Abstract
Sepsis, a life-threatening condition caused by infection, is characterized by the dysregulation of immune responses and activation of monocytes. Plac8, a protein, has been implicated in various inflammatory conditions. This study aimed to investigate the effect of Plac8 upregulation on monocyte proliferation and activation in sepsis patients. Peripheral blood samples were collected from healthy individuals and sepsis patients. Monocytes were stimulated with lipopolysaccharide (LPS) to create an in vitro sepsis model, while a murine sepsis model was established using cecal ligation and puncture (CLP). The levels of monocyte markers, proliferation index (PI), and pro-inflammatory cytokines were assessed using flow cytometry and qPCR, respectively. Plac8 and phosphorylated ERK protein levels were determined by western blot, and TNF-α, IL-6, and IL-10 levels were quantified using ELISA. The CCK-8 assay was used to evaluate PBMC proliferation and activation. The results showed that Plac8 was highly expressed in sepsis models, promoting the survival, proliferation, and activation of monocytes. Plac8 upregulation activated the ERK pathway, leading to increased phosphorylation of ERK protein and elevated levels of CD14, CD16, TNF-α, IL-6, Plac8, and IL-10. In sepsis mice, Plac8 overexpression similarly activated the ERK pathway and promoted the survival, proliferation, and activation of monocytes. In conclusion, the upregulation of Plac8 enhances the activation of the ERK pathway and promotes monocyte proliferation and activation in sepsis patients.
Similar content being viewed by others
Introduction
Sepsis is a life-threatening disease caused by an uncontrolled host response to infection, and it remains a major cause of death and severe illness worldwide, imposing a significant burden on global healthcare systems [1,2,3]. Despite advances in antimicrobial therapy and intensive care techniques, the incidence of sepsis continues to rise, attributed to the aging population and the increasing prevalence of antibiotic-resistant pathogens [2, 4, 5]. The complexity of sepsis arises from its heterogeneity, with diverse clinical manifestations ranging from systemic inflammatory response to severe septic shock and multiple organ dysfunction syndrome [6, 7]. Dysregulation of immune responses plays a central role in the pathophysiology of sepsis, and peripheral blood mononuclear cells (PBMCs) play a key role in this process [8,9,10]. Cells, including monocytes, lymphocytes, and dendritic cells, play important roles in balancing pro-inflammatory and anti-inflammatory responses [11, 12]. However, due to a limited understanding of the regulatory mechanisms governing PBMC function during the acute and recovery phases of sepsis, the clinical management of sepsis has become complex, highlighting the need for innovative research in this field.
Monocytes, a crucial subset of PBMCs, play a dual role in sepsis by directing innate immune responses through pathogen recognition, cytokine production, and initiation of adaptive immunity. These cells exhibit significant plasticity, differentiating into various effector cells, including macrophages and dendritic cells, to adapt to the ever-changing septic environment [13,14,15]. The functional state of monocytes is a key factor determining the progression and outcomes of sepsis [16]. A deeper understanding of the biology of monocytes in sepsis may reveal new therapeutic strategies to modulate their activity [17]. The bidirectional nature of monocytes in promoting inflammation and resolution and repair emphasizes the need to elucidate the regulatory networks influencing their function [18]. Despite extensive research on various signaling pathways, such as NF-κB and STAT, exploring alternative pathways regulating monocyte responses in sepsis remains a crucial research area [19].
Plac8 is a multifunctional protein involved in cell proliferation, differentiation, and immune regulation. Recently, it has garnered attention due to its potential role in infectious diseases [20, 21]. Preliminary research indicates that Plac8 plays a role in the regulation of immune cell functions; however, the exact mechanism by which it operates in this capacity is not fully comprehended [22, 23]. The involvement of this protein in autophagy, cell cycle control, and apoptosis implies its potential key role in sepsis pathogenesis [24]. Particularly in the context of monocytes, understanding how Plac8 influences their activity by modulating intracellular signaling is crucial [23, 25]. Within the extracellular signal-regulated kinase (ERK) signaling pathway, ERK activation promotes cell survival, proliferation, and differentiation, and is associated with the development of various diseases including infections and autoimmune disorders [26, 27]. In innate immune responses, the ERK pathway plays a crucial role in regulating cytokine production, cell migration, and functional effector differentiation. Therefore, the ERK pathway stands as a potent candidate mechanism for modulating monocyte function and shaping sepsis immune responses [28]. Previous studies have suggested that Plac8 participates in cell fate determination through the ERK pathway [29, 30]. Furthermore, the Plac8/ERK signaling pathway has been confirmed to be involved in regulating cell proliferation [31], yet its role in sepsis remains insufficiently explored. Thus, further research on the interaction between the ERK pathway and Plac8 is crucial for unraveling the complex network of immune cell function in sepsis pathology, providing new insights that could potentially form the basis for future targeted intervention strategies.
This study aims to enhance our understanding of the mechanisms underlying monocyte responses in sepsis, facilitate the translation from basic science to clinical practice, and strengthen our ability to combat this formidable enemy. Ultimately, this research intends to bridge the gap between basic science and clinical practice and enhance our arsenal against sepsis.
Results
Characteristics and blood evaluation of patients with septicemia
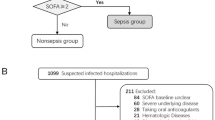
A total of 28 septicemia patients were included, comprising 15 males and 13 females, with an age range of 18–60 years and a mean age of 38.18 ± 12.97. Additionally, 18 normal controls were recruited, including 10 males and 8 females, with an age range of 28–64 years and a mean age of 45.44 ± 12.34. Statistical analysis revealed no significant differences in age or gender between septicemia patients and normal controls. The sepsis patients had a Sequential Organ Failure Assessment (SOFA) score of 6.54 ± 2.89. Details regarding complications and site of infection in septicemia patients can be found in Table S2.
Altered phenotype and function of PBMCs in patients with septicemia
Monocytes were isolated from the peripheral blood of septic patients and healthy individuals using Ficoll density gradient centrifugation. Compared to monocytes from healthy individuals, the proportion of CD14+ and CD16+ monocytes in the peripheral blood of septic patients was higher (Fig. 1A, B) (P < 0.05). qRT-PCR analysis revealed higher expression levels of the marker genes CD14, CD16, and plac8 (Fig. 1C) (P < 0.05). Western blot analysis demonstrated higher expression levels of the plac8 protein (Fig. 1E, F) (P < 0.05), with increased phosphorylation of ERK1/2 protein without significant changes in its expression levels (P < 0.05). ELISA results indicated higher expression levels of monocyte chemoattractant protein-1 (MCP-1), tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and anti-inflammatory cytokine IL-10 (Fig. 1D) (P < 0.05). Flow cytometry analysis showed a decrease in the proportion of cells in the G0/G1 phase and an increase in the proportion of cells in the S phase, accompanied by an elevated cell proliferation index (PI) with statistical significance (P < 0.05) (Fig. 1G, H). No significant differences were observed in the G2 phase of the cells (P > 0.05).
