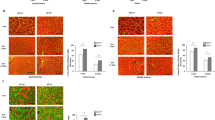
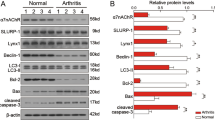
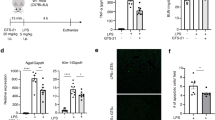
Abstract
Denervation induces severe muscle atrophy characterized by inflammatory responses and tissue degradation, with limited effective therapeutic options. This study investigates the role of the α7 nicotinic acetylcholine receptor (α7nAChR) in denervation-induced muscle atrophy and evaluates electroacupuncture (EA) as a potential treatment strategy. Using a sciatic nerve transection mouse model, we observe that denervation decreases α7nAChR expression, activates proteolytic pathways. We find that α7nAChR degradation is associated with the activation of inflammatory cytokines and the caspase pathway. In α7nAChR knockout mice, we demonstrate that α7nAChR modulates mitochondrial metabolism and fiber-type composition. It exerts protective effects by activating the AKT-FOXO1 pathway, thereby reducing inflammation and apoptosis, processes that are critical for muscle regeneration. Additionally, treatment with PNU120596 or EA restores α7nAChR function and alleviates muscle atrophy. Our findings suggest that targeting α7nAChR offers a promising therapeutic approach for muscle wasting following denervation, with potential implications for clinical management and future intervention strategies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The RNA-seq data supporting this study have been deposited in the Gene Expression Omnibus (GEO) under accession numbers GSE315922, GSE253469 and GSE304280. The other data generated or analyzed during this study are available upon reasonable request from the corresponding author.
References
Lopes B, Sousa P, Alvites R, Branquinho M, Sousa AC, Mendonca C, et al. Peripheral nerve injury treatments and advances: one health perspective. Int J Mol Sci. 2022;23:918.
Laumonier T, Menetrey J. Muscle injuries and strategies for improving their repair. J Exp Orthop. 2016;3:15.
Khaled MM, Ibrahium AM, Abdelgalil AI, El-Saied MA, El-Bably SH. Regenerative strategies in treatment of peripheral nerve injuries in different animal models. Tissue Eng Regen Med. 2023;20:839–77.
Grinsell D, Keating CP. Peripheral nerve reconstruction after injury: a review of clinical and experimental therapies. Biomed Res Int. 2014;2014:698256.
Bakooshli MA, Wang YX, Monti E, Su S, Kraft P, Nalbandian M, et al. Regeneration of neuromuscular synapses after acute and chronic denervation by inhibiting the gerozyme 15-prostaglandin dehydrogenase. Sci Transl Med. 2023;15:eadg1485.
Castets P, Rion N, Theodore M, Falcetta D, Lin S, Reischl M, et al. mTORC1 and PKB/Akt control the muscle response to denervation by regulating autophagy and HDAC4. Nat Commun. 2019;10:3187.
Sartori R, Hagg A, Zampieri S, Armani A, Winbanks CE, Viana LR, et al. Perturbed BMP signaling and denervation promote muscle wasting in cancer cachexia. Sci Transl Med. 2021;13:eaay9592.
Chen X, Ji Y, Liu R, Zhu X, Wang K, Yang X, et al. Mitochondrial dysfunction: roles in skeletal muscle atrophy. J Transl Med. 2023;21:503.
Kedlian VR, Wang Y, Liu T, Chen X, Bolt L, Tudor C, et al. Human skeletal muscle aging atlas. Nat Aging. 2024;4:727–44.
Romanello V, Sandri M. The connection between the dynamic remodeling of the mitochondrial network and the regulation of muscle mass. Cell Mol Life Sci. 2021;78:1305–28.
Abdon B, Liang Y, da Luz Scheffer D, Torres M, Shrestha N, Reinert RB, et al. Muscle-specific ER-associated degradation maintains postnatal muscle hypertrophy and systemic energy metabolism. JCI Insight. 2023;8:e170387.
Milan G, Romanello V, Pescatore F, Armani A, Paik JH, Frasson L, et al. Regulation of autophagy and the ubiquitin-proteasome system by the FoxO transcriptional network during muscle atrophy. Nat Commun. 2015;6:6670.
Le Guerroue F, Youle RJ. Ubiquitin signaling in neurodegenerative diseases: an autophagy and proteasome perspective. Cell Death Differ. 2021;28:439–54.
