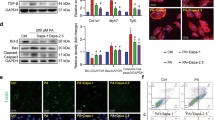
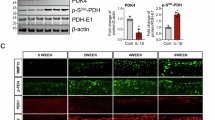
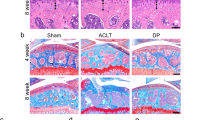
Abstract
Osteoarthritis (OA) is a degenerative joint disease characterized by progressive cartilage degradation and a complex pathogenesis. Degenerated chondrocytes exhibit an imbalance between catabolism and anabolism, leading to cartilage matrix loss. Currently, there are no effective clinical therapies to halt or reverse this degeneration. This study investigated the therapeutic potential of Dapagliflozin (DAPA) for OA. We demonstrated that DAPA exerts protective effects on cartilage explants from patients with OA as well as in surgically induced OA models in mice. In vitro studies revealed that DAPA ameliorates OA by restoring chondrocyte metabolic homeostasis. Transcriptome sequencing showed that DAPA activated the AMP-activated protein kinase (AMPK) signaling pathway while suppressing MAPK signaling. Mechanistically, AMPKα was identified as a novel target of DAPA. DAPA alleviated excessive catabolism by targeting both AMPKα and SGLT2, while promoting anabolic processes through AMPKα activation. Furthermore, DAPA rescued impaired autophagy caused by SGLT2 upregulation in degenerated chondrocytes. Our findings demonstrated that DAPA regulates cartilage metabolism by concurrently modulating AMPKα and SGLT2, underscoring the therapeutic promise of combined AMPK activation and SGLT2 inhibition in OA treatment.

Mechanism of DAPA in treating osteoarthritis. Created with BioRender.com.
Similar content being viewed by others
Data availability
Data are available from the corresponding author upon reasonable request.
References
Kloppenburg M, Namane M, Cicuttini F. Osteoarthritis. Lancet. 2025;405:71–85.
Tang S, Zhang C, Oo WM, Fu K, Risberg MA, Bierma-Zeinstra SM, et al. Osteoarthritis. Nat Rev Dis Prim. 2025;11:10.
Fu W, Hettinghouse A, Chen Y, Hu W, Ding X, Chen M, et al. 14-3-3 epsilon is an intracellular component of TNFR2 receptor complex and its activation protects against osteoarthritis. Ann Rheum Dis. 2021;80:1615–27.
Wang KD, Ding X, Jiang N, Zeng C, Wu J, Cai XY, et al. Digoxin targets low density lipoprotein receptor-related protein 4 and protects against osteoarthritis. Ann Rheum Dis. 2022;81:544–55.
Courties A, Kouki I, Soliman N, Mathieu S, Sellam J. Osteoarthritis year in review 2024: epidemiology and therapy. Osteoarthr Cartil. 2024;32:1397–404.
Cho Y, Jeong S, Kim H, Kang D, Lee J, Kang SB, et al. Disease-modifying therapeutic strategies in osteoarthritis: current status and future directions. Exp Mol Med. 2021;53:1689–96.
Chen J, Williams S, Ho S, Loraine H, Hagan D, Whaley JM, et al. Quantitative PCR tissue expression profiling of the human SGLT2 gene and related family members. Diab Ther. 2010;1:57–92.
Marfella R, Scisciola L, D’Onofrio N, Maiello C, Trotta MC, Sardu C, et al. Sodium-glucose cotransporter-2 (SGLT2) expression in diabetic and non-diabetic failing human cardiomyocytes. Pharm Res. 2022;184:106448.
Cowie MR, Fisher M. SGLT2 inhibitors: mechanisms of cardiovascular benefit beyond glycaemic control. Nat Rev Cardiol. 2020;17:761–72.
Vallon V, Verma S. Effects of SGLT2 inhibitors on kidney and cardiovascular function. Annu Rev Physiol. 2021;83:503–28.
Katsuumi G, Shimizu I, Suda M, Yoshida Y, Furihata T, Joki Y, et al. SGLT2 inhibition eliminates senescent cells and alleviates pathological aging. Nat Aging. 2024;4:926–38.
Zhao XY, Li SS, He YX, Yan LJ, Lv F, Liang QM, et al. SGLT2 inhibitors alleviated podocyte damage in lupus nephritis by decreasing inflammation and enhancing autophagy. Ann Rheum Dis. 2023;82:1328–40.
Mashayekhi M, Safa BI, Gonzalez MSC, Kim SF, Echouffo-Tcheugui JB. Systemic and organ-specific anti-inflammatory effects of sodium-glucose cotransporter-2 inhibitors. Trends Endocrinol Metab. 2024;35:425–38.
Liu W, Guo NY, Wang JQ, Xu BB. Osteoarthritis: mechanisms and therapeutic advances. MedComm (2020). 2025;6:e70290.