A Flow cytometry analysis of peripheral blood CD14+ and CD16+ cell subsets. B Flow cytometry analysis of CD14+ and CD16+ cell subsets. C qRT-PCR analysis of gene expression. D ELISA analysis of cytokine expression. E Western blot analysis of protein expression. F Western blot analysis of protein expression. G Flow cytometry analysis of cell cycle. H Cell proliferation index PI = (S + G2M)/(G0/1 + S + G2M). * indicates P < 0.05 compared to the normal group. Data is presented as mean ± standard deviation. Data analysis was performed using an independent samples t-test, and the experiment was repeated three times.
In vitro model of septicemia using LPS-stimulated PBMCs from healthy individuals
To construct an in vitro sepsis model, PBMCs from healthy individuals were stimulated with lipopolysaccharide (LPS). To confirm the model’s success, qRT-PCR analysis revealed higher expression levels of CD14, CD16, and plac8 genes in the LPS-stimulated group compared to the normal group (Fig. S1A, P < 0.05). Western blot analysis showed elevated expression of plac8 protein in the LPS-stimulated group compared to the normal group (Fig. S1B, P < 0.05), with no significant changes in the protein content of ERK1/2 but an increased phosphorylation level (P < 0.05). ELISA results demonstrated increased expression levels of TNF-α, IL-6, IL-10, and MCP-1 in the LPS-stimulated group compared to the normal group (Fig. S1C, P < 0.05). Furthermore, using the CCK-8 assay, it was observed that cell proliferation in the LPS-stimulated group was enhanced in comparison to the normal group (Fig. S1D, P < 0.05). These results indicate that LPS stimulation induces sepsis-like responses in mononuclear cells and influences their proliferation.
Plac8 modulation influences survival and proliferation of monocytes in an in vitro model of septicemia
In this study, we established an in vitro sepsis model by stimulating PBMCs from healthy individuals with LPS. The qRT-PCR results (Fig. 2A) showed a significant decrease in the expression levels of plac8, CD14, and CD16 genes in the si-Plac8 group compared to the si-plac8-NC group (P < 0.05). In contrast, the plac8 group exhibited a significant increase in the expression levels of plac8, CD14, and CD16 genes compared to the plac8-NC group (P < 0.05). Western blot analysis (Fig. 2B, C) revealed a decrease in the protein expression levels of plac8, CD14, and CD16 in the si-Plac8 group compared to the si-plac8-NC group (P < 0.05). Conversely, the plac8 group exhibited an increase in the protein expression levels of plac8, CD14, and CD16 compared to the plac8-NC group (P < 0.05). ELISA results (Fig. 2D) demonstrated a decrease in the protein expression levels of TNF-α, IL-6, IL-10, and MCP-1 in the si-Plac8 group compared to the si-plac8-NC group (P < 0.05). Similarly, the plac8 group exhibited an increase in the protein expression levels of TNF-α, IL-6, IL-10, and MCP-1 compared to the plac8-NC group (P < 0.05). Additionally, the CCK-8 assay (Fig. 2E) showed a decrease in cell proliferation of mononuclear cells in the si-Plac8 group compared to the si-plac8-NC group (P < 0.05). Conversely, the plac8 group exhibited increased mononuclear cell proliferation compared to the plac8-NC group (P < 0.05). These findings indicate that upregulation of plac8 enhances the survival and proliferation of mononuclear cells in sepsis patients, while downregulation of plac8 reduces their survival and proliferation, suggesting that plac8 influences the survival and proliferation of mononuclear cells in sepsis patients.
A qRT-PCR analysis of gene expression. B Western blot analysis of protein band. C Western blot analysis of protein expression. D ELISA analysis of cytokine expression. E Cell proliferation measured by CCK-8 assay, cell proliferation (%) = [OD (treatment group) − OD (blank group)] / [OD (control group) − OD (blank group)] × 100%. * indicates P < 0.05 compared to si-Plac8-NC or Plac8-NC group. Data is presented as mean ± standard deviation. An independent samples t-test was used to compare two groups, and two-way ANOVA was used to compare at different time points. The experiment was repeated three times.
Plac8 modulation via ERK pathway affects survival, proliferation, and activation of monocytes in septicemia
Western blot analysis revealed that (Fig. 3A, B), compared to the Plac8 + DMSO group, the Plac8 + GDC-0994 group demonstrated a significant decrease in plac8 protein levels (P < 0.05). Additionally, the phosphorylation levels of ERK1/2 proteins decreased in the Plac8 + GDC-0994 group, while the protein content remained unchanged. In contrast, in the si-Plac8 + EGF group, plac8 protein expression levels were significantly increased (P < 0.05). Similarly, the protein content of ERK1/2 remained unchanged, but the phosphorylation levels increased. ELISA results (Fig. 3C) showed that, compared to the Plac8 + DMSO group, the Plac8 + GDC-0994 group had decreased protein levels of pro-inflammatory factors TNF-α, IL-6, IL-10, and MCP-1 (P < 0.05). Conversely, compared to the si-Plac8 + DMSO group, the si-Plac8 + EGF group exhibited increased protein levels of these pro-inflammatory factors (P < 0.05). Cell proliferation, measured using the CCK-8 assay (Fig. 3D), showed a significant decrease in monocyte proliferation in the Plac8 + GDC-0994 group when compared to the Plac8 + DMSO group (P < 0.05). Conversely, the si-Plac8 + EGF group demonstrated significantly increased monocyte proliferation compared to the si-Plac8 + DMSO group (P < 0.05). These findings suggest that Plac8 influences the survival, proliferation, and activation of peripheral blood monocytes in septic patients through the ERK pathway.
A Western blot analysis of protein band. B Western blot analysis of protein expression. C ELISA analysis of cytokine expression. D Cell proliferation was measured by CCK-8 assay. * indicates P < 0.05 compared to Plac + DMSO or si-Plac + DMSO group. Data is presented as mean ± standard deviation. Independent samples t-test was used to compare two groups, and two-way ANOVA was used for comparison at different time points. The experiment was repeated three times.
