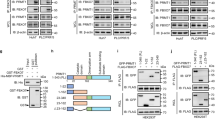
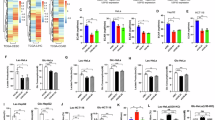
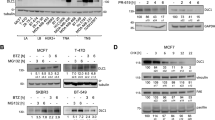
Abstract
Our previous studies demonstrated that the fat mass and obesity-associated protein (FTO) is upregulated in colorectal cancer (CRC). It demethylates G6PD/PARP1 and SLC7A11/GPX4 mRNAs, thereby protecting CRC from DNA damage and ferroptotic cell death. However, the mechanisms underlying FTO upregulation in CRC remain unclear. Unexpectedly, we show Ubiquitin-specific peptidase 30 (USP30) binds serine/glycine and senses their levels to protect FTO from proteosome degradation. Stabilized FTO demethylates 3-phosphoglycerate dehydrogenase (PHGDH) and phosphoserine aminotransferase 1 (PSAT1) mRNAs and inhibits their degradation in an m6A-YTHDF2-dependent manner, thereby promoting serine synthesis and CRC tumor growth. Furthermore, we identify sodium 2, 2-dichloroacetate (DCA) as a novel inhibitor of USP30, and DCA inhibits CRC serine synthesis and tumor growth. Clinically, USP30, FTO, PHGDH, and PSAT1 levels are highly correlated in CRC tissues. This study provides mechanistic insights into how USP30 senses serine/glycine levels to regulate serine synthesis via the FTO-PHGDH/PSAT1 axis, offering a potential therapeutic strategy for targeting serine/glycine metabolism in cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Data availability
Raw sequencing data have been deposited at the NCBI Sequence Read Archive and are publicly accessible. Other origin data, including origin image of western blot about this paper, have been deposited at Mendeley and are publicly available. Accession numbers or DOIs are listed in the key resources table. Any additional information required to reanalyze the data reported in this paper is available from the lead contact upon request. All the data supporting the findings of this study are available within the article and its supplemental files. Any data generated can be requested from the corresponding authors.
References
Luo L, Wu X, Fan J, Dong L, Wang M, Zeng Y, et al. FBXO7 ubiquitinates PRMT1 to suppress serine synthesis and tumor growth in hepatocellular carcinoma. Nat Commun. 2024;15:4790.
Wang K, Luo L, Fu S, Wang M, Wang Z, Dong L, et al. PHGDH arginine methylation by PRMT1 promotes serine synthesis and represents a therapeutic vulnerability in hepatocellular carcinoma. Nat Commun. 2023;14:1011.
Yamamoto T, Hayashida T, Masugi Y, Oshikawa K, Hayakawa N, Itoh M, et al. PRMT1 sustains De Novo fatty acid synthesis by methylating PHGDH to drive chemoresistance in triple-negative breast cancer. Cancer Res. 2024;84:1065–83.
Maddocks ODK, Athineos D, Cheung EC, Lee P, Zhang T, van den Broek NJF, et al. Modulating the therapeutic response of tumours to dietary serine and glycine starvation. Nature. 2017;544:372–6.
Sun W, Zhao E, Cui H. Target enzymes in serine-glycine-one-carbon metabolic pathway for cancer therapy. Int J Cancer. 2023;152:2446–63.
Jia G, Fu Y, Zhao X, Dai Q, Zheng G, Yang Y, et al. N6-methyladenosine in nuclear RNA is a major substrate of the obesity-associated FTO. Nat Chem Biol. 2011;7:885–7.
Huang Y, Su R, Sheng Y, Dong L, Dong Z, Xu H, et al. Small-molecule targeting of oncogenic FTO demethylase in acute myeloid leukemia. Cancer cell. 2019;35:677–691 e610.
Niu Y, Lin Z, Wan A, Chen H, Liang H, Sun L, et al. RNA N6-methyladenosine demethylase FTO promotes breast tumor progression through inhibiting BNIP3. Mol cancer. 2019;18:46.
