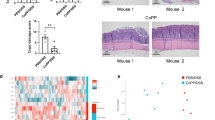
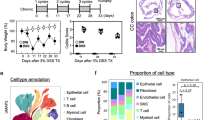
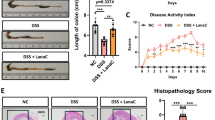
Abstract
To elucidate the role and regulatory mechanisms of macrophage-derived cystatin C (CST3) in Crohn’s disease (CD), focusing on colonic inflammation, macrophage–epithelial interactions, and barrier dysfunction. Colonic samples from CD patients, including inflamed and non-inflamed regions, were subjected to scRNA-seq. In vitro macrophage–epithelial co-culture models and untargeted metabolomics were employed, and the findings were validated using macrophage-specific CST3 knockout (KO) and overexpression mice under TNBS-induced and IL-10 KO colitis conditions. Mechanistic investigations included Co-IP, ChIP-qPCR, ubiquitination assays, rescue experiments, and functional analyses of efferocytosis, macrophage polarization, and barrier integrity. CST3 expression was considerably reduced in macrophages from inflamed CD tissues through suppressor of SMAD5-dependent transcriptional repression and MYCBP2-mediated K48-linked ubiquitination and degradation. Loss of CST3 impaired efferocytosis and M2 polarization by inhibiting the ACVR1C/TGF-β/SMAD pathway. CST3 deficiency also disrupted intestinal epithelial proliferation, compromised barrier function, and increased apoptosis via enhanced NAMPT-INSR signaling and accumulation of the inflammatory cytokines. In mice, macrophage-specific CST3 deletion exacerbated colitis, whereas its overexpression alleviated inflammation and restored epithelial integrity. These findings establish macrophage CST3 as a key regulator of immune–metabolic–epithelial crosstalk in CD, and indicate that restoring CST3 function or targeting its regulatory axis may represent a novel therapeutic strategy for CD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
Data availability
Single-cell sequences and RNA-sequences were deposited to GEO database (GSE319204, GSE318527). Please contact the corresponding author upon reasonable request.
References
Torres J, Mehandru S, Colombel JF, Peyrin-Biroulet L. Crohn’s disease. Lancet. 2017;389:1741–55.
Rogler G, Singh A, Kavanaugh A, Rubin DT. Extraintestinal manifestations of inflammatory bowel disease: current concepts, treatment, and implications for disease management. Gastroenterology. 2021;161:1118–32.
Gisbert JP, Chaparro M. De-escalation of biologic treatment in inflammatory bowel disease: a comprehensive review. J Crohns Colitis. 2024;18:642–58.
Dharmasiri S, Garrido-Martin EM, Harris RJ, Bateman AC, Collins JE, Cummings J, et al. Human intestinal macrophages are involved in the pathology of both ulcerative colitis and Crohn disease. Inflamm Bowel Dis. 2021;27:1641–52.
Han X, Ding S, Jiang H, Liu G. Roles of macrophages in the development and treatment of gut inflammation. Front Cell Dev Biol. 2021;9:625423.
Elliott TR, Hudspith BN, Rayment NB, Prescott NJ, Petrovska L, Hermon-Taylor J, et al. Defective macrophage handling of Escherichia coli in Crohn’s disease. J Gastroenterol Hepatol. 2015;30:1265–74.
Pastorelli L, De Salvo C, Mercado JR, Vecchi M, Pizarro TT. Central role of the gut epithelial barrier in the pathogenesis of chronic intestinal inflammation: lessons learned from animal models and human genetics. Front Immunol. 2013;4:280.
Zhao J, Li Y, Ying P, Zhou Y, Xu Z, Wang D, et al. ITLN1 exacerbates Crohn’s colitis by driving ZBP1-dependent PANoptosis in intestinal epithelial cells through antagonizing TRIM8-mediated CAPN2 ubiquitination. Int J Biol Sci. 2025;21:3705–25.
Best WR, Becktel JM, Singleton JW, Kern F Jr. Development of a Crohn’s disease activity index. National Cooperative Crohn’s Disease Study. Gastroenterology. 1976;70:439–44.
Daperno M, D'haens G, Van Assche G, Baert F, Bulois P, Maunoury V, et al. Development and validation of a new, simplified endoscopic activity score for Crohn’s disease: the SES-CD. Gastrointest Endosc. 2004;60:505–12.
Macosko EZ, Basu A, Satija R, Nemesh J, Shekhar K, Goldman M, et al. Highly parallel genome-wide expression profiling of individual cells using nanoliter droplets. Cell. 2015;161:1202–14.
Butler A, Hoffman P, Smibert P, Papalexi E, Satija R. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat Biotechnol. 2018;36:411–20.