Construction of a mouse model of sepsis
The success rate of constructing a mouse model of sepsis was 88.89%, with all mice surviving within 6 h. Results obtained from qRT-PCR analysis (Fig. 4A) indicated that, compared to the normal group, the expression levels of plac8, CD14, and CD16 genes were increased in the sepsis model (P < 0.05). Western blot analysis (Fig. 4C, D) showed that, compared to the normal group, the protein expression level of plac8 was elevated in the sepsis model (P < 0.05), whereas the ERK protein content remained unchanged, but phospho-ERK increased (P < 0.05). ELISA results (Fig. 4B) revealed that, compared to the normal group, the protein levels of TNF-α, IL-6, IL-10, and MCP-1 were increased in the sepsis model (P < 0.05).
A qRT-PCR analysis of gene expression. B ELISA analysis of relevant factors in mouse serum. C Western blot analysis of protein band. D Western blot analysis of protein expression. E Results of cell proliferation assay using CCK-8 method. * indicates P < 0.05 compared to the sham group. Each group consisted of ten mice, and data is presented as mean ± standard deviation. Independent samples t-test was used to compare two groups, and two-way ANOVA was used for comparison at different time points.
The influence of Plac8 gene silencing on peripheral blood mononuclear cells in septicemia mice
In the experimental setup, septic mice were injected with Plac8-siRNA/Plac8 and the corresponding negative control virus, leading to the isolation of mononuclear cells. Flow cytometry analysis demonstrated a decrease in the proportion of CD14+ and CD16+ monocytes in the si-Plac8 group compared to the si-plac8-NC group (P < 0.05), whereas an increase was observed in the Plac8 group compared to the Plac8-NC group (P < 0.05) (Fig. 5A, B). Western blot analysis revealed a decrease in plac8 protein expression levels in the si-Plac8 group compared to the si-plac8-NC group (P < 0.05), accompanied by unchanged levels of ERK protein but decreased phosphorylation (P < 0.05). Conversely, an increase in plac8 protein expression levels along with unchanged levels of ERK protein but increased phosphorylation was observed in the Plac8 group compared to the Plac8-NC group (P < 0.05) (Fig. 5C, D). ELISA results demonstrated a decrease in the protein expression levels of TNF-α, IL-6, IL-10, and MCP-1 in the si-Plac8 group compared to the si-plac8-NC group (P < 0.05), as well as an increase in the Plac8 group compared to the Plac8-NC group (P < 0.05) (Fig. 5E). Flow cytometry analysis revealed that the si-Plac8 group exhibited an increase in the proportion of cells in the G0/G1 phase and a decrease in the proportion of cells in the S phase, resulting in a decreased PI with statistical significance (P < 0.05) compared to the si-plac8-NC group. On the other hand, the Plac8 group showed a decrease in the proportion of cells in the G0/G1 phase, an increase in the proportion of cells in the S phase, and an elevated PI, all statistically significant (P < 0.05) compared to the Plac8-NC group (Fig. 5F–H). These findings strongly suggest the impact of Plac8 on the survival and proliferation of mononuclear cells in the septic mouse model.
A Flow cytometry analysis of CD14+ and CD16+ cell subsets. B Flow cytometry analysis of peripheral blood CD14+ and CD16+ cell subsets. C Western blot analysis of protein band. D Western blot analysis of protein expression. E ELISA analysis of cytokine expression. F Detection of cell proliferation. G Flow cytometry analysis of cell cycle. H Cell proliferation index PI. * indicates P < 0.05 compared to si-Plac8-NC or Plac8-NC group. Data is presented as mean ± standard deviation. Independent samples t-test was used to compare two groups, and two-way ANOVA was used for comparison at different time points. The experiment was repeated three times.
Discussion
The regulation of Plac8 protein expression and its functions in various cell types has been a research focus [32, 33]. Historically, it has been found to play a role in tumor cell proliferation and differentiation, as well as in immune responses. In the inflammatory disease model of sepsis, Plac8 is involved in the activation and apoptosis of inflammatory cells.
Early research has shown that the ERK pathway plays a central role in cell signaling, particularly in regulating cell proliferation, differentiation, and survival [34,35,36]. The involvement of the ERK pathway has been widely recognized in monocyte activation and regulation [37, 38]. However, previous studies have not provided a detailed explanation of how Plac8 regulates the ERK pathway [22, 23, 39]. We observed that as the expression of Plac8 increased, the phosphorylation level of ERK protein also increased. It was verified in an in vitro model of LPS-stimulated monocytes and a cecal ligation and puncture (CLP)-induced septic mouse model. It suggests that Plac8 may play a role in sepsis through the signaling pathway mediated by ERK (Fig. 6).
The role and mechanism of Plac8-mediated ERK signaling pathway activation in the proliferation and activation of peripheral blood monocytes in septic patients.
CD14 and CD16, important markers for classifying monocyte subsets, have been extensively studied for their roles in inflammatory responses [40,41,42]. We observed that the upregulation of Plac8 is not only associated with overall monocyte proliferation activity but also related to changes in the expression of specific phenotypic markers. It may explain the different functions and pathophysiological roles of monocytes in sepsis.
In investigating the regulation of inflammatory responses, we also found that Plac8 may have a bidirectional regulatory function in pro-inflammatory and anti-inflammatory processes. Inflammation is a complex process involving multiple signaling pathways and cell types, and balancing pro-inflammatory and anti-inflammatory signals is crucial [43, 44]. However, further exploration is needed to understand how Plac8 maintains this balance in sepsis, which could have important implications for developing new treatment strategies [24].
In vitro models provide a simplified system for studying cell responses, but they may not fully replicate the complex environment in the human body [45,46,47]. Similarly, although animal models are indispensable in studying complex disease mechanisms, their results may not always directly extrapolate to humans due to inter-species differences [48, 49]. Future research needs to be conducted in a broader population to validate the role of Plac8 and evaluate its reliability as a biomarker in patients of different ethnicities, ages, and genders [50].
Activation of the ERK pathway significantly influences the proliferation and activation of monocytes by Plac8, providing a novel perspective for understanding the pathophysiology of sepsis. Furthermore, the regulatory role of Plac8 is meaningful for altering the phenotype of monocytes, aiding in uncovering the functional heterogeneity of monocytes in various inflammatory states. These findings expand our scientific understanding of the immune response in sepsis and offer new target molecules for designing therapeutic strategies aimed at modulating monocytes. Building upon the theoretical foundation of this study, clinical applications could explore interventions targeting Plac8 specific to PBMCs to provide effective solutions for precise sepsis treatment.
Limitations of this study primarily include a limited sample size and demographic diversity, potentially impacting the generalizability of the results. While the use of in vitro and animal models facilitates mechanistic exploration, these models do not fully replicate the complex physiological states and immune responses in humans. Moreover, this study has yet to elucidate the molecular mechanisms of Plac8 regulation or consider the interactions of other signaling pathways and their long-term effects. Additionally, uncontrolled variables such as individual differences and lifestyle habits may have influenced the results. Future research efforts should focus on validating the conclusions of this study through increasing sample sizes and conducting multi-center clinical trials. Further investigation into the molecular mechanisms, exploring the specific role of Plac8 in the ERK pathway and its interactions with other signaling pathways to uncover its comprehensive role in immune regulation, will be a key area of future research. Finally, translational medical research should apply these discoveries to the development of therapeutic strategies, ultimately achieving the goal of personalized medicine.
Materials and methods
Healthy and sepsis peripheral blood data
Collection of peripheral blood from healthy individuals: fresh peripheral venous blood was collected from 18 healthy voluntary blood donors in our hospital, including 10 males and 8 females, aged 28–64 years and an average age of 45.44 ± 12.34 years.
Collection of peripheral blood from sepsis patients: peripheral blood samples were obtained from 28 sepsis patients admitted to our hospital from January 2020 to December 2020. Among them, 15 were males and 13 were females, with ages ranging from 18 to 60 years and an average age of 38.18 ± 12.97 years. During the collection process, the purpose of the research was explained to the patients or their family members, and informed consent was obtained by having them sign a consent form. Adequate amounts of peripheral blood were drawn from each case.
Inclusion criteria were as follows: (1) patients with a confirmed diagnosis of sepsis; (2) clinical and pathological data available; (3) patients receiving their initial treatment and having not undergone any relevant therapy before. The severity of sepsis was evaluated using the SOFA scoring system. All experiments were conducted in accordance with the principles of the Helsinki Declaration.
Isolation and culture of blood monocytes
After collecting 1 mL of peripheral blood from patients or healthy individuals and mixing it thoroughly with 1 mL of physiological saline solution, the mixture was slowly added to a test tube containing 2 mL of Ficoll separation solution (F5415, Sigma). The tube was then centrifuged at 1800 rpm for 20 min at room temperature in a horizontal centrifuge (Centrifuge 5804, Eppendorf). Subsequently, the second layer of milky white lymphocytes was carefully transferred to another test tube using a pipette. An equal volume of physiological saline solution was added, and the tube was centrifuged at 1800 rpm for another 20 min using the same centrifuge. This process was repeated three times to isolate monocytes from peripheral blood samples of both healthy individuals and septic patients. Once isolated, the monocytes were suspended in RPMI1640 medium containing 15% FBS (11875119, ThermoFisher) and a 10 μL sample was taken for a trypan blue exclusion test to ensure a cell viability of over 95%. Subsequently, the cells were seeded into 35 mm culture dishes (430165, Corning) and incubated at 37 °C with 5% CO2 for 2 h. The culture medium was then aspirated, and the cells were washed five times with preheated 37 °C blank RPMI1640 medium to remove non-adherent cells. A small portion of adherent cells was collected for Wright staining to confirm them as monocytes, comprising 85% of the total cell population. The adherent cells were gently scraped off the culture dish using a cell scraper and transferred to RPMI1640 medium containing 10% FBS (A5669701, ThermoFisher) and 1% P/S (15070063, ThermoFisher), with the cell density adjusted to 1 × 106/ml, and then seeded into 24-well culture plates, 200 μl/well.
Flow cytometric analysis of MFI in CD14+ CD16+ monocytes
The mean fluorescence intensity (MFI) of CD14+ CD16+ monocytes was measured by flow cytometry. A total of 150 µL of EDTA anticoagulated peripheral whole blood was added to the bottoms of flow tubes, with one tube reserved as a blank control tube without fluorescence antibodies. The remaining tubes were each added with respective antibodies (CD14-PE (ab25390, Abcam, UK) and CD16-FITC (ab124042, Abcam, UK), 10 µL each), and then gently mixed by shaking and incubated at room temperature in the dark. Red blood cell lysis buffer was added to each tube, followed by cell washing. Each tube was then fixed with paraformaldehyde, and the MFI of CD14+ CD16+ was measured using a flow cytometer. Isotype controls were used concurrently to ensure antibody specificity.
LPS stimulation in an in vitro model
To establish an in vitro model of sepsis, PBMCs from healthy individuals were isolated and stimulated with 8 μg/ml of LPS from Sigma-Aldrich (Merck KGaA, Darmstadt, Germany) for 12 h [51].
Plasmid transfection
The steps for plasmid transfection were conducted as follows: DNA oligonucleotides were chemically synthesized by Shanghai Jikai Gene Chemistry Technology Co., Ltd. The synthesized DNA was then inserted into a plasmid containing the reporter gene cGFP and the neomycin resistance gene. The day before transfection, the culture medium was replaced with an antibiotic-free medium supplemented with 10% fetal bovine serum. When the cell density reached 70%–80%, transfection was performed according to the instructions provided with Lipofectamine 2000 (Invitrogen, USA, Cat. No. 11668019). The transfected cells were incubated for 4–6 h before replacing the medium with a serum-containing medium. Successful transfection was confirmed by the expression of green fluorescent protein within the cells. After 48 h of transfection, cells were observed using an inverted fluorescence microscope (CFM-500E/CFM-500Z, Shanghai Changfang Optical Instrument Co., Ltd., Shanghai, China) to assess transfection efficiency, and cells were collected for subsequent experiments. The experimental groups for cell transfection were as follows: (1) Plac8-NC group: transfected with the plasmid carrying a nonspecific sequence for Plac8; (2) Plac8 group: transfected with the plasmid overexpressing Plac8; (3) si-Plac8-NC group: transfected with the Plac8 siRNA nonspecific sequence; (4) si-Plac8 group: transfected with the Plac8 siRNA; (5) Plac8 + DMSO group: transfected with the plasmid overexpressing Plac8 and treated with the ERK pathway inhibitor solvent DMSO; (6) Plac8 + GDC-0994 group: transfected with the plasmid overexpressing Plac8 and treated with the ERK pathway inhibitor GDC-0994; (7) si-Plac8 + DMSO group: transfected with Plac8 siRNA and treated with the ERK pathway activator solvent DMSO; (8) si-Plac8 + EGF group: transfected with Plac8 siRNA and treated with the ERK pathway activator EGF ((H)Y-15947, MCE for 24 h; EGF treatment at 200 pg/mL; (H)Y-P7109, MCE) for cells.