Safaie N, Masoumi S, Alizadeh S, Mirzajanzadeh P, Nejabati HR, Hajiabbasi M, et al. SGLT2 inhibitors and AMPK: the road to cellular housekeeping? Cell Biochem Funct. 2024;42:e3922.
Kogot-Levin A, Riahi Y, Abramovich I, Mosenzon O, Agranovich B, Kadosh L, et al. Mapping the metabolic reprogramming induced by sodium-glucose cotransporter 2 inhibition. JCI Insight. 2023;8:e164296.
Herzig S, Shaw RJ. AMPK: guardian of metabolism and mitochondrial homeostasis. Nat Rev Mol Cell Biol. 2018;19:121–35.
Yao Q, Wu X, Tao C, Gong W, Chen M, Qu M, et al. Osteoarthritis: pathogenic signaling pathways and therapeutic targets. Signal Transduct Target Ther. 2023;8:56.
Zheng L, Zhang Z, Sheng P, Mobasheri A. The role of metabolism in chondrocyte dysfunction and the progression of osteoarthritis. Ageing Res Rev. 2021;66:101249.
Liu Z, Huang J, Wang X, Deng S, Zhou J, Gong Z, et al. Dapagliflozin suppress endoplasmic reticulum stress mediated apoptosis of chondrocytes by activating Sirt1. Chem Biol Interact. 2023;384:110724.
Wang T, He C. Pro-inflammatory cytokines: the link between obesity and osteoarthritis. Cytokine Growth Factor Rev. 2018;44:38–50.
Hu W, Chen Y, Dou C, Dong S. Microenvironment in subchondral bone: predominant regulator for the treatment of osteoarthritis. Ann Rheum Dis. 2021;80:413–22.
Zhang H, Wang L, Cui J, Wang S, Han Y, Shao H, et al. Maintaining hypoxia environment of subchondral bone alleviates osteoarthritis progression. Sci Adv. 2023;9:eabo7868.
Liang W, Feng R, Li X, Duan X, Feng S, Chen J, et al. A RANKL-UCHL1-sCD13 negative feedback loop limits osteoclastogenesis in subchondral bone to prevent osteoarthritis progression. Nat Commun. 2024;15:8792.
Li J, Zhang B, Liu WX, Lu K, Pan H, Wang T, et al. Metformin limits osteoarthritis development and progression through activation of AMPK signalling. Ann Rheum Dis. 2020;79:635–45.
Petursson F, Husa M, June R, Lotz M, Terkeltaub R, Liu-Bryan R. Linked decreases in liver kinase B1 and AMP-activated protein kinase activity modulate matrix catabolic responses to biomechanical injury in chondrocytes. Arthritis Res Ther. 2013;15:R77.
Schonberger E, Mihaljevic V, Steiner K, Saric S, Kurevija T, Majnaric LT, et al. Immunomodulatory effects of SGLT2 inhibitors-targeting inflammation and oxidative stress in aging. Int J Environ Res Public Health. 2023;20:6671.
Lomenick B, Hao R, Jonai N, Chin RM, Aghajan M, Warburton S, et al. Target identification using drug affinity responsive target stability (DARTS). Proc Natl Acad Sci USA. 2009;106:21984–9.
Hasan S, Ghani N, Zhao X, Good J, Huang A, Wrona HL, et al. Dietary pyruvate targets cytosolic phospholipase A2 to mitigate inflammation and obesity in mice. Protein Cell. 2024;15:661–85.
Li L, Li J, Li JJ, Zhou H, Zhu XW, Zhang PH, et al. Chondrocyte autophagy mechanism and therapeutic prospects in osteoarthritis. Front Cell Dev Biol. 2024;12:1472613.
Fu W, Vasylyev D, Bi Y, Zhang M, Sun G, Khleborodova A, et al. Na(v)1.7 as a chondrocyte regulator and therapeutic target for osteoarthritis. Nature. 2024;625:557–65.
Dhillon S. Dapagliflozin: a review in type 2 diabetes. Drugs. 2019;79:1135–46.
Wu Q, Yao Q, Hu T, Yu J, Jiang K, Wan Y, et al. Dapagliflozin protects against chronic heart failure in mice by inhibiting macrophage-mediated inflammation, independent of SGLT2. Cell Rep Med. 2023;4:101334.
Xu J, Kitada M, Ogura Y, Liu H, Koya D Dapagliflozin restores impaired autophagy and suppresses inflammation in high glucose-treated HK-2 cells. Cells. 2021;10:1457.
Tsai KL, Hsieh PL, Chou WC, Cheng HC, Huang YT, Chan SH. Dapagliflozin attenuates hypoxia/reoxygenation-caused cardiac dysfunction and oxidative damage through modulation of AMPK. Cell Biosci. 2021;11:44.
Alsereidi FR, Khashim Z, Marzook H, Al-Rawi AM, Salomon T, Almansoori MK, et al. Dapagliflozin mitigates cellular stress and inflammation through PI3K/AKT pathway modulation in cardiomyocytes, aortic endothelial cells, and stem cell-derived beta cells. Cardiovasc Diabetol. 2024;23:388.
