Abstract
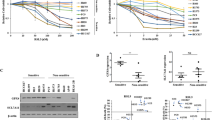
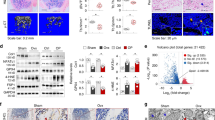
Osteosarcoma is an aggressive and highly metastatic cancer that arises in bones. Ferroptosis, an iron-dependent form of cell death, is critically controlled by glutathione peroxidase 4 (GPX4). In this study, we characterized GPX4 and its upstream regulator xCT across five osteosarcoma cell lines (U2OS, MG-63, HOS, Saos2, and 143B). We also demonstrated that chemical inhibition of these proteins using RSL3 (GPX4 inhibitor) and erastin (xCT inhibitor) significantly suppressed osteosarcoma cell growth. In U2OS and MG-63 cells, GPX4 inhibition triggered lipid peroxidation, NRF2 activation, and upregulated antioxidant genes including, AKR1C1, HO-1, and NQO1, indicating a compensatory response to oxidative stress. Furthermore, GPX4 inhibition, through RSL3 treatment or transient GPX4 knockdown, increased ferritin heavy chain 1 and ferroportin expression, promoting intracellular iron depletion and conferring resistance to ferroptosis. RSL3 exposure also elevated post-translational regulators such as HIF-1α, c-MET and MSPR/RON, associated with cell survival pathways. Pharmacological inhibition of these signaling molecules synergistically enhanced RSL3-induced cytotoxicity. Collectively, our findings reveal that GPX4 inhibition initiates ferroptosis while simultaneously activating NRF2-driven antioxidant defenses, iron homeostasis mechanisms, and adaptive cell survival signaling. The results highlight potential therapeutic strategies that combine GPX4 inhibition with targeted disruption of compensatory pathways to overcome ferroptosis resistance in osteosarcoma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in the main manuscript or supplementary materials. The RNA-seq data analyzed in this study were obtained from the National Cancer Institute Genomic Data Commons Data Portal, specifically from the TARGET-OS project (dbGap accession: phs000218). These data are publicly available at https://portal.gdc.cancer.gov/. Other data relevant to this study are available upon request from the corresponding author.
References
Cole S, Gianferante DM, Zhu B, Mirabello L. Osteosarcoma: a surveillance, epidemiology, and end results program-based analysis from 1975 to 2017. Cancer. 2022;128:2107–18.
Longhi A, Errani C, De Paolis M, Mercuri M, Bacci G. Primary bone osteosarcoma in the pediatric age: state of the art. Cancer Treat Rev. 2006;32:423–36.
Desai S, Guddati AK. Bimodal age distribution in cancer incidence. World J Oncol. 2022;13:329–36.
Gianferante DM, Mirabello L, Savage SA. Germline and somatic genetics of osteosarcoma - connecting aetiology, biology and therapy. Nat Rev Endocrinol. 2017;13:480–91.
Meltzer PS, Helman LJ. New horizons in the treatment of osteosarcoma. N Engl J Med. 2021;385:2066–76.
Bielack SS, Kempf-Bielack B, Delling G, Exner GU, Flege S, Helmke K, et al. Prognostic factors in high-grade osteosarcoma of the extremities or trunk: an analysis of 1,702 patients treated on neoadjuvant cooperative osteosarcoma study group protocols. J Clin Oncol. 2002;20:776–90.
Bacci G, Bertoni F, Longhi A, Ferrari S, Forni C, Biagini R, et al. Neoadjuvant chemotherapy for high-grade central osteosarcoma of the extremity. Histologic response to preoperative chemotherapy correlates with histologic subtype of the tumor. Cancer. 2003;97:3068–75.
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149:1060–72.
Lee JY, Kim WK, Bae KH, Lee SC, Lee EW. Lipid metabolism and ferroptosis. Biology. 2021;10:184.
Stockwell BR. Ferroptosis turns 10: emerging mechanisms, physiological functions, and therapeutic applications. Cell. 2022;185:2401–21.
Dixon SJ, Olzmann JA. The cell biology of ferroptosis. Nat Rev Mol Cell Biol. 2024;25:424–42.
Li FJ, Long HZ, Zhou ZW, Luo HY, Xu SG, Gao LC, et al. Front Pharmacol. 2022;13:910292.
Yang WS, SriRamaratnam R, Welsch ME, Shimada K, Skouta R, Viswanathan VS, et al. Regulation of ferroptotic cancer cell death by GPX4. Cell. 2014;156:317–31.
Chen X, Yu C, Kang R, Tang D. Iron metabolism in ferroptosis. Front Cell Dev Biol. 2020;8:590226.
Cardona CJ, Montgomery MR. Iron regulatory proteins: players or pawns in ferroptosis and cancer? Front Mol Biosci. 2023;10:1229710.
Liu X, Du S, Wang S, Ye K. Ferroptosis in osteosarcoma: a promising future. Front Oncol. 2022;12:1031779.
