Abstract
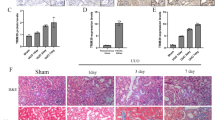
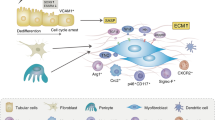
Chronic kidney disease (CKD) is a major global health concern and the third leading cause of premature death. Renal fibrosis is the primary process driving the progression of CKD, but the mechanisms behind it are not fully understood, making treatment options limited. Here, we find that the E3 ligase TRIM65 is a positive regulator of renal fibrosis. Deletion of TRIM65 results in a reduction of pathological lesions and renal fibrosis in mouse models of kidney fibrosis induced by unilateral ureteral obstruction (UUO)- and folic acid. Through screening with a yeast-hybrid system, we identify a new interactor of TRIM65, the mammalian cleavage factor I subunit CFIm25 (NUDT21), which plays a crucial role in fibrosis through alternative polyadenylation (APA). TRIM65 interacts with NUDT21 to induce K48-linked polyubiquitination of lysine 56 and proteasomal degradation, leading to the inhibition of TGF-β1-mediated SMAD and ERK1/2 signaling pathways. The degradation of NUDT21 subsequently altered the length and sequence content of the 3′UTR (3′UTR-APA) of several pro-fibrotic genes including Col1a1, Fn-1, Tgfbr1, Wnt5a, and Fzd2. Furthermore, reducing NUDT21 expression via hydrodynamic renal pelvis injection of adeno-associated virus 9 (AAV9) exacerbated UUO-induced renal fibrosis in the normal mouse kidneys and blocked the protective effect of TRIM65 deletion. These findings suggest that TRIM65 promotes renal fibrosis by regulating NUDT21-mediated APA and highlight TRIM65 as a potential target for reducing renal fibrosis in CKD patients.

Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
The data analyzed during this study are included in this published article. Additional supporting data are available from the corresponding authors upon reasonable request.
References
Romagnani P, Remuzzi G, Glassock R, Levin A, Jager KJ, Tonelli M, et al. Chronic kidney disease. Nat Rev Dis Prim. 2017;3:17088.
Carney EF. The impact of chronic kidney disease on global health. Nat Rev Nephrol. 2020;16:251.
Glassock RJ, Warnock DG, Delanaye P. The global burden of chronic kidney disease: estimates, variability and pitfalls. Nat Rev Nephrol. 2017;13:104–14.
Mutsaers HA, Olinga P. Editorial: organ fibrosis: triggers, pathways, and cellular plasticity. Front Med. 2016;3:55.
Huang R, Fu P, Ma L. Kidney fibrosis: from mechanisms to therapeutic medicines. Signal Transduct Target Ther. 2023;8:129.
Liu BC, Tang TT, Lv LL, Lan HY. Renal tubule injury: a driving force toward chronic kidney disease. Kidney Int. 2018;93:568–79.
Leaf IA, Duffield JS. What can target kidney fibrosis? Nephrol Dial Transpl. 2017;32:i89–97.
Meng XM, Nikolic-Paterson DJ, Lan HY. Inflammatory processes in renal fibrosis. Nat Rev Nephrol. 2014;10:493–503.
Yang J, Liu Y. Dissection of key events in tubular epithelial to myofibroblast transition and its implications in renal interstitial fibrosis. Am J Pathol. 2001;159:1465–75.
Corbett AH. Post-transcriptional regulation of gene expression and human disease. Curr Opin Cell Biol. 2018;52:96–104.
Ren F, Zhang N, Zhang L, Miller E, Pu JJ. Alternative Polyadenylation: a new frontier in post transcriptional regulation. Biomark Res. 2020;8:67.
Hao Y, Cai T, Liu C, Zhang X, Fu XD. Sequential Polyadenylation to Enable Alternative mRNA 3′ End Formation. Mol Cells. 2023;46:57–64.
Yuan F, Hankey W, Wagner EJ, Li W, Wang Q. Alternative polyadenylation of mRNA and its role in cancer. Genes Dis. 2021;8:61–72.
Xiao S, Gu H, Deng L, Yang X, Qiao D, Zhang X, et al. Relationship between NUDT21 mediated alternative polyadenylation process and tumor. Front Oncol. 2023;13:1052012.
Weng T, Ko J, Masamha CP, Xia Z, Xiang Y, Chen NY, et al. Cleavage factor 25 deregulation contributes to pulmonary fibrosis through alternative polyadenylation. J Clin Investig. 2019;129:1984–99.
