Abstract
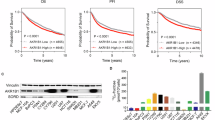
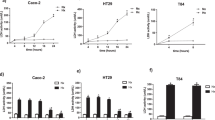
Excessive fructose intake has been associated with the development and progression of pancreatic cancer. This study aimed to elucidate the relationship between fructose utilization and pancreatic cancer progression. Our findings revealed that pancreatic cancer cells have a high capacity to utilize fructose and are capable of converting glucose to fructose via the AKR1B1-mediated polyol pathway, in addition to uptake via the fructose transporter GLUT5. Fructose metabolism exacerbates pancreatic cancer proliferation by enhancing glycolysis and accelerating the production of key metabolites that regulate angiogenesis. However, pharmacological blockade of fructose metabolism has been shown to slow pancreatic cancer progression and synergistically enhance anti-tumor capabilities when combined with anti-angiogenic agents. Overall, targeting fructose metabolism may prove to be a promising therapeutic approach in the treatment of pancreatic cancer.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Qin C, Yang G, Yang J, Ren B, Wang H, Chen G, et al. Metabolism of pancreatic cancer: paving the way to better anticancer strategies. Mol Cancer. 2020;19:50.
Vincent A, Herman J, Schulick R, Hruban RH, Goggins M. Pancreatic cancer. Lancet. 2011;378:607–20.
Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74:12–49.
Halbrook CJ, Lyssiotis CA, Pasca di Magliano M, Maitra A. Pancreatic cancer: Advances and challenges. Cell. 2023;186:1729–54.
Huang L, Jansen L, Balavarca Y, Molina-Montes E, Babaei M, van der Geest L, et al. Resection of pancreatic cancer in Europe and USA: an international large-scale study highlighting large variations. Gut. 2019;68:130–9.
Strobel O, Neoptolemos J, Jäger D, Büchler MW. Optimizing the outcomes of pancreatic cancer surgery. Nat Rev Clin Oncol. 2019;16:11–26.
Herman MA, Birnbaum MJ. Molecular aspects of fructose metabolism and metabolic disease. Cell Metab. 2021;33:2329–54.
Jensen T, Abdelmalek MF, Sullivan S, Nadeau KJ, Green M, Roncal C, et al. Fructose and sugar: A major mediator of non-alcoholic fatty liver disease. J Hepatol. 2018;68:1063–75.
Mirtschink P, Jang C, Arany Z, Krek W. Fructose metabolism, cardiometabolic risk, and the epidemic of coronary artery disease. Eur Heart J. 2018;39:2497–505.
Wu Y, Wong CW, Chiles EN, Mellinger AL, Bae H, Jung S, et al. Glycerate from intestinal fructose metabolism induces islet cell damage and glucose intolerance. Cell Metab. 2022;34:1042–53.e6.
Carreño DV, Corro NB, Cerda-Infante JF, Echeverría CE, Asencio-Barría CA, Torres-Estay VA, et al. Dietary Fructose Promotes Prostate Cancer Growth. Cancer Res. 2021;81:2824–32.
Kuehm LM, Khojandi N, Piening A, Klevorn LE, Geraud SC, McLaughlin NR, et al. Fructose Promotes Cytoprotection in Melanoma Tumors and Resistance to Immunotherapy. Cancer Immunol Res. 2021;9:227–38.
Kim J, Kang J, Kang YL, Woo J, Kim Y, Huh J, et al. Ketohexokinase-A acts as a nuclear protein kinase that mediates fructose-induced metastasis in breast cancer. Nat Commun. 2020;11:5436.
Bu P, Chen KY, Xiang K, Johnson C, Crown SB, Rakhilin N, et al. Aldolase B-Mediated Fructose Metabolism Drives Metabolic Reprogramming of Colon Cancer Liver Metastasis. Cell Metab. 2018;27:1249–62.e4.
Chen WL, Wang YY, Zhao A, Xia L, Xie G, Su M, et al. Enhanced Fructose Utilization Mediated by SLC2A5 Is a Unique Metabolic Feature of Acute Myeloid Leukemia with Therapeutic Potential. Cancer Cell. 2016;30:779–91.
Chen WL, Jin X, Wang M, Liu D, Luo Q, Tian H, et al. GLUT5-mediated fructose utilization drives lung cancer growth by stimulating fatty acid synthesis and AMPK/mTORC1 signaling. JCI Insight. 2020;5:e131596.
