Abstract
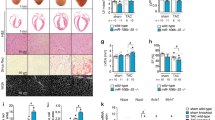
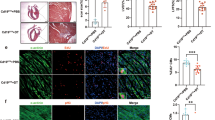
Activation of the intrinsic regenerative potential of adult mammalian hearts by promoting cardiomyocyte proliferation holds great potential in heart repair. CAND1 (Cullin-associated and neddylation-dissociated protein 1) functions as a critical regulator of cellular protein homeostasis by fine-tuning the ubiquitinated degradation of specific abnormally expressed protein substrates. Here, we identified that cardiac-specific transgenic overexpression of CAND1 reduced the infarct size, restored cardiac function, and promoted cardiomyocyte proliferation after myocardial infarction in juvenile (7-day-old) and adult (8-week-old) mice. Conversely, CAND1 deficiency blunted the regenerative capacity of neonatal hearts after apex resection. MS and functional verification demonstrated that CAND1 enhanced the assembly of Cullin1, FBXW11(F-box/WD repeat-containing protein 11), and Mob1b (Mps one binder kinase activator 1b) complexes, and thus promotes the degradation of Mob1b. The ubiquitination of Mob1b occurred at K108 and was linked by K48 of ubiquitin. Mob1b deletion partially rescued the loss of regenerative capacity in neonatal hearts induced by CAND1 deficiency and improved cardiac function in adult mice post-MI. Moreover, CAND1 promoted the proliferation of human induced pluripotent stem cell-derived cardiomyocytes (iPSC-CMs). Our data demonstrate that CAND1 promotes cardiomyocyte proliferation via FBXW11-mediated K48-linked ubiquitination degradation of Mob1b, and improves heart regeneration after cardiac injury. The findings provide a novel strategy to promote cardiac regeneration and repair.

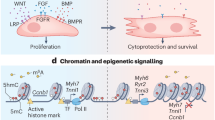
Schematic diagram of the role of CAND1 in regulating ubiquitination and degradation of Mob1b and cardiomyocyte proliferation and heart regeneration. Under CAND1-High condition, CAND1 promotes the incorporation of Cullin1, FBXW11, and Mob1b complexes, and accelerates SCFFBXW11-mediated K48-linked ubiquitination of Mob1b at the K108 site, which leads to the degradation of Mob1b and thus suppresses the Hippo signaling pathway and facilitates cardiomyocyte proliferation and heart regeneration post-MI.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its Supplementary Information files. Additional supporting data are available from the corresponding authors upon reasonable request.
References
Abouleisa RRE, Salama ABM, Ou Q, Tang XL, Solanki M, Guo Y, et al. Transient cell cycle induction in cardiomyocytes to treat subacute ischemic heart failure. Circulation. 2022;145:1339–55.
Chi C, Song K. Cellular reprogramming of fibroblasts in heart regeneration. J Mol Cell Cardiol. 2023;180:84–93.
Nishiyama C, Saito Y, Sakaguchi A, Kaneko M, Kiyonari H, Xu Y, et al. Prolonged myocardial regenerative capacity in neonatal opossum. Circulation. 2022;146:125–39.
Porrello ER, Mahmoud AI, Simpson E, Hill JA, Richardson JA, Olson EN, et al. Transient regenerative potential of the neonatal mouse heart. Science. 2011;331:1078–80.
Chen Y, Luttmann FF, Schoger E, Scholer HR, Zelarayan LC, Kim KP, et al. Reversible reprogramming of cardiomyocytes to a fetal state drives heart regeneration in mice. Science. 2021;373:1537–40.
Huang H, Huang GN, Payumo AY. Two decades of heart regeneration research: Cardiomyocyte proliferation and beyond. WIREs Mech Dis. 2024;16:e1629.
Mohamed TMA, Ang YS, Radzinsky E, Zhou P, Huang Y, Elfenbein A, et al. Regulation of cell cycle to stimulate adult cardiomyocyte proliferation and cardiac regeneration. Cell. 2018;173:104–16.e112.
Xiang FL, Guo M, Yutzey KE. Overexpression of Tbx20 in adult cardiomyocytes promotes proliferation and improves cardiac function after myocardial infarction. Circulation. 2016;133:1081–92.
