Abstract
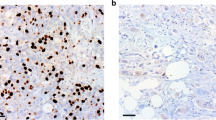
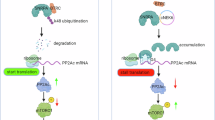
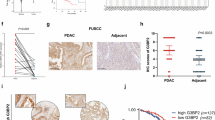
Gemcitabine is a cornerstone chemotherapeutic for pancreatic ductal adenocarcinoma (PDAC); however, the frequent development of resistance compromises its efficacy and poses a significant challenge to patient prognosis. Here, we report that nuclear pore protein NUP93 is upregulated in PDAC and correlates with poor patient survival. Functional studies demonstrated that NUP93 promotes PDAC cell proliferation and confers gemcitabine resistance by enhancing DNA damage repair. Mechanistically, NUP93 interacts with the transcription factor SOX2 by recognizing its nuclear localization sequence and facilitates its nuclear import. Nuclear SOX2 transcriptionally activates the key stress granule component G3BP1 by directly binding to its promoter. Subsequently, G3BP1 stabilizes the mRNA of RAD51, a crucial homologous recombination repair factor, thereby promoting DNA damage repair and gemcitabine resistance. In vivo, disruption of the NUP93/SOX2/G3BP1 axis suppressed tumor growth and synergized with gemcitabine. Our findings unveil the novel NUP93-SOX2-G3BP1 signaling axis as a critical driver of gemcitabine resistance in PDAC, presenting a promising therapeutic target for overcoming chemoresistance.
Similar content being viewed by others
Data availability
All data are included within the main text and supplementary files.
References
Siegel RL, Kratzer TB, Giaquinto AN, Sung H, Jemal A. Cancer statistics, 2025. CA Cancer J Clin. 2025;75:10–45.
Mizrahi JD, Surana R, Valle JW, Shroff RT. Pancreatic cancer. Lancet. 2020;395:2008–20.
du Toit-Thompson T, Leck L, Gillson J, Pavlakis N, Gill AJ, Samra JS, et al. Overcoming therapy resistance in pancreatic cancer: challenges and emerging strategies. Adv Drug Deliv Rev. 2025;224:115647.
Park W, Chawla A, O’Reilly EM. Pancreatic cancer: a review. JAMA. 2021;326:851–62.
Xu L, Ma X, Zhang X, Zhang C, Zhang Y, Gong S, et al. hsa_circ_0007919 induces LIG1 transcription by binding to FOXA1/TET1 to enhance the DNA damage response and promote gemcitabine resistance in pancreatic ductal adenocarcinoma. Mol Cancer. 2023;22:195.
Perkhofer L, Gout J, Roger E, Kude de Almeida F, Baptista Simoes C, Wiesmuller L, et al. DNA damage repair as a target in pancreatic cancer: state-of-the-art and future perspectives. Gut. 2021;70:606–17.
Mini E, Nobili S, Caciagli B, Landini I, Mazzei T. Cellular pharmacology of gemcitabine. Ann Oncol. 2006;17:v7–12.
Calheiros J, Silva R, Barbosa F, Morais J, Moura SR, Almeida S, et al. A first-in-class inhibitor of homologous recombination DNA repair counteracts tumour growth, metastasis and therapeutic resistance in pancreatic cancer. J Exp Clin Cancer Res. 2025;44:129.
Liang YY, Khalid K, Le HV, Teo HMV, Raitelaitis M, Gerault MA, et al. MS CETSA deep functional proteomics uncovers DNA repair programs leading to gemcitabine resistance. Nat Commun. 2025;16:4234.
Im MM, Flanagan SA, Ackroyd JJ, Knapp B, Kramer A, Shewach DS. Late DNA damage mediated by homologous recombination repair results in radiosensitization with gemcitabine. Radiat Res. 2016;186:466–77.
Xing F, Qin Y, Xu J, Wang W, Zhang B. Stress granules dynamics and promising functions in pancreatic cancer. Biochim Biophys Acta Rev Cancer. 2023;1878:188885.
Xing FL, Li BR, Fang YJ, Liang C, Liu J, Wang W, et al. G3BP2 promotes tumor progression and gemcitabine resistance in PDAC via regulating PDIA3-DKC1-hENT in a stress granule-dependent manner. Acta Pharmacol Sin. 2025;46:474–88.
Zhan Y, Wang H, Ning Y, Zheng H, Liu S, Yang Y, et al. Understanding the roles of stress granule during chemotherapy for patients with malignant tumors. Am J Cancer Res. 2020;10:2226–41.