Dutka M, Bobinski R, Francuz T, Garczorz W, Zimmer K, Ilczak T, et al. SGLT-2 inhibitors in cancer treatment-mechanisms of action and emerging new perspectives. Cancers. 2022;14:5811.
Steinberg GR, Hardie DG. New insights into activation and function of the AMPK. Nat Rev Mol Cell Biol. 2023;24:255–72.
Trefts E, Shaw RJ. AMPK: restoring metabolic homeostasis over space and time. Mol Cell. 2021;81:3677–90.
Lin SC, Hardie DG. AMPK: sensing glucose as well as cellular energy status. Cell Metab. 2018;27:299–313.
Chen Y, Liu Y, Jiang K, Wen Z, Cao X, Wu S. Linear ubiquitination of LKB1 activates AMPK pathway to inhibit NLRP3 inflammasome response and reduce chondrocyte pyroptosis in osteoarthritis. J Orthop Transl. 2023;39:1–11.
Li J, Wang Y, Chen D, Liu-Bryan R. Oral administration of berberine limits post-traumatic osteoarthritis development and associated pain via AMP-activated protein kinase (AMPK) in mice. Osteoarthr Cartil. 2022;30:160–71.
Terkeltaub R, Yang B, Lotz M, Liu-Bryan R. Chondrocyte AMP-activated protein kinase activity suppresses matrix degradation responses to proinflammatory cytokines interleukin-1beta and tumor necrosis factor alpha. Arthritis Rheum. 2011;63:1928–37.
Lee Y, Kim HE, Kwak JS, Park CS, Chun JS. The cereblon-AMPK (AMP-activated protein kinase) axis in chondrocytes regulates the pathogenesis of osteoarthritis. Osteoarthr Cartil. 2024;32:1579–90.
Wan Y, Shen K, Yu H, Fan W. Baicalein limits osteoarthritis development by inhibiting chondrocyte ferroptosis. Free Radic Biol Med. 2023;196:108–20.
Gong Y, Li S, Wu J, Zhang T, Fang S, Feng D, et al. Autophagy in the pathogenesis and therapeutic potential of post-traumatic osteoarthritis. Burns Trauma. 2023;11:tkac060.
Yang L, Liang B, Li J, Zhang X, Chen H, Sun J, et al. Dapagliflozin alleviates advanced glycation end product induced podocyte injury through AMPK/mTOR mediated autophagy pathway. Cell Signal. 2022;90:110206.
Park JM, Lee DH, Kim DH. Redefining the role of AMPK in autophagy and the energy stress response. Nat Commun. 2023;14:2994.
Feng Y, Chen Y, Wu X, Chen J, Zhou Q, Liu B, et al. Interplay of energy metabolism and autophagy. Autophagy. 2024;20:4–14.
Lazzaroni E, Ben Nasr M, Loretelli C, Pastore I, Plebani L, Lunati ME, et al. Anti-diabetic drugs and weight loss in patients with type 2 diabetes. Pharm Res. 2021;171:105782.
Geng N, Fan M, Kuang B, Zhang F, Xian M, Deng L, et al. 10-hydroxy-2-decenoic acid prevents osteoarthritis by targeting aspartyl beta hydroxylase and inhibiting chondrocyte senescence in male mice preclinically. Nat Commun. 2024;15:7712.
Glasson SS, Blanchet TJ, Morris EA. The surgical destabilization of the medial meniscus (DMM) model of osteoarthritis in the 129/SvEv mouse. Osteoarthr Cartil. 2007;15:1061–9.
Acknowledgements
We thank the Translational Medicine Core Facility of Shandong University for consultation and instrument availability that supported this work. We also extend our sincere thanks to You Zuo (University of Groningen, The Netherlands), as well as Xuehui Zheng, Anda Shi, and Yuan Zhang (Shandong University) for their guidance in statistical analysis and computational work.
Funding
This work was supported partly by the Natural Science Foundation of Shandong Province (ZR2023MH006).
Author information
Authors and Affiliations
Contributions
Conceptualization: KL; Methodology: KL and ZL; Investigation: KL and ZL; Formal analysis: CW and HS; Visualization: CW and HS; Supervision: JZ and MS; Writing—original draft: KL; Writing—review & editing: JZ and MS; Funding acquisition: MS.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008 [5]. Informed consent was obtained from all patients for being included in the study. All Institutional and National Guidelines for the care and use of animals (fisheries) were followed. Patients involved in the study provided consent and all experiments involving animals were conducted according to the ethical policies and procedures approved by the Medical Ethical Committee of Qilu Hospital of Shandong University [Approval no. KYLL-2023(ZM)-182].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, K., Li, Z., Wang, C. et al. Dapagliflozin regulates chondrocyte homeostasis and protects against osteoarthritis via targets AMPKα and SGLT2. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-03016-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-03016-y