Raudvere U, Kolberg L, Kuzmin I, Arak T, Adler P, Peterson H, et al. g:Profiler: a web server for functional enrichment analysis and conversions of gene lists (2019 update). Nucleic Acids Res. 2019;47:W191–W8.
Prianichnikov N, Koch H, Koch S, Lubeck M, Heilig R, Brehmer S, et al. MaxQuant Software for Ion Mobility Enhanced Shotgun Proteomics. Mol Cell Proteomics. 2020;19:1058–69.
UniProt: the universal protein knowledgebase in 2021. Nucleic Acids Res. 2021. 49: D480-D9.
Wirtz S, Popp V, Kindermann M, Gerlach K, Weigmann B, Fichtner-Feigl S, et al. Chemically induced mouse models of acute and chronic intestinal inflammation. Nat Protoc. 2017;12:1295–309.
Wirtz S, Neufert C, Weigmann B, Neurath MF. Chemically induced mouse models of intestinal inflammation. Nat Protoc. 2007;2:541–6.
Chiriac MT, Hracsko Z, Günther C, Gonzalez-Acera M, Atreya R, Stolzer I, et al. IL-20 controls resolution of experimental colitis by regulating epithelial IFN/STAT2 signalling. Gut. 2024;73:282–97.
Huang S, Xie Z, Han J, Wang H, Yang G, Li M, et al. Protocadherin 20 maintains intestinal barrier function to protect against Crohn’s disease by targeting ATF6. Genome Biol. 2023;24:159.
Singh UP, Singh S, Taub DD, Lillard JW Jr. Inhibition of IFN-gamma-inducible protein-10 abrogates colitis in IL-10-/- mice. J Immunol. 2003;171:1401–6.
Yu M, Luo Y, Cong Z, Mu Y, Qiu Y, Zhong M. MicroRNA-590-5p Inhibits Intestinal Inflammation by Targeting YAP. J Crohns Colitis. 2018;12:993–1004.
Arrieta MC, Madsen K, Doyle J, Meddings J. Reducing small intestinal permeability attenuates colitis in the IL10 gene-deficient mouse. Gut. 2009;58:41–8.
Looijer-van Langen M, Hotte N, Dieleman LA, Albert E, Mulder C, Madsen KL. Estrogen receptor-β signaling modulates epithelial barrier function. Am J Physiol Gastrointest Liver Physiol. 2011;300:G621–6.
Putt KK, Pei R, White HM, Bolling BW. Yogurt inhibits intestinal barrier dysfunction in Caco-2 cells by increasing tight junctions. Food Funct. 2017;8:406–14.
Ahmad R, Rah B, Bastola D, Dhawan P, Singh AB. Obesity-induces organ and tissue specific tight junction restructuring and barrier deregulation by claudin switching. Sci Rep. 2017;7:5125.
Zhao J, Wang H, Zhou J, Qian J, Yang H, Zhou Y, et al. miR-130a-3p, a Preclinical Therapeutic Target for Crohn’s Disease. J Crohns Colitis. 2021;15:647–64.
Wang L, Li H, Huang A, Zhao Y, Xiao C, Dong J, et al. Mutual regulation between TRIM21 and TRIM8 via K48-linked ubiquitination. Oncogene. 2023;42:3708–18.
Li Z, Zhang W, Wei XY, Hu JZ, Hu X, Liu H, et al. TRIM15 drives chondrocyte senescence and osteoarthritis progression. Sci Transl Med. 2025;17:eadq1735.
Jiang X, Wang J, Lin L, Du L, Ding Y, Zheng F, et al. Macrophages promote pre-metastatic niche formation of breast cancer through aryl hydrocarbon receptor activity. Signal Transduct Target Ther. 2024;9:352.
Hong H, Han H, Wang L, Cao W, Hu M, Li J, et al. ABCF1-K430-Lactylation promotes HCC malignant progression via transcriptional activation of HIF1 signaling pathway. Cell Death Differ. 2025;32:613–31.
Cerda MB, Lloyd R, Batalla M, Giannoni F, Casal M, Policastro L. Silencing peroxiredoxin-2 sensitizes human colorectal cancer cells to ionizing radiation and oxaliplatin. Cancer Lett. 2017;388:312–9.
Rana N, Privitera G, Kondolf HC, Bulek K, Lechuga S, De Salvo C, et al. GSDMB is increased in IBD and regulates epithelial restitution/repair independent of pyroptosis. Cell. 2022;185:283–98.e17.
Wang Y, Liu XY, Wang Y, Zhao WX, Li FD, Guo PR, et al. NOX2 inhibition stabilizes vulnerable plaques by enhancing macrophage efferocytosis via MertK/PI3K/AKT pathway. Redox Biol. 2023;64:102763.
Fritsch SD, Sukhbaatar N, Gonzales K, Sahu A, Tran L, Vogel A, et al. Metabolic support by macrophages sustains colonic epithelial homeostasis. Cell Metab. 2023;35:1931–43.e8.
