Abstract
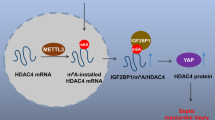
Sepsis, a life-threatening systemic inflammatory condition, frequently leads to myocardial injury-a complication for which current therapeutic strategies demonstrate limited efficacy. Here, we explored the potential role and therapeutic implications of Deltex E3 ubiquitin ligase 2 (DTX2) in sepsis-induced myocardial injury. Our results demonstrated that DTX2 expression was significantly upregulated in septic patients, mice models, and lipopolysaccharide (LPS)-stimulated cardiomyocytes. Notably, Dtx2 deficiency markedly aggravated sepsis-induced myocardial hypertrophy, fibrosis, ferroptosis, and mitochondrial dysfunction. In contrast, cardiac-specific overexpression of Dtx2 improved cardiac function in vivo, highlighting its protective role in septic cardiomyopathy. Mechanistically, DTX2 was found to directly interact with transferrin receptor 1 (TfR1) through its DTC domain, mediating K27-linked ubiquitination at lysine 39, which facilitated TfR1 degradation and regulated iron metabolism. Importantly, pharmacological inhibition of ferroptosis counteracted the detrimental effects of Dtx2 deficiency in both LPS-challenged cells and mice. Moreover, genetic silencing of TfR1 considerably suppressed ferroptosis and ameliorated myocardial injury in Dtx2 knockout septic mice. The findings indicate that DTX2 exerts protective effects against abnormal iron accumulation and ferroptosis, thereby alleviating myocardial injury induced by sepsis. These insights could have therapeutic implications for patients with reduced DTX2 expression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
References
Carey MG, Valcin EK, Lent D, White M. Nursing care for the initial resuscitation of severe sepsis patients. Crit Care Nurs Clin North Am. 2021;33:263–74.
Hollenberg SM, Singer M. Pathophysiology of sepsis-induced cardiomyopathy. Nat Rev Cardiol. 2021;18:424–34.
Dong Y, Wei S, Liu Y, Ji X, Yin X, Wu Z, et al. Aspirin is associated with improved outcomes in patients with sepsis-induced myocardial injury: an analysis of the MIMIC-IV database. J Clin Anesth. 2024;99:111597.
Frencken JF, Donker DW, Spitoni C, Koster-Brouwer ME, Soliman IW, Ong DSY, et al. Myocardial injury in patients with sepsis and its association with long-term outcome. Circ Cardiovasc Qual Outcomes. 2018;11:e004040.
Wang Z, Shen N, Wang Z, Yu L, Yang S, Wang Y, et al. TRIM3 facilitates ferroptosis in non-small cell lung cancer through promoting SLC7A11/xCT K11-linked ubiquitination and degradation. Cell Death Differ. 2024;31:53–64.
Cai W, Liu L, Shi X, Liu Y, Wang J, Fang X, et al. Alox15/15-HpETE aggravates myocardial ischemia-reperfusion injury by promoting cardiomyocyte ferroptosis. Circulation. 2023;147:1444–60.
Li K, Liu P, Han L, Tian J, Zheng Z, Sha M, et al. Elucidating ferroptosis mechanisms in heart failure through transcriptomics, single-cell sequencing, and experimental validation. Cell Signal. 2024;124:111416.
Kazmirczak F, Vogel NT, Prisco SZ, Patterson MT, Annis J, Moon RT, et al. Ferroptosis-mediated inflammation promotes pulmonary hypertension. Circ Res. 2024;135:1067–83.
Zhang H, Pan J, Huang S, Chen X, Chang A, Wang C, et al. Hydrogen sulfide protects cardiomyocytes from doxorubicin-induced ferroptosis through the SLC7A11/GSH/GPx4 pathway by Keap1 S-sulfhydration and Nrf2 activation. Redox Biol. 2024;70:103066.
Zhao M, Shen Z, Zheng Z, Xu Y, Zhang J, Liu J, et al. Cardiomyocyte LGR6 alleviates ferroptosis in diabetic cardiomyopathy via regulating mitochondrial biogenesis. Metabolism. 2024;159:155979.