Liu L, Gu M, Ma J, Wang Y, Li M, Wang H, et al. CircGPR137B/miR-4739/FTO feedback loop suppresses tumorigenesis and metastasis of hepatocellular carcinoma. Mol cancer. 2022;21:149.
Chen J, Du B. Novel positioning from obesity to cancer: FTO, an m(6)A RNA demethylase, regulates tumour progression. J Cancer Res Clin Oncol. 2019;145:19–29.
Qiao Y, Su M, Zhao H, Liu H, Wang C, Dai X, et al. Targeting FTO induces colorectal cancer ferroptotic cell death by decreasing SLC7A11/GPX4 expression. J Exp Clin Cancer Res. 2024;43:108.
Wang J, Qiao Y, Sun M, Sun H, Xie F, Chang H, et al. FTO promotes colorectal cancer progression and chemotherapy resistance via demethylating G6PD/PARP1. Clin Transl Med. 2022;12:e772.
Lee JM, Hammaren HM, Savitski MM, Baek SH. Control of protein stability by post-translational modifications. Nat Commun. 2023;14:201.
Dagar G, Kumar R, Yadav KK, Singh M, Pandita TK. Ubiquitination and deubiquitination: Implications on cancer therapy. Biochim Biophys Acta Gene Regul Mech. 2023;1866:194979.
Wang F, Gao Y, Zhou L, Chen J, Xie Z, Ye Z, et al. USP30: structure, emerging physiological role, and target inhibition. Front Pharm. 2022;13:851654.
Sun K, Liao S, Yao X, Yao F. USP30 promotes the progression of breast cancer by stabilising Snail. Cancer Gene Ther. 2024;31:472–83.
Gu L, Zhu Y, Lin X, Li Y, Cui K, Prochownik EV, et al. Amplification of glyceronephosphate O-acyltransferase and recruitment of USP30 stabilize DRP1 to promote hepatocarcinogenesis. Cancer Res. 2018;78:5808–19.
Gu L, Zhu Y, Lin X, Lu B, Zhou X, Zhou F, et al. The IKKbeta-USP30-ACLY axis controls lipogenesis and tumorigenesis. Hepatology. 2021;73:160–74.
Wagner SA, Beli P, Weinert BT, Nielsen ML, Cox J, Mann M, et al. A proteome-wide, quantitative survey of in vivo ubiquitylation sites reveals widespread regulatory roles. Mol Cell Proteom. 2011;10:M111 013284.
Ruan DY, Li T, Wang YN, Meng Q, Li Y, Yu K, et al. FTO downregulation mediated by hypoxia facilitates colorectal cancer metastasis. Oncogene. 2021;40:5168–81.
Akimov V, Barrio-Hernandez I, Hansen SVF, Hallenborg P, Pedersen AK, Bekker-Jensen DB, et al. UbiSite approach for comprehensive mapping of lysine and N-terminal ubiquitination sites. Nat Struct Mol Biol. 2018;25:631–40.
Boeing S, Williamson L, Encheva V, Gori I, Saunders RE, Instrell R, et al. Multiomic analysis of the UV-induced DNA damage response. Cell Rep. 2016;15:1597–610.
Udeshi ND, Svinkina T, Mertins P, Kuhn E, Mani DR, Qiao JW, et al. Refined preparation and use of anti-diglycine remnant (K-epsilon-GG) antibody enables routine quantification of 10,000s of ubiquitination sites in single proteomics experiments. Mol Cell Proteom. 2013;12:825–31.
Povlsen LK, Beli P, Wagner SA, Poulsen SL, Sylvestersen KB, Poulsen JW, et al. Systems-wide analysis of ubiquitylation dynamics reveals a key role for PAF15 ubiquitylation in DNA-damage bypass. Nat Cell Biol. 2012;14:1089–98.
Wang T, Kong S, Tao M, Ju S. The potential role of RNA N6-methyladenosine in Cancer progression. Mol Cancer. 2020;19:88.