Flannagan RS, Jaumouillé V, Grinstein S. The cell biology of phagocytosis. Annu Rev Pathol. 2012;7:61–98.
Fan W, Qu Y, Yuan X, Shi H, Liu G, Loureirin B. Accelerates diabetic wound healing by promoting TGFβ/Smad-dependent macrophage M2 polarization: a concerted analytical approach through single-cell RNA sequencing and experimental verification. Phytother Res. 2024;39:5450–63 .
Geng K, Ma X, Jiang Z, Gu J, Huang W, Wang W, et al. WDR74 facilitates TGF-β/Smad pathway activation to promote M2 macrophage polarization and diabetic foot ulcer wound healing in mice. Cell Biol Toxicol. 2023;39:1577–91.
Ramos GP, Papadakis KA. Mechanisms of disease: inflammatory bowel diseases. Mayo Clin Proc. 2019;94:155–65.
Liu J, Chen Y. Cell-cell crosstalk between fat cells and immune cells. Am J Physiol Endocrinol Metab. 2024;327:E371–E83.
Serretti A, Olgiati P, De Ronchi D. Genetics of Alzheimer’s disease. A rapidly evolving field. J Alzheimers Dis. 2007;12:73–92.
Gren ST, Janciauskiene S, Sandeep S, Jonigk D, Kvist PH, Gerwien JG, et al. The protease inhibitor cystatin C down-regulates the release of IL-β and TNF-α in lipopolysaccharide activated monocytes. J Leukoc Biol. 2016;100:811–22.
Lin Z, Zhuang J, He L, Zhu S, Kong W, Lu W, et al. Exploring Smad5: a review to pave the way for a deeper understanding of the pathobiology of common respiratory diseases. Mol Med. 2024;30:225.
Allaire JM, Darsigny M, Marcoux SS, Roy SA, Schmouth JF, Umans L, et al. Loss of Smad5 leads to the disassembly of the apical junctional complex and increased susceptibility to experimental colitis. Am J Physiol Gastrointest Liver Physiol. 2011;300:G586–97.
Szondy Z, Garabuczi E, Joós G, Tsay GJ, Sarang Z. Impaired clearance of apoptotic cells in chronic inflammatory diseases: therapeutic implications. Front Immunol. 2014;5:354.
Kawano M, Nagata S. Efferocytosis and autoimmune disease. Int Immunol. 2018;30:551–8.
Wu J, Jiang L, Wang S, Peng L, Zhang R, Liu Z. TGF β1 promotes the polarization of M2-type macrophages and activates PI3K/mTOR signaling pathway by inhibiting ISG20 to sensitize ovarian cancer to cisplatin. Int Immunopharmacol. 2024;134:112235.
Su J, Song Y, Zhu Z, Huang X, Fan J, Qiao J, et al. Cell-cell communication: new insights and clinical implications. Signal Transduct Target Ther. 2024;9:196.
Naik S, Larsen SB, Gomez NC, Alaverdyan K, Sendoel A, Yuan S, et al. Inflammatory memory sensitizes skin epithelial stem cells to tissue damage. Nature. 2017;550:475–80.
Zhao D, Ravikumar V, Leach TJ, Kraushaar D, Lauder E, Li L, et al. Inflammation-induced epigenetic imprinting regulates intestinal stem cells. Cell Stem Cell. 2024;31:1447–64.e6.
Funding
This work was supported by National Natural Science Foundation of China (82300609, 82000492), Jiangsu Provincial Commission of Health and Family Planning (No.M2025006), Changzhou Sci&Tech Program (2022CZBJ066, CJ20245024), Changzhou Medical Center of Nanjing Medical University Program (CMC2024PY17), Yanzhen Talent Program for Emerging Academic Leaders (YZ-HBDTR-SJ-2025), Taizhou School of Clinical Medicine, Nanjing Medical University (TZKY20230308, TZKY20230102).
Author information
Authors and Affiliations
Contributions
HGW, CYJ, HLX, and JZ: Study design and data analysis. SQC, CC, WLJ, and ZWX: Patient recruitment, data collection and writing up of the first draft of the paper. HGW, JBY, JS, and JZ: scientific advice, supervision and drafting of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The clinical study was approved by the ethics committee of The First Affiliated Hospital of Nanjing Medical University (No. 2023-SR-115). All the experimental protocols were approved by the Ethics Committee of the Laboratory Animal Center of Nantong University (NO.S20200323-289) according to the guidelines by the Chinese Council on Animal Care.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, H., Jiao, C., Xing, H. et al. Transcriptional and ubiquitinative suppression of macrophage CST3 disrupts colonic homeostasis through defective efferocytosis. Cell Death Differ (2026). https://doi.org/10.1038/s41418-026-01750-5
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41418-026-01750-5