Liu C, Zou Q, Tang H, Liu J, Zhang S, Fan C, et al. Melanin nanoparticles alleviate sepsis-induced myocardial injury by suppressing ferroptosis and inflammation. Bioact Mater. 2022;24:313–21.
Dutt S, Hamza I, Bartnikas TB. Molecular mechanisms of iron and heme metabolism. Annu Rev Nutr. 2022;42:311–35.
Lan T, Hu L, Sun T, Wang X, Xiao Z, Shen D, et al. H3K9 trimethylation dictates neuronal ferroptosis through repressing Tfr1. J Cereb Blood Flow Metab. 2023;43:1365–81.
Sanguigno L, Guida N, Anzilotti S, Cuomo O, Mascolo L, Serani A, et al. Stroke by inducing HDAC9-dependent deacetylation of HIF-1 and Sp1, promotes TfR1 transcription and GPX4 reduction, thus determining ferroptotic neuronal death. Int J Biol Sci. 2023;19:2695–710.
Wu Y, Jiao H, Yue Y, He K, Jin Y, Zhang J, et al. Ubiquitin ligase E3 HUWE1/MULE targets transferrin receptor for degradation and suppresses ferroptosis in acute liver injury. Cell Death Differ. 2022;29:1705–18.
Shao M, Qi K, Wang L, Yu X, Zhang Q, Yu L, et al. E3 ubiquitin ligase CHIP interacts with transferrin receptor 1 for degradation and promotes cell proliferation through inhibiting ferroptosis in hepatocellular carcinoma. Cell Signal. 2024;118:111148.
Ahmed SF, Buetow L, Gabrielsen M, Lilla S, Chatrin C, Sibbet GJ, et al. DELTEX2 C-terminal domain recognizes and recruits ADP-ribosylated proteins for ubiquitination. Sci Adv. 2020;6:eabc0629.
Liu Z, Liu C, Fan C, Li R, Zhang S, Liu J, et al. E3 ubiquitin ligase DTX2 fosters ferroptosis resistance via suppressing NCOA4-mediated ferritinophagy in non-small cell lung cancer. Drug Resist Updat. 2024;77:101154.
Wu X, Chen J, Chen Y, Song S, Fang Y, Mao S, et al. Targeting Deltex E3 ubiquitin ligase 2 inhibits tumor-associated neutrophils and sensitizes hepatocellular carcinoma cells to immunotherapy. Adv Sci (Weinh). 2025;12:e2408233.
Cui Y, Wei J, Fan H, Li W, Zhao L, Wilkinson E, et al. Targeting DTX2/UFD1-mediated FTO degradation to regulate antitumor immunity. Proc Natl Acad Sci USA. 2024;121:e2407910121.
Zhou Z, Li Y, Xu H, Xie X, He Z, Lin S, et al. An inducible CRISPR/Cas9 screen identifies DTX2 as a transcriptional regulator of human telomerase. iScience. 2022;25:103813.
Wu S, Shen S, Lu F, Zheng P, Lin K, Liao J, et al. Bromodomain containing 4 transcriptionally activated Deltex E3 ubiquitin ligase 2 contributes to glioma progression and predicts an unfavorable prognosis. Ann Transl Med. 2022;10:313.
Chen H, Huang Y, Yeh T, Chang C, Shen Y, Chen Y, et al. Dtx2 deficiency induces ependymo-radial glial cell proliferation and improves spinal cord motor function recovery. Stem Cells Dev. 2024;33:540–50.
Djerir B, Marois I, Dubois JC, Findlay S, Morin T, Senoussi I, et al. An E3 ubiquitin ligase localization screen uncovers DTX2 as a novel ADP-ribosylation-dependent regulator of DNA double-strand break repair. J Biol Chem. 2024;300:107545.
Ji N, Li H, Zhang Y, Li Y, Wang P, Chen X, et al. Lansoprazole (LPZ) reverses multidrug resistance (MDR) in cancer through impeding ATP-binding cassette (ABC) transporter-mediated chemotherapeutic drug efflux and lysosomal sequestration. Drug Resist Updat. 2024;76:101100.
