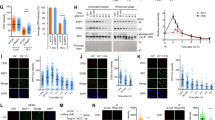
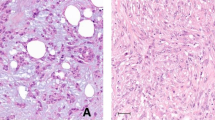
Abstract
BTAF1, an ATP-dependent remodeler of the TBP-DNA complex, is frequently mutated in gastric cancer. However, its role in DNA repair and therapeutic relevance remains largely undefined. Here, we show that BTAF1 knockout leads to accumulation of double-strand breaks (DSBs) by impairing DNA end-resection process of homologous recombination (HR) repair, thereby sensitizing cells to genotoxic agents both in vitro and in vivo. Mechanistically, BTAF1 prevents ubiquitin-mediated degradation of MRE11, maintaining its protein stability, promoting DNA end resection and HR, and consequently enhancing cellular resistance to DNA-damaging stress. Notably, the interaction between BTAF1 and MRE11 is dynamically regulated by PARP1-mediated PARylation of BTAF1 during the DNA damage response. Loss of BTAF1 also increases chemosensitivity in gastric cancer xenograft and organoid models. Clinically, high BTAF1 expression correlates with poor prognosis in gastric cancer patients receiving neoadjuvant chemotherapy. Collectively, our findings identify BTAF1 as a critical regulator of HR repair through stabilization of MRE11 and propose BTAF1 as a potential biomarker for predicting response to genotoxic chemotherapy.

BTAF1 is frequently mutated in gastric cancer, yet its role in DNA repair remains unclear. BTAF1 promotes homologous recombination and chemoresistance in gastric cancer by stabilizing MRE11. BTAF1 loss impairs DNA end resection, sensitizing cells to genotoxic agents. The BTAF1-MRE11 interaction is regulated by PARP1 mediated PARylation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request. Uncropped original western blot images are provided in the Supplementary Information.
References
Brown JS, O’Carrigan B, Jackson SP, Yap TA. Targeting DNA repair in cancer beyond PARP inhibitors. Cancer Discov. 2017;7:20–37.
Negrini S, Gorgoulis VG, Halazonetis TD. Genomic instability—an evolving hallmark of cancer. Nat Rev Mol Cell Biol. 2010;11:220–8.
Jackson SP, Bartek J. The DNA-damage response in human biology and disease. Nature. 2009;461:1071–8.
Chang HH, Pannunzio NR, Adachi N, Lieber MR. Non-homologous DNA end joining and alternative pathways to double-strand break repair. Nat Rev Mol Cell Biol. 2017;18:495–506.
Chapman JR, Taylor MR, Boulton SJ. Playing the end game: DNA double-strand break repair pathway choice. Mol Cell. 2012;47:497–510.
Lord CJ, Ashworth A. PARP inhibitors: synthetic lethality in the clinic. Science. 2017;355:1152–8.
Groelly FJ, Fawkes M, Dagg RA, Blackford AN, Tarsounas M. Targeting DNA damage response pathways in cancer. Nat Rev Cancer. 2023;23:78–94.
Bouwman P, Jonkers J. The effects of deregulated DNA damage signalling on cancer chemotherapy response and resistance. Nat Rev Cancer. 2012;12:587–98.
Gogola E, Rottenberg S, Jonkers J. Resistance to PARP inhibitors: lessons from preclinical models of BRCA-associated cancer. Annu Rev Cancer Biol. 2019;3:235–54.
Raderschall E, Stout K, Freier S, Suckow V, Schweiger S, Haaf T. Elevated levels of Rad51 recombination protein in tumor cells. Cancer Res. 2002;62:219–25.
Ma CX, Janetka JW, Piwnica-Worms H. Death by releasing the breaks: CHK1 inhibitors as cancer therapeutics. Trends Mol Med. 2011;17:88–96.
Galluzzi L, Senovilla L, Vitale I, Michels J, Martins I, Kepp O, et al. Molecular mechanisms of cisplatin resistance. Oncogene. 2012;31:1869–83.
Nitiss JL. Targeting DNA topoisomerase II in cancer chemotherapy. Nat Rev Cancer. 2009;9:338–50.
Pilié PG, Tang C, Mills GB, Yap TA. State-of-the-art strategies for targeting the DNA damage response in cancer. Nat Rev Clin Oncol. 2019;16:81–104.