Shen Z, Li Z, Liu Y, Li Y, Feng X, Zhan Y, et al. GLUT5-KHK axis-mediated fructose metabolism drives proliferation and chemotherapy resistance of colorectal cancer. Cancer Lett. 2022;534:215617.
Goncalves MD, Lu C, Tutnauer J, Hartman TE, Hwang SK, Murphy CJ, et al. High-fructose corn syrup enhances intestinal tumor growth in mice. Science. 2019;363:1345–9.
Jeong S, Savino AM, Chirayil R, Barin E, Cheng Y, Park SM, et al. High Fructose Drives the Serine Synthesis Pathway in Acute Myeloid Leukemic Cells. Cell Metab. 2021;33:145–59.e6.
Hannou SA, Haslam DE, McKeown NM, Herman MA. Fructose metabolism and metabolic disease. J Clin Invest. 2018;128:545–55.
Han B, Wang L, Zhang J, Wei M, Rajani C, Wei R, et al. Fructose Metabolism Contributes to the Warburg effect. bioRxiv. 2021. https://www.biorxiv.org/content/10.1101/2020.06.04.132902v4.
Kang YL, Kim J, Kwak SB, Kim YS, Huh J, Park JW. The polyol pathway and nuclear ketohexokinase A signaling drive hyperglycemia-induced metastasis of gastric cancer. Exp Mol Med. 2024;56:220–34.
Schwab A, Siddiqui A, Vazakidou ME, Napoli F, Böttcher M, Menchicchi B, et al. Polyol Pathway Links Glucose Metabolism to the Aggressiveness of Cancer Cells. Cancer Res. 2018;78:1604–18.
An YF, Pu N, Jia JB, Wang WQ, Liu L. Therapeutic advances targeting tumor angiogenesis in pancreatic cancer: Current dilemmas and future directions. Biochim Biophys Acta Rev Cancer. 2023;1878:188958.
Kerr J, Anderson C, Lippman SM. Physical activity, sedentary behaviour, diet, and cancer: an update and emerging new evidence. Lancet Oncol. 2017;18:e457–e71.
Malik VS, Hu FB. The role of sugar-sweetened beverages in the global epidemics of obesity and chronic diseases. Nat Rev Endocrinol. 2022;18:205–18.
Chen CH, Tsai MK, Lee JH, Lin RT, Hsu CY, Wen C, et al. Sugar-Sweetened Beverages” Is an Independent Risk From Pancreatic Cancer: Based on Half a Million Asian Cohort Followed for 25 Years. Front Oncol. 2022;12:835901.
Aune D, Chan DSM, Vieira AR, Navarro Rosenblatt DA, Vieira R, Greenwood DC, et al. Dietary fructose, carbohydrates, glycemic indices and pancreatic cancer risk: a systematic review and meta-analysis of cohort studies. Ann Oncol. 2012;23:2536–46.
Huang Y, Chen Z, Chen B, Li J, Yuan X, Li J, et al. Dietary sugar consumption and health: umbrella review. Bmj. 2023;381:e071609.
Krause N, Wegner A. Fructose Metabolism in Cancer. Cells. 2020;9:2635.
Yabe-Nishimura C. Aldose reductase in glucose toxicity: a potential target for the prevention of diabetic complications. Pharmacol Rev. 1998;50:21–33.
Zhang KR, Zhang YF, Lei HM, Tang YB, Ma CS, Lv QM, et al. Targeting AKR1B1 inhibits glutathione de novo synthesis to overcome acquired resistance to EGFR-targeted therapy in lung cancer. Sci Transl Med. 2021;13:eabg6428.
Wu X, Li X, Fu Q, Cao Q, Chen X, Wang M, et al. AKR1B1 promotes basal-like breast cancer progression by a positive feedback loop that activates the EMT program. J Exp Med. 2017;214:1065–79.
Kindler HL, Ioka T, Richel DJ, Bennouna J, Létourneau R, Okusaka T, et al. Axitinib plus gemcitabine versus placebo plus gemcitabine in patients with advanced pancreatic adenocarcinoma: a double-blind randomised phase 3 study. Lancet Oncol. 2011;12:256–62.
Kindler HL, Niedzwiecki D, Hollis D, Sutherland S, Schrag D, Hurwitz H, et al. Gemcitabine plus bevacizumab compared with gemcitabine plus placebo in patients with advanced pancreatic cancer: phase III trial of the Cancer and Leukemia Group B (CALGB 80303). J Clin Oncol. 2010;28:3617–22.
