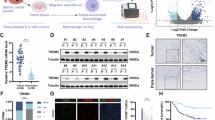
Abstract
Background
The epithelial-mesenchymal axis frames gastric cancer heterogeneity but mainly captures phenotypic extremes. CCNE1 gain, encompassing amplification or Cyclin E1 overexpression, represents one intermediate state linked to chromosomal instability and therapeutic resistance. However, its clinicopathologic identity and immune features in gastric cancer remain insufficiently characterised.
Methods
We analysed 1273 gastric cancer patients across six independent cohorts: ZSHS (n = 453), TCGA (n = 410), ACRG (n = 300), SMC (n = 43), MSKCC (n = 22), and ZSHS NGS cohort (n = 45). Tumours were classified into CCNE1 gain, epithelial, or mesenchymal subtypes using protein, copy-number, or transcript-level data. Clinicopathologic features, survival outcomes, treatment responses, and immune contexture were evaluated across subtypes.
Results
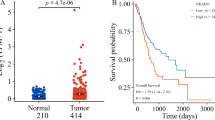
CCNE1 gain tumours retained E-cadherin positivity but showed increased proliferation, more nerve and venous invasion. They were associated with poor prognosis and reduced response to adjuvant chemotherapy, HER2-targeted therapy, anti-angiogenic agents, and PD-1 blockade independent of epithelial-mesenchymal classification. The immune contexture exhibited an immune-desert phenotype with reduced lymphocyte infiltration, impaired cytotoxicity, increased M2 macrophage polarisation, and elevated TGF-β.
Conclusion
CCNE1 gain delineates a clinic-ready, therapy-refractory subtype of gastric cancer beyond the epithelial-mesenchymal framework, representing over one in ten patients. These tumours preserve epithelial morphology yet exhibit aggressive proliferative and invasive behaviour, coupled with immune desert and myeloid-driven suppression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data and materials generated that are relevant to the results are included in this article. Other data are available from the corresponding author Prof. Xu upon reasonable request.
References
Gullo I, Carneiro F, Oliveira C, Almeida GM. Heterogeneity in gastric cancer: from pure morphology to molecular classifications. Pathobiology. 2018;85:50–63.
Smyth EC, Nilsson M, Grabsch HI, van Grieken NC, Lordick F. Gastric cancer. Lancet. 2020;396:635–48.
Lauren P. The two histological main types of gastric carcinoma: diffuse and so-called intestinal-type carcinoma. An attempt at a histo-clinical classification. Acta Pathol Microbiol Scand. 1965;64:31–49.
Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014;513:202–9.
Cristescu R, Lee J, Nebozhyn M, Kim KM, Ting JC, Wong SS, et al. Molecular analysis of gastric cancer identifies subtypes associated with distinct clinical outcomes. Nat Med. 2015;21:449–56.
Walch HS, Borpatragohain R, Jee J, Chatila W, Fong C, Maron SB, et al. Clinical Implications of The Cancer Genome Atlas molecular classification system in esophagogastric cancer. Clin Cancer Res. 2025;31:1912–21.
Pastushenko I, Blanpain C. EMT transition states during tumor progression and metastasis. Trends Cell Biol. 2019;29:212–26.
Dongre A, Weinberg RA. New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer. Nat Rev Mol Cell Biol. 2019;20:69–84.
Pastushenko I, Brisebarre A, Sifrim A, Fioramonti M, Revenco T, Boumahdi S, et al. Identification of the tumour transition states occurring during EMT. Nature. 2018;556:463–8.
Ruscetti M, Quach B, Dadashian EL, Mulholland DJ, Wu H. Tracking and functional characterization of epithelial-mesenchymal transition and mesenchymal tumor cells during prostate cancer metastasis. Cancer Res. 2015;75:2749–59.
Tan TZ, Miow QH, Miki Y, Noda T, Mori S, Huang RY, et al. Epithelial-mesenchymal transition spectrum quantification and its efficacy in deciphering survival and drug responses of cancer patients. EMBO Mol Med. 2014;6:1279–93.
Creighton CJ, Li X, Landis M, Dixon JM, Neumeister VM, Sjolund A, et al. Residual breast cancers after conventional therapy display mesenchymal as well as tumor-initiating features. Proc Natl Acad Sci USA. 2009;106:13820–5.
Oh SC, Sohn BH, Cheong JH, Kim SB, Lee JE, Park KC, et al. Clinical and genomic landscape of gastric cancer with a mesenchymal phenotype. Nat Commun. 2018;9:1777.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–74.
Evan GI, Vousden KH. Proliferation, cell cycle and apoptosis in cancer. Nature. 2001;411:342–8.
Hwang HC, Clurman BE. Cyclin E in normal and neoplastic cell cycles. Oncogene. 2005;24:2776–86.
