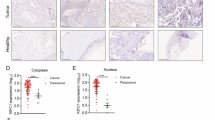
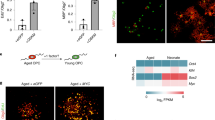
Abstract
Oncogene-induced senescence (OIS) is regarded a tumor suppressive mechanism in normal cells. Accumulated evidences, however, demonstrate that OIS would play a role in cancer promotion through the secretion of senescence associated secretory phenotypes (SASP). The underlying mechanisms remain to be addressed. In this study, we found that c-Myc oncogene could induce senescence in human diploid lung fibroblasts and non-small cell lung cancer cells (NSCLC) without concomitant emergence of apoptosis. c-Myc-induced senescence (cMIS) caused morphological enlargement, increased F-actin and nuclear G-actin that generally detected in senescent cells. These events were found to be associated with increased expression of cofilin-1, an actin-binding protein required for actin dynamics. Transfection of c-Myc could induce cofilin-1, but transfection of truncated Myc-Nick mutant and inhibition of c-Myc reduced cofilin-1 expression. Additionally, knockdown of cofilin-1 could suppress cMIS. The chromatin immunoprecipitation-quantitative polymerase chain reaction (ChIP-qPCR) assay showed that the endogenous c-Myc mainly bound to two out of three predicted E-boxes located in middle and proximity to the transcription initiation site of the CFL1 promoter. Interestingly, ectopic expression of c-Myc bound to all E-boxes, especially the distal one. Furthermore, the conditioned medium (CM) collected from cells with cMIS could enhance the proliferation and migration of other NSCLC cells, whereas that obtained from cofilin-1 silencing cells with forced expression of c-Myc diminished these capacities. The c-Myc transactivated cofilin-1 could also be triggered by H2O2 through the middle E-box. Surprisingly, a physical interaction between c-Myc and cofilin-1 was detected, and H2O2 increased this effect. Clinically, high expression of both c-Myc and CFL1 genes correlated to worse survival rates among NSCLC patients, especially those with the adenocarcinoma subtype. Taken together, the c-Myc-cofilin-1 regulatory axis would explain the mechanism of OIS promoted cancer progression, and it may be a potent target for design of treatments.
Similar content being viewed by others
References
Serrano M, Lin AW, McCurrach ME, Beach D, Lowe SW. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell. 1997;88:593–602. https://doi.org/10.1016/s0092-8674(00)81902-9.
Zhu H, Blake S, Kusuma FK, Pearson RB, Kang J, Chan KT. Oncogene-induced senescence: from biology to therapy. Mech Ageing Dev. 2020;187:111229 https://doi.org/10.1016/j.mad.2020.111229.
Liu XL, Ding J, Meng LH. Oncogene-induced senescence: a double edged sword in cancer. Acta Pharm Sin. 2018;39:1553–8. https://doi.org/10.1038/aps.2017.198.
Ko A, Han SY, Choi CH, Cho H, Lee MS, Kim SY, et al. Oncogene-induced senescence mediated by c-Myc requires USP10 dependent deubiquitination and stabilization of p14ARF. Cell Death Differ. 2018;25:1050–62. https://doi.org/10.1038/s41418-018-0072-0.
Astle MV, Hannan KM, Ng PY, Lee RS, George AJ, Hsu AK, et al. AKT induces senescence in human cells via mTORC1 and p53 in the absence of DNA damage: implications for targeting mTOR during malignancy. Oncogene. 2012;31:1949–62. https://doi.org/10.1038/onc.2011.394.
Rattanavirotkul N, Kirschner K, Chandra T. Induction and transmission of oncogene-induced senescence. Cell Mol Life Sci. 2021;78:843–52. https://doi.org/10.1007/s00018-020-03638-0.
Patel PL, Suram A, Mirani N, Bischof O, Herbig U. Derepression of hTERT gene expression promotes escape from oncogene-induced cellular senescence. Proc Natl Acad Sci USA. 2016;113:E5024–5033. https://doi.org/10.1073/pnas.1602379113.
Schmitt CA, Wang B, Demaria M. Senescence and cancer - role and therapeutic opportunities. Nat Rev Clin Oncol. 2022;19:619–36. https://doi.org/10.1038/s41571-022-00668-4.
Pan Y, van der Watt PJ, Kay SA. E-box binding transcription factors in cancer. Front Oncol. 2023;13:1223208 https://doi.org/10.3389/fonc.2023.1223208.
Li Z, Owonikoko TK, Sun SY, Ramalingam SS, Doetsch PW, Xiao ZQ, et al. c-Myc suppression of DNA double-strand break repair. Neoplasia. 2012;14:1190–202. https://doi.org/10.1593/neo.121258.
Gao FY, Li XT, Xu K, Wang RT, Guan XX. c-MYC mediates the crosstalk between breast cancer cells and tumor microenvironment. Cell Commun Signal. 2023;21:28. https://doi.org/10.1186/s12964-023-01043-1.
Melnik S, Werth N, Boeuf S, Hahn EM, Gotterbarm T, Anton M, et al. Impact of c-MYC expression on proliferation, differentiation, and risk of neoplastic transformation of human mesenchymal stromal cells. Stem Cell Res Ther. 2019;10:73 https://doi.org/10.1186/s13287-019-1187-z.
Yoshida GJ. Emerging roles of Myc in stem cell biology and novel tumor therapies. J Exp Clin Cancer Res. 2018;37:173 https://doi.org/10.1186/s13046-018-0835-y.