Hanson RL, Ho RS, Wiseberg JJ, Simpson R, Younathan ES, Blair JB. Inhibition of gluconeogenesis and glycogenolysis by 2,5-anhydro-D-mannitol. J Biol Chem. 1984;259:218–23.
Tordoff MG, Rafka R, DiNovi MJ, Friedman MI. 2,5-anhydro-D-mannitol: a fructose analogue that increases food intake in rats. Am J Physiol. 1988;254:R150–3.
Caligiuri G. CD31 as a Therapeutic Target in Atherosclerosis. Circ Res. 2020;126:1178–89.
Park S, Sorenson CM, Sheibani N. PECAM-1 isoforms, eNOS and endoglin axis in regulation of angiogenesis. Clin Sci (Lond). 2015;129:217–34.
Zhang YY, Kong LQ, Zhu XD, Cai H, Wang CH, Shi WK, et al. CD31 regulates metastasis by inducing epithelial-mesenchymal transition in hepatocellular carcinoma via the ITGB1-FAK-Akt signaling pathway. Cancer Lett. 2018;429:29–40.
Dunleavey JM, Xiao L, Thompson J, Kim MM, Shields JM, Shelton SE, et al. Vascular channels formed by subpopulations of PECAM1+ melanoma cells. Nat Commun. 2014;5:5200.
Cui Y, Liu H, Wang Z, Zhang H, Tian J, Wang Z, et al. Fructose promotes angiogenesis by improving vascular endothelial cell function and upregulating VEGF expression in cancer cells. J Exp Clin Cancer Res. 2023;42:184.
Fang JH, Chen JY, Zheng JL, Zeng HX, Chen JG, Wu CH, et al. Fructose Metabolism in Tumor Endothelial Cells Promotes Angiogenesis by Activating AMPK Signaling and Mitochondrial Respiration. Cancer Res. 2023;83:1249–63.
Zhou J, Liu T, Guo H, Cui H, Li P, Feng D, et al. Lactate potentiates angiogenesis and neurogenesis in experimental intracerebral hemorrhage. Exp Mol Med. 2018;50:1–12.
Brown TP, Ganapathy V. Lactate/GPR81 signaling and proton motive force in cancer: Role in angiogenesis, immune escape, nutrition, and Warburg phenomenon. Pharmacol Ther. 2020;206:107451.
Eelen G, Dubois C, Cantelmo AR, Goveia J, Brüning U, DeRan M, et al. Role of glutamine synthetase in angiogenesis beyond glutamine synthesis. Nature. 2018;561:63–9.
Acknowledgements
This work was supported by Guangdong-Hong Kong-Macao Joint Laboratory for New Drug Screening (2023B1212120005).
Author information
Authors and Affiliations
Contributions
W.J. conceptualized the study. W.J and C.W. designed the study and coordinated the experimental planning. C.W. and L.W. performed the experiments. C.W., J.M., X.Y, and Y.L. were responsible for mouse experiments. C.W. was responsible for cell studies. C.W. and L.W. were responsible for sample preparation and metabolomics analysis. C.W. K.C. and L.W. performed the data preprocessing and statistical analysis. C.W. and W.J. drafted the manuscript and produced the figures. G.M., A.L., H.B., Q.Z., J.K. and K.C. provided valuable suggestions in data analysis and interpretation. W.J. and C.W. critically revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All animal experiments were approved by the Research Ethics Committee of the Hong Kong Baptist University (REC/20–21/0584) and were performed in accordance with relevant guidelines and regulations. The relevant licenses were also approved by the Department of Health, Hong Kong, China (22-145 in DH/HT&A/8/2/6 Pt.6. and 21-189 in DH/HT&A/8/2/6 Pt.4.).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, C., Wang, L., Zhao, Q. et al. Exploring fructose metabolism as a potential therapeutic approach for pancreatic cancer. Cell Death Differ 31, 1625–1635 (2024). https://doi.org/10.1038/s41418-024-01394-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41418-024-01394-3
This article is cited by
-
New hope for the world cancer day
Biology Direct (2025)
-
Evidence of fructose metabolism in colorectal cancer
Cell Death Discovery (2025)
-
Role of fructose in renal cell carcinoma progression
Discover Oncology (2025)