Abstract
Background
Almost half of the patients with gastroesophageal cancer treated with curative intent develop recurrence. It is unknown whether the effectiveness of curative treatment is associated with the outcomes of subsequent first-line systemic therapy.
Methods
From the Netherlands Cancer Registry, we identified patients with metachronous metastatic gastroesophageal adenocarcinoma(mGEA) initially treated for nonmetastatic disease(2015–2017) with perioperative chemotherapy or neoadjuvant chemoradiotherapy, who later received first-line systemic therapy. Effectiveness of the treatment with curative intent was assessed by time-to-treatment-failure(TTF) and by pathological response. First-line systemic therapy outcomes were assessed by TTF and overall survival(OS). Associations were analysed using Kaplan–Meier curves and multivariable Cox models.
Results
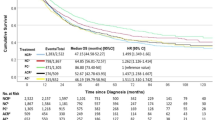
Patients treated with perioperative chemotherapy (n = 81) and neoadjuvant chemoradiotherapy (n = 249) with a TTF longer than the median (19.6 and 14.9 months, respectively) had significantly longer first-line TTF(HR 1.94 95% CI: 1.18–3.19; HR 1.36, 95%CI: 1.04–1.78). This also translated into longer OS for neoadjuvant chemoradiotherapy (HR 1.35 95% CI 1.03–1.77). Pathological response was not associated with systemic therapy outcomes.
Conclusions
A longer TTF of curative treatment was positively associated with improved first-line systemic therapy outcomes in patients with metachronous mGEA. When counselling patients, TTF of their curative treatment may be considered, whereas pathological response may not.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data can be made available by the Netherlands Comprehensive Cancer Organisation on justified request.
References
Pape M, Vissers PAJ, Bertwistle D, McDonald L, Beerepoot LV, van Berge Henegouwen MI, et al. Population-based study of treatment and outcome of recurrent oesophageal or junctional cancer. Br J Surg. 2022;109:1264–73.
Pape M, Vissers PAJ, Bertwistle D, McDonald L, Slingerland M, Haj Mohammad N, et al. A population-based study in synchronous versus metachronous metastatic esophagogastric adenocarcinoma. Ther Adv Med Oncol. 2022;14:17588359221085557.
Ter Veer E, van Kleef JJ, Schokker S, van der Woude SO, Laarman M, Haj Mohammad N, et al. Prognostic and predictive factors for overall survival in metastatic oesophagogastric cancer: a systematic review and meta-analysis. Eur J Cancer. 2018;103:214–26.
van den Boorn HG, Abu-Hanna A, Haj Mohammad N, Hulshof M, Gisbertz SS, Klarenbeek BR, et al. SOURCE: prediction models for overall survival in patients with metastatic and potentially curable esophageal and gastric cancer. J Natl Compr Canc Netw. 2021;19:403–10.
Kuijper SC, Pape M, Haj Mohammad N, van Voorthuizen T, Verhoeven RHA, van Laarhoven HWM. SOURCE beyond first-line: a survival prediction model for patients with metastatic esophagogastric adenocarcinoma after failure of first-line palliative systemic therapy. Int J Cancer. 2023;152:1202–9.
Hoeppner J, Brunner T, Schmoor C, Bronsert P, Kulemann B, Claus R, et al. Perioperative chemotherapy or preoperative chemoradiotherapy in esophageal cancer. N Engl J Med. 2025;392:323–35.
Obermannova R, Alsina M, Cervantes A, Leong T, Lordick F, Nilsson M, et al. Oesophageal cancer: ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann Oncol. 2022;33:992–1004.
Lordick F, Carneiro F, Cascinu S, Fleitas T, Haustermans K, Piessen G, et al. Gastric cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2022;33:1005–20.
van Velzen MJM, Pape M, Dijksterhuis WPM, Slingerland M, van Voorthuizen T, Beerepoot LV, et al. The association between effectiveness of first-line treatment and second-line treatment in gastro-oesophageal cancer. Eur J Cancer. 2021;156:60–9.