Wong H, Cvijanovich N, Lin R, Allen G, Thomas NJ, Willson DF, et al. Identification of pediatric septic shock subclasses based on genome-wide expression profiling. BMC Med. 2009;7:34.
Wong H, Cvijanovich N, Allen L, Lin R, Anas N, Meyer K, et al. Genomic expression profiling across the pediatric systemic inflammatory response syndrome, sepsis, and septic shock spectrum. Crit Care Med. 2009;37:1558–66.
Ajoolabady A, Aslkhodapasandhokmabad H, Libby P, Tuomilehto J, Lip GYH, Penninger JM, et al. Ferritinophagy and ferroptosis in the management of metabolic diseases. Trends Endocrinol Metab. 2021;32:444–62.
Rochette L, Dogon G, Rigal E, Zeller M, Cottin Y, Vergely C. Lipid peroxidation and iron metabolism: two corner stones in the homeostasis control of ferroptosis. Int J Mol Sci. 2022;24:449.
Sheng SY, Li JM, Hu XY, Wang Y. Regulated cell death pathways in cardiomyopathy. Acta Pharm Sin. 2023;44:1521–35.
Cho HM, Cho JY. Cardiomyocyte death and genome-edited stem cell therapy for ischemic heart disease. Stem Cell Rev Rep. 2021;17:1264–79.
Chiong M, Wang ZV, Pedrozo Z, Cao DJ, Troncoso R, Ibacache M, et al. Cardiomyocyte death: mechanisms and translational implications. Cell Death Dis. 2011;2:e244.
Kerr JF, Wyllie AH, Currie AR. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer. 1972;26:239–57.
Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22:266–82.
Wang C, Yuan W, Hu A, Lin J, Xia Z, Yang CF, et al. Dexmedetomidine alleviated sepsis‑induced myocardial ferroptosis and septic heart injury. Mol Med Rep. 2020;22:175–84.
Wu X, Li Y, Zhang S, Zhou X. Ferroptosis as a novel therapeutic target for cardiovascular disease. Theranostics. 2021;11:3052–9.
Lu Y, Yang Q, Su Y, Ji Y, Li G, Yang X, et al. MYCN mediates TFRC-dependent ferroptosis and reveals vulnerabilities in neuroblastoma. Cell Death Dis. 2021;12:511.
Wang N, Ma H, Li J, Meng C, Zou J, Wang H, et al. HSF1 functions as a key defender against palmitic acid-induced ferroptosis in cardiomyocytes. J Mol Cell Cardiol. 2021;150:65–76.
Tang D, Chen X, Kang R, Kroemer G. Ferroptosis: molecular mechanisms and health implications. Cell Res. 2021;31:107–25.
Lange SM, Armstrong LA, Kulathu Y. Deubiquitinases: from mechanisms to their inhibition by small molecules. Mol Cell. 2022;82:15–29.
Acknowledgements
We would like to thank Xi Wang (Tiantan Hospital of Capital Medical University, Beijing, China) for providing ubiquitin and its mutants plasmids.
Funding
This work was supported by the National Natural Science Foundation of China (No. 82272221, 82202403, 82172170, 82372195); the Tianjin Natural Science Foundation (No. 24JCQNJC00600, 24JCYBJC01010); the National College Students’ Innovative Entrepreneurial Training Plan Program (No. 202510062034).
Author information
Authors and Affiliations
Contributions
CL conceived and performed the study idea, analyzed the data, and wrote the manuscript. JL performed the experiments and analyzed the data. MY, RL, CF, YC, and LG performed the research. ZQ and YS conceived and supervised the project, and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
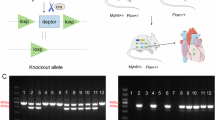
All methods were performed in accordance with the relevant guidelines and regulations. All animal experiments were approved by the Experimental Animal Ethics Committee of Tianjin Medical University. No human subjects involved in this study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, C., Liu, J., Yin, M. et al. Deltex E3 ubiquitin ligase 2 prevents sepsis-induced myocardial injury through degrading TfR1 via promoting K27-linked ubiquitination. Cell Death Differ (2026). https://doi.org/10.1038/s41418-026-01690-0
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41418-026-01690-0