Lin K, Wei L, Wang R, Li L, Song S, Wang F, et al. Disrupted methionine cycle triggers muscle atrophy in cancer cachexia through epigenetic regulation of REDD1. Cell Metab. 2024;37:460–76. e8.
Svandova E, Vesela B, Janeckova E, Chai Y, Matalova E. Exploring caspase functions in mouse models. Apoptosis. 2024;29:938–66.
Finkbeiner S. The autophagy lysosomal pathway and neurodegeneration. Cold Spring Harb Perspect Biol. 2020;12:a033993.
Sartori R, Romanello V, Sandri M. Mechanisms of muscle atrophy and hypertrophy: implications in health and disease. Nat Commun. 2021;12:330.
Sirago G, Pellegrino MA, Bottinelli R, Franchi MV, Narici MV. Loss of neuromuscular junction integrity and muscle atrophy in skeletal muscle disuse. Ageing Res Rev. 2023;83:101810.
Zhang Y, Liao Q, Wen X, Fan J, Yuan T, Tong X, et al. Hijacking of the nervous system in cancer: mechanism and therapeutic targets. Mol Cancer. 2025;24:44.
Ho TNT, Abraham N, Lewis RJ. Structure-function of neuronal nicotinic acetylcholine receptor inhibitors derived from natural toxins. Front Neurosci. 2020;14:609005.
Unwin N. Nicotinic acetylcholine receptor and the structural basis of neuromuscular transmission: insights from Torpedo postsynaptic membranes. Q Rev Biophys. 2013;46:283–322.
Tian ZL, Jiang SK, Zhang M, Wang M, Li JY, Zhao R, et al. alpha7nAChR is expressed in satellite cells at different myogenic status during skeletal muscle wound healing in rats. J Mol Histol. 2015;46:499–509.
Wang Y, Jiang Q, Xia YY, Huang ZH, Huang C. Involvement of alpha7nAChR in electroacupuncture relieving neuropathic pain in the spinal cord of rat with spared nerve injury. Brain Res Bull. 2018;137:257–64.
Sousa-Soares C, Noronha-Matos JB, Correia-de-Sa P. Purinergic tuning of the tripartite neuromuscular synapse. Mol Neurobiol. 2023;60:4084–104.
Hua Y, Yang B, Chen Q, Zhang J, Hu J, Fan Y. Activation of alpha7 nicotinic acetylcholine receptor protects against 1-Methyl-4-phenylpyridinium-induced astroglial apoptosis. Front Cell Neurosci. 2019;13:507.
Han X, Li W, Li P, Zheng Z, Lin B, Zhou B, et al. Stimulation of alpha7 nicotinic acetylcholine receptor by nicotine suppresses decidual M1 macrophage polarization against inflammation in lipopolysaccharide-induced preeclampsia-like mouse model. Front Immunol. 2021;12:642071.
Chen Y, Lian P, Peng Z, Wazir J, Ma C, Wei L, et al. Alpha-7 nicotinic acetylcholine receptor agonist alleviates psoriasis-like inflammation through inhibition of the STAT3 and NF-kappaB signaling pathway. Cell Death Discov. 2022;8:141.
Xie H, Yepuri N, Meng Q, Dhawan R, Leech CA, Chepurny OG, et al. Therapeutic potential of alpha7 nicotinic acetylcholine receptor agonists to combat obesity, diabetes, and inflammation. Rev Endocr Metab Disord. 2020;21:431–47.
Shi H, Li F, Zhang F, Wei X, Liu C, Duan R. An electrical stimulation intervention protocol to prevent disuse atrophy and muscle strength decline: an experimental study in rat. J Neuroeng Rehabil. 2023;20:84.
Inoue DS, Janini Gomes M. Integrative insights into PNI: Low-grade chronic inflammation, skeletal muscle wasting, and brain impairments. Brain Behav Immun Health. 2024;40:100838.
Pu W, Su Z, Wazir J, Zhao C, Wei L, Wang R, et al. Protective effect of alpha7 nicotinic acetylcholine receptor activation on experimental colitis and its mechanism. Mol Med. 2022;28:104.
Goodman CA, Hornberger TA. Measuring protein synthesis with SUnSET: a valid alternative to traditional techniques? Exerc Sport Sci Rev. 2013;41:107–15.
Cisterna BA, Cardozo C, Saez JC. Neuronal involvement in muscular atrophy. Front Cell Neurosci. 2014;8:405.
Wu P, Chawla A, Spinner RJ, Yu C, Yaszemski MJ, Windebank AJ, et al. Key changes in denervated muscles and their impact on regeneration and reinnervation. Neural Regen Res. 2014;9:1796–809.