Bian L, Meng Y, Zhang M, Li D. MRE11-RAD50-NBS1 complex alterations and DNA damage response: implications for cancer treatment. Mol Cancer. 2019;18:169.
Huertas P, Jackson SP. Human CtIP mediates cell cycle control of DNA end resection and double strand break repair. J Biol Chem. 2009;284:9558–65.
Anand R, Ranjha L, Cannavo E, Cejka P. Phosphorylated CtIP functions as a co-factor of the MRE11-RAD50-NBS1 endonuclease in DNA end resection. Mol Cell. 2016;64:940–50.
Stracker TH, Petrini JH. The MRE11 complex: starting from the ends. Nat Rev Mol Cell Biol. 2011;12:90–103.
Garcia V, Phelps SE, Gray S, Neale MJ. Bidirectional resection of DNA double-strand breaks by Mre11 and Exo1. Nature. 2011;479:241–4.
Zhang T, Yang H, Zhou Z, Bai Y, Wang J, Wang W. Crosstalk between SUMOylation and ubiquitylation controls DNA end resection by maintaining MRE11 homeostasis on chromatin. Nat Commun. 2022;13:5133.
Cejka P, Symington LS. DNA end resection: mechanism and control. Annu Rev Genet. 2021;55:285–307.
Jachimowicz RD, Beleggia F, Isensee J, Velpula BB, Goergens J, Bustos MA, et al. UBQLN4 represses homologous recombination and is overexpressed in aggressive tumors. Cell. 2019;176:505–19.e22.
Belmonte-Fernández A, Herrero-Ruíz J, Galindo-Moreno M, Limón-Mortés MC, Mora-Santos M, Sáez C, et al. Cisplatin-induced cell death increases the degradation of the MRE11-RAD50-NBS1 complex through the autophagy/lysosomal pathway. Cell Death Differ. 2023;30:488–99.
Nicholson J, Jevons SJ, Groselj B, Ellermann S, Konietzny R, Kerr M, et al. E3 ligase cIAP2 mediates downregulation of MRE11 and radiosensitization in response to HDAC inhibition in bladder cancer. Cancer Res. 2017;77:3027–39.
Sanchez-Bailon M, Choi S, Dufficy E, Sharma K, McNee G, Gunnell E, et al. Arginine methylation and ubiquitylation crosstalk controls DNA end-resection and homologous recombination repair. Nat Commun. 2021;12:6313.
Yu Z, Vogel G, Coulombe Y, Dubeau D, Spehalski E, Hébert J, et al. The MRE11 GAR motif regulates DNA double-strand break processing and ATR activation. Cell Res. 2012;22:305–20.
Chen Y, Wu J, Zhai L, Zhang T, Yin H, Gao H, et al. Metabolic regulation of homologous recombination repair by MRE11 lactylation. Cell. 2024;187:294–311.e21.
Zeng X, Zhao F, Cui G, Zhang Y, Deshpande RA, Chen Y, et al. METTL16 antagonizes MRE11-mediated DNA end resection and confers synthetic lethality to PARP inhibition in pancreatic ductal adenocarcinoma. Nat Cancer. 2022;3:1088–104.
Bai Y, Wang W, Li S, Zhan J, Li H, Zhao M, et al. C1QBP promotes homologous recombination by stabilizing MRE11 and controlling the assembly and activation of the MRE11/RAD50/NBS1 complex. Mol Cell. 2019;75:1299–1314.e6.
Liu W, Zheng M, Zhang R, Jiang Q, Du G, Wu Y, et al. RNF126-mediated MRE11 ubiquitination activates the DNA damage response and confers resistance of triple-negative breast cancer to radiotherapy. Adv Sci. 2023;10:2203884.
Chicca JJ, Auble DT, Pugh BF. Cloning and biochemical characterization of TAF-172, a human homolog of yeast Mot1. Mol Cell Biol. 1998;18:1701–10.
Pereira LA, Klejman MP, Timmers HTM. Roles for BTAF1 and Mot1p in dynamics of TATA-binding protein and regulation of RNA polymerase II transcription. Gene. 2003;315:1–13.
