Abstract
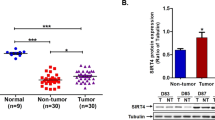
Hepatitis B virus (HBV) infection remains a leading etiological driver of hepatocellular carcinoma (HCC). Cuproptosis is a recently defined copper-dependent form of regulated cell death that selectively eliminates mitochondria-dependent cells; whether HBV rewires this vulnerability remains unknown. Here we unveil a novel HBV X protein (HBx)-driven mechanism of cuproptosis evasion. Integrative analysis of clinical specimens, HBx-transgenic (HBx-Tg) mice, and multi-omics datasets revealed marked downregulation of STEAP4 (six-transmembrane epithelial antigen of prostate 4), a metalloreductase essential for cuproptosis sensitivity, in HBV-positive HCC. Mechanistically, HBx attenuates sirtuin 3 (SIRT3), impairing deacetylation of STEAP4 at lysine 404 and abolishing its mitochondrial targeting. Consequently, cells switch from the tricarboxylic acid (TCA) cycle respiration to glycolysis, reducing sensitivity to the copper ionophore elesclomol (ES). Restoring STEAP4 expression or pharmacological activation of SIRT3 with honokiol (HKL) re-instated mitochondrial STEAP4 localization and re-sensitized HBV-related HCC cells to cuproptosis; combination with ES produced synergistic tumor suppression in vitro and in orthotopic models. Collectively, our findings establish the SIRT3–STEAP4 axis as a novel regulator of cuproptosis resistance in HBV-related HCC. HBx-mediated repression of SIRT3 disrupts STEAP4 deacetylation and mitochondrial targeting, fostering metabolic reprogramming and evasion of copper-induced cell death. The results provide a pre-clinical rationale for copper-directed combination strategies in HBV-associated HCC.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
All data supporting the conclusions of this study are contained within the article and its supplementary files. Raw data and unique biological materials are available from the corresponding authors upon reasonable request.
References
Jia L, Gao Y, He Y, Hooper JD, Yang P. HBV induced hepatocellular carcinoma and related potential immunotherapy. Pharmacol Res. 2020;159:104992.
Hsu YC, Huang DQ, Nguyen MH. Global burden of hepatitis B virus: current status, missed opportunities and a call for action. Nat Rev Gastroenterol Hepatol. 2023;20:524–37.
Tu T, McQuaid TJ, Jacobson IM. HBV-induced carcinogenesis: mechanisms, correlation with viral suppression, and implications for treatment. Liver Int. 2025;45:e16202.
Yang YM, Kim SY, Seki E. Inflammation and liver cancer: molecular mechanisms and therapeutic targets. Semin Liver Dis. 2019;39:26–42.
Sivasudhan E, Blake N, Lu Z, Meng J, Rong R. Hepatitis B viral protein HBx and the molecular mechanisms modulating the hallmarks of hepatocellular carcinoma: a comprehensive review. Cells. 2022;11:741.
Ma M, Yi L, Pei Y, Zhang Q, Tong C, Zhao M, et al. USP26 as a hepatitis B virus-induced deubiquitinase primes hepatocellular carcinogenesis by epigenetic remodeling. Nat Commun. 2024;15:7856.
Wang Y, Zhao M, Zhao L, Geng Y, Li G, Chen L, et al. HBx-induced HSPA8 stimulates HBV replication and suppresses ferroptosis to support liver cancer progression. Cancer Res. 2023;83:1048–61.
Wang Y, Guan X, Lv F, Rong Y, Meng X, Tong Y, et al. HBx integration in diffuse large B-cell lymphoma inhibits Caspase-3-PARP related apoptosis. Tumour Virus Res. 2024;18:200290.
Huang FY, Wong DK, Seto WK, Mak LY, Cheung TT, Yuen MF. Tumor suppressive role of mitochondrial sirtuin 4 in induction of G2/M cell cycle arrest and apoptosis in hepatitis B virus-related hepatocellular carcinoma. Cell Death Discov. 2021;7:88.
He C, Qiu Y, Han P, Chen Y, Zhang L, Yuan Q, et al. ER stress regulating protein phosphatase 2A-B56gamma, targeted by hepatitis B virus X protein, induces cell cycle arrest and apoptosis of hepatocytes. Cell Death Dis. 2018;9:762.
Dong J, Zhang R, Xia Y, Jiang X, Zhou K, Li J, et al. The necroptosis related gene LGALS3 can be used as a biomarker for the adverse progression from chronic HBV infection to HCC. Front Immunol. 2023;14:1142319.
Xiao C, Gong J, Jie Y, Liang W, Tai Y, Qin W, et al. E2F1-mediated up-regulation of NCAPG promotes hepatocellular carcinoma development by inhibiting pyroptosis. J Clin Transl Hepatol. 2024;12:25–35.
Zhao Q, Chen DP, Chen HD, Wang YZ, Shi W, Lu YT, et al. NK-cell-elicited gasdermin-D-dependent hepatocyte pyroptosis induces neutrophil extracellular traps that facilitate HBV-related acute-on-chronic liver failure. Hepatology. 2025;81:917–31.
Xie WH, Ding J, Xie XX, Yang XH, Wu XF, Chen ZX, et al. Hepatitis B virus X protein promotes liver cell pyroptosis under oxidative stress through NLRP3 inflammasome activation. Inflamm Res. 2020;69:683–96.
Tsvetkov P, Coy S, Petrova B, Dreishpoon M, Verma A, Abdusamad M, et al. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science. 2022;375:1254–61.
Kim BE, Nevitt T, Thiele DJ. Mechanisms for copper acquisition, distribution and regulation. Nat Chem Biol. 2008;4:176–85.
Yang Y, Wu J, Wang L, Ji G, Dang Y. Copper homeostasis and cuproptosis in health and disease. MedComm. 2024;5:e724.
Tang D, Kroemer G, Kang R. Targeting cuproplasia and cuproptosis in cancer. Nat Rev Clin Oncol. 2024;21:370–88.
Ge EJ, Bush AI, Casini A, Cobine PA, Cross JR, DeNicola GM, et al. Connecting copper and cancer: from transition metal signalling to metalloplasia. Nat Rev Cancer. 2022;22:102–13.
Rashed MN. The role of trace elements on hepatitis virus infections: a review. J Trace Elem Med Biol. 2011;25:181–7.
Zheng P, Zhou C, Lu L, Liu B, Ding Y. Elesclomol: a copper ionophore targeting mitochondrial metabolism for cancer therapy. J Exp Clin Cancer Res. 2022;41:271.
Kirubakaran S, Sureshkumar D, Chandrasekaran S. Tetrathiomolybdate and tetraselenotungstate as sulfur/selenium transfer reagents: applications in the synthesis of new thio/seleno sugars. Chem Rec. 2021;21:3076–86.
Ohgami RS, Campagna DR, McDonald A, Fleming MD. The Steap proteins are metalloreductases. Blood. 2006;108:1388–94.
Scarl RT, Lawrence CM, Gordon HM, Nunemaker CS. STEAP4: its emerging role in metabolism and homeostasis of cellular iron and copper. J Endocrinol. 2017;234:R123–R34.
Ju MH, Jang EJ, Kang SH, Roh YH, Jeong JS, Han SH. Six-transmembrane epithelial antigen of prostate 4: an indicator of prognosis and tumor immunity in hepatocellular carcinoma. J Hepatocell Carcinoma. 2023;10:643–58.
Liao Y, Zhao J, Bulek K, Tang F, Chen X, Cai G, et al. Inflammation mobilizes copper metabolism to promote colon tumorigenesis via an IL-17-STEAP4-XIAP axis. Nat Commun. 2020;11:900.
Kim HY, Cho HK, Yoo SK, Cheong JH. Hepatic STAMP2 decreases hepatitis B virus X protein-associated metabolic deregulation. Exp Mol Med. 2012;44:622–32.
Che L, Du ZB, Wang WH, Wu JS, Han T, Chen YY, et al. Intracellular antibody targeting HBx suppresses invasion and metastasis in hepatitis B virus-related hepatocarcinogenesis via protein phosphatase 2A-B56gamma-mediated dephosphorylation of protein kinase B. Cell Prolif. 2022;55:e13304.
Du ZB, Wu XM, Han T, Cai YX, Qian B, Shen YS, et al. MALAT1 promotes malignancy of HBV-related hepatocellular carcinoma by regulating IGF2BP3-mediated nuclear-cytoplasmic shuttling. Int J Biol Sci. 2025;21:4942–60.
Lan Y, Qian B, Huang HY, Wang P, Li T, Yuan Q, et al. Hepatocyte-derived prostaglandin E2-modulated macrophage M1-type polarization via mTOR-NPC1 axis-regulated cholesterol transport from lysosomes to the endoplasmic reticulum in Hepatitis B Virus x protein-related nonalcoholic steatohepatitis. Int J Mol Sci. 2022;23:11660.
Liu YT, Chen L, Li SJ, Wang WY, Wang YY, Yang QC, et al. Dysregulated Wnt/beta-catenin signaling confers resistance to cuproptosis in cancer cells. Cell Death Differ. 2024;31:1452–66.
Zhang JF, Xiong HL, Cao JL, Wang SJ, Guo XR, Lin BY, et al. A cell-penetrating whole molecule antibody targeting intracellular HBx suppresses hepatitis B virus via TRIM21-dependent pathway. Theranostics. 2018;8:549–62.
Hou Z, Quan J. Hepatitis B virus X protein increases microRNA‑21 expression and accelerates the development of hepatoma via the phosphatase and tensin homolog/phosphoinositide 3‑kinase/protein kinase B signaling pathway. Mol Med Rep. 2017;15:3285–91.
Tang F, Zhang JN, Zhao XL, Xu LY, Ao H, Peng C. Unlocking the dual role of autophagy: a new strategy for treating lung cancer. J Pharm Anal. 2025;15:101098.
Kalyanaraman B. Exploiting the tumor immune microenvironment and immunometabolism using mitochondria-targeted drugs: challenges and opportunities in racial disparity and cancer outcome research. FASEB J. 2022;36:e22226.
Zoulim F, Chen PJ, Dandri M, Kennedy PT, Seeger C. Hepatitis B virus DNA integration: implications for diagnostics, therapy, and outcome. J Hepatol. 2024;81:1087–99.
Liu W, Yao Q, Su X, Deng Y, Yang M, Peng B, et al. Molecular insights into Spindlin1-HBx interplay and its impact on HBV transcription from cccDNA minichromosome. Nat Commun. 2023;14:4663.
Wang Y, Chen Y, Zhang J, Yang Y, Fleishman JS, Wang Y, et al. Cuproptosis: a novel therapeutic target for overcoming cancer drug resistance. Drug Resist Updat. 2024;72:101018.
Xie J, Yang Y, Gao Y, He J. Cuproptosis: mechanisms and links with cancers. Mol Cancer. 2023;22:46.
Fang AP, Chen PY, Wang XY, Liu ZY, Zhang DM, Luo Y, et al. Serum copper and zinc levels at diagnosis and hepatocellular carcinoma survival in the Guangdong Liver Cancer Cohort. Int J Cancer. 2019;144:2823–32.
Liao Q, Deng J, Tong J, Gan Y, Hong W, Dong H, et al. p53 induces circFRMD4A to suppress cancer development through glycolytic reprogramming and cuproptosis. Mol Cell. 2025;85:132–49.e7.
Sun Z, Xu H, Lu G, Yang C, Gao X, Zhang J, et al. AKT1 phosphorylates FDX1 to promote cuproptosis resistance in triple-negative breast cancer. Adv Sci. 2025;12:e2408106.
Schollmeier A, Glitscher M, Hildt E. Relevance of HBx for Hepatitis B virus-associated pathogenesis. Int J Mol Sci. 2023;24:4964.
Yan Y, Wei Z, Zheng M, Lu M, Wang X. HBV and host metabolic crosstalk: Reprogramming pathways for viral replication and pathogenesis. Virol Sin. 2025;40:685–93.
Chen YY, Wang WH, Che L, Lan Y, Zhang LY, Zhan DL, et al. BNIP3L-dependent mitophagy promotes HBx-induced cancer stemness of hepatocellular carcinoma cells via glycolysis metabolism reprogramming. Cancers. 2020;12:655.
Lu K, Wijaya CS, Yao Q, Jin H, Feng L. Cuproplasia and cuproptosis, two sides of the coin. Cancer Commun. 2025;45:505–24.
Wang T, Cao Y, Zheng Q, Tu J, Zhou W, He J, et al. SENP1-Sirt3 signaling controls mitochondrial protein acetylation and metabolism. Mol Cell. 2019;75:823–34.e5.
Zhang J, Xiang H, Liu J, Chen Y, He RR, Liu B. Mitochondrial sirtuin 3: new emerging biological function and therapeutic target. Theranostics. 2020;10:8315–42.
Overmyer KA, Evans CR, Qi NR, Minogue CE, Carson JJ, Chermside-Scabbo CJ, et al. Maximal oxidative capacity during exercise is associated with skeletal muscle fuel selection and dynamic changes in mitochondrial protein acetylation. Cell Metab. 2015;21:468–78.
Tarin M, Babaie M, Eshghi H, Matin MM, Saljooghi AS. Elesclomol, a copper-transporting therapeutic agent targeting mitochondria: from discovery to its novel applications. J Transl Med. 2023;21:745.
Jin J, Bai L, Wang D, Ding W, Cao Z, Yan P, et al. SIRT3-dependent delactylation of cyclin E2 prevents hepatocellular carcinoma growth. EMBO Rep. 2023;24:e56052.
Acknowledgements
We thanks all colleagues who provided reagents, technical support or intellectual input.
Funding
This work was supported by the National Natural Science Foundation of China (Nos. 82273667, 82272935, 82574139, 82573484), the Fujian Province Special Fund Project for Promoting High-Quality Development of Marine and Fishery Industries (FJHYF-L-2025-27), the Industry-University-Research Cooperation Project of Fujian Science and Technology Plan (No. 2022Y4009), the Fundamental Research Funds for the Central Universities (No. 20720250004), Natural Science Foundation of Xiamen, China (No. 3502Z202573032), and Scientific Research Foundation of State Key Laboratory of Vaccines for Infectious Diseases, Xiang An Biomedicine Laboratory (2023XAKJ0101024).
Author information
Authors and Affiliations
Contributions
ZD, XW, ZL, and BQ conceived and designed the study and wrote the manuscript with comments from all authors; JL, YC, JH, XH, WH, and YL performed experiments. XX, HZ, DG, and YY acquired and analyzed the data; WL, Y-CL, and ZL provided reagents and technical assistance. ZD, and ZL wrote, reviewed, and edited the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
We confirm that all methods were performed in accordance with the relevant guidelines and regulations. Animal procedures were approved by the Animal Ethics Committee of Xiamen University (XMULAC20220282; 12 March 2022) and conducted in accordance with institutional guidelines.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Du, ZB., Wu, XM., Lei, JM. et al. SIRT3 deacetylates STEAP4 to modulate cuproptosis sensitivity via mitochondrial metabolic reprogramming in HBV-related HCC. Cell Death Differ (2026). https://doi.org/10.1038/s41418-026-01713-w
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41418-026-01713-w