McMahon SB. MYC and the control of apoptosis. Cold Spring Harb Perspect Med. 2014;4:a014407 https://doi.org/10.1101/cshperspect.a014407.
van Riggelen J, Yetil A, Felsher DW. MYC as a regulator of ribosome biogenesis and protein synthesis. Nat Rev Cancer. 2010;10:301–9. https://doi.org/10.1038/nrc2819.
Miller DM, Thomas SD, Islam A, Muench D, Sedoris K. c-Myc and cancer metabolism. Clin Cancer Res. 2012;18:5546–53. https://doi.org/10.1158/1078-0432.CCR-12-0977.
Casey SC, Baylot V, Felsher DW. The MYC oncogene is a global regulator of the immune response. Blood. 2018;131:2007–15. https://doi.org/10.1182/blood-2017-11-742577.
Garcia-Gutierrez L, Delgado MD, Leon J. MYC oncogene contributions to release of cell cycle brakes. Genes (Basel). 2019;10. https://doi.org/10.3390/genes10030244
Madden SK, de Araujo AD, Gerhardt M, Fairlie DP, Mason JM. Taking the Myc out of cancer: toward therapeutic strategies to directly inhibit c-Myc. Mol Cancer. 2021;20:3. https://doi.org/10.1186/s12943-020-01291-6.
Larsson LG, Henriksson MA. The Yin and Yang functions of the Myc oncoprotein in cancer development and as targets for therapy. Exp Cell Res. 2010;316:1429–37. https://doi.org/10.1016/j.yexcr.2010.03.025.
Grandori C, Wu KJ, Fernandez P, Ngouenet C, Grim J, Clurman BE, et al. Werner syndrome protein limits MYC-induced cellular senescence. Genes Dev. 2003;17:1569–74. https://doi.org/10.1101/gad.1100303.
Campaner S, Doni M, Verrecchia A, Fagà G, Bianchi L, Amati B. Myc, Cdk2 and cellular senescence: old players, new game. Cell Cycle. 2010;9:3655–61.
Campaner S, Doni M, Hydbring P, Verrecchia A, Bianchi L, Sardella D, et al. Cdk2 suppresses cellular senescence induced by the c-myc oncogene. Nat Cell Biol. 2010;12:54–59.
Zindy F, Eischen CM, Randle DH, Kamijo T, Cleveland JL, Sherr CJ, et al. Myc signaling via the ARF tumor suppressor regulates p53-dependent apoptosis and immortalization. Genes Dev. 1998;12:2424–33. https://doi.org/10.1101/gad.12.15.2424.
Bernstein BW, Bamburg JR. ADF/cofilin: a functional node in cell biology. Trends Cell Biol. 2010;20:187–95.
Bamburg JR, McGough A, Ono S. Putting a new twist on actin: ADF/cofilins modulate actin dynamics. Trends Cell Biol. 1999;9:364–70.
Bamburg JR, Bernstein BW. Roles of ADF/cofilin in actin polymerization and beyond. F1000 Biol Rep. 2010;2:62 https://doi.org/10.3410/B2-62.
Tsai CH, Chang CY, Lin BZ, Wu YL, Wu MH, Lin LT, et al. Up-regulation of cofilin-1 in cell senescence associates with morphological change and p27(kip1) -mediated growth delay. Aging Cell. 2021;20:13288 https://doi.org/10.1111/acel.13288.
Du HQ, Chen L, Wang Y, Wang LJ, Yan H, Liu HY, et al. Increasing radiosensitivity with the downregulation of cofilin-1 in U251 human glioma cells. Mol Med Rep. 2015;11:3354–60. https://doi.org/10.3892/mmr.2014.3125.
Satoh M, Takano S, Sogawa K, Noda K, Yoshitomi H, Ishibashi M, et al. Immune-complex level of cofilin-1 in sera is associated with cancer progression and poor prognosis in pancreatic cancer. Cancer Sci. 2017;108:795–803. https://doi.org/10.1111/cas.13181.
Cho HJ, Baek GO, Yoon MG, Ahn HR, Son JA, Kim SS, et al. Overexpressed proteins in HCC cell-derived exosomes, CCT8, and cofilin-1 are potential biomarkers for patients with HCC. Diagnostics. 2021;11:1221.
Nishimura S, Tsuda H, Kataoka F, Arao T, Nomura H, Chiyoda T, et al. Overexpression of cofilin 1 can predict progression-free survival in patients with epithelial ovarian cancer receiving standard therapy. Hum Pathol. 2011;42:516–21. https://doi.org/10.1016/j.humpath.2010.07.019.
Howard J, Goh CY, Gorzel KW, Higgins M, McCann A. The potential role of cofilin-1 in promoting triple negative breast cancer (TNBC) metastasis via the extracellular vesicles (EVs). Transl Oncol. 2022;15:101247. https://doi.org/10.1016/j.tranon.2021.101247.
Peng XC, Gong FM, Zhao YW, Zhou LX, Xie YW, Liao HL, et al. Comparative proteomic approach identifies PKM2 and cofilin-1 as potential diagnostic, prognostic and therapeutic targets for pulmonary adenocarcinoma. PLoS One. 2011;6:e27309. https://doi.org/10.1371/journal.pone.0027309.
Müller CB, de Barros RL, Castro MA, Lopes FM, Meurer RT, Roehe A, et al. Validation of cofilin-1 as a biomarker in non-small cell lung cancer: application of quantitative method in a retrospective cohort. J Cancer Res Clin Oