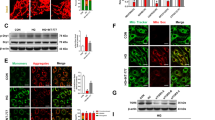
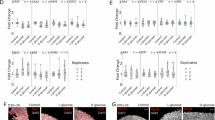
Abstract
This study elucidated the mechanistic role of the Suppressor of Cytokine Signaling 5 (SOCS5) in diabetic retinopathy (DR), focusing on DNA damage and cellular senescence pathways. Utilizing both in vitro (high glucose (HG)-induced human retinal microvascular endothelial cells (HRMECs)) and in vivo (streptozotocin-induced DR mouse models) approaches, we demonstrated that SOCS5 was significantly upregulated in DR. SOCS5 knockdown mitigated retinal tissue damage, vascular leakage, and apoptosis in DR mice while reducing DNA damage and cellular senescence in HG-stimulated HRMECs. Mechanistically, SOCS5 promoted DR progression by regulating the expression of Cyclin-Dependent Kinase Inhibitor 1 A (CDKN1A), a key mediator of cell cycle arrest and senescence. Furthermore, we identified POU Class 2 Homeobox 1 (POU2F1) as an upstream transcriptional activator of SOCS5, forming a novel POU2F1-SOCS5-CDKN1A axis that drove DR pathogenesis. Inhibition of POU2F1 and SOCS5 ameliorated DR-related pathology in mice, suggesting a novel therapeutic strategy. These findings reveal a previously unrecognized signaling pathway in DR and highlight SOCS5 as a promising target for intervention.
Similar content being viewed by others
Data availability
All data related to the article can be obtained from the corresponding author upon reasonable request.
References
Monzo L, Kobayashi M, Ferreira JP, Lamiral Z, Delles C, Clark AL, et al. Echocardiographic and biomarker characteristics in diabetes, coronary artery disease or both: insights from the HOMAGE trial. Cardiovasc Diabetol. 2025;24:111.
Mouzaka AE, Chandrinos A, Chatziralli I, Chatzichristou E, Gialelis TK. Eye axial length: is there a protective link to diabetic retinopathy? Cureus 2024;16:e75712.
D’Angelo A, Lixi F, Vitiello L, Gagliardi V, Pellegrino A, Giannaccare G, et al. The role of diet and oral supplementation for the management of diabetic retinopathy and diabetic macular edema: a narrative review. BioMed Res Int. 2025;2025:6654976.
Kamath S, Charlotte A, Nandan N. Silicone Oil droplets in the vitreous after an anti-vascular endothelial growth factor (VEGF) injection: a complication of syringe lubrication. Cureus 2025;17:e78715.
Godani K, Vidyasagar S, Biradar P, Prabhu V, Hande P, Gandhi P, et al. Coats-like reaction post-vitreoretinal surgery for PDR managed with laser photocoagulation and adjunctive intravitreal steroids– a case report. BMC Ophthalmol. 2025;25:32.
Ahamad Khan MM, Ganguly A, Barman S, Das C, Ganesan SK. Unveiling ferroptosis genes and inhibitors in diabetic retinopathy through single-cell analysis and docking simulations. Biochem Biophys Rep. 2025;41:101932.
Taylor OB, DeGroff N, El-Hodiri HM, Gao C, Fischer AJ. Sphingosine-1-phosphate signaling regulates the ability of Müller glia to become neurogenic, proliferating progenitor-like cells. eLife. 2025;13:RP102151.
Flindris K, Lagkada V, Christodoulou A, Gazouli M, Moschos M, Markozannes G, et al. Investigation of UCHL3 and HNMT gene polymorphisms in Greek patients with type 2 diabetes mellitus and diabetic retinopathy. Biomedicines. 2025;13:341.
Zhu S, Zhang M, Qu Z, Xu S, Peng J, Jiang F. Moscatilin alleviates oxidative stress and inflammatory response of Müller cells in diabetic retinopathy through suppressing the p38 mitogen-activated protein kinase/c-Jun N-terminal kinase and nuclear factor kappa-B signaling pathways. J Cell Commun Signal. 2025;19:e12059.
Lei X, Wang X, Zhang X. YY2 mediates transcriptional repression of PHGDH and expedites oxidative stress in retinal pigment epithelial cells in diabetic retinopathy. J Diabetes Investig. 2025;16:775–90.
Zhang M, Liu S, Chua M-S, Li H, Luo D, Wang S, et al. SOCS5 inhibition induces autophagy to impair metastasis in hepatocellular carcinoma cells via the PI3K/Akt/mTOR pathway. Cell Death Dis. 2019;10:612.
