Abstract
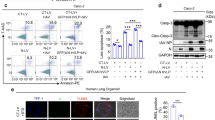
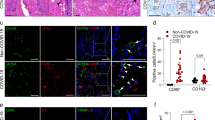
Deaths caused by coronavirus disease 2019 (COVID-19) are largely due to the lungs edema resulting from the disruption of the lung alveolo-capillary barrier, induced by SARS-CoV-2-triggered pulmonary cell apoptosis. However, the molecular mechanism underlying the proapoptotic role of SARS-CoV-2 is still unclear. Here, we revealed that SARS-CoV-2 membrane (M) protein could induce lung epithelial cells mitochondrial apoptosis. Notably, M protein stabilized B-cell lymphoma 2 (BCL-2) ovarian killer (BOK) via inhibiting its ubiquitination and promoted BOK mitochondria translocation. The endodomain of M protein was required for its interaction with BOK. Knockout of BOK by CRISPR/Cas9 increased cellular resistance to M protein-induced apoptosis. BOK was rescued in the BOK-knockout cells, which led to apoptosis induced by M protein. M protein induced BOK to trigger apoptosis in the absence of BAX and BAK. Furthermore, the BH2 domain of BOK was required for interaction with M protein and proapoptosis. In vivo M protein recombinant lentivirus infection induced caspase-associated apoptosis and increased alveolar-capillary permeability in the mouse lungs. BOK knockdown improved the lung edema due to lentivirus-M protein infection. Overall, M protein activated the BOK-dependent apoptotic pathway and thus exacerbated SARS-CoV-2 associated lung injury in vivo. These findings proposed a proapoptotic role for M protein in SARS-CoV-2 pathogenesis, which may provide potential targets for COVID-19 treatments.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, et al. Lancet Respir Med. 2020;8:420–22.
Goshua G, Pine AB, Meizlish ML, Chang CH, Zhang H, Bahel P, et al. Endotheliopathy in COVID-19-associated coagulopathy: evidence from a single-centre, cross-sectional study. Lancet Haematol. 2020;7:e575–82.
Matthay MA, Leligdowicz A, Liu KD. Biological mechanisms of COVID-19 acute respiratory distress syndrome. Am J Respir Crit Care Med. 2020;202:1489–91.
Galluzzi L, Vitale I, Aaronson SA, Abrams JM, Adam D, Agostinis P, et al. Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018;25:486–541.
Danthi P. Viruses and the diversity of cell death. Annu Rev Virol. 2016;3:533–53.
Fung TS, Liu DX. Human coronavirus: host-pathogen interaction. Annu Rev Microbiol. 2019;73:529–57.
Zhang J, Han Y, Shi H, Chen J, Zhang X, Wang X, et al. Swine acute diarrhea syndrome coronavirus-induced apoptosis is caspase- and cyclophilin D- dependent. Emerg Microbes Infect. 2020;9:439–56.
Colmenero I, Santonja C, Alonso-Riano M, Noguera-Morel L, Hernandez-Martin A, Andina D, et al. SARS-CoV-2 endothelial infection causes COVID-19 chilblains: histopathological, immunohistochemical and ultrastructural study of seven paediatric cases. Br J Dermatol. 2020;183:729–37.
Katsura H, Sontake V, Tata A, Kobayashi Y, Edwards CE, Heaton BE, et al. Human lung stem cell-based alveolospheres provide insights into SARS-CoV-2-mediated interferon responses and pneumocyte dysfunction. Cell Stem Cell. 2020;27:890–904.
Bao L, Deng W, Huang B, Gao H, Liu J, Ren L, et al. The pathogenicity of SARS-CoV-2 in hACE2 transgenic mice. Nature.2020;583:830–3.
Chan JF, Zhang AJ, Yuan S, Poon VK, Chan CC, Lee AC, et al. Simulation of the clinical and pathological manifestations of coronavirus disease 2019 (COVID-19) in a golden syrian hamster model: implications for disease pathogenesis and transmissibility. Clin Infect Dis. 2020;71:2428–46.
Jiang RD, Liu MQ, Chen Y, Shan C, Zhou YW, Shen XR, et al. Pathogenesis of SARS-CoV-2 in transgenic mice expressing human angiotensin-converting enzyme 2. Cell. 2020;182:50–8.
Lin P, Wang M, Wei Y, Kim T, Wei X. Coronavirus in human diseases: mechanisms and advances in clinical treatment. MedComm (Beijing). 2020;1:270–301.
Lai CW, Chan ZR, Yang DG, Lo WH, Lai YK, Chang MD, et al. Accelerated induction of apoptosis in insect cells by baculovirus-expressed SARS-CoV membrane protein. FEBS Lett. 2006;580:3829–34.
Padhan K, Minakshi R, Towheed MAB, Jameel S. Severe acute respiratory syndrome coronavirus 3a protein activates the mitochondrial death pathway through p38 MAP kinase activation. J Gen Virol. 2008;89:1960–9.
Hekman RM, Hume AJ, Goel RK, Abo KM, Huang J, Blum BC, et al. Actionable cytopathogenic host responses of human alveolar type 2 cells to SARS-CoV-2. Mol Cell. 2021;81:212.
Moldoveanu T, Czabotar PE. BAX, BAK, and BOK: a coming of age for the BCL-2 family effector proteins. Cold Spring Harb Perspect Biol. 2020;12:a036319.
Zhang J, Cruz-Cosme R, Zhuang MW, Liu D, Liu Y, Teng S, et al. A systemic and molecular study of subcellular localization of SARS-CoV-2 proteins. Signal Transduct Target Ther. 2020;5:269.
Zheng Y, Zhuang MW, Han L, Zhang J, Nan ML, Zhan P, et al. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) membrane (M) protein inhibits type I and III interferon production by targeting RIG-I/MDA-5 signaling. Signal Transduct Target Ther. 2020;5:299.
Neupane AS, Willson M, Chojnacki AK, Vargas ESCF, Morehouse C, Carestia A, et al. Patrolling alveolar macrophages conceal bacteria from the immune system to maintain homeostasis. Cell. 2020;183:110–25.
Bierman A, Yerrapureddy A, Reddy NM, Hassoun PM, Reddy SP. Epidermal growth factor receptor (EGFR) regulates mechanical ventilation-induced lung injury in mice. Transl Res. 2008;152:265–72.
Xu M, Wang F, Li G, Wang X, Fang X, Jin H, et al. MED12 exerts an emerging role in actin-mediated cytokinesis via LIMK2/cofilin pathway in NSCLC. Mol Cancer. 2019;18:93.
Bi HL, Zhang XL, Zhang YL, Xie X, Xia YL, Du J, et al. The deubiquitinase UCHL1 regulates cardiac hypertrophy by stabilizing epidermal growth factor receptor. Sci Adv. 2020;6:eaax4826.
Yang Y, Kuang L, Li L, Wu Y, Zhong B, Huang X. Distinct mitochondria-mediated T-cell apoptosis responses in children and adults with coronavirus disease 2019. J Infect Dis. 2021;224:1333–44.
Zamzami N, Marchetti P, Castedo M, Zanin C, Vayssiere JL, Petit PX, et al. Reduction in mitochondrial potential constitutes an early irreversible step of programmed lymphocyte death in vivo. J Exp Med. 1995;181:1661–72.
Dawood AA. Glycosylation, ligand binding sites and antigenic variations between membrane glycoprotein of COVID-19 and related coronaviruses. Vacunas.2021;22:1–9.
Snijder EJ, Bredenbeek PJ, Dobbe JC, Thiel V, Ziebuhr J, Poon LL, et al. Unique and conserved features of genome and proteome of SARS-coronavirus, an early split-off from the coronavirus group 2 lineage. J Mol Biol. 2003;331:991–1004.
Llambi F, Wang YM, Victor B, Yang M, Schneider DM, Gingras S, et al. BOK is a non-canonical BCL-2 family effector of apoptosis regulated by ER-associated degradation. Cell. 2016;165:421–33.
Lindsten T, Ross AJ, King A, Zong WX, Rathmell JC, Shiels HA, et al. The combined functions of proapoptotic Bcl-2 family members bak and bax are essential for normal development of multiple tissues. Mol Cell. 2000;6:1389–99.
Wei MC, Zong WX, Cheng EH, Lindsten T, Panoutsakopoulou V, Ross AJ, et al. Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science. 2001;292:727–30.
Kushimoto S, Taira Y, Kitazawa Y, Okuchi K, Sakamoto T, Ishikura H, et al. The clinical usefulness of extravascular lung water and pulmonary vascular permeability index to diagnose and characterize pulmonary edema: a prospective multicenter study on the quantitative differential diagnostic definition for acute lung injury/acute respiratory distress syndrome. Crit Care. 2012;16:R232.
Villar J, Zhang H, Slutsky AS. Lung repair and regeneration in ARDS: role of PECAM1 and Wnt signaling. Chest. 2019;155:587–94.
Michalick L, Weidenfeld S, Grimmer B, Fatykhova D, Solymosi PD, Behrens F, et al. Plasma mediators in patients with severe COVID-19 cause lung endothelial barrier failure. Eur Respir J. 2021;57:2002384.
Varga Z, Flammer AJ, Steiger P, Haberecker M, Andermatt R, Zinkernagel AS, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395:1417–8.
Herzog EL, Van Arnam J, Hu B, Krause DS. Threshold of lung injury required for the appearance of marrow-derived lung epithelia. Stem Cells. 2006;24:1986–92.
Tan KS, Lim RL, Liu J, Ong HH, Tan VJ, Lim HF, et al. Respiratory viral infections in exacerbation of chronic airway inflammatory diseases: novel mechanisms and insights from the upper airway epithelium. Front Cell Dev Biol. 2020;8:99.