Tan Y, Zheng T, Zhang R, Chen S, Cheng Q, Zhang J, et al. Alternative polyadenylation writer CSTF2 forms a positive loop with FGF2 to promote tubular epithelial-mesenchymal transition and renal fibrosis. Biochim Biophys Acta Mol Basis Dis. 2022;1868:166541.
Zheng T, Tan Y, Qiu J, Xie Z, Hu X, Zhang J, et al. Alternative polyadenylation trans-factor FIP1 exacerbates UUO/IRI-induced kidney injury and contributes to AKI-CKD transition via ROS-NLRP3 axis. Cell Death Dis. 2021;12:512.
Masamha CP. The emerging roles of CFIm25 (NUDT21/CPSF5) in human biology and disease. Wiley Interdiscip Rev RNA. 2023;14:e1757.
De Silva ARI, Page RC. Ubiquitination detection techniques. Exp Biol Med. 2023;248:1333–46.
Carroll EC, Marqusee S. Site-specific ubiquitination: deconstructing the degradation tag. Curr Opin Struct Biol. 2022;73:102345.
Meyer-Schwesinger C. The ubiquitin-proteasome system in kidney physiology and disease. Nat Rev Nephrol. 2019;15:393–411.
Chen J, Feng X, Zhou X, Li Y. Role of the tripartite motif-containing (TRIM) family of proteins in insulin resistance and related disorders. Diabetes Obes Metab. 2023;26:3–15.
Huang Y, Xiao Y, Zhang X, Huang X, Li Y. The emerging roles of Tripartite Motif Proteins (TRIMs) in acute lung injury. J Immunol Res. 2021;2021:1007126.
Huang N, Sun X, Li P, Liu X, Zhang X, Chen Q, et al. TRIM family contribute to tumorigenesis, cancer development, and drug resistance. Exp Hematol Oncol. 2022;11:75.
Shen Z, Wei L, Yu ZB, Yao ZY, Cheng J, Wang YT, et al. The roles of TRIMs in antiviral innate immune signaling. Front Cell Infect Microbiol. 2021;11:628275.
Huang Y, Chen T, Jiang M, Xiong C, Mei C, Nie J, et al. E3 ligase TRIM65 alleviates intestinal ischemia/reperfusion injury through inhibition of TOX4-mediated apoptosis. Cell Death Dis. 2024;15:29.
Lu Y, Xiao Y, Yang J, Su H, Zhang X, Su F, et al. TRIM65 promotes malignant cell behaviors in triple-negative breast cancer by impairing the stability of LATS1 protein. Oxid Med Cell Longev. 2022;2022:4374978.
Li Y, Huang X, Guo F, Lei T, Li S, Monaghan-Nichols P, et al. TRIM65 E3 ligase targets VCAM-1 degradation to limit LPS-induced lung inflammation. J Mol Cell Biol. 2020;12:190–201.
Liu B, Tang Y, Yang P, Wu C, Huang Y. TRIM65 in white matter lesions, innate immunity, and tumor. Curr Mol Pharm. 2021;14:798–805.
Woodard LE, Welch RC, Williams FM, Luo W, Cheng J, Wilson MH. Hydrodynamic renal pelvis injection for non-viral expression of proteins in the kidney. J Vis Exp. 2018;131:e56324.
Grgic I, Campanholle G, Bijol V, Wang C, Sabbisetti VS, Ichimura T, et al. Targeted proximal tubule injury triggers interstitial fibrosis and glomerulosclerosis. Kidney Int. 2012;82:172–83.
Li S, Wang L, Fu B, Berman MA, Diallo A, Dorf ME. TRIM65 regulates microRNA activity by ubiquitination of TNRC6. Proc Natl Acad Sci USA. 2014;111:6970–5.
Peng Z, Wang H, Zheng J, Wang J, Xiang Y, Liu C, et al. Is the proximal tubule the focus of tubulointerstitial fibrosis? Heliyon. 2023;9:e13508.
Zhang WR, Parikh CR. Biomarkers of acute and chronic kidney disease. Annu Rev Physiol. 2019;81:309–33.
Ren LL, Miao H, Wang YN, Liu F, Li P, Zhao YY. TGF-beta as a master regulator of aging-associated tissue fibrosis. Aging Dis. 2023;14:1633–50.
Chen X, Shi W, Zhu L, Zhou X, Wang Y. Mammalian cleavage factor 25 targets KLF14 to inhibit hepatic stellate cell activation and liver fibrosis. Cell Signal. 2023;109:110752.
Weng T, Huang J, Wagner EJ, Ko J, Wu M, Wareing NE, et al. Downregulation of CFIm25 amplifies dermal fibrosis through alternative polyadenylation. J Exp Med. 2020;217:e20181384.