Luo D, Wang Y, Zhang M, Li H, Zhao D, Li H, et al. SOCS5 knockdown suppresses metastasis of hepatocellular carcinoma by ameliorating HIF-1α-dependent mitochondrial damage. Cell Death Dis. 2022;13:918.
Patnaik BB, Kim BB, Jo YH, Bang IS. Molecular cloning and expression analysis of three suppressors of cytokine signaling genes (SOCS5, SOCS6, SOCS7) in the mealworm beetle Tenebrio molitor. Insects. 2019;10:76.
Hernández C, Bogdanov P, Gómez-Guerrero C, Sampedro J, Solà-Adell C, Espejo C et al. SOCS1-derived peptide administered by eye drops prevents retinal neuroinflammation and vascular leakage in experimental diabetes. Int J Mol Sci. 2019;20:3615.
Yang P-W, Chang Y-H, Wong L-F, Lin C-C, Huang P-M, Hsieh M-S, et al. The genetic effect and molecular function of the SOCS5 in the prognosis of esophageal squamous cell carcinoma. J Cancer. 2021;12:2216–29.
Rong E, Dry I, Dalziel RG, Tan WS. Bovine transcription factor POU class 2 homeobox 1 (POU2F1/Oct1) protein promotes BoHV-1 replication in MDBK cells. Viruses. 2024;16:1549.
Zhu Y, Li S, Wang H, Ren W, Chi K, Wu J, et al. Molecular subtypes, predictive markers and prognosis in small-cell lung carcinoma. J Clin Pathol. 2023;78:42–50.
Lyssenko V, Vaag A. Genetics of diabetes-associated microvascular complications. Diabetologia. 2023;66:1601–13.
Voukeng I, Chen J, Lafontaine DLJ. The natural alkaloid nitidine chloride targets RNA polymerase I to inhibit ribosome biogenesis and repress cancer cell growth. Cell Death Discov. 2025;11:116.
Ishibashi M, Shibata K, Terasaki M, Saito Y, Yamagishi S-i, Hasumi K, et al. SMTP-44D alleviates diabetic retinopathy by suppressing inflammation and oxidative stress in in vivo and in vitro models. J Pharmacol Sci. 2025;157:57–64.
Chondrozoumakis G, Chatzimichail E, Habra O, Vounotrypidis E, Papanas N, Gatzioufas Z, et al. Retinal biomarkers in diabetic retinopathy: from early detection to personalized treatment. J Clin Med. 2025;14:1343.
Sato N, Tamada Y, Yu G, Okuno Y. CBNplot: Bayesian network plots for enrichment analysis. Bioinformatics. 2022;38:2959–60.
Yang Q, Zhang Y, Zhang X, Li X. Prevalence of diabetic retinopathy, proliferative diabetic retinopathy and non-proliferative diabetic retinopathy in Asian T2DM patients: a systematic review and Meta-analysis. Int J Ophthalmol. 2019;12:302–11.
Maroufizadeh S, Almasi-Hashiani A, Hosseini M, Sepidarkish M, Omani Samani R. Prevalence of diabetic retinopathy in Iran: a systematic review and meta-analysis. Int J Ophthalmol. 2017;10:782–9.
Leroith D, Nissley P. Knock your SOCS off! J Clin Investig. 2005;115:233–6.
Herrera-Uribe J, Convery O, Weinberg DAL, Stevenson FI. NJ: the neglected suppressor of cytokine signalling (SOCS): SOCS4-7. Inflammation. 2025;48:1607–23.
Brosius FC 3rd, Banes-Berceli A. A new pair of SOCS for diabetic nephropathy. J Am Soc Nephrol. 2010;21:723–4.
Özmen Ö, Şahi NM, Topsakal Ş, Taşan Ş, Şahi NU. Lycopene ameliorates diabetes-induced pancreatic, hepatic, and renal damage by modulating the JAK/STAT/SOCS signaling pathway in rats. Iran J Basic Med Sci. 2025;28:461–8.
Suchy D, Łabuzek K, Machnik G, Kozłowski M, Okopień B. SOCS and diabetes-ups and downs of a turbulent relationship. Cell Biochem Funct. 2013;31:181–95.
Thomas J, Garg ML, Smith DW. Dietary resveratrol supplementation normalizes gene expression in the hippocampus of streptozotocin-induced diabetic C57Bl/6 mice. J Nutr Biochem. 2014;25:313–8.