Xu Z, Chen L, Wang C, Zhang L, Xu W. MicroRNA-1287-5p promotes ferroptosis of osteosarcoma cells through inhibiting GPX4. Free Radic Res. 2021;55:1119–29.
Zhao J, Zhao Y, Ma X, Zhang B, Feng H. Targeting ferroptosis in osteosarcoma. J Bone Oncol. 2021;30:100380.
Liu J, Lou C, Zhen C, Wang Y, Shang P, Lv H. Iron plays a role in sulfasalazine-induced ferroptosis with autophagic flux blockage in K7M2 osteosarcoma cells. Metallomics 2022;14:mfac027.
Shen Y, Zhang B, Su Y, Badshah SA, Wang X, Li X, et al. Iron promotes dihydroartemisinin cytotoxicity via ROS production and blockade of autophagic flux via lysosomal damage in osteosarcoma. Front Pharmacol. 2020;11:444.
Dodson M, Castro-Portuguez R, Zhang DD. NRF2 plays a critical role in mitigating lipid peroxidation and ferroptosis. Redox Biol. 2019;23:101107.
Penning TM, Jonnalagadda S, Trippier PC, Rižner TL. Aldo-keto reductases and cancer drug resistance. Pharmacol Rev. 2021;73:1150–71.
Torti SV, Torti FM. Iron: the cancer connection. Mol Aspects Med. 2020;75:100860.
Lee J, Hyun DH. The interplay between intracellular iron homeostasis and neuroinflammation in neurodegenerative diseases. Antioxidants 2023;12:918.
Yapici FI, Bebber CM, von Karstedt S. A guide to ferroptosis in cancer. Mol Oncol. 2024;18:1378–96.
Lin H, Chen X, Zhang C, Yang T, Deng Z, Song Y, et al. EF24 induces ferroptosis in osteosarcoma cells through HMOX1. Biomed Pharmacother. 2021;136:111202.
Liu Q, Wang K. The induction of ferroptosis by impairing STAT3/Nrf2/GPx4 signaling enhances the sensitivity of osteosarcoma cells to cisplatin. Cell Biol Int. 2019;43:1245–56.
Yuan C, Fan R, Zhu K, Wang Y, Xie W, Liang Y. Curcumin induces ferroptosis and apoptosis in osteosarcoma cells by regulating Nrf2/GPX4 signaling pathway. Exp Biol Med. 2023;248:2183–97.
Wen RJ, Dong X, Zhuang HW, Pang FX, Ding SC, Li N, et al. Baicalin induces ferroptosis in osteosarcomas through a novel Nrf2/xCT/GPX4 regulatory axis. Phytomedicine. 2023;116:154881.
Qin Q, Zhang H, Lai M, Wei J, Qian J, Chen X, et al. Sulfasalazine induces ferroptosis in osteosarcomas by regulating Nrf2/SLC7A11/GPX4 signaling axis. Sci Rep. 2025;15:30197.
Liu W, Zhao Y, Wang G, Feng S, Ge X, Ye W, et al. TRIM22 inhibits osteosarcoma progression through destabilizing NRF2 and thus activation of ROS/AMPK/mTOR/autophagy signaling. Redox Biol. 2022;53:102344.
Tang D, Chen X, Kang R, Kroemer G. Ferroptosis: molecular mechanisms and health implications. Cell Res. 2021;31:107–25.
Anandhan A, Dodson M, Schmidlin CJ, Liu P, Zhang DD. Breakdown of an ironclad defense system: the critical role of NRF2 in mediating ferroptosis. Cell Chem Biol. 2020;27:436–47.
Luo Z, Tian M, Yang G, Tan Q, Chen Y, Li G, et al. Hypoxia signaling in human health and diseases: implications and prospects for therapeutics. Signal Transduct Target Ther. 2022;7:218.
Yang Z, Su W, Wei X, Qu S, Zhao D, Zhou J, et al. HIF-1α drives resistance to ferroptosis in solid tumors by promoting lactate production and activating SLC1A1. Cell Rep. 2023;42:112945.
Jiang L, Kon N, Li T, Wang SJ, Su T, Hibshoosh H, et al. Ferroptosis as a p53-mediated activity during tumour suppression. Nature. 2015;520:57–62.
Gao M, Monian P, Quadri N, Ramasamy R, Jiang X. Glutaminolysis and transferrin regulate ferroptosis. Mol Cell. 2015;59:298–308.
Ou Y, Wang SJ, Li D, Chu B, Gu W. Activation of SAT1 engages polyamine metabolism with p53-mediated ferroptotic responses. Proc Natl Acad Sci USA. 2016;113:E6806–E12.
Tarangelo A, Magtanong L, Bieging-Rolett KT, Li Y, Ye J, Attardi LD, et al. p53 suppresses metabolic stress-induced ferroptosis in cancer cells. Cell Rep. 2018;22:569–75.