Chen L, Sun K, Qin W, Huang B, Wu C, Chen J, et al. LIMK1 m(6)A-RNA methylation recognized by YTHDC2 induces 5-FU chemoresistance in colorectal cancer via endoplasmic reticulum stress and stress granule formation. Cancer Lett. 2023;576:216420.
Zhao Z, Qing Y, Dong L, Han L, Wu D, Li Y, et al. QKI shuttles internal m(7)G-modified transcripts into stress granules and modulates mRNA metabolism. Cell. 2023;186:3208–3226 e3227.
Li L, Garg M, Wang Y, Wang W, Godbout R. DEAD box 1 (DDX1) protein binds to and protects cytoplasmic stress response mRNAs in cells exposed to oxidative stress. J Biol Chem. 2022;298:102180.
Beck M, Hurt E. The nuclear pore complex: understanding its function through structural insight. Nat Rev Mol Cell Biol. 2017;18:73–89.
Huang G, Zhan X, Zeng C, Zhu X, Liang K, Zhao Y, et al. Cryo-EM structure of the nuclear ring from Xenopus laevis nuclear pore complex. Cell Res. 2022;32:349–58.
Furusawa Y, Yamada S, Kawaoka Y. Host factor nucleoporin 93 is involved in the nuclear export of influenza virus RNA. Front Microbiol. 2018;9:1675.
Gomez GN, Abrar F, Dodhia MP, Gonzalez FG, Nag A. SARS coronavirus protein nsp1 disrupts localization of Nup93 from the nuclear pore complex. Biochem Cell Biol. 2019;97:758–66.
Pan L, Song XW, Song JC, Shi CY, Wang ZK, Huang SQ, et al. Downregulation of NUP93 aggravates hypoxia-induced death of cardiomyocytes in vitro through abnormal regulation of gene transcription. Acta Pharmacol Sin. 2023;44:969–83.
Braun DA, Sadowski CE, Kohl S, Lovric S, Astrinidis SA, Pabst WL, et al. Mutations in nuclear pore genes NUP93, NUP205 and XPO5 cause steroid-resistant nephrotic syndrome. Nat Genet. 2016;48:457–65.
Moudry P, Lukas C, Macurek L, Neumann B, Heriche JK, Pepperkok R, et al. Nucleoporin NUP153 guards genome integrity by promoting nuclear import of 53BP1. Cell Death Differ. 2012;19:798–807.
Gaillard H, Santos-Pereira JM, Aguilera A. The Nup84 complex coordinates the DNA damage response to warrant genome integrity. Nucleic Acids Res. 2019;47:4054–67.
Rodriguez-Berriguete G, Granata G, Puliyadi R, Tiwana G, Prevo R, Wilson RS, et al. Nucleoporin 54 contributes to homologous recombination repair and post-replicative DNA integrity. Nucleic Acids Res. 2018;46:7731–46.
Nataraj NB, Noronha A, Lee JS, Ghosh S, Mohan Raju HR, Sekar A, et al. Nucleoporin-93 reveals a common feature of aggressive breast cancers: robust nucleocytoplasmic transport of transcription factors. Cell Rep. 2022;38:110418.
Lin CS, Liang Y, Su SG, Zheng YL, Yang X, Jiang N, et al. Nucleoporin 93 mediates beta-catenin nuclear import to promote hepatocellular carcinoma progression and metastasis. Cancer Lett. 2022;526:236–47.
Wang Z, Zhang J, Luo L, Zhang C, Huang X, Liu S, et al. Nucleoporin 93 regulates cancer cell growth and stemness in bladder cancer via Wnt/beta-catenin signaling. Mol Biotechnol. 2025;67:2072–84.
Ouyang X, Hao X, Liu S, Hu J, Hu L. Expression of Nup93 is associated with the proliferation, migration and invasion capacity of cervical cancer cells. Acta Biochim Biophys Sin. 2019;51:1276–85.
Gong R, Chen X, Sun X, Zhang Y, Wang J, Yu Q, et al. Identification of FOXP3(+) epithelial cells contributing to pancreatic proliferation and angiogenesis. Am J Physiol Cell Physiol. 2024;326:C294–C303.
Zhang Y, Chen XN, Zhang H, Wen JK, Gao HT, Shi B, et al. CDK13 promotes lipid deposition and prostate cancer progression by stimulating NSUN5-mediated m5C modification of ACC1 mRNA. Cell Death Differ. 2023;30:2462–76.
Sun H, Yang Z, Teng Z, Zhang Y, Han Z, Xu C, et al. DDX58 expression promotes inflammation and growth arrest in Sertoli cells by stabilizing p65 mRNA in patients with Sertoli cell-only syndrome. Front Immunol. 2023;14:1135753.