Yao XH, He ZC, Li TY, Zhang HR, Wang Y, Mou H, et al. Pathological evidence for residual SARS-CoV-2 in pulmonary tissues of a ready-for-discharge patient. Cell Res. 2020;30:541–3.
Stehle D, Grimm M, Einsele-Scholz S, Ladwig F, Johanning J, Fischer G, et al. Contribution of BH3-domain and transmembrane-domain to the activity and interaction of the pore-forming Bcl-2 Proteins Bok, Bak, and Bax. Sci Rep. 2018;8:12434.
Echeverry N, Bachmann D, Ke F, Strasser A, Simon HU, Kaufmann T. Intracellular localization of the BCL-2 family member BOK and functional implications. Cell Death Differ. 2013;20:785–99.
Zheng JH, Grace CR, Guibao CD, McNamara DE, Llambi F, Wang YM, et al. Intrinsic instability of BOK enables membrane permeabilization in apoptosis. Cell Rep. 2018;23:2083–94.
Gordon DE, Jang GM, Bouhaddou M, Xu J, Obernier K, O’Meara MJ, et al. A SARS-CoV-2-human protein–protein interaction map reveals drug targets and potential drug-repurposing. bioRxiv. 2020. Preprint.
Fernandez-Marrero Y, Bleicken S, Das KK, Bachmann D, Kaufmann T, Garcia-Saez AJ. The membrane activity of BOK involves formation of large, stable toroidal pores and is promoted by cBID. FEBS J. 2017;284:711–24.
Zha H, Aime-Sempe C, Sato T, Reed JC. Proapoptotic protein Bax heterodimerizes with Bcl-2 and homodimerizes with Bax via a novel domain (BH3) distinct from BH1 and BH2. J Biol Chem. 1996;271:7440–4.
Einsele-Scholz S, Malmsheimer S, Bertram K, Stehle D, Johanning J, Manz M, et al. Bok is a genuine multi-BH-domain protein that triggers apoptosis in the absence of Bax and Bak. J Cell Sci. 2016;129:3054.
Zhang F, Ren L, Zhou S, Duan P, Xue J, Chen H, et al. Role of B-Cell Lymphoma 2 Ovarian Killer (BOK) in acute toxicity of human lung epithelial cells caused by cadmium chloride. Med Sci Monit. 2019;25:5356–68.
Acknowledgements
We thank Dr. Li Huiyan for providing plentiCRISPR v2 and pLVX-nCoV-M-mCherry.
Funding
This work was supported by grants from the National Natural Science Foundation of China (31470877), the National Science and Technology Key Projects for Major Infectious Diseases (2017ZX10302301-002), the Support Scheme of Guangzhou for Leading Talents in Innovation and Entrepreneurship (No.2017004), the Guangdong Scientific and Technological Research Project for Prevention and Treatment of COVID-19 (2020A111128022, 2020B111112003), the Guangdong Scientific and Technological Research Special Fund for COVID-19 (202020012612200001), the Zhuhai Scientific and Technological Research Project for COVID-19 containment (ZH22036302200029PWC), the Zhuhai Industrial Technological Research and Development Project for Prevention and Treatment of COVID-19 (ZH22046301200018PWC), the Three Major Scientific Research Projects of Sun Yat-sen University (20200326236) (X.H.), the Science and Technology Planning Project of Guangzhou (201704020226 and 201604020006), and Development Project of Foshan Fourth People’s Hospital (FSSYKF-2020003, FSSYKF-2020017).
Author information
Authors and Affiliations
Contributions
YY, WYJ, and HX conceptualized study. WYJ, YY, MXJ and WZY collected, analyzed the data and edited the manuscript. All co-authors made comments to the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Our studies did not include human participants, human data or human tissue. The animal studies were approved by the Ethics Committee of Sun-Yat Sen University (No.00109), in accordance with UK Animals (Scientific Procedures) Act 1986.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by DL Vaux
Rights and permissions
About this article
Cite this article
Yang, Y., Wu, Y., Meng, X. et al. SARS-CoV-2 membrane protein causes the mitochondrial apoptosis and pulmonary edema via targeting BOK. Cell Death Differ 29, 1395–1408 (2022). https://doi.org/10.1038/s41418-022-00928-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41418-022-00928-x
This article is cited by
-
Production of SARS-CoV-2 virus-like particles as a vaccine candidate in stable cell lines through inducible E and M protein expression
BMC Biotechnology (2025)
-
The role of programmed cell death in organ dysfunction induced by opportunistic pathogens
Critical Care (2025)
-
Mitochondrial haplogroup A2 is associated with increased COVID-19 mortality in an admixed Brazilian population
Scientific Reports (2025)
-
SARS-CoV-2 membrane protein induces neurodegeneration via affecting Golgi-mitochondria interaction
Translational Neurodegeneration (2024)
-
A glimpse into viral warfare: decoding the intriguing role of highly pathogenic coronavirus proteins in apoptosis regulation
Journal of Biomedical Science (2024)