Li Y, Feng J, Song S, Li H, Yang H, Zhou B, et al. gp130 controls cardiomyocyte proliferation and heart regeneration. Circulation. 2020;142:967–82.
Rigaud VOC, Hoy RC, Kurian J, Zarka C, Behanan M, Brosious I, et al. RNA-binding protein LIN28a regulates new myocyte formation in the heart through long noncoding RNA-H19. Circulation. 2023;147:324–37.
Nguyen NUN, Canseco DC, Xiao F, Nakada Y, Li S, Lam NT, et al. A calcineurin-Hoxb13 axis regulates growth mode of mammalian cardiomyocytes. Nature. 2020;582:271–6.
Zhang Y, Gago-Lopez N, Li N, Zhang Z, Alver N, Liu Y, et al. Single-cell imaging and transcriptomic analyses of endogenous cardiomyocyte dedifferentiation and cycling. Cell Discov. 2019;5:30.
Song L, Luo ZQ. Post-translational regulation of ubiquitin signaling. J Cell Biol. 2019;218:1776–86.
Xie Y, Zhao M. CAND1 orchestrates CRLs through rock and roll. Cell. 2023;186:1817–8.
Liu X, Reitsma JM, Mamrosh JL, Zhang Y, Straube R, Deshaies RJ. Cand1-mediated adaptive exchange mechanism enables variation in F-Box protein expression. Mol Cell. 2018;69:773–86.e776.
Shaaban M, Clapperton JA, Ding S, Kunzelmann S, Maeots ME, Maslen SL, et al. Structural and mechanistic insights into the CAND1-mediated SCF substrate receptor exchange. Mol Cell. 2023;83:2332–46.e2338.
Dubiel D, Ordemann J, Pratschke J, Dubiel W, Naumann M. CAND1 exchange factor promotes Keap1 integration into cullin 3-RING ubiquitin ligase during adipogenesis. Int J Biochem Cell Biol. 2015;66:95–100.
Zhang H, Xia P, Yang Z, Liu J, Zhu Y, Huang Z, et al. Cullin-associated and neddylation-dissociated 1 regulate reprogramming of lipid metabolism through SKP1-Cullin-1-F-box(FBXO11) -mediated heterogeneous nuclear ribonucleoprotein A2/B1 ubiquitination and promote hepatocellular carcinoma. Clin Transl Med. 2023;13:e1443.
Huang X, Liu X, Li X, Zhang Y, Gao J, Yang Y, et al. Cullin-associated and neddylation-dissociated protein 1 (CAND1) alleviates NAFLD by reducing ubiquitinated degradation of ACAA2. Nat Commun. 2023;14:4620.
Li X, Zhang Y, Zhao Y, Zhou Y, Han Q, Yang Y, et al. Cullin-associated and neddylation-dissociated 1 protein (CAND1) governs cardiac hypertrophy and heart failure partially through regulating calcineurin degradation. Pharmacol Res. 2022;182:106284.
Lam NT, Sadek HA. Neonatal heart regeneration: comprehensive literature review. Circulation. 2018;138:412–23.
Mahmoud AI, Kocabas F, Muralidhar SA, Kimura W, Koura AS, Thet S, et al. Meis1 regulates postnatal cardiomyocyte cell cycle arrest. Nature. 2013;497:249–53.
Couzens AL, Xiong S, Knight JDR, Mao DY, Guettler S, Picaud S, et al. MOB1 mediated phospho-recognition in the core mammalian hippo pathway. Mol Cell Proteom. 2017;16:1098–110.
Bothos J, Tuttle RL, Ottey M, Luca FC, Halazonetis TD. Human LATS1 is a mitotic exit network kinase. Cancer Res. 2005;65:6568–75.
Mudhasani R, Tran JP, Retterer C, Kota KP, Whitehouse CA, Bavari S. Protein kinase R degradation is essential for Rift Valley Fever virus infection and is regulated by SKP1-CUL1-F-box (SCF)FBXW11-NSs E3 ligase. PLoS Pathog. 2016;12:e1005437.