Li J, Xie H, Ying Y, Chen H, Yan H, He L, et al. YTHDF2 mediates the mRNA degradation of the tumor suppressors to induce AKT phosphorylation in N6-methyladenosine-dependent way in prostate cancer. Mol Cancer. 2020;19:152.
Sun L, Song L, Wan Q, Wu G, Li X, Wang Y, et al. cMyc-mediated activation of serine biosynthesis pathway is critical for cancer progression under nutrient deprivation conditions. Cell Res. 2015;25:429–44.
Zhang C, Yu JJ, Yang C, Yuan ZL, Zeng H, Wang JJ, et al. Wild-type IDH1 maintains NSCLC stemness and chemoresistance through activation of the serine biosynthetic pathway. Sci Transl Med. 2023;15:eade4113.
Tajan M, Hennequart M, Cheung EC, Zani F, Hock AK, Legrave N, et al. Serine synthesis pathway inhibition cooperates with dietary serine and glycine limitation for cancer therapy. Nat Commun. 2021;12:366.
Gupta R, Laxman S. tRNA wobble-uridine modifications as amino acid sensors and regulators of cellular metabolic state. Curr Genet. 2020;66:475–80.
Forteza MJ, Berg M, Edsfeldt A, Sun J, Baumgartner R, Kareinen I, et al. Pyruvate dehydrogenase kinase regulates vascular inflammation in atherosclerosis and increases cardiovascular risk. Cardiovasc Res. 2023;119:1524–36.
Matsuoka K, Bakiri L, Bilban M, Toegel S, Haschemi A, Yuan H, et al. Metabolic rewiring controlled by c-Fos governs cartilage integrity in osteoarthritis. Ann Rheum Dis. 2023;82:1227–39.
Fujiwara S, Kawano Y, Yuki H, Okuno Y, Nosaka K, Mitsuya H, et al. PDK1 inhibition is a novel therapeutic target in multiple myeloma. Br J Cancer. 2013;108:170–8.
Kolb D, Kolishetti N, Surnar B, Sarkar S, Guin S, Shah AS, et al. Metabolic modulation of the tumor microenvironment leads to multiple checkpoint inhibition and immune cell infiltration. ACS Nano. 2020;14:11055–66.
Wang D, Xu C, Yang W, Chen J, Ou Y, Guan Y, et al. E3 ligase RNF167 and deubiquitinase STAMBPL1 modulate mTOR and cancer progression. Mol Cell. 2022;82:770–784.e779.
Li Z, Zhang H. Reprogramming of glucose, fatty acid and amino acid metabolism for cancer progression. Cell Mol Life Sci. 2016;73:377–92.
Nam A, Jain S, Wu C, Campos A, Shepard RM, Yu Z, et al. Integrin alphavbeta3 upregulation in response to nutrient stress promotes lung cancer cell metabolic plasticity. Cancer Res. 2024;84:1630–42.
Fernandez MR, Schaub FX, Yang C, Li W, Yun S, Schaub SK, et al. Disrupting the MYC-TFEB circuit impairs amino acid homeostasis and provokes metabolic anergy. Cancer Res. 2022;82:1234–50.
Jin HR, Wang J, Wang ZJ, Xi MJ, Xia BH, Deng K, et al. Lipid metabolic reprogramming in tumor microenvironment: from mechanisms to therapeutics. J Hematol Oncol. 2023;16:103.
Muthusamy T, Cordes T, Handzlik MK, You L, Lim EW, Gengatharan J, et al. Serine restriction alters sphingolipid diversity to constrain tumour growth. Nature. 2020;586:790–5.
Amelio I, Cutruzzola F, Antonov A, Agostini M, Melino G. Serine and glycine metabolism in cancer. Trends Biochem Sci. 2014;39:191–8.
Geeraerts SL, Heylen E, De Keersmaecker K, Kampen KR. The ins and outs of serine and glycine metabolism in cancer. Nat Metab. 2021;3:131–41.
Locasale JW. Serine, glycine and one-carbon units: cancer metabolism in full circle. Nat Rev Cancer. 2013;13:572–83.
