grade 1 HFSR, and one patient with grade 3 HFSR. We observed a higher sorafenib plasma level in patients with HFSR compared with patients without this AE (p = 0.017). No patients with a geometric mean of <4100 ng/mL had HFSR events (Fig. 5). No significant difference was seen for other skin toxicities, probably due to the small sample size. Given our findings and the observed safety profile, initiation of sorafenib at a reduced dose of 200 mg BID and therapeutic drug monitoring are potential strategies to optimise sorafenib tolerability in this setting.
Angiogenesis is a complex process that is crucial for tumour growth and metastasis, and is regulated by a balance of pro- and antiangiogenic factors secreted by cancer cells and the tumour microenvironment [36]. Local ablative therapies such as SIRT can induce tumour hypoxia, which in turn increases the synthesis and release of proangiogenic factors, and thus angiogenesis. We observed an increase in plasma levels of most proangiogenic factors during the first two weeks post-SIRT in the DIC, which was reversed by the initiation of sorafenib on Days 11 or 14. PDGF-AB/BB and TSP-1 were among the most affected factors, while VEGF-A, FGF-basic and angiopoietin-2 were less affected. Interestingly, TSP-2 had a distinct response profile, increasing soon after SIRT and remaining elevated. This pattern is consistent with TSP-2 synthesis by both endothelial and stromal cells, such as fibroblasts [37]. Angiostatin, which is generated by proteolysis of plasminogen, was not affected by SIRT in our study.
An angiogenic response after SIRT has been reported in other malignancies [38,39,40]. In a trial in 22 patients with HCC or colorectal cancer (CRC) and liver metastasis treated with SIRT, without sorafenib, plasma level increases from baseline of >50% were observed for VEGF, PDGF, angiopoietin-2, and TSP-1 in most patients [38]. Another study reported increases in VEGF, hepatocyte growth factor (HGF) and angiopoietin-2 soon after SIRT in 42 patients with CRC and liver metastases [39]. Finally, increased levels of VEGF (36%), PDGF (24%), angiopoietin-2 (36%) and HGF (26%) were observed two weeks after SIRT in a study of patients with HCC [40]. These trials also negatively correlated increases in angiogenic factors after SIRT with treatment response [39] and prognosis [40].
Sorafenib appeared to inhibit the initial angiogenic response in the DIC once started on Days 11 or 14, and largely abolished the response when initiated before or shortly after SIRT in the EIC. In a randomised study in HCC, similar results were observed, with decreased levels of angiopoietin-2 and PDGF, without significant effects on VEGF [40]. We observed the strongest effect of sorafenib on PDGF-AB/BB and TSP-1, which are key mediators of vascular smooth muscle cell proliferation and migration. The close association of these two factors in our clustering analysis is consistent with a report showing their tight and specific binding [41]. Given the poor prognostic significance of SIRT-associated increases in VEGF, PDGF-BB and angiopoetin-2 [38, 39], the potential role for antiangiogenic drugs in combination with SIRT to improve outcomes warrants further evaluation.
Study limitations include the lack of SIRT-only control group, small sample size and high inter-individual variability in angiogenic factors. Furthermore, the relevance of differences in plasma levels of angiogenic factors for tumour vascularisation has yet to be determined in the context of SIRT.
Conclusions
SIRT plus sorafenib is feasible for patients with MUM and liver metastases but appeared more toxic than sorafenib alone, presumably due to altered pharmacokinetics. Antiangiogenic drugs hold promise for attenuating the angiogenic response and hence enhancing SIRT efficacy, particularly when initiated before or shortly after SIRT.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Rodriguez-Vidal C, Fernandez-Diaz D, Fernandez-Marta B, Lago-Baameiro N, Pardo M, Silva P, et al. Treatment of Metastatic Uveal Melanoma: Systematic Review. Cancers (Basel). 2020;12:1–22.
Rossi E, Croce M, Reggiani F, Schinzari G, Ambrosio M, Gangemi R, et al. Uveal Melanoma Metastasis. Cancers (Basel). 2021;13:5684.
Willson JKV, Albert DM, Diener-West M, McCaffrey L, Mo CS, Scully RE, et al. Assessment of Metastatic Disease Status at Death in 435 Patients With Large Choroidal Melanoma in the Collaborative Ocular Melanoma Study (COMS): COMS Report No. 15. Arch Ophthalmol. 2001;119:670–6.
Kujala E, Mäkitie T, Kivelä T. Very long-term prognosis of patients with malignant uveal melanoma. Invest Ophthalmol Vis Sci. 2003;44:4651–9.
Diener-West M, Reynolds SM, Agugliaro DJ, Caldwell R, Cumming K, Earle JD, et al. Development of Metastatic Disease After Enrollment in the COMS Trials for Treatment of Choroidal Melanoma: Collaborative Ocular Melanoma Study Group Report No. 26. Arch Ophthalmol. 2005;123:1639–43.
Garg G, Finger PT, Kivelä TT, Simpson R, Gallie BL, Saakyan S, et al. Patients presenting with metastases: stage IV uveal melanoma, an international study. Br J Ophthalmol. 2022;106:510–7.