Cats A, Jansen EPM, van Grieken NCT, Sikorska K, Lind P, Nordsmark M, et al. Chemotherapy versus chemoradiotherapy after surgery and preoperative chemotherapy for resectable gastric cancer (CRITICS): an international, open-label, randomised phase 3 trial. Lancet Oncol. 2018;19:616–28.
Mandard AM, Dalibard F, Mandard JC, Marnay J, Henry-Amar M, Petiot JF, et al. Pathologic assessment of tumor regression after preoperative chemoradiotherapy of esophageal carcinoma. Clinicopathologic correlations. Cancer. 1994;73:2680–6.
von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP, et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. 2007;370:1453–7.
Steins A, Ebbing EA, Creemers A, van der Zalm AP, Jibodh RA, Waasdorp C, et al. Chemoradiation induces epithelial-to-mesenchymal transition in esophageal adenocarcinoma. Int J Cancer. 2019;145:2792–803.
Luan S, Zeng X, Zhang C, Qiu J, Yang Y, Mao C, et al. Advances in drug resistance of esophageal cancer: from the perspective of tumor microenvironment. Front Cell Dev Biol. 2021;9:664816.
Li S, Hoefnagel SJM, Krishnadath KK. Molecular biology and clinical management of esophageal adenocarcinoma. Cancers (Basel). 2023;15:5410.
Eyck BM, Jansen MP, Noordman BJ, Atmodimedjo PN, van der Wilk BJ, Martens JW, et al. Detection of circulating tumour DNA after neoadjuvant chemoradiotherapy in patients with locally advanced oesophageal cancer. J Pathol. 2023;259:35–45.
Ng, Ko HY, Lam KO JMY, Kwong DLW, Lo AWI, Wong IYH, et al. Circulating tumor DNA dynamics as prognostic markers in locally advanced and metastatic esophageal squamous cell carcinoma. JAMA Surg. 2023;158:1141–50.
Haffner I, Schierle K, Raimundez E, Geier B, Maier D, Hasenauer J, et al. HER2 expression, test deviations, and their impact on survival in metastatic gastric cancer: results from the prospective multicenter VARIANZ study. J Clin Oncol. 2021;39:1468–78.
Zhou KI, Peterson B, Serritella A, Thomas J, Reizine N, Moya S, et al. Spatial and temporal heterogeneity of PD-L1 expression and tumor mutational burden in gastroesophageal adenocarcinoma at baseline diagnosis and after chemotherapy. Clin Cancer Res. 2020;26:6453–63.
Liu DHW, Grabsch HI, Gloor B, Langer R, Dislich B. Programmed death-ligand 1 (PD-L1) expression in primary gastric adenocarcinoma and matched metastases. J Cancer Res Clin Oncol. 2023;149:13345–52.
Choi E, Shin J, Ryu MH, Kim HD, Park YS. Heterogeneity of claudin 18.2 expression in metastatic gastric cancer. Sci Rep. 2024;14:17648.
Eyck BM, van Lanschot JJB, Hulshof M, van der Wilk BJ, Shapiro J, van Hagen P, et al. Ten-year outcome of neoadjuvant chemoradiotherapy plus surgery for esophageal cancer: the randomized controlled CROSS trial. J Clin Oncol. 2021;39:1995–2004.
Smyth EC, Fassan M, Cunningham D, Allum WH, Okines AF, Lampis A, et al. Effect of pathologic tumor response and nodal status on survival in the medical research council adjuvant gastric infusional chemotherapy trial. J Clin Oncol. 2016;34:2721–7.
Blum Murphy M, Xiao L, Patel VR, Maru DM, Correa AM, Fatemeh GA, et al. Pathological complete response in patients with esophageal cancer after the trimodality approach: the association with baseline variables and survival-The University of Texas MD Anderson Cancer Center experience. Cancer. 2017;123:4106–13.