Pereira LA, Klejman MP, Ruhlmann C, Kavelaars F, Oulad-Abdelghani M, Timmers HTM, et al. Molecular architecture of the basal transcription factor B-TFIID. J Biol Chem. 2004;279:21802–7.
Klejman MP, Zhao X, van Schaik FM, Herr W, Timmers HTM. Mutational analysis of BTAF1-TBP interaction: BTAF1 can rescue DNA-binding defective TBP mutants. Nucleic Acids Res. 2005;33:5426–36.
Butryn A, Schuller JM, Stoehr G, Runge-Wollmann P, Förster F, Auble DT, et al. Structural basis for recognition and remodeling of the TBP:DNA:NC2 complex by Mot1. eLife. 2015;4:e07432.
Cai MY, Hou JH, Rao HL, Luo RZ, Li M, Pei XQ, et al. High expression of H3K27me3 in human hepatocellular carcinomas correlates closely with vascular invasion and predicts worse prognosis in patients. Mol Med. 2011;17:12–20.
Olivieri M, Cho T, Álvarez-Quilón A, Li K, Schellenberg MJ, Zimmermann M, et al. A genetic map of the response to DNA damage in human cells. Cell. 2020;182:481–96.e21.
van der Knaap JA, van den Boom V, Kuipers J, van Eijk MJ, van der Vliet PC, Timmers HTM. The gene for human TATA-binding-protein-associated factor (TAFII) 170: structure, promoter and chromosomal localization. Biochem J. 2000;345:521–7.
van der Knaap JA, Borst JW, van der Vliet PC, Gentz R, Timmers HTM. Cloning of the cDNA for the TATA-binding protein-associated factorII170 subunit of transcription factor B-TFIID reveals homology to global transcription regulators in yeast and Drosophila. Proc Natl Acad Sci USA. 1997;94:11827–32.
Pereira LA, van der Knaap JA, van den Boom V, van den Heuvel FA, Timmers HTM. TAFII170 interacts with the concave surface of TATA-binding protein to inhibit its DNA binding activity. Mol Cell Biol. 2001;21:7523–34.
Li X, Heyer WD. Homologous recombination in DNA repair and DNA damage tolerance. Cell Res. 2008;18:99–113.
Ray Chaudhuri A, Nussenzweig A. The multifaceted roles of PARP1 in DNA repair and chromatin remodelling. Nat Rev Mol Cell Biol. 2017;18:610–21.
Slade D. PARP and PARG inhibitors in cancer treatment. Genes Dev. 2020;34:360–94.
Lo Monte M, Manelfi C, Gemei M, Corda D, Beccari AR. ADPredict: ADP-ribosylation site prediction based on physicochemical and structural descriptors. Bioinformatics. 2018;34:2566–74.
Lord CJ, Ashworth A. The DNA damage response and cancer therapy. Nature. 2012;481:287–94.
Acknowledgements
This work was supported by the grants from the National Key R&D Program of China (No. 2021YFA1300201), the National Natural Science Foundation of China (grant No. 81972227, 82172646, 82202905 and 82373190), and Guangdong basic and applied basic research foundation (No. 2024A1515012828). The patient-derived GCOs was a kind gift from Prof. Xiaoxing Li (Institute of Precision Medicine, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, China).
Author information
Authors and Affiliations
Contributions
Conceptualization, M-YC, DX, and J-JX; Methodology, J-JX, J-LD, J-LL and Z-CX; Software, J-JX, C-HQ, and X-XC; Investigation, R-CN, PL; Formal Analysis, Y-XY; Writing – Original Draft, J-JX, J-LD, Z-CX, and J-LL; Writing – Review & Editing, DX and M-YC; Funding Acquisition, DX, M-YC, J-LD and PL; Resources, DX and M-YC; Supervision, DX, M-YC and J-LD.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Institutional Review Board approval (Sun Yat-sen University Cancer Center) the protocol (B2023-335-01) and informed consent were obtained prior to tissue acquisition and analysis. Animal ethics are listed in the Materials and Methods section.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xie, JJ., Lin, JL., Xiang, ZC. et al. BTAF1: a key regulator of DNA end resection and predictor of chemotherapy sensitivity in gastric cancer. Cell Death Differ (2026). https://doi.org/10.1038/s41418-026-01709-6
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41418-026-01709-6