Gonsalves CF, Adamo RD, Eschelman DJ. Locoregional Therapies for the Treatment of Uveal Melanoma Hepatic Metastases. Semin Interv Radio. 2020;37:508–17.
Hassel JC, Piperno-Neumann S, Rutkowski P, Baurain JF, Schlaak M, Butler MO, et al. Three-Year Overall Survival with Tebentafusp in Metastatic Uveal Melanoma. N Engl J Med. 2023;389:2256–66.
Jager MJ, Shields CL, Cebulla CM, Abdel-Rahman MH, Grossniklaus HE, Stern MH, et al. Uveal melanoma. Nat Rev Dis Prim. 2020;6:24.
Yang H, Jager MJ, Grossniklaus HE. Bevacizumab Suppression of Establishment of Micrometastases in Experimental Ocular Melanoma. Invest Ophthalmol Vis Sci. 2010;51:2835–42.
Grossniklaus HE, Zhang Q, You S, McCarthy C, Heegaard S, Coupland SE. Metastatic ocular melanoma to the liver exhibits infiltrative and nodular growth patterns. Hum Pathol. 2016;57:165–75.
Wilhelm SM, Adnane L, Newell P, Villanueva A, Llovet JM, Lynch M. Preclinical overview of sorafenib, a multikinase inhibitor that targets both Raf and VEGF and PDGF receptor tyrosine kinase signaling. Mol Cancer Ther. 2008;7:3129–40.
Mouriaux F, Servois V, Parienti JJ, Lesimple T, Thyss A, Dutriaux C, et al. Sorafenib in metastatic uveal melanoma: efficacy, toxicity and health-related quality of life in a multicentre phase II study. Br J Cancer. 2016;115:20–4.
Mahipal A, Tijani L, Chan K, Laudadio M, Mastrangelo MJ, Sato T. A pilot study of sunitinib malate in patients with metastatic uveal melanoma. Melanoma Res. 2012;22:440–6.
Gonsalves CF. Immunoembolization for the Treatment of Uveal Melanoma Hepatic Metastases. Semin Interv Radio. 2024;41:20–26.
Höppener DJ, Grünhagen DJ, Eggermont AMM, van der Veldt AAM, Verhoef C. An Overview of Liver Directed Locoregional Therapies. Hematol Oncol Clin North Am. 2025;39:103–23.
Alexander H, Wen D, Chu M, Han C, Hadden P, Thomas R, et al. Selective internal radiation therapy for hepatic metastases of uveal melanoma: a systematic review. Br J Radio. 2022;95:20210200.
Munshi A, Ramesh R. Mitogen-Activated Protein Kinases and Their Role in Radiation Response. Genes Cancer. 2013;4:401–8.
Kanthou C, Tozer G. Targeting the vasculature of tumours: combining VEGF pathway inhibitors with radiotherapy. Br J Radio. 2019;92:20180405.
Rana N, Ju AW, Bazylewicz M, Kallakury B, He AR, Unger KR, et al. Yttrium-90 radioembolization in patients with hepatocellular carcinoma who have previously received sorafenib. Front Oncol. 2013;3:323.
Winkler F, Kozin SV, Tong RT, Chae SS, Booth MF, Garkavtsev I, et al. Kinetics of vascular normalization by VEGFR2 blockade governs brain tumor response to radiation: Role of oxygenation, angiopoietin-1, and matrix metalloproteinases. Cancer Cell. 2004;6:553–63.
Plastaras JP, Kim SH, Liu YY, Dicker DT, Dorsey JF, McDonough J, et al. Cell cycle dependent and schedule-dependent antitumor effects of sorafenib combined with radiation. Cancer Res. 2007;67:9443–54.
Suen AW, Galoforo S, Marples B, McGonagle M, Downing L, Martinez AA, et al. Sorafenib and radiation: a promising combination in colorectal cancer. Int J Radiat Oncol Biol Phys. 2010;78:213–20.
Liu D, Westcott M, Garcia-Monaco R, Abraham R, Gandhi R. Down and dirty with dosimetry. Endovasc Today. 2016;15:70–6.
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228–47.
Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: Evolving Considerations for PET response criteria in solid tumors. J Nucl Med. 2009;50:122S–50S.
Haouala A, Zanolari B, Rochat B, Montemurro M, Zaman K, Duchosal MA, et al. Therapeutic Drug Monitoring of the new targeted anticancer agents imatinib, nilotinib, dasatinib, sunitinib, sorafenib and lapatinib by LC tandem mass spectrometry. J Chromatogr B Anal Technol Biomed Life Sci. 2009;877:1982–96.
Jain L, Woo S, Gardner ER, Dahut WL, Kohn EC, Kummar S, et al. Population pharmacokinetic analysis of sorafenib in patients with solid tumours. Br J Clin Pharm. 2011;72:294–305.