Tsai YC, Kuo PL, Hung WW, Wu LY, Wu PH, Chang WA, et al. Angpt2 induces mesangial cell apoptosis through the MicroRNA-33-5p-SOCS5 loop in diabetic nephropathy. Mol Ther Nucleic Acids. 2018;13:543–55.
Dutto I, Tillhon M, Cazzalini O, Stivala LA, Prosperi E. Biology of the cell cycle inhibitor p21(CDKN1A): molecular mechanisms and relevance in chemical toxicology. Arch Toxicol. 2015;89:155–78.
Elmitwalli O, Darwish R, Al-Jabery L, Algahiny A, Roy S, Butler AE, et al. The emerging role of p21 in diabetes and related metabolic disorders. Int J Mol Sci. 2024;25:13209.
Tesch GH, Ma FY, Ozols E, Nikolic-Paterson DJ. Intervention treatment reducing cellular senescence inhibits tubulointerstitial fibrosis in diabetic mice following acute kidney injury. Clin Sci. 2024;138:309–26.
Yin Q, Yang G, Su R, Bu J, Li Y, Zhang H, et al. Zi Shen Wan Fang repaired blood-brain barrier integrity in diabetic cognitive impairment mice by preventing cerebrovascular cells' senescence. Chin Med. 2024;19:169.
Al-Dabet MM, Shahzad K, Elwakiel A, Sulaj A, Kopf S, Bock F, et al. Reversal of the renal hyperglycemic memory in diabetic kidney disease by targeting sustained tubular p21 expression. Nat Commun. 2022;13:5062.
Ng MC, Lam VK, Tam CH, Chan AW, So WY, Ma RC, et al. Association of the POU class 2 homeobox 1 gene (POU2F1) with susceptibility to Type 2 diabetes in Chinese populations. Diabet Med. 2010;27:1443–49.
Batool A, Jahan N, Sun Y, Hanif A, Xue H. Genetic association of IDE, POU2F1, PON1, IL1α and IL1β with type 2 diabetes in the Pakistani population. Mol Biol Rep. 2014;41:3063–69.
Xia L, Lin J, Peng M, Jiang X, Peng Q, Cui S, et al. Diallyl disulfide induces DNA damage and growth inhibition in colorectal cancer cells by promoting POU2F1 ubiquitination. Int J Biol Sci. 2024;20:1125–41.
Schulze AB, Wenge DV, Evers G, Heitkötter B, Bleckmann A, Schmidt LH, et al. High expression of transcription factor POU2F1 confers improved survival on smokers with lung adenocarcinoma: a retrospective study of two cohorts. Transl Lung Cancer Res. 2023;12:727–41.
de Lemos L, Antas P, Ferreira IS, Santos IP, Felgueiras B, Gomes CM, et al. Modelling neurodegeneration and inflammation in early diabetic retinopathy using 3D human retinal organoids. Vitr Model. 2024;3:33–48.
Móvio MI, de Lima-Vasconcellos TH, Dos Santos GB, Echeverry MB, Colombo E, Mattos LS, et al. Retinal organoids from human-induced pluripotent stem cells: from studying retinal dystrophies to early diagnosis of Alzheimer’s and Parkinson’s disease. Semin Cell Dev Biol. 2023;144:77–86.
Funding
This work was supported by Yunnan Fundamental Research Projects (grant NO. 202301AT070092), Priority Union Foundation of Yunnan Provincial Science and Technology Department and Kunming Medical University (grant NO. 202201AY070001-077), Yunnan Provincial Youth Talent Research (grant NO. RLQB20220002), 535 Talent Project of First Affiliated Hospital of Kunming Medical University (grant NO. 2023535D19) the First-Class Discipline Team of Kunming Medical University (grant NO. 2024XKTDTS01) and Key Clinical Specialty of Ophthalmology in Yunnan Province (grant NO. 300070).
Author information
Authors and Affiliations
Contributions
Di Yang: Conceptualization, Methodology, Writing—review and editing. Siduo Lu: Data curation, Formal analysis, Writing—original draft. Hongmei Liu: Investigation, Formal analysis. You Zhou: Investigation, Data curation. Hua Zhong: Conceptualization, Methodology, Writing - review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, D., Lu, S., Liu, H. et al. Mechanistic insights into SOCS5-related DNA damage and cellular senescence in diabetic retinopathy. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-03011-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-03011-3