Zhang Q, Li Y, Zhu Q, Xie T, Xiao Y, Zhang F, et al. TRIM65 promotes renal cell carcinoma through ubiquitination and degradation of BTG3. Cell Death Dis. 2024;15:355.
Xiao Y, Liu R, Li N, Li Y, Huang X. Role of the ubiquitin-proteasome system on macrophages in the tumor microenvironment. J Cell Physiol. 2024;239:e31180.
Li Y, Li S, Wu H. Ubiquitination-Proteasome System (UPS) and autophagy two main protein degradation machineries in response to cell stress. Cells. 2022;11:851.
Huang X, Dixit VM. Drugging the undruggables: exploring the ubiquitin system for drug development. Cell Res. 2016;26:484–98.
Liu W, Yi Y, Zhang C, Zhou B, Liao L, Liu W, et al. The expression of TRIM6 activates the mTORC1 pathway by regulating the ubiquitination of TSC1-TSC2 to promote renal fibrosis. Front Cell Dev Biol. 2020;8:616747.
Liao L, Duan L, Guo Y, Zhou B, Xu Q, Zhang C, et al. TRIM46 upregulates Wnt/beta-catenin signaling by inhibiting Axin1 to mediate hypoxia-induced epithelial-mesenchymal transition in HK2 cells. Mol Cell Biochem. 2022;477:2829–39.
Zhang J, Cao L, Wang X, Li Q, Zhang M, Cheng C, et al. The E3 ubiquitin ligase TRIM31 plays a critical role in hypertensive nephropathy by promoting proteasomal degradation of MAP3K7 in the TGF-beta1 signaling pathway. Cell Death Differ. 2022;29:556–67.
Tamaddon M, Shokri G, Hosseini Rad SMA, Rad I, Emami Razavi A, Kouhkan F. Involved microRNAs in alternative polyadenylation intervene in breast cancer via regulation of cleavage factor “CFIm25”. Sci Rep. 2020;10:11608.
Shimazu T, Horinouchi S, Yoshida M. Multiple histone deacetylases and the CREB-binding protein regulate pre-mRNA 3′-end processing. J Biol Chem. 2007;282:4470–8.
Sartini BL, Wang H, Wang W, Millette CF, Kilpatrick DL. Pre-messenger RNA cleavage factor I (CFIm): potential role in alternative polyadenylation during spermatogenesis. Biol Reprod. 2008;78:472–82.
Ren LL, Li XJ, Duan TT, Li ZH, Yang JZ, Zhang YM, et al. Transforming growth factor-beta signaling: From tissue fibrosis to therapeutic opportunities. Chem Biol Interact. 2023;369:110289.
Acknowledgements
We appreciate Dr. Hongbin Shu and Dr. Shu Li (School of Medicine, Wuhan University) for providing HA-Ub mutant plasmids. We thank Alan Jiang (The First Affiliated Hospital, Jiangxi Medical College, Nanchang University) for microscopy assistance.
Funding
This work was supported by grants from the National Natural Science Foundation of China (82160133, 31960147, and 32170793), Jiangxi Provincial Natural Science Foundation (20212ACB216005, 20224ACB216013, 20224BAB206007, and 20212BAB206086).
Author information
Authors and Affiliations
Contributions
YL conceived and designed the study, prepared the figures, analyzed data, and participated in the paper writing. SW and XH performed the experiments, analyzed the data, and wrote the first draft of the manuscript. QZhu, TC, JT, TP, and XK conducted experiments during the study. LZ, TX, and QZ bred and genotyped the mice. YZ critically revised the manuscript for important intellectual content. EL reviewed the manuscript. All authors approved and contributed to the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All animal procedures were approved by The Institutional Animal Care and Use Committee of The First Affiliated Hospital of Nanchang University and performed in accordance with the ARRIVE guideline. All methods were carried out in accordance with relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wei, S., Huang, X., Zhu, Q. et al. TRIM65 deficiency alleviates renal fibrosis through NUDT21-mediated alternative polyadenylation. Cell Death Differ 31, 1422–1438 (2024). https://doi.org/10.1038/s41418-024-01336-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41418-024-01336-z
This article is cited by
-
The role of TRIM proteins in the pathogenesis of mycobacterium tuberculosis
Biology Direct (2025)
-
TRIM2 inhibits apoptosis by ubiquitinating BNIP3 to protect the intestine against ischemia-reperfusion injury in mice
Communications Biology (2025)
-
USP7 promotes endothelial activation to aggravate sepsis-induced acute lung injury through PDK1/AKT/NF-κB signaling pathway
Cell Death Discovery (2025)