Meredith KL, Weber JM, Turaga KK, Siegel EM, McLoughlin J, Hoffe S, et al. Pathologic response after neoadjuvant therapy is the major determinant of survival in patients with esophageal cancer. Ann Surg Oncol. 2010;17:1159–67.
Li Z, Shan F, Wang Y, Zhang Y, Zhang L, Li S, et al. Correlation of pathological complete response with survival after neoadjuvant chemotherapy in gastric or gastroesophageal junction cancer treated with radical surgery: a meta-analysis. PLoS One. 2018;13:e0189294.
Janjigian YY, Al-Batran SE, Wainberg ZA, Muro K, Molena D, Van Cutsem E, et al. Perioperative durvalumab in gastric and gastroesophageal junction cancer. N Engl J Med. 2025;393:217–30.
Al-Batran SE, Homann N, Pauligk C, Goetze TO, Meiler J, Kasper S, et al. Perioperative chemotherapy with fluorouracil plus leucovorin, oxaliplatin, and docetaxel versus fluorouracil or capecitabine plus cisplatin and epirubicin for locally advanced, resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4): a randomised, phase 2/3 trial. Lancet. 2019;393:1948–57.
Gu YM, Lyu SM, Shang QX, Zhang HL, Yang YS, Wang WP, et al. Is tumor regression grade sufficient to predict survival in esophageal cancer with trimodal therapy? J Invest Surg. 2022;35:1818–23.
Acknowledgements
The authors acknowledge the use of ChatGPT (OpenAI) to improve the readability and clarity of the manuscript. No text was generated or copied directly from the tool; all scientific content and wording were conceived and written by the authors.
Funding
This work was financially supported by UMC Utrecht & Wilhelmina Kinderziekenhuis Foundation [project number 1522166]. The funder was not involved in the study design, collection, analysis and interpretation of data, writing of the report or the decision to submit the article for publication.
Author information
Authors and Affiliations
Contributions
DK: writing—original draft, investigation, formal analysis, data curation, methodology. AMM: writing—review & editing, supervision, methodology, investigation. HWMvL: writing—review & editing, supervision, methodology, investigation, conceptualisation. NHM: writing—review & editing, supervision, methodology, investigation, conceptualisation, resources. MJMvV: writing—review & editing, conceptualisation, investigation. SM, JEF, BM, RHAV: writing—review & editing, investigation
Corresponding author
Ethics declarations
Competing interests
HWMvL has acted as a consultant or in an advisory role for Auristone, Incyte, Merck, Myeloid, Servier; and has received research funding and/or medication/material supply from Amphera, Anocca, Astellas, AstraZeneca, Beigene, Boehringer, BMS, Daiichy-Sankyo, Dragonfly, MSD, MyeloidTx, ORCA, Servier and speaker roles for Astellas, Beigene, Benecke, BMS, Daiichy-Sankyo, JAAP, Medtalks, Novartis, Springer, Travel Congress Management B.V, and has received travel support from AstraZeneca. NHM has acted as a consultant or in an advisory role for BMS, Astra Zeneca, Servier, MSD, and Eli Lilly; and has received research funding and/or medication/material supply from Servier. RHAV has received research funding from BMS and Amgen and has acted as consultant for Daiichi Sankyo. BM: has received research funding and/or medication supply from BMS and Pfizer; and has acted as a consultant or in an advisory role for Amgen. AMM, MJMvV, SM, JEF and DK declare no conflicts of interest.
Ethics approval
This study did not require approval from an Institutional Review Board in the Netherlands, as determined by the Central Committee on Research Involving Human Subjects. However, it was approved by the Privacy Review Board of the Netherlands Cancer Registry (request number K19.288) and the scientific committee of the Dutch Upper GI Cancer Group. The study was approved in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kamp, D., May, A.M., van Velzen, M.J.M. et al. Association between effectiveness of treatment with curative intent and outcomes of first-line systemic therapy in metachronous metastatic gastroesophageal adenocarcinoma. Br J Cancer (2026). https://doi.org/10.1038/s41416-026-03374-5
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41416-026-03374-5