Chu C, Geng Y, Zhou Y, Sicinski P. Cyclin E in normal physiology and disease states. Trends Cell Biol. 2021;31:732–46.
Zeng J, Hills SA, Ozono E, Diffley JFX. Cyclin E-induced replicative stress drives p53-dependent whole-genome duplication. Cell. 2023;186:528–42.e14.
Kuhn E, Wang TL, Doberstein K, Bahadirli-Talbott A, Ayhan A, Sehdev AS, et al. CCNE1 amplification and centrosome number abnormality in serous tubal intraepithelial carcinoma: further evidence supporting its role as a precursor of ovarian high-grade serous carcinoma. Mod Pathol. 2016;29:1254–61.
Span PN, Tjan-Heijnen VC, Manders P, Beex LV, Sweep CG. Cyclin-E is a strong predictor of endocrine therapy failure in human breast cancer. Oncogene. 2003;22:4898–904.
Cancer Genome Atlas Research Network. Integrated genomic analyses of ovarian carcinoma. Nature. 2011;474:609–15.
Alsina M, Landolfi S, Aura C, Caci K, Jimenez J, Prudkin L, et al. Cyclin E amplification/overexpression is associated with poor prognosis in gastric cancer. Ann Oncol. 2015;26:438–9.
Xing R, Zhou Y, Yu J, Yu Y, Nie Y, Luo W, et al. Whole-genome sequencing reveals novel tandem-duplication hotspots and a prognostic mutational signature in gastric cancer. Nat Commun. 2019;10:2037.
Rustgi N, Wu S, Samec T, Walker P, Xiu J, Lou E, et al. Molecular landscape and clinical implication of CCNE1-amplified Esophagogastric Cancer. Cancer Res Commun. 2024;4:1399–409.
Garsed DW, Pandey A, Fereday S, Kennedy CJ, Takahashi K, Alsop K, et al. The genomic and immune landscape of long-term survivors of high-grade serous ovarian cancer. Nat Genet. 2022;54:1853–64.
Guerrero Llobet S, Bhattacharya A, Everts M, Kok K, van der Vegt B, Fehrmann RSN, et al. An mRNA expression-based signature for oncogene-induced replication-stress. Oncogene. 2022;41:1216–24.
Gu Y, Zhang P, Wang J, Lin C, Liu H, Li H, et al. Somatic ARID1A mutation stratifies patients with gastric cancer to PD-1 blockade and adjuvant chemotherapy. Cancer Immunol Immunother. 2023;72:1199–208.
Chan AM, Enwere E, McIntyre JB, Wilson H, Nwaroh C, Wiebe N, et al. Combined CCNE1 high-level amplification and overexpression is associated with unfavourable outcome in tubo-ovarian high-grade serous carcinoma. J Pathol Clin Res. 2020;6:252–62.
Kim ST, Cristescu R, Bass AJ, Kim KM, Odegaard JI, Kim K, et al. Comprehensive molecular characterization of clinical responses to PD-1 inhibition in metastatic gastric cancer. Nat Med. 2018;24:1449–58.
Thorsson V, Gibbs DL, Brown SD, Wolf D, Bortone DS, Ou Yang TH, et al. The immune landscape of cancer. Immunity. 2018;48:812–30.e14.
Bagaev A, Kotlov N, Nomie K, Svekolkin V, Gafurov A, Isaeva O, et al. Conserved pan-cancer microenvironment subtypes predict response to immunotherapy. Cancer Cell. 2021;39:845–65.e7.
Jin K, Cao Y, Gu Y, Fang H, Fei Y, Wang J, et al. Poor clinical outcomes and immunoevasive contexture in CXCL13+CD8+ T cells enriched gastric cancer patients. Oncoimmunology. 2021;10:1915560.
Wang J, Li R, Cao Y, Gu Y, Fang H, Fei Y, et al. Intratumoral CXCR5(+)CD8(+)T associates with favorable clinical outcomes and immunogenic contexture in gastric cancer. Nat Commun. 2021;12:3080.
Li R, Liu H, Cao Y, Wang J, Chen Y, Qi Y, et al. Identification and validation of an immunogenic subtype of gastric cancer with abundant intratumoural CD103(+)CD8(+) T cells conferring favourable prognosis. Br J Cancer. 2020;122:1525–34.
Yu K, Cao Y, Zhang Z, Wang L, Gu Y, Xu T, et al. Blockade of CLEVER-1 restrains immune evasion and enhances anti-PD-1 immunotherapy in gastric cancer. J Immunother Cancer. 2025;13:e011080.
Wang J, Liang Y, Xue A, Xiao J, Zhao X, Cao S, et al. Intratumoral CXCL13(+) CD160(+) CD8(+) T cells promote the formation of tertiary lymphoid structures to enhance the efficacy of immunotherapy in advanced gastric cancer. J Immunother Cancer. 2024;12:e009603.