Inaba H, Panetta JC, Pounds SB, Wang L, Li L, Navid F, et al. Sorafenib Population Pharmacokinetics and Skin Toxicities in Children and Adolescents with Refractory/Relapsed Leukemia or Solid Tumor Malignancies. Clin Cancer Res. 2019;25:7320–30.
Ricke J, Klümpen HJ, Amthauer H, Bargellini I, Bartenstein P, de Toni EN, et al. Impact of combined selective internal radiation therapy and sorafenib on survival in advanced hepatocellular carcinoma. J Hepatol. 2019;71:1164–74.
Li Y, Gao ZH, Qu XJ. The Adverse Effects of Sorafenib in Patients with Advanced Cancers. Basic Clin Pharm Toxicol. 2015;116:216–21.
Schutz FAB, Je Y, Choueiri TK. Hematologic toxicities in cancer patients treated with the multi-tyrosine kinase sorafenib: A meta-analysis of clinical trials. Crit Rev Oncol Hematol. 2011;80:291–300.
Fukudo M, Ito T, Mizuno T, Shinsako K, Hatano E, Uemoto S, et al. Exposure-toxicity relationship of sorafenib in Japanese patients with renal cell carcinoma and hepatocellular carcinoma. Clin Pharmacokinet. 2014;53:185–96.
Labeur TA, Hofsink Q, Takkenberg RB, van Delden OM, Mathôt RAA, Schinner R, et al. The value of sorafenib trough levels in patients with advanced hepatocellular carcinoma - a substudy of the SORAMIC trial. Acta Oncol. 2020;59:1028–35.
Blanchet B, Billemont B, Cramard J, Benichou AS, Chhun S, Harcouet L, et al. Validation of an HPLC-UV method for sorafenib determination in human plasma and application to cancer patients in routine clinical practice. J Pharm Biomed Anal. 2009;49:1109–14.
Liu ZL, Chen HH, Zheng LL, Sun LP, Shi L. Angiogenic signaling pathways and anti-angiogenic therapy for cancer. Signal Transduct Target Ther. 2023;8:198.
Streit M, Riccardi L, Velasco P, Brown LF, Hawighorst T, Bornstein P, et al. Thrombospondin-2: a potent endogenous inhibitor of tumor growth and angiogenesis. Proc Natl Acad Sci USA. 1999;96:14888–93.
Carpizo DR, Gensure RH, Yu X, Gendel VM, Greene SJ, Moore DF, et al. Pilot study of angiogenic response to yttrium-90 radioembolization with resin microspheres. J Vasc Inter Radio. 2014;25:297–306.
Rosenbaum CE, van den Hoven AF, Braat MN, Koopman M, Lam MG, Zonnenberg BA, et al. Yttrium-90 radioembolization for colorectal cancer liver metastases: a prospective cohort study on circulating angiogenic factors and treatment response. EJNMMI Res. 2016;6:92.
Lewandowski RJ, Andreoli JM, Hickey R, Kallini JR, Gabr A, Baker T, et al. Angiogenic Response following Radioembolization: Results from a Randomized Pilot Study of Yttrium-90 with or without Sorafenib. J Vasc Inter Radio. 2016;27:1329–36.
Hogg PJ, Hotchkiss KA, Jiménez BM, Stathakis P, Chesterman CN. Interaction of platelet-derived growth factor with thrombospondin 1. Biochem J. 1997;326:709–16.
Acknowledgements
Medical writing support was provided by Mark Dyson, DPhil (Berlin, Germany). Gilles Willemin, Center for Integrative Genomics, Faculty of Biology and Medicine, University of Lausanne, Lausanne, Switzerland.
Funding
This work was supported by a grant from Bayer Healthcare Pharmaceuticals, Berlin, Germany. SIR-Spheres were provided by Sirtex Medical, Sydney, Australia. This trial was also sponsored by the Gonella Foundation, Lausanne, Switzerland.
Author information
Authors and Affiliations
Contributions
Grégoire Berthod and Serge Leyvraz were responsible for the trial conceptualisation and design, protocol writing, fundraising, data collection and manuscript writing. Christophe Cisarovsky and Tatiana Petrova conducted the methodology of angiogenesis analysis. Dan Celestini and Michel A. Cuendet conducted the statistical analysis and figures. Laurent A. Decosterd and Eva Choong conducted the methodology and statistics of pharmacological analysis and figures. Sarah Boughdad and John O. Prior conducted the SIRT procedure and PET-CT evaluation and response. Jean-Yves Meuwly conducted the radiological response assessment. Olivier Michielin was involved in the patient recruitment and data collection. William D. Figg was involved in the pharmacological data interpretation. All co-authors were responsible for manuscript revision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The trial was approved by the local institutional ethics committee (CER-VD/ Reference number 2013DR1073). Patients provided written informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Berthod, G., Cisarovsky, C., Petrova, T. et al. Sorafenib plus selective internal radiotherapy with 90Y resin microspheres for the treatment of uveal melanoma with liver metastasis: a phase I trial. Br J Cancer 134, 589–597 (2026). https://doi.org/10.1038/s41416-025-03279-9
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-025-03279-9