Hui X, Cao L, Xu T, Zhao L, Huang K, Zou Z, et al. PSMD12-mediated M1 ubiquitination of influenza A virus at K102 regulates viral replication. J Virol. 2022;96:e0078622.
Kodron A, Mussulini BH, Pilecka I, Chacinska A. The ubiquitin-proteasome system and its crosstalk with mitochondria as therapeutic targets in medicine. Pharmacol Res. 2021;163:105248.
Pohl C, Dikic I. Cellular quality control by the ubiquitin-proteasome system and autophagy. Science. 2019;366:818–22.
Xiong Y, Yu C, Zhang Q. Ubiquitin-proteasome system-regulated protein degradation in spermatogenesis. Cells. 2022;11:1058.
Li YF, Wang YX, Wang H, Ma Y, Wang LS. Posttranslational modifications: emerging prospects for cardiac regeneration therapy. J Cardiovasc Transl Res. 2022;15:49–60.
Dong W, Xie F, Chen XY, Huang WL, Zhang YZ, Luo WB, et al. Inhibition of Smurf2 translation by miR-322/503 protects from ischemia-reperfusion injury by modulating EZH2/Akt/GSK3beta signaling. Am J Physiol Cell Physiol. 2019;317:C253–61.
Huang S, Li X, Zheng H, Si X, Li B, Wei G, et al. Loss of super-enhancer-regulated circRNA Nfix induces cardiac regeneration after myocardial infarction in adult mice. Circulation. 2019;139:2857–76.
Pierce NW, Lee JE, Liu X, Sweredoski MJ, Graham RL, Larimore EA, et al. Cand1 promotes assembly of new SCF complexes through dynamic exchange of F box proteins. Cell. 2013;153:206–15.
Enchev RI, Schulman BA, Peter M. Protein neddylation: beyond cullin-RING ligases. Nat Rev Mol Cell Biol. 2015;16:30–44.
Harper JW, Schulman BA. Cullin-RING ubiquitin ligase regulatory circuits: a quarter century beyond the F-box hypothesis. Annu Rev Biochem. 2021;90:403–29.
Hua Z, Vierstra RD. The cullin-RING ubiquitin-protein ligases. Annu Rev Plant Biol. 2011;62:299–334.
Jiang L, Li J, Zhang C, Shang Y, Lin J. YAP‑mediated crosstalk between the Wnt and Hippo signaling pathways (Review). Mol Med Rep. 2020;22:4101–6.
Driskill JH, Pan D. Control of stem cell renewal and fate by YAP and TAZ. Nat Rev Mol Cell Biol. 2023;24:895–911.
Xin M, Kim Y, Sutherland LB, Murakami M, Qi X, McAnally J, et al. Hippo pathway effector Yap promotes cardiac regeneration. Proc Natl Acad Sci USA. 2013;110:13839–44.
Leach JP, Heallen T, Zhang M, Rahmani M, Morikawa Y, Hill MC, et al. Hippo pathway deficiency reverses systolic heart failure after infarction. Nature. 2017;550:260–4.
Monroe TO, Hill MC, Morikawa Y, Leach JP, Heallen T, Cao S, et al. YAP partially reprograms chromatin accessibility to directly induce adult cardiogenesis in vivo. Dev Cell. 2019;48:765–79.e767.
Cai B, Ma W, Wang X, Sukhareva N, Hua B, Zhang L, et al. Targeting LncDACH1 promotes cardiac repair and regeneration after myocardium infarction. Cell Death Differ. 2020;27:2158–75.
Hergovich A. MOB control: reviewing a conserved family of kinase regulators. Cell Signal. 2011;23:1433–40.
Nishio M, Sugimachi K, Goto H, Wang J, Morikawa T, Miyachi Y, et al. Dysregulated YAP1/TAZ and TGF-beta signaling mediate hepatocarcinogenesis in Mob1a/1b-deficient mice. Proc Natl Acad Sci USA. 2016;113:E71–80.
Otsubo K, Goto H, Nishio M, Kawamura K, Yanagi S, Nishie W, et al. MOB1-YAP1/TAZ-NKX2.1 axis controls bronchioalveolar cell differentiation, adhesion and tumour formation. Oncogene. 2017;36:4201–11.