Lv K, Sun M, Fang H, Wang J, Lin C, Liu H, et al. Targeting myeloid checkpoint Siglec-10 reactivates antitumor immunity and improves anti-programmed cell death 1 efficacy in gastric cancer. J Immunother Cancer. 2023;11:e007669.
Zhang P, Gu Y, Wang J, Lv K, Lin C, Zhang H, et al. Complement receptor C5aR1 blockade reprograms tumor-associated macrophages and synergizes with anti-PD-1 therapy in gastric cancer. Int J Cancer. 2023;153:224–37.
Liu X, Lv K, Wang J, Lin C, Liu H, Zhang H, et al. C-type lectin receptor Dectin-1 blockade on tumour-associated macrophages improves anti-PD-1 efficacy in gastric cancer. Br J Cancer. 2023;129:721–32.
Lordick F, Carneiro F, Cascinu S, Fleitas T, Haustermans K, Piessen G, et al. Gastric cancer: ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann Oncol. 2022;33:1005–20.
Rizvi NA, Hellmann MD, Snyder A, Kvistborg P, Makarov V, Havel JJ, et al. Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science. 2015;348:124–8.
Pfirschke C, Engblom C, Rickelt S, Cortez-Retamozo V, Garris C, Pucci F, et al. Immunogenic chemotherapy sensitizes tumors to checkpoint blockade therapy. Immunity. 2016;44:343–54.
Gallo D, Young JTF, Fourtounis J, Martino G, Álvarez-Quilón A, Bernier C, et al. CCNE1 amplification is synthetic lethal with PKMYT1 kinase inhibition. Nature. 2022;604:749–56.
Fu S, Yao S, Yuan Y, Previs RA, Elias AD, Carvajal RD, et al. Multicenter phase II trial of the WEE1 inhibitor adavosertib in refractory solid tumors harboring CCNE1 amplification. J Clin Oncol. 2023;41:1725–34.
Dietrich C, Trub A, Ahn A, Taylor M, Ambani K, Chan KT, et al. INX-315, a selective CDK2 inhibitor, induces cell cycle arrest and senescence in solid tumors. Cancer Discov. 2024;14:446–67.
Melero I, de Miguel Luken M, de Velasco G, Garralda E, Martín-Liberal J, Joerger M, et al. Neutralizing GDF-15 can overcome anti-PD-1 and anti-PD-L1 resistance in solid tumours. Nature. 2025;637:1218–27.
Kang EY, Weir A, Meagher NS, Farrington K, Nelson GS, Ghatage P, et al. CCNE1 and survival of patients with tubo-ovarian high-grade serous carcinoma: an ovarian tumor tissue analysis consortium study. Cancer. 2023;129:697–713.
Thiery JP, Acloque H, Huang RY, Nieto MA. Epithelial-mesenchymal transitions in development and disease. Cell. 2009;139:871–90.
Acknowledgements
We would like to thank Dr. Lingli Chen (Department of Pathology, Zhongshan Hospital, Fudan University, Shanghai, China) and Dr. Yunyi Kong (Department of Pathology, Shanghai Cancer Center, Fudan University, Shanghai, China) for their excellent pathological technology help.
Funding
This study was funded by grants from National Natural Science Foundation of China (82203201, 82272786, 82303966, 82373417, 82503945), China Postdoctoral Science Foundation (2023M742327), Shanghai Municipal Natural Science Foundation (23ZR1409900), Shanghai Municipal Health Commission (20254Y0166), Shanghai Pujiang Talents Program (24PJD013), Chenguang Program of Shanghai Education Development Foundation and Shanghai Municipal Education Commission (25CGA05), Natural Science Foundation of Fujian Province (2023J05294) and Clinical Research Fund of Zhongshan Hospital Fudan University (ZSLCYJ202343). All these study sponsors have no roles in the study design, in the collection, analysis and interpretation of data.
Author information
Authors and Affiliations
Contributions
Y Gu and J Wang for the acquisition of data, interpretation of data, statistical analysis and drafting of the manuscript; Z Ling, J Liu, C Lin, H Liu and H He for technical and material support; R Li, F Shao and J Xu for study concept and design, analysis and interpretation of data, drafting of the manuscript, obtained funding and study supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
The study was approved by the Clinical Research Ethics Committee of Zhongshan Hospital, Fudan University, with the approval number B2025-295. Written informed consent was obtained from each patient included and this study was performed in accordance with the Declaration of Helsinki.
Consent for publication
All the information from individual patients of Zhongshan Hospital has gained written informed consent for publication.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gu, Y., Wang, J., Ling, Z. et al. Lethal clinical outcome and immune desert contexture in refractory gastric cancer harboring CCNE1 amplification and overexpression. Br J Cancer (2026). https://doi.org/10.1038/s41416-026-03461-7
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41416-026-03461-7