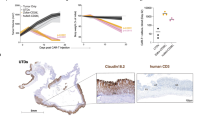
Abstract
Background
Claudin18.2 (CLDN18.2)-specific CAR-T cell therapy has demonstrated promise in advanced gastric cancer (GC). However, the impact of concomitant medications on the efficacy outcomes remains unclear.
Methods
We retrospectively analyzed advanced GC patients receiving CLDN18.2-specific CAR-T cell therapy from a phase I trial. Concomitant medications were defined as any drugs administered within 30 days before and after CAR-T cell infusion, including corticosteroids, antibiotics, tocilizumab, granulocyte colony-stimulating factor (G-CSF), thrombopoietin (TPO), and erythropoietin. Metagenomic sequencing was employed to elucidate the differences in gut microbiome signatures between responders and non-responders.
Results
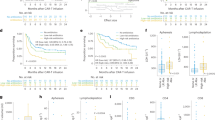
Of 72 patients included in the study, 6 (8.3%) received corticosteroids, 49 (68.1%) received tocilizumab, and 22 (30.6%) received antibiotics, 15 (20.8%) received G-CSF, 5 (6.9%) received thrombopoietin, and no patient received erythropoietin. The median progression-free survival (PFS) (2.6 vs. 5.8 months; P < 0.001) and overall survival (OS) (3.9 vs. 9.5 months; P < 0.001) were significantly shorter for patients who received antibiotics for infection compared to those who did not. No significant differences were observed in objective response rate (ORR), PFS, and OS between patients who received corticosteroids, tocilizumab, antibiotics for prophylaxis, G-CSF, or TPO and those who did not. A higher abundance of Fusobacterium nucleatum, Lactobacillus mucosae, Prevotella pallens, and Streptococcus pseudopneumoniae in gut microbiome was associated with a superior treatment response.
Conclusions
The study indicates that the use of antibiotics for infection reduces the efficacy outcomes of CLDN18.2-specific CAR-T cell therapy for advanced GC, while other concomitant medications do not affect the outcomes. Further research is needed to clarify the optimal administration of these medications and the underlying mechanisms of the gut microbiome in impacting CAR-T treatment response.
Trial registration
NCT03874897
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Requests for individual participant-level data from this study should be submitted via email to the corresponding author with detailed proposals.
References
Nakayama I, Qi C, Chen Y, Nakamura Y, Shen L, Shitara K. Claudin 18.2 as a novel therapeutic target. Nat Rev Clin Oncol. 2024;21:354–69.
Li J, Liu C, Zhang P, Shen L, Qi C. Optimizing CAR T cell therapy for solid tumours: a clinical perspective. Nat Rev Clin Oncol. 2025;22:953–68.
Qi C, Gong J, Li J, Liu D, Qin Y, Ge S, et al. Claudin18.2-specific CAR T cells in gastrointestinal cancers: phase 1 trial interim results. Nature Med. 2022;28:1189–98.
Qi C, Liu C, Gong J, Liu D, Wang X, Zhang P, et al. Claudin18.2-specific CAR T cells in gastrointestinal cancers: phase 1 trial final results. Nature Med. 2024;30:2224–34.
Qi C, Liu C, Peng Z, Zhang Y, Wei J, Qiu W, et al. Claudin-18 isoform 2-specific CART-cell therapy (satri-cel) versus treatment of physician’s choice for previously treated advanced gastric or gastro-oesophageal junction cancer (CT041-ST-01): a randomised, open-label, phase 2 trial. Lancet. 2025;405:2049–60.
Santomasso BD, Nastoupil LJ, Adkins S, Lacchetti C, Schneider BJ, Anadkat M, et al. Management of immune-related adverse events in patients treated with chimeric antigen receptor T-cell therapy: ASCO Guideline. J Clin Oncol. 2021;39:3978–92.
Li J, Yang K, Zhao L, Bai C, Sun Z. Impact of corticosteroids use on efficacy of immune checkpoint inhibitors in cancer patients: A meta-analysis. Journal Clin Oncol. 2020;38:e15234-e.