Yang HB, Li Y, Li XH, Yan QM, Han XZ, Cao J, et al. The compensatory increase of Gli-similar 3 inhibited neuronal apoptosis through regulating Mps one binder kinase activator 1b (MOB1b): a possible strategy for the functional recovery after spinal cord injury. Exp Anim. 2024;73:61–72.
Si YJ, Ren QH, Bi L. miR-135b-5p regulates human mesenchymal stem cell osteogenic differentiation by facilitating the Hippo signaling pathway. Int J Clin Exp Pathol. 2017;10:7767–75.
Hirabayashi S, Nakagawa K, Sumita K, Hidaka S, Kawai T, Ikeda M, et al. Threonine 74 of MOB1 is a putative key phosphorylation site by MST2 to form the scaffold to activate nuclear Dbf2-related kinase 1. Oncogene. 2008;27:4281–92.
Miller DR, Thorburn A. Autophagy and organelle homeostasis in cancer. Dev Cell. 2021;56:906–18.
Xie F, Xu S, Lu Y, Wong KF, Sun L, Hasan KMM, et al. Metformin accelerates zebrafish heart regeneration by inducing autophagy. NPJ Regen Med. 2021;6:62.
Hao Z, Sheng Y, Duncan GS, Li WY, Dominguez C, Sylvester J, et al. K48-linked KLF4 ubiquitination by E3 ligase Mule controls T-cell proliferation and cell cycle progression. Nat Commun. 2017;8:14003.
Bae J, Salamon RJ, Brandt EB, Paltzer WG, Zhang Z, Britt EC, et al. Malonate Promotes Adult Cardiomyocyte Proliferation and Heart Regeneration. Circulation. 2021;143:1973–86.
Ma W, Tian Y, Shi L, Liang J, Ouyang Q, Li J, et al. N-Acetyltransferase 10 represses Uqcr11 and Uqcrb independently of ac4C modification to promote heart regeneration. Nat Commun. 2024;15:2137.
Ma W, Wang X, Sun H, Xu B, Song R, Tian Y, et al. Oxidant stress-sensitive circRNA Mdc1 controls cardiomyocyte chromosome stability and cell cycle re-entry during heart regeneration. Pharmacol Res. 2022;184:106422.
Funding
This work was supported by the Ministry of Science and Technology of China (YFA1800902 to ZP), National Natural Science Foundation of China (82430017, 82270245 to ZP, 82170393, 82470308 to YZ, 82070283 to YL, and 82330011, U21A20339 to BFY), Natural Science Foundation of Heilongjiang (LH2023H047 to XDL), and China Postdoctoral Science Foundation (2023MD744212 to XDL), Spring Goose Support Program: Young Talents Project funding of Heilongjiang Province (CYQN24041 to YZ).
Author information
Authors and Affiliations
Contributions
WP, BY, BC, and YL conceived the study concept. XL, LZ, and TT performed the animal experiments, conducted Western blot and qPCR analysis. YP, KW, and SW performed the histological examination of mice heart samples. XN, PZ, YQ, and HG performed cell culture. CL, XL, JY, and YZ helped culture hiPSCs-derived cardiomyocytes. HG, LX, and YZ performed IF staining. XL, LZ, and TT carried out the data analysis. WP and XL wrote the manuscript. WP and BY provided the funding. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Throughout the study, all experiments involving animals were approved by the Ethics Committees of Harbin Medical University and conformed to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85–23, revised 1996). Mice in the experiment were randomly assigned to experimental groups using computer-generated randomization. All experiments were permitted by the Animal Care and Use Committee of Harbin Medical University.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, X., Zhang, L., Tian, T. et al. Cullin-associated and neddylation-dissociated protein 1 (CAND1) promotes cardiomyocyte proliferation and heart regeneration by enhancing the ubiquitinated degradation of Mps one binder kinase activator 1b (Mob1b). Cell Death Differ 32, 2384–2398 (2025). https://doi.org/10.1038/s41418-025-01540-5
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41418-025-01540-5