Fernandez-Gonzalo R, Tesch PA, Lundberg TR, Alkner BA, Rullman E, Gustafsson T. Three months of bed rest induce a residual transcriptomic signature resilient to resistance exercise countermeasures. FASEB J. 2020;34:7958–69.
Lukasiewicz CJ, Tranah GJ, Evans DS, Coen PM, Barnes HN, Huo Z, et al. Higher expression of denervation-responsive genes is negatively associated with muscle volume and performance traits in the study of muscle, mobility, and aging (SOMMA). Aging Cell. 2024;23:e14115.
Calvo C, Swoboda CO, Montecino-Morales F, Nagar S, Petrany MJ, Sun C, et al. The multimodal transcriptional response of denervated skeletal muscle involves regulation of Gramd1 genes impacting muscle size. Proc Natl Acad Sci USA. 2025;122:e2424246122.
Fan YY, Zhang ST, Yu LS, Ye GH, Lin KZ, Wu SZ, et al. The time-dependent expression of alpha7nAChR during skeletal muscle wound healing in rats. Int J Leg Med. 2014;128:779–86.
Borkar NA, Roos B, Prakash YS, Sathish V, Pabelick CM. Nicotinic alpha7 acetylcholine receptor (alpha7nAChR) in human airway smooth muscle. Arch Biochem Biophys. 2021;706:108897.
Madaro L, Passafaro M, Sala D, Etxaniz U, Lugarini F, Proietti D, et al. Denervation-activated STAT3-IL-6 signalling in fibro-adipogenic progenitors promotes myofibres atrophy and fibrosis. Nat Cell Biol. 2018;20:917–27.
Haddad F, Zaldivar F, Cooper DM, Adams GR. IL-6-induced skeletal muscle atrophy. J Appl Physiol (1985). 2005;98:911–7.
Lepore E, Casola I, Dobrowolny G, Musaro A. Neuromuscular Junction as an Entity of Nerve-Muscle Communication. Cells. 2019;8:906.
Guo J, Yang G, He Y, Xu H, Fan H, An J, et al. Involvement of alpha7nAChR in the protective effects of genistein against beta-amyloid-induced oxidative stress in neurons via a PI3K/Akt/Nrf2 pathway-related mechanism. Cell Mol Neurobiol. 2021;41:377–93.
Wang Q, Zhang C, Jiang H, He W. Targeting CAMK2N1/CAMK2 inhibits invasion, migration and angiogenesis of non-small cell lung cancer by promoting autophagy and apoptosis via AKT/mTOR signaling pathway. Gene. 2024;913:148375.
Okuno S, Kitani T, Matsuzaki H, Konishi H, Kikkawa U, Fujisawa H. Studies on the phosphorylation of protein kinase B by Ca(2+)/calmodulin-dependent protein kinases. J Biochem. 2000;127:965–70.
Brocca L, Longa E, Cannavino J, Seynnes O, de Vito G, McPhee J, et al. Human skeletal muscle fibre contractile properties and proteomic profile: adaptations to 3 weeks of unilateral lower limb suspension and active recovery. J Physiol. 2015;593:5361–85.
Pang X, Zhang P, Chen X, Liu W. Ubiquitin-proteasome pathway in skeletal muscle atrophy. Front Physiol. 2023;14:1289537.
Bell RA, Al-Khalaf M, Megeney LA. The beneficial role of proteolysis in skeletal muscle growth and stress adaptation. Skelet Muscle. 2016;6:16.
Yadav A, Dabur R. Skeletal muscle atrophy after sciatic nerve damage: mechanistic insights. Eur J Pharm. 2024;970:176506.
Brock Symons T, Park J, Kim JH, Kwon EH, Delacruz J, Lee J, et al. Attenuation of skeletal muscle atrophy via acupuncture, electro-acupuncture, and electrical stimulation. Integr Med Res. 2023;12:100949.
Kostrominova TY. Skeletal muscle denervation: past, present and future. Int J Mol Sci. 2022;23:7489.
Zhang YT, Jin H, Wang JH, Wen LY, Yang Y, Ruan JW, et al. Tail nerve electrical stimulation and electro-acupuncture can protect spinal motor neurons and alleviate muscle atrophy after spinal cord transection in rats. Neural Plast. 2017;2017:7351238.
Chen F, Zhang Z, Zhang H, Guo P, Feng J, Shen H, et al. Activation of alpha7 nicotinic acetylcholine receptor improves muscle endurance by upregulating orosomucoid expression and glycogen content in mice. J Cell Biochem. 2024;125:e30630.
Zhu S, Nagashima M, Khan MA, Yasuhara S, Kaneki M, Martyn JA. Lack of caspase-3 attenuates immobilization-induced muscle atrophy and loss of tension generation along with mitigation of apoptosis and inflammation. Muscle Nerve. 2013;47:711–21.
Wadman RI, van der Pol WL, Bosboom WM, Asselman FL, van den Berg LH, Iannaccone ST, et al. Drug treatment for spinal muscular atrophy types II and III. Cochrane Database Syst Rev. 2020;1:CD006282.
Liu YP, Luo ZR, Wang C, Cai H, Zhao TT, Li H, et al. Electroacupuncture promoted nerve repair after peripheral nerve injury by regulating Mir-1B and its target brain-derived neurotrophic factor. Front Neurosci. 2020;14:525144.
Kubiak CA, Grochmal J, Kung TA, Cederna PS, Midha R, Kemp SWP. Stem-cell-based therapies to enhance peripheral nerve regeneration. Muscle Nerve. 2020;61:449–59.
Eggers R, de Winter F, Tannemaat MR, Malessy MJA, Verhaagen J. GDNF gene therapy to repair the injured peripheral nerve. Front Bioeng Biotechnol. 2020;8:583184.
Murphy RNA, de Schoulepnikoff C, Chen JHC, Columb MO, Bedford J, Wong JK, et al. The incidence and management of peripheral nerve injury in England (2005-2020). J Plast Reconstr Aesthet Surg. 2023;80:75–85.
Tuffaha SH, Budihardjo JD, Sarhane KA, Khusheim M, Song D, Broyles JM, et al. Growth hormone therapy accelerates axonal regeneration, promotes motor reinnervation, and reduces muscle atrophy following peripheral nerve injury. Plast Reconstr Surg. 2016;137:1771–80.
Wan Q, Zhang L, Huang Z, Zhang H, Gu J, Xu H, et al. Aspirin alleviates denervation-induced muscle atrophy via regulating the Sirt1/PGC-1alpha axis and STAT3 signaling. Ann Transl Med. 2020;8:1524.
Lee JI, Hur JM, You J, Lee DH. Functional recovery with histomorphometric analysis of nerves and muscles after combination treatment with erythropoietin and dexamethasone in acute peripheral nerve injury. PLoS One. 2020;15:e0238208.
Hargreaves M, Spriet LL. Skeletal muscle energy metabolism during exercise. Nat Metab. 2020;2:817–28.
Gergalova G, Lykhmus O, Kalashnyk O, Koval L, Chernyshov V, Kryukova E, et al. Mitochondria express alpha7 nicotinic acetylcholine receptors to regulate Ca2+ accumulation and cytochrome c release: study on isolated mitochondria. PLoS One. 2012;7:e31361.
Tian MY, Yang YD, Qin WT, Liu BN, Mou FF, Zhu J, et al. Electroacupuncture promotes nerve regeneration and functional recovery through regulating lncRNA GAS5 targeting miR-21 after sciatic nerve injury. Mol Neurobiol. 2024;61:935–49.
Wang Y, Xue M, Xia Y, Jiang Q, Huang Z, Huang C. Electroacupuncture treatment upregulates alpha7nAChR and inhibits JAK2/STAT3 in dorsal root ganglion of rat with spared nerve injury. J Pain Res. 2019;12:1947–55.
Penniman CM, Bhardwaj G, Nowers CJ, Brown CU, Junck TL, Boyer CK, et al. Loss of FoxOs in muscle increases strength and mitochondrial function during aging. J Cachexia Sarcopenia Muscle. 2023;14:243–59.
Fan BS, Zhang EH, Wu M, Guo JM, Su DF, Liu X, et al. Activation of alpha7 nicotinic acetylcholine receptor decreases on-site mortality in crush syndrome through insulin signaling-Na/K-ATPase pathway. Front Pharm. 2016;7:79.
Mahmassani ZS, Reidy PT, McKenzie AI, Stubben C, Howard MT, Drummond MJ. Age-dependent skeletal muscle transcriptome response to bed rest-induced atrophy. J Appl Physiol (1985). 2019;126:894–902.
Zhang XF, Xiang SY, Geng WY, Cong WJ, Lu J, Jiang CW, et al. Electro-acupuncture regulates the cholinergic anti-inflammatory pathway in a rat model of chronic obstructive pulmonary disease. J Integr Med. 2018;16:418–26.
McIlwain DR, Berger T, Mak TW. Caspase functions in cell death and disease. Cold Spring Harb Perspect Biol. 2013;5:a008656.
May S, Locke S, Kingsley M. Gastrocnemius muscle architecture in elite basketballers and cyclists: a cross-sectional cohort study. Front Sports Act Living. 2021;3:768846.
Rodriguez-Quiroga S. Late adult-onset Niemann Pick type C (NPC): An “atypical” typical presentation at the age of 62. Expert commentary. Parkinsonism Relat Disord. 2024;120:105981.
Uwada J, Nakazawa H, Mikami D, Islam MS, Muramatsu I, Taniguchi T, et al. PNU-120596, a positive allosteric modulator of alpha7 nicotinic acetylcholine receptor, directly inhibits p38 MAPK. Biochem Pharm. 2020;182:114297.
Jun MH, Kim YM, Kim JU. Modern acupuncture-like stimulation methods: a literature review. Integr Med Res. 2015;4:195–219.
Wang TQ, Li LR, Tan CX, Yang JW, Shi GX, Wang LQ, et al. Effect of electroacupuncture on gut microbiota in participants with knee osteoarthritis. Front Cell Infect Microbiol. 2021;11:597431.
Wei L, Wang R, Lin K, Jin X, Li L, Wazir J, et al. Creatine modulates cellular energy metabolism and protects against cancer cachexia-associated muscle wasting. Front Pharm. 2022;13:1086662.
Niu M, Song S, Su Z, Wei L, Li L, Pu W, et al. Inhibition of heat shock protein (HSP) 90 reverses signal transducer and activator of transcription (STAT) 3-mediated muscle wasting in cancer cachexia mice. Br J Pharm. 2021;178:4485–4500.
Schmidt EK, Clavarino G, Ceppi M, Pierre P. SUnSET, a nonradioactive method to monitor protein synthesis. Nat Methods. 2009;6:275–7.
Heinz S, Benner C, Spann N, Bertolino E, Lin YC, Laslo P, et al. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol Cell. 2010;38:576–89.
Acknowledgements
We appreciate the contributions of all our lab members and the valuable insights provided by Prof. Muthu Periasamy in the scientific discussions. We thank BioRender for the drawing tools provided.
Funding
This research was funded by grants from the National Natural Science Foundation of China (No. 82571040, 82370899 and 82070912); State Key Laboratory of Analytical Chemistry for Life Science (5431ZZXM2404); Visiting Researcher Fund Program of State Key Laboratory of Metabolism and Regulation in Complex Organisms (Grant No.KF20250005); Medical Research Priority Project Establishment of Yancheng Municipal Health Commission (YK2023003); NUS-NJU Research Collaboration Fund; Jiangsu Province Leading Talents Cultivation Project for Traditional Chinese Medicine (SLJ0316) and Jiangsu Province Basic Research Funds (No. BK20251988, BK20251803). It was also supported by the Fundamental Research Funds for the Central Universities (No. 14380538, 14380550 to Z.H.) and by team building and scientific research start-up funds of Nanjing University (No. 14912217 to Z.H.). R.F. was supported by EFSD Novo Nordisk future leaders award, Swedish Research Council (2023-02311), Swedish Cancer Society (232891 Pj), and Karolinska Institute Strategic Research Program in Diabetes (SRP) Rolf Luft Grants.
Author information
Authors and Affiliations
Contributions
ZH, HW, MS, and GS supervised and designed the experiments and revised the manuscript. XJ, YZ, and LS performed the experiments, analyzed the data and wrote the manuscript. DH, QZ, and RF analyzed the data. LL, CZ, ZS, and CD provided essential technical instructions and intellectual support. HW and ZH provided funding support. XJ, YZ, and LS contributed equally as first authors.
Corresponding authors
Ethics declarations
Ethics approval
All animal procedures were conducted at Nanjing University in accordance with the Law of the People’s Republic of China on Progress of Science and Technology and the Law of the People’s Republic of China on Animal Epidemic Prevention, concerning the protection of animals used for scientific purposes. All methods were carried out in compliance with the relevant guidelines and regulations.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jin, X., Zhou, Y., Sun, L. et al. Activation of α7nAChR reduces inflammation and apoptosis, promoting muscle regeneration through the AKT-FOXO1 pathway. Cell Death Differ (2026). https://doi.org/10.1038/s41418-026-01738-1
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41418-026-01738-1