Strati P, Ahmed S, Furqan F, Fayad LE, Lee HJ, Iyer SP, et al. Prognostic impact of corticosteroids on efficacy of chimeric antigen receptor T-cell therapy in large B-cell lymphoma. Blood. 2021;137:3272–6.
Stein-Thoeringer CK, Saini NY, Zamir E, Blumenberg V, Schubert M-L, Mor U, et al. A non-antibiotic-disrupted gut microbiome is associated with clinical responses to CD19-CAR-T cell cancer immunotherapy. Nature Med. 2023;29:906–16.
Smith M, Dai A, Ghilardi G, Amelsberg KV, Devlin SM, Pajarillo R, et al. Gut microbiome correlates of response and toxicity following anti-CD19 CAR T cell therapy. Nat Med. 2022;28:713–23.
Costa BA, Flynn J, Nishimura N, Devlin SM, Farzana T, Rajeeve S, et al. Prognostic impact of corticosteroid and tocilizumab use following chimeric antigen receptor T-cell therapy for multiple myeloma. Blood Cancer J. 2024;14:84.
Wang X, Zhang B, Zhang Q, Zhou H, Sun Q, Zhou Y, et al. Impact of tocilizumab on anti-CD19 chimeric antigen receptor T-cell therapy in B-cell acute lymphoblastic leukemia. Cancer. 2024;130:2660–9.
Lee DW, Santomasso BD, Locke FL, Ghobadi A, Turtle CJ, Brudno JN, et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol Blood Marrow Transpl. 2019;25:625–38.
Kultima JR, Coelho LP, Forslund K, Huerta-Cepas J, Li SS, Driessen M, et al. MOCAT2: a metagenomic assembly, annotation and profiling framework. Bioinformatics. 2016;32:2520–3.
Martin M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnetjournal. 2011;17:3.
Cox MP, Peterson DA, Biggs PJ. SolexaQA: At-a-glance quality assessment of Illumina second-generation sequencing data. BMC Bioinforma. 2010;11:485.
Li R, Yu C, Li Y, Lam TW, Yiu SM, Kristiansen K, et al. SOAP2: an improved ultrafast tool for short read alignment. Bioinformatics. 2009;25:1966–7.
Segata N, Waldron L, Ballarini A, Narasimhan V, Jousson O, Huttenhower C. Metagenomic microbial community profiling using unique clade-specific marker genes. Nat Methods. 2012;9:811–4.
Luo W, Friedman MS, Shedden K, Hankenson KD, Woolf PJ. GAGE: generally applicable gene set enrichment for pathway analysis. BMC Bioinform. 2009;10:161.
Libert C, Dejager L. How steroids steer T cells. Cell Rep. 2014;7:938–9.
Oluwole OO, Bouabdallah K, Munoz J, De Guibert S, Vose JM, Bartlett NL, et al. Prophylactic corticosteroid use in patients receiving axicabtagene ciloleucel for large B-cell lymphoma. Br J Haematol. 2021;194:690–700.
Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. New Engl J Med. 2017;377:2531–44.
Wang X, Qi Y, Li H, Liu F, Cao J, Chen W, et al. Impact of glucocorticoids on short-term and long-term outcomes in patients with relapsed/refractory multiple myeloma treated with CAR-T therapy. Front. Immunol. 2022;13:943004.
Le RQ, Li L, Yuan W, Shord SS, Nie L, Habtemariam BA, et al. FDA approval summary: tocilizumab for treatment of chimeric antigen receptor T cell-induced severe or life-threatening cytokine release syndrome. Oncologist. 2018;23:943–7.
Si S, Teachey DT. Spotlight on tocilizumab in the treatment of CAR-T-cell-induced cytokine release syndrome: clinical evidence to date. Ther Clin Risk Manag. 2020;16:705–14.
Cortellini A, Tucci M, Adamo V, Stucci LS, Russo A, Tanda ET, et al. Integrated analysis of concomitant medications and oncological outcomes from PD-1/PD-L1 checkpoint inhibitors in clinical practice. J Immunother Cancer. 2020;8:e001361.
Routy B, Le Chatelier E, Derosa L, Duong CPM, Alou MT, Daillere R, et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science. 2018;359:91–7.