Xu R, Wang W, Zhang W. Ferroptosis and the bidirectional regulatory factor p53. Cell Death Discov. 2023;9:197.
Jin W, Zhou L, Yan B, Yan L, Liu F, Tong P, et al. Theabrownin triggers DNA damage to suppress human osteosarcoma U2OS cells by activating p53 signalling pathway. J Cell Mol Med. 2018;22:4423–36.
Spina A, Sorvillo L, Di Maiolo F, Esposito A, D’Auria R, Di Gesto D, et al. Inorganic phosphate enhances sensitivity of human osteosarcoma U2OS cells to doxorubicin via a p53-dependent pathway. J Cell Physiol. 2013;228:198–206.
Danilkovitch A, Leonard EJ. Kinases involved in MSP/RON signaling. J Leukoc Biol. 1999;65:345–8.
Zhang Y, Xia M, Jin K, Wang S, Wei H, Fan C, et al. Function of the c-Met receptor tyrosine kinase in carcinogenesis and associated therapeutic opportunities. Mol Cancer. 2018;17:45.
Crielaard BJ, Lammers T, Rivella S. Targeting iron metabolism in drug discovery and delivery. Nat Rev Drug Discov. 2017;16:400–23.
Galaris D, Pantopoulos K. Oxidative stress and iron homeostasis: mechanistic and health aspects. Crit Rev Clin Lab Sci. 2008;45:1–23.
Ba Q, Hao M, Huang H, Hou J, Ge S, Zhang Z, et al. Iron deprivation suppresses hepatocellular carcinoma growth in experimental studies. Clin Cancer Res. 2011;17:7625–33.
Nyffenegger N, Flace A, Doucerain C, Dürrenberger F, Manolova V. The oral ferroportin inhibitor VIT-2763 improves erythropoiesis without interfering with iron chelation therapy in a mouse model of β-thalassemia. Int J Mol Sci. 2021;22:873.
Ma S, Henson ES, Chen Y, Gibson SB. Ferroptosis is induced following siramesine and lapatinib treatment of breast cancer cells. Cell Death Dis. 2016;7:e2307.
Hassannia B, Vandenabeele P, Vanden Berghe T. Targeting ferroptosis to iron out cancer. Cancer Cell. 2019;35:830–49.
Wang W, Green M, Choi JE, Gijón M, Kennedy PD, Johnson JK, et al. CD8. Nature. 2019;569:270–4.
Weichhart T. Transferrin: the iron transporter takes control. Blood. 2024;144:9–10.
Ganz T. Macrophages and iron metabolism. Microbiol Spectr. 2016;4:492–504.
Boyle WJ, Simonet WS, Lacey DL. Osteoclast differentiation and activation. Nature. 2003;423:337–42.
Gao YM, Pei Y, Zhao FF, Wang L. Osteoclasts in Osteosarcoma: mechanisms, interactions, and therapeutic prospects. Cancer Manag Res. 2023;15:1323–37.
Russo S, Scotto di Carlo F, Gianfrancesco F. The osteoclast traces the route to bone tumors and metastases. Front Cell Dev Biol. 2022;10:886305.
Cersosimo F, Lonardi S, Bernardini G, Telfer B, Mandelli GE, Santucci A, et al. Tumor-associated macrophages in osteosarcoma: from mechanisms to therapy. Int J Mol Sci. 2020;21:5207.
Zheng S, Wang W, Aldahdooh J, Malyutina A, Shadbahr T, Tanoli Z, et al. SynergyFinder plus: toward better interpretation and annotation of drug combination screening datasets. Genomics Proteomics Bioinf. 2022;20:587–96.
Acknowledgements
We acknowledge the UF ICBR Gene Expression & Genotyping Core (RRID: SCR_019145) for RNA-seq and the Cytometry Core (RRID: SCR_019119) for flow cytometry. Figure 6 was crafted using Biorender (biorender.com). We also acknowledge the UF HiPerGator Research Computing for computational resources and technical support. This work was partly supported by the UF AI Initiative.
Author information
Authors and Affiliations
Contributions
Conception and design: MA and JHK; Development of methodology: MA, DHL, and JHK; Acquisition of data: MA, DHL, EN, JCG, and JHK; Analysis and interpretation of data: MA, DHL, and JHK; Writing, review, and/or revision of the manuscript: MA, DHL, EN, JCG, AMC, OHM, MES, RNR, RJM, and JHK; Administrative, technical, or material support: MA, EN, and JHK; Study supervision: JHK.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study did not involve any live animals, human participants, or human specimens. Therefore, ethics approval and informed consent were not required. All methods were performed in accordance with the relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abdullah, M., Lee, D., Nipa, E. et al. Osteosarcoma cells promote intracellular iron detoxification to mitigate GPX4-mediated ferroptosis. Cancer Gene Ther (2026). https://doi.org/10.1038/s41417-026-01021-y
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41417-026-01021-y