Construction of sepsis mouse model
A total of 80 C57BL/6 laboratory mice were randomly chosen for the study. Ten mice were assigned to the sham operation group, while the remaining 70 mice were used to construct the CLP sepsis mouse model, with 10 mice in each group. The modeling process was conducted in a room at an ambient temperature of 22 °C. For the CLP model group, male C57BL/6 mice were selected and fasted for 8 h without water access. Anesthesia was induced by intraperitoneal injection of the prepared anesthetic agent. The mice were placed supine and fixed on an operating table. The abdominal area was disinfected with 75% ethanol. A midline incision of 2–3 cm in length was made below the xiphoid process along the linea alba to expose the abdominal area. Next, the cecum and the junction of the small and large intestines were located. After the cecum was identified, a circumferential ligation was performed at the distal part of the cecum using 3-0 silk thread, with the ligation width being two-thirds of the width of the ligation site. Then, the intestine was pierced twice with an 8-gauge needle, and the two needle holes were ~3 mm apart to extrude an appropriate amount of intestinal contents. The intestinal contents and cecum were replaced in situ, and the abdominal area was closed layer by layer, with the ligation site being sutured separately. Immediately after the surgery, a subcutaneous injection of 0.9% saline solution at a dose of 5 mL/100 g of body weight was administered to replenish fluid loss during the surgery. The sham operation group was subjected to exposure of intestinal contents without intestinal ligation or puncture.
After successfully constructing the CLP sepsis mouse model, 10 model mice were randomly selected as the CLP control group. Subsequently, an additional 60 mice were randomly divided into four groups, with 15 mice in each group. During the breeding process, each group of mice was injected with their respective lentiviral vectors (1 × 109 pfu/100 μL) via tail injection twice a week for 6 weeks. The mice in the four groups were injected with the following: si-Plac8-NC group (mice injected with si-Plac8-negative control lentiviral vector via tail vein), si-Plac8 group (mice injected with si-Plac8 lentiviral vector via tail vein), Plac8-NC group (mice injected with Plac8-negative control lentiviral vector via tail vein), Plac8 group (mice injected with Plac8 lentiviral vector via tail vein). After the last injection, blood samples were collected from the mice and stored for subsequent experiments 24 h post-injection. Sibiono GeneTech Co., Ltd. provided all lentiviral vectors.
ELISA
Blood was collected from the abdominal aorta of experimental mice. After 30 min of static incubation, the blood was centrifuged at 3500 rpm for 10 min. ELISA testing was performed to measure the serum levels of pro-inflammatory cytokines TNF-α, IL-6, anti-inflammatory cytokine IL-10, and monocyte chemoattractant protein-1 (MCP-1), following the instructions provided in the BMS623/2 kit (Bender, USA). The known antigens were diluted to a concentration of 1–10 μg/mL and added to each well at a volume of 0.1 mL, which was then kept at 4 °C overnight. The plates were washed three times the next day. Subsequently, a certain volume of the diluted supernatant was added to the coated wells and incubated at 37 °C for 1 h, followed by washing.
Meanwhile, blank, negative, and positive control wells were set up in the reaction wells, and 0.1 mL of freshly diluted enzyme-linked secondary antibody (Abcam) was added. The plates were then incubated at 37 °C for 35–60 min and washed. Finally, the plates were washed again with ddH2O provided by Beijing Dingguo Changsheng Biotechnology Co., Ltd. (Batch No. PER018-1). A total of 0.1 mL of TMB substrate solution (Batch No. EL0001 provided by Huzhou Yingchuang Biotechnology Co., Ltd.) was added to each reaction well and incubated at 37 °C for 10–30 min. The reaction was stopped by adding 50 μL of stop solution, and the absorbance (OD value) of each well was measured at a wavelength of 450 nm within 20 min.
qRT-PCR
The total RNA of each group of cells was extracted using the one-step Trizol method (15596026, Invitrogen, Car, Cal, USA). The purity and concentration of the RNA were assessed using a UV spectrophotometer (DU640; Backman, USA), and a purity ratio of A260/A280 between 1.8 and 2.0 was considered high. The RNA was then reverse transcribed into cDNA using the PrimeScript RT reagent Kit (RR047A, Takara, Japan) according to the manufacturer’s instructions. The reaction conditions were as follows: 37 °C for 15 min, 85 °C for 5 s, with a total volume of 20 μL. Subsequently, PCR reactions were conducted using the SYBR Premix EX Taq kit (RR420A, Takara) and a real-time fluorescence quantitative PCR instrument (ABI 7500, ABI; Foster City, CA, USA). The reaction mixture contained SYBR Mix 9 μL, upstream primer 0.5 μL, downstream primer 0.5 μL, cDNA 2 μL, and RNase Free dH2O 8 μL. The reaction conditions were as follows: 95 °C for 10 min, followed by 40 cycles of 95 °C for 15 s and 60 °C for 1 min. Each well was set up with 3 replicates. The primers were synthesized by Shanghai Jikai Gene Chemical Technology Co., Ltd. (primer sequences are listed in Table S1). The Ct values of each well were recorded, and the relative expression level of the target gene was calculated using the 2−ΔΔCt method with GAPDH as the internal reference [52]. ΔΔCt = (average Ct value of the target gene in the experimental group − average Ct value of the housekeeping gene in the experimental group) − (average Ct value of the target gene in the control group − average Ct value of the housekeeping gene in the control group).
Western blot
The cells were cultured until 80% confluency and then lysed on ice for 5 min using RIPA lysis buffer (P0013B, BiyunTian). The lysates were centrifuged at 14,000 rpm at 4 °C, and the supernatant was collected. The protein concentration of the samples was determined using the BCA method (23225, Pierce, USA). Electrophoresis and transfer were performed using 4% stacking gel and 10% resolving gel. After blocking with 0.5% BSA, the membranes were incubated overnight with primary antibodies: rabbit anti-CD14 (ab182032, 1:1000, Abcam, UK), CD16 (Cat No. 16559-1-AP, 1:1000, Proteintech), plac8 (Cat No. 12284-1-AP, 1:200, Proteintech), ERK1/2 (ab17942, 1:1000, Abcam, UK), p-ERK1/2 (ab223500, 1:1000, Abcam, UK), and GAPDH (ab8245, 1:1000, Abcam, UK). After washing, the membranes were incubated with HRP-conjugated goat anti-rabbit IgG (1:2000, ab6721, Abcam, UK) at room temperature for 2 h, followed by visualization using the ECL method (32209, Invitrogen, USA). Images were captured using the Bio-Rad imaging system and analyzed using Image Lab. The experiments above were repeated three times. All the Original WB gels are available at Supplementary files.