Han Z, Cheng S, Dai D, Kou Y, Zhang X, Li F, et al. The gut microbiome affects response of treatments in HER2-negative advanced gastric cancer. Clin Transl Med. 2023;13:e1312.
Wang X, Fang Y, Liang W, Wong CC, Qin H, Gao Y, et al. Fusobacterium nucleatum facilitates anti-PD-1 therapy in microsatellite stable colorectal cancer. Cancer Cell. 2024;42:1729–1746.e8.
Peng Z, Cheng S, Kou Y, Wang Z, Jin R, Hu H, et al. The gut microbiome is associated with clinical response to anti-PD-1/PD-L1 immunotherapy in gastrointestinal cancer. Cancer Immunol Res. 2020;8:1251–61.
Kim CG, Koh JY, Shin SJ, Shin JH, Hong M, Chung HC, et al. Prior antibiotic administration disrupts anti-PD-1 responses in advanced gastric cancer by altering the gut microbiome and systemic immune response. Cell Rep. Med. 2023;4:101251.
Hamada T, Zhang X, Mima K, Bullman S, Sukawa Y, Nowak JA, et al. Fusobacterium nucleatum in colorectal cancer relates to immune response differentially by tumor microsatellite instability status. Cancer Immunol Res. 2018;6:1327–36.
Rejeski K, Jain MD, Shah NN, Perales MA, Subklewe M. Immune effector cell-associated haematotoxicity after CAR T-cell therapy: from mechanism to management. Lancet Haematol. 2024;11:e459–e70.
Miller KC, Johnson PC, Abramson JS, Soumerai JD, Yee AJ, Branagan AR, et al. Effect of granulocyte colony-stimulating factor on toxicities after CAR T cell therapy for lymphoma and myeloma. Blood Cancer J. 2022;12:146.
Clark OA, Lyman GH, Castro AA, Clark LG, Djulbegovic B. Colony-stimulating factors for chemotherapy-induced febrile neutropenia: a meta-analysis of randomized controlled trials. J Clin Oncol. 2005;23:4198–214.
Barreto JN, Bansal R, Hathcock MA, Doleski CJ, Hayne JR, Truong TA, et al. The impact of granulocyte colony stimulating factor on patients receiving chimeric antigen receptor T-cell therapy. Am J Hematol. 2021;96:E399–E402.
Lievin R, Di Blasi R, Morin F, Galli E, Allain V, De Jorna R, et al. Effect of early granulocyte-colony-stimulating factor administration in the prevention of febrile neutropenia and impact on toxicity and efficacy of anti-CD19 CAR-T in patients with relapsed/refractory B-cell lymphoma. Bone Marrow Transpl. 2022;57:431–9.
Acknowledgements
We thank all the participants and their families in this trial.
Funding
This study was funded by National Natural Science Foundation of China (No. 92459302, No. 82522068, and No. U22A20327), National Key Research and Development Program of China (No. 2025YFC3409800, No. 2023YFC3403700, and No. 2022YFA0912400), Beijing Natural Science Foundation (L232080), Beijing Hospitals Authority Youth Program (QMS20201101), Science Foundation of Peking University Cancer Hospital (JC202406), Clinical Medicine Plus X - Young Scholars Project of Peking University, Peking University Clinical Scientist Training Program, Fundamental Research Funds for the Central Universities, and CARsgen Therapeutics Co., Ltd.
Author information
Authors and Affiliations
Contributions
JL, LL, MT, ZH, and MM collected the data. JL drafted the manuscript. JL and ZH performed the statistical analyses. CQ, XZ, and LS conceived and designed the trial. CQ, XZ, and LS reviewed and revised the manuscript. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was approved by the Ethics Committee of Peking University Cancer Hospital (2018YJZ75). The study was performed in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. All patients provided written informed consent before participation in this trial
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, J., Liu, L., Tao, M. et al. Impact of concomitant medications on efficacy of CLDN18.2-specific CAR-T cell therapy in advanced gastric cancer. Br J Cancer 134, 439–446 (2026). https://doi.org/10.1038/s41416-025-03289-7
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41416-025-03289-7