Liu X, Zhang S, An Y, Xu B, Yan G, Sun M. USP10/XAB2/ANXA2 axis promotes DNA damage repair to enhance chemoresistance to oxaliplatin in colorectal cancer. J Exp Clin Cancer Res. 2025;44:94.
Wen H, Qu C, Wang Z, Gao H, Liu W, Wang H, et al. Cuproptosis enhances docetaxel chemosensitivity by inhibiting autophagy via the DLAT/mTOR pathway in prostate cancer. FASEB J. 2023;37:e23145.
Guo M, Chen S, Sun J, Xu R, Qi Z, Li J, et al. PIP5K1A suppresses ferroptosis and induces sorafenib resistance by stabilizing NRF2 in hepatocellular carcinoma. Adv Sci. 2025;12:e04372.
Qi JC, Yang Z, Lin T, Ma L, Wang YX, Zhang Y, et al. CDK13 upregulation-induced formation of the positive feedback loop among circCDK13, miR-212-5p/miR-449a and E2F5 contributes to prostate carcinogenesis. J Exp Clin Cancer Res. 2021;40:2.
Zhu X, Qi C, Wang R, Lee JH, Shao J, Bei L, et al. Acute depletion of human core nucleoporin reveals direct roles in transcription control but dispensability for 3D genome organization. Cell Rep. 2022;41:111576.
Vollmer B, Antonin W. The diverse roles of the Nup93/Nic96 complex proteins - structural scaffolds of the nuclear pore complex with additional cellular functions. Biol Chem. 2014;395:515–28.
Labade AS, Salvi A, Kar S, Karmodiya K, Sengupta K. Nup93 and CTCF modulate spatiotemporal dynamics and function of the HOXA gene locus during differentiation. J Cell Sci. 2021, 134:jcs259307.
Schoenfelder S, Fraser P. Long-range enhancer-promoter contacts in gene expression control. Nat Rev Genet. 2019;20:437–55.
Sanyal A, Lajoie BR, Jain G, Dekker J. The long-range interaction landscape of gene promoters. Nature. 2012;489:109–13.
Huang G, Zhan X, Zeng C, Liang K, Zhu X, Zhao Y, et al. Cryo-EM structure of the inner ring from the Xenopus laevis nuclear pore complex. Cell Res. 2022;32:451–60.
Chen B, Xu F, Gao Y, Hu G, Zhu K, Lu H, et al. DNA damage-induced translocation of mitochondrial factor HIGD1A into the nucleus regulates homologous recombination and radio/chemo-sensitivity. Oncogene. 2022;41:1918–30.
Monwan W, Kawasaki T, Hasan MZ, Ori D, Kawai T. Identification of nucleoporin 93 (Nup93) that mediates antiviral innate immune responses. Biochem Biophys Res Commun. 2020;521:1077–82.
Liu L, Du X, Fang J, Zhao J, Guo Y, Zhao Y, et al. Development of an interferon gamma response-related signature for prediction of survival in clear cell renal cell carcinoma. J Inflamm Res. 2021;14:4969–85.
Singh MK, Jamal F, Dubey AK, Shivam P, Kumari S, Pushpanjali, et al. Visceral leishmaniasis: a novel nuclear envelope protein ‘nucleoporins-93 (NUP-93)’ from Leishmania donovani prompts macrophage signaling for T-cell activation towards host protective immune response. Cytokine. 2019;113:200–15.
Bonilla B, Hengel SR, Grundy MK, Bernstein KA. RAD51 gene family structure and function. Annu Rev Genet. 2020;54:25–46.
Li L, Lai F, Hu X, Liu B, Lu X, Lin Z, et al. Multifaceted SOX2-chromatin interaction underpins pluripotency progression in early embryos. Science. 2023;382:eadi5516.
Roy S, Dukic T, Keepers Z, Bhandary B, Lamichhane N, Molitoris J, et al. SOX2 and OCT4 mediate radiation and drug resistance in pancreatic tumor organoids. Cell Death Discov. 2024;10:106.
Jia Y, Gu D, Wan J, Yu B, Zhang X, Chiorean EG, et al. The role of GLI-SOX2 signaling axis for gemcitabine resistance in pancreatic cancer. Oncogene. 2019;38:1764–77.
Cattoglio C, Zhang ET, Grubisic I, Chiba K, Fong YW, Tjian R. Functional and mechanistic studies of XPC DNA-repair complex as transcriptional coactivator in embryonic stem cells. Proc Natl Acad Sci USA. 2015;112:E2317–2326.
Fong YW, Inouye C, Yamaguchi T, Cattoglio C, Grubisic I, Tjian R. A DNA repair complex functions as an Oct4/Sox2 coactivator in embryonic stem cells. Cell. 2011;147:120–31.