Flow cytometry for cell cycle detection
First, cells were transfected for 48 h and in the logarithmic growth phase, they were washed twice with PBS solution, and the supernatant was discarded. Then, 1 mL of pre-chilled 70% ethanol was added, and cells were fixed overnight at 4 °C. Subsequently, cells were washed twice with PBS solution and suspended in 500 μL of PBS. Propidium iodide (88378, Sigma, USA) and RNase A (GE101-01, Full Shine Biological Co., Ltd., Beijing, China) were added to the cell suspension to reach a final concentration of 50 μg/mL and incubated at 37 °C in a water bath for 30 min. Finally, flow cytometry with red fluorescence detection at an excitation wavelength of 488 nm was used to determine the cell cycle. The PI was calculated using the formula PI = (S + G2M) / (G0/1 + S + G2M).
CCK-8
The cells were collected and digested using 0.25% trypsin. After counting, they were seeded into a 96-well plate, with a density of 1 × 104 cells/well and 3 replicate wells per group. Each well was added with 10 µL of CCK-8 reagent (40203ES60, Shanghai Yisheng Biotechnology Co., Ltd., China). Subsequently, the absorbance values at 450 nm were measured using an automated microplate reader (MK3, Thermo, USA) at 0, 24, 48, and 72 h to determine cell viability. Cell viability was calculated using the formula (%) = [(OD treatment group) − (OD blank)] / [(OD control group) − (OD blank)] × 100%. The experiment was replicated three times.
Statistical analysis
All data were processed using the GraphPad prism statistical software. Following normality assessment (Shapiro–Wilk test) and homogeneity of variances test (Levene’s test), quantitative data conforming to a normal distribution were presented as mean ± standard deviation. Differences between the two groups were compared using the unpaired Student’s t-test, while one-way analysis of variance (ANOVA) was utilized for comparisons among multiple groups. Additionally, Tukey’s correction and other multiple comparison methods were employed to determine specific intergroup differences’ significance. In the case of non-normally distributed or heteroscedastic data, significant differences were evaluated using the Mann–Whitney U test or the Kruskal–Wallis H test. Cell viability at different time points was analyzed using two-way analysis of variance. A p value < 0.05 indicated statistically significant differences. All experiments were conducted independently at least three times.
Data availability
The data that support the findings of this study are available on request from the corresponding author.
References
Rudd KE, Johnson SC, Agesa KM, Shackelford KA, Tsoi D, Kievlan DR, et al. Global, regional, and national sepsis incidence and mortality, 1990-2017: analysis for the Global Burden of Disease Study. Lancet. 2020;395:200–11. https://doi.org/10.1016/S0140-6736(19)32989-7.
Liu D, Huang SY, Sun JH, Zhang HC, Cai QL, Gao C, et al. Sepsis-induced immunosuppression: mechanisms, diagnosis and current treatment options. Mil Med Res. 2022;9:56. https://doi.org/10.1186/s40779-022-00422-y.
van der Poll T, Shankar-Hari M, Wiersinga WJ. The immunology of sepsis. Immunity. 2021;54:2450–64. https://doi.org/10.1016/j.immuni.2021.10.012.
Purcarea A, Sovaila S. Sepsis, a 2020 review for the internist. Rom J Intern Med. 2020;58:129–37. https://doi.org/10.2478/rjim-2020-0012.
Gavelli F, Castello LM, Avanzi GC. Management of sepsis and septic shock in the emergency department. Intern Emerg Med. 2021;16:1649–61. https://doi.org/10.1007/s11739-021-02735-7.
Leise BS, Fugler LA. Laminitis updates: sepsis/systemic inflammatory response syndrome-associated laminitis. Vet Clin Equine Pract. 2021;37:639–56. https://doi.org/10.1016/j.cveq.2021.08.003.
Meyr AJ, Dougherty M, Kwaadu KY. An evaluation of patient characteristics associated with medical disposition in the surgical treatment of ankle fractures. J Foot Ankle Surg. 2022;61:72–78. https://doi.org/10.1053/j.jfas.2021.06.009.
Wang T, Zhang X, Liu Z, Yao T, Zheng D, Gan J, et al. Single-cell RNA sequencing reveals the sustained immune cell dysfunction in the pathogenesis of sepsis secondary to bacterial pneumonia. Genomics. 2021;113:1219–33. https://doi.org/10.1016/j.ygeno.2021.01.026.
Pickering H, Sen S, Arakawa-Hoyt J, Ishiyama K, Sun Y, Parmar R, et al. NK and CD8+ T cell phenotypes predict onset and control of CMV viremia after kidney transplant. JCI Insight. 2021;6:e153175. https://doi.org/10.1172/jci.insight.153175.
Wu T, Liang X, Jiang Y, Chen Q, Zhang H, Zhang S, et al. Comprehensive transcriptome profiling of peripheral blood mononuclear cells from patients with sepsis. Int J Med Sci. 2020;17:2077–86. https://doi.org/10.7150/ijms.46910.
Schenz J, Tamulyte S, Nusshag C, Brenner T, Poschet G, Weigand MA, et al. Population-specific metabolic alterations in professional antigen-presenting cells contribute to sepsis-associated immunosuppression. Shock. 2020;53:5–15. https://doi.org/10.1097/SHK.0000000000001337.
Washburn ML, Wang Z, Walton AH, Goedegebuure SP, Figueroa DJ, Van Horn S, et al. T cell- and monocyte-specific RNA-sequencing analysis in septic and nonseptic critically Ill patients and in patients with cancer. J Immunol. 2019;203:1897–908. https://doi.org/10.4049/jimmunol.1900560.
Chang JG, Tu SJ, Huang CM, Chen YC, Chiang HS, Lee YT, et al. Single-cell RNA sequencing of immune cells in patients with acute gout. Sci Rep. 2022;12:22130. https://doi.org/10.1038/s41598-022-25871-2.
Crawford LB, Diggins NL, Caposio P, Hancock MH. Advances in model systems for human cytomegalovirus latency and reactivation. mBio. 2022;13:e0172421. https://doi.org/10.1128/mbio.01724-21.
Hettema JM, van den Oord EJCG, Zhao M, et al. Methylome-wide association study of anxiety disorders. Mol Psychiatry. 2023;28:3484–92. https://doi.org/10.1038/s41380-023-02205-w.
Li LL, Dai B, Sun YH, Zhang TT. Monocytes undergo functional reprogramming to generate immunosuppression through HIF-1α signaling pathway in the late phase of sepsis. Mediators Inflamm. 2020;2020:4235909. https://doi.org/10.1155/2020/4235909.
Trzeciak A, Lerman YV, Kim TH, Kim MR, Mai N, Halterman MW, et al. Long-term microgliosis driven by acute systemic inflammation. J Immunol. 2019;203:2979–89. https://doi.org/10.4049/jimmunol.1900317.
Avendaño-Ortiz J, Redondo-Calvo FJ, Lozano-Rodríguez R, Terrón-Arcos V, Bergón-Gutiérrez M, Rodríguez-Jiménez C, et al. Thiosulfinate-enriched Allium sativum extract exhibits differential effects between healthy and sepsis patients: the implication of HIF-1α. Int J Mol Sci. 2023;24:6234. https://doi.org/10.3390/ijms24076234.
He F, Wu X, Zhang Q, Li Y, Ye Y, Li P, et al. Bacteriostatic potential of melatonin: therapeutic standing and mechanistic insights. Front Immunol. 2021;12:683879. https://doi.org/10.3389/fimmu.2021.683879.
Lee H, Kim JI, Park JS, Roh JI, Lee J, Kang BC, et al. CRISPR/Cas9-mediated generation of a Plac8 knockout mouse model. Lab Anim Res. 2018;34:279–87. https://doi.org/10.5625/lar.2018.34.4.279.
Pang J, Li H, Zhang X, Luo Z, Chen Y, Zhao H, et al. Application of novel transcription factor machine learning model and targeted drug combination therapy strategy in triple negative breast cancer. Int J Mol Sci. 2023;24:13497. https://doi.org/10.3390/ijms241713497.
Mao M, Chen Y, Yang J, Cheng Y, Xu L, Ji F, et al. Modification of PLAC8 by UFM1 affects tumorous proliferation and immune response by impacting PD-L1 levels in triple-negative breast cancer. J Immunother Cancer. 2022;10:e005668. https://doi.org/10.1136/jitc-2022-005668.
Kemp SB, Steele NG, Carpenter ES, Donahue KL, Bushnell GG, Morris AH, et al. Pancreatic cancer is marked by complement-high blood monocytes and tumor-associated macrophages. Life Sci Alliance. 2021;4:e202000935. https://doi.org/10.26508/lsa.202000935.
Gravrand V, Mellot F, Ackermann F, Ballester MC, Zuber B, Kirk JT, et al. Stratification of COVID-19 severity using SeptiCyte RAPID, a novel host immune response test. Viruses. 2023;15:419. https://doi.org/10.3390/v15020419.
Tsuboi H, Segawa S, Yagishita M, Toko H, Honda F, Kitada A, et al. Activation mechanisms of monocytes/macrophages in adult-onset still disease. Front Immunol. 2022;13:953730. https://doi.org/10.3389/fimmu.2022.953730.
Yang Q, Ni L, Imani S, Xiang Z, Hai R, Ding R, et al. Anlotinib suppresses colorectal cancer proliferation and angiogenesis via inhibition of AKT/ERK signaling cascade. Cancer Manag Res. 2020;12:4937–48. https://doi.org/10.2147/CMAR.S252181.
Javaid S, Schaefer A, Goodwin CM, Nguyen VV, Massey FL, Pierobon M, et al. Concurrent inhibition of ERK and farnesyltransferase suppresses the growth of HRAS mutant head and neck squamous cell carcinoma. Mol Cancer Ther. 2022;21:762–74. https://doi.org/10.1158/1535-7163.MCT-21-0142.
Ren M, Zhao Y, He Z, Lin J, Xu C, Liu F, et al. Baicalein inhibits inflammatory response and promotes osteogenic activity in periodontal ligament cells challenged with lipopolysaccharides. BMC Complement Med Ther. 2021;21:43. https://doi.org/10.1186/s12906-021-03213-5.
Zhao C, Zou T, Tang R, Zhu C. Placenta-specific 8 (PLAC8) mediates inflammation and mobility of the hPDLCs via MEK/ERK signaling pathway. Int Immunopharmacol. 2022;103:108459. https://doi.org/10.1016/j.intimp.2021.108459.
Mao M, Hu D, Yang J, Chen Y, Zhang X, Shen J, et al. Regulation of tamoxifen sensitivity by the PLAC8/MAPK pathway axis is antagonized by curcumin-induced protein stability change. J Mol Med. 2021;99:845–58. https://doi.org/10.1007/s00109-021-02047-5.
Zeng X, Liu Q, Yang Y, Jia W, Li S, He D, et al. Placenta-specific protein 8 promotes the proliferation of lung adenocarcinoma PC-9 cells and their tolerance to an epidermal growth factor receptor tyrosine kinase inhibitor by activating the ERK signaling pathway. Oncol Lett. 2019;18:5621–7. https://doi.org/10.3892/ol.2019.10911.
Li R, Li M, Chen L. [Knockdown of placental-specific protein 8 (PLAC8) inhibits proliferation and promotes apoptosis of human embryonic stem cells]. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2020;36:1089–94.
An H, Liu Y, Fang L, Shu M, Zhai Q, Chen J. Placenta-specific 8 facilitates the infection of duck hepatitis A virus type 1 by inhibiting the TLR7 MyD88-dependent signaling pathway. Poult Sci. 2023;102:102724. https://doi.org/10.1016/j.psj.2023.102724.
Yang L, Zhou R, Tong Y, Chen P, Shen Y, Miao S, et al. Neuroprotection by dihydrotestosterone in LPS-induced neuroinflammation. Neurobiol Dis. 2020;140:104814. https://doi.org/10.1016/j.nbd.2020.104814.
Tan H, Chen J, Li Y, Li Y, Zhong Y, Li G, et al. Glabridin, a bioactive component of licorice, ameliorates diabetic nephropathy by regulating ferroptosis and the VEGF/Akt/ERK pathways. Mol Med. 2022;28:58. https://doi.org/10.1186/s10020-022-00481-w.
de Los Reyes Corrales T, Losada-Pérez M, Casas-Tintó S. JNK Pathway in CNS Pathologies. Int J Mol Sci. 2021;22:3883. https://doi.org/10.3390/ijms22083883.
Tang L, Wu M, Lu S, Zhang H, Shen Y, Shen C, et al. Fgf9 negatively regulates bone mass by inhibiting osteogenesis and promoting osteoclastogenesis via MAPK and PI3K/AKT signaling. J Bone Miner Res. 2021;36:779–91. https://doi.org/10.1002/jbmr.4230.
Wu X, Wang A, Zhang S, Wang X, Guo D, Zhu W, et al. Multiomic landscape of immune pathogenesis in Kimura’s disease. iScience. 2023;26:106559. https://doi.org/10.1016/j.isci.2023.106559.
Ugalde AP, Bretones G, Rodríguez D, Quesada V, Llorente F, Fernández-Delgado R, et al. Autophagy-linked plasma and lysosomal membrane protein PLAC8 is a key host factor for SARS-CoV-2 entry into human cells. EMBO J. 2022;41:e110727. https://doi.org/10.15252/embj.2022110727.
Almubarak A, Tanagala KKK, Papapanou PN, Lalla E, Momen-Heravi F. Disruption of monocyte and macrophage homeostasis in periodontitis. Front Immunol. 2020;11:330. https://doi.org/10.3389/fimmu.2020.00330.
Ye C, Zhu J, Wang J, Chen D, Meng L, Zhan Y, et al. Single-cell and spatial transcriptomics reveal the fibrosis-related immune landscape of biliary atresia. Clin Transl Med. 2022;12:e1070. https://doi.org/10.1002/ctm2.1070.
Chi M, Tian Z, Ma K, Li Y, Wang L, Nasser MI, et al. The diseased kidney: aging and senescent immunology. Immun Ageing. 2022;19:58. https://doi.org/10.1186/s12979-022-00313-9.
Hadjadj J, Yatim N, Barnabei L, Corneau A, Boussier J, Smith N, et al. Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients. Science. 2020;369:718–24. https://doi.org/10.1126/science.abc6027.
Wang XL, Chen F, Shi H, Zhang M, Yan L, Pei XY, et al. Oxymatrine inhibits neuroinflammation byRegulating M1/M2 polarization in N9 microglia through the TLR4/NF-κB pathway. Int Immunopharmacol. 2021;100:108139. https://doi.org/10.1016/j.intimp.2021.108139.
Zheng L, Tang X, Lu M, Sun S, Xie S, Cai J, et al. microRNA-421-3p prevents inflammatory response in cerebral ischemia/reperfusion injury through targeting m6A Reader YTHDF1 to inhibit p65 mRNA translation. Int Immunopharmacol. 2020;88:106937. https://doi.org/10.1016/j.intimp.2020.106937.
Long Y, Wang L, Li Z. SP1-induced SNHG14 aggravates hypertrophic response in in vitro model of cardiac hypertrophy via up-regulation of PCDH17. J Cell Mol Med. 2020;24:7115–26. https://doi.org/10.1111/jcmm.15073.
Persich P, Engels GE, van Oeveren W, Galia E, Benay S, Thun S. Development of an in vitro system and model-based translational framework to assess haemolysis risk following intravenous abuse of medications containing polyethylene oxide. Toxicol Vitro. 2020;65:104776. https://doi.org/10.1016/j.tiv.2020.104776.
Tasnim F, Huang X, Lee CZW, Ginhoux F, Yu H. Recent advances in models of immune-mediated drug-induced liver injury. Front Toxicol. 2021;3:605392. https://doi.org/10.3389/ftox.2021.605392.
Du K, Li S, Li C, Li P, Miao C, Luo T, et al. Modeling nonalcoholic fatty liver disease on a liver lobule chip with dual blood supply. Acta Biomater. 2021;134:228–39. https://doi.org/10.1016/j.actbio.2021.07.013.
Huang ML, Zou Y, Yang R, Jiang Y, Sheng JF, Han JB, et al. Placenta specific 8 gene induces epithelial-mesenchymal transition of nasopharyngeal carcinoma cells via the TGF-β/Smad pathway. Exp Cell Res. 2019;374:172–80. https://doi.org/10.1016/j.yexcr.2018.11.021.
Sranujit RP, Noysang C, Tippayawat P, Kooltheat N, Luetragoon T, Usuwanthim K. Phytochemicals and immunomodulatory effect of Nelumbo nucifera flower extracts on human macrophages. Plants. 2021;10:2007. https://doi.org/10.3390/plants10102007.
Song R, He S, Wu Y, Chen W, Song J, Zhu Y, et al. Validation of reference genes for the normalization of the RT-qPCR in peripheral blood mononuclear cells of septic patients. Heliyon. 2023;9:e15269. https://doi.org/10.1016/j.heliyon.2023.e15269.
Funding
This work was supported by Tianjin Municipal Health Commission (RC20145 to TZ).
Author information
Authors and Affiliations
Contributions
Teng Zhang contributed to conceptualization, data curation, methodology, formal analysis, and the initial drafting of the manuscript. Jing-nan Fu and Xiu Zhang participated in data curation, investigation, visualization, and contributed to reviewing and editing the manuscript. Gui-bing Chen provided supervision, managed project administration, secured funding, and contributed to reviewing and editing the manuscript. All authors actively contributed to the study’s design and execution, provided critical feedback during manuscript preparation, and gave their final approval for publication.
Corresponding author
Ethics declarations
Competing interests
The author declare no competing interests.
Ethical approval
All procedures involving the use of animals in research were authorized by the ethics committee of Tianjin Medical University General Hospital (approval no. IRB2022-DWFL-44).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, T., Fu, Jn., Chen, Gb. et al. Plac8-ERK pathway modulation of monocyte function in sepsis. Cell Death Discov. 10, 308 (2024). https://doi.org/10.1038/s41420-024-02012-4
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41420-024-02012-4
This article is cited by
-
Docosahexaenoic acid supplementation inhibits monocyte exhaustion memory formation during sepsis
Inflammation Research (2026)
-
RNASEH2C enhances TRAF3IP1 to degrade RAI14 in lysosomes thus hindering macrophage antigen presentation and advancing liver cancer
Cell Death & Disease (2025)
-
Single cell RNA-seq reveals cellular and transcriptional heterogeneity in the splenic CD11b+Ly6Chigh monocyte population expanded in sepsis-surviving mice
Molecular Medicine (2024)