Banh RS, Biancur DE, Yamamoto K, Sohn ASW, Walters B, Kuljanin M, et al. Neurons release serine to support mRNA translation in pancreatic cancer. Cell. 2020;183:1202–1218 e1225.
Bingol B, Tea JS, Phu L, Reichelt M, Bakalarski CE, Song Q, et al. The mitochondrial deubiquitinase USP30 opposes parkin-mediated mitophagy. Nature. 2014;510:370–5.
Fang TZ, Sun Y, Pearce AC, Eleuteri S, Kemp M, Luckhurst CA, et al. Knockout or inhibition of USP30 protects dopaminergic neurons in a Parkinson’s disease mouse model. Nat Commun. 2023;14:7295.
Cunningham CN, Baughman JM, Phu L, Tea JS, Yu C, Coons M, et al. USP30 and parkin homeostatically regulate atypical ubiquitin chains on mitochondria. Nat Cell Biol. 2015;17:160–9.
Liu Y, Yao C, Sheng B, Zhi S, Chen X, Ding P, et al. Inhibition of USP30 promotes mitophagy by regulating ubiquitination of MFN2 by Parkin to attenuate early brain injury after SAH. Transl Stroke Res. 2023.
Yang M, Vousden KH. Serine and one-carbon metabolism in cancer. Nat Rev Cancer. 2016;16:650–62.
Lau A, Blenis J, Burgos-Barragan G. Decoding serine metabolism: unveiling novel pathways for evolving cancer therapies. Cancer Res. 2024;84:1191–4.
Maddocks OD, Berkers CR, Mason SM, Zheng L, Blyth K, Gottlieb E, et al. Serine starvation induces stress and p53-dependent metabolic remodelling in cancer cells. Nature. 2013;493:542–6.
Ma Y, Zhang X, Han X, Hao Z, Ji R, Li J, et al. PDK1 regulates the progression of esophageal squamous cell carcinoma through metabolic reprogramming. Mol Carcinog. 2023;62:866–81.
Khan AUH, Allende-Vega N, Gitenay D, Gerbal-Chaloin S, Gondeau C, Vo DN, et al. The PDK1 inhibitor dichloroacetate controls cholesterol homeostasis through the ERK5/MEF2 pathway. Sci Rep. 2017;7:10654.
Lu H, Lu Y, Xie Y, Qiu S, Li X, Fan Z. Rational combination with PDK1 inhibition overcomes cetuximab resistance in head and neck squamous cell carcinoma. JCI Insight. 2019;4.
Acknowledgements
We thank Guo Chen (China Pharmaceutical University, Nanjing) for sharing plasmid DNA of UPSs and Yan Chen (Shandong Normal University, Jinan) for sharing plasmid DNA of HA-Ubiquitin mutants.
Funding
This work was supported by grants from National Nature Science Foundation of China (82473806, 82373906, and 82273963), Beijing Natural Science Foundation of China (grant no. J230016), National Science and Technology Major Project (2024ZD0521203), Beijing-Tianjin-Hebei Basic Research Cooperation Project (23JCZXJC00020), China Postdoctoral Science Foundation (2023M741829), Guangdong Basic and Applied Basic Research Foundation (2022A1515111102), the Youth Innovation Talent Project of Ordinary university in Guangdong Province, China (2022KQNCX061), and Guangzhou Science and Technology Plan Project (SL2022A04J00697).
Author information
Authors and Affiliations
Contributions
C.S., S.Z., C.Z., and Y.Q. conceived and designed the study and wrote the manuscript with comments from all authors. Y.Q., C.W., H.L., H.S., H.Z., Q.Z., X.D., M.S., T.W., and T.H. performed experiments. Z.L., Y.L., J.X. provided reagents and technical assistance. C.S., S.Z., and C.Z. wrote, reviewed, and edited the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflicts of interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qiao, Y., Wang, C., Liu, H. et al. USP30 senses serine/glycine levels to regulate serine biosynthesis and colorectal tumorigenesis by deubiquitinating FTO. Cell Death Differ (2026). https://doi.org/10.1038/s41418-026-01680-2
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41418-026-01680-2