Xie CK, Liao CY, Lin HY, Wu YD, Lu FC, Huang XX, et al. Sulindac (K-80003) with nab-paclitaxel and gemcitabine overcomes drug-resistant pancreatic cancer. Mol Cancer. 2024;23:215.
Lin K, Zhou E, Shi T, Zhang S, Zhang J, Zheng Z, et al. m6A eraser FTO impairs gemcitabine resistance in pancreatic cancer through influencing NEDD4 mRNA stability by regulating the PTEN/PI3K/AKT pathway. J Exp Clin Cancer Res. 2023;42:217.
Golan T, O’Kane GM, Denroche RE, Raitses-Gurevich M, Grant RC, Holter S, et al. Genomic features and classification of homologous recombination-deficient pancreatic ductal adenocarcinoma. Gastroenterology. 2021;160:2119–2132 e2119.
Zhang C, Chen L, Peng D, Jiang A, He Y, Zeng Y, et al. METTL3 and N6-methyladenosine promote homologous recombination-mediated repair of DSBs by modulating DNA-RNA hybrid accumulation. Mol Cell. 2020;79:425–442 e427.
Yang J, Xu P, Chen Z, Zhang X, Xia Y, Fang L, et al. N6-methyadenosine modified SUV39H2 regulates homologous recombination through epigenetic repression of DUSP6 in gastric cancer. Cancer Lett. 2023;558:216092.
Xie Z, Li W, Ai J, Xie J, Zhang X. C2orf40 inhibits metastasis and regulates chemo-resistance and radio-resistance of nasopharyngeal carcinoma cells by influencing cell cycle and activating the PI3K/AKT/mTOR signaling pathway. J Transl Med. 2022;20:264.
Zhou B, Yang Y, Pang X, Shi J, Jiang T, Zheng X. Quercetin inhibits DNA damage responses to induce apoptosis via SIRT5/PI3K/AKT pathway in non-small cell lung cancer. Biomed Pharmacother. 2023;165:115071.
Lopez BS. RAD51-mediated homologous recombination is a pro-tumour driver pathway. Oncogene. 2025;44:4006–16.
Demeyer A, Benhelli-Mokrani H, Chenais B, Weigel P, Fleury F. Inhibiting homologous recombination by targeting RAD51 protein. Biochim Biophys Acta Rev Cancer. 2021;1876:188597.
Al-Ejeh F, Pajic M, Shi W, Kalimutho M, Miranda M, Nagrial AM, et al. Gemcitabine and CHK1 inhibition potentiate EGFR-directed radioimmunotherapy against pancreatic ductal adenocarcinoma. Clin Cancer Res. 2014;20:3187–97.
Kausar T, Schreiber JS, Karnak D, Parsels LA, Parsels JD, Davis MA, et al. Sensitization of pancreatic cancers to gemcitabine chemoradiation by WEE1 kinase inhibition depends on homologous recombination repair. Neoplasia. 2015;17:757–66.
Tsai MS, Kuo YH, Chiu YF, Su YC, Lin YW. Down-regulation of Rad51 expression overcomes drug resistance to gemcitabine in human non-small-cell lung cancer cells. J Pharmacol Exp Ther. 2010;335:830–40.
Funding
This study was partially supported by the National Natural Science Foundation of China (No. 82472667); Taishan Scholars Young Experts Project (No. tsqn202312380); China Higher Education Innovation Fund project (No. 2024GR041); the Natural Science Foundation of Shandong Province (ZR2025MS1491).
Author information
Authors and Affiliations
Contributions
ZY, HS, and CX carry out the design and conception. HS, CXY, JTD, and JSC implement the methodology. HS, CXY, and JTD acquire the data. HS, CX, YC, and JSC carry out analysis and interpretation of the data. HS, CXY, and RNG conducted animal models. ZY, HS, and NXY carry out writing, review, and/or revision of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics Statement
All methods in this study were performed in accordance with the relevant guidelines and regulations. The study involving human participants was reviewed and approved by the Ethics Committee of the Affiliated Hospital of Qingdao University (Permission No. QYFYWZLL28017). All patients provided written informed consent prior to participation. All animal experimental procedures were approved by the Committee for the Care and Use of Laboratory Animals at the Affiliated Hospital of Qingdao University (Permission No. AHQU-MAL20201016) and were carried out in compliance with the Guide for the Care and Use of Laboratory Animals (National Research Council).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Professor Nickolai Barley
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sun, H., Yang, C., Du, J. et al. NUP93 facilitates the nuclear import of SOX2 to activate G3BP1 transcription and impairs gemcitabine response in pancreatic cancer. Cell Death Dis (2026). https://doi.org/10.1038/s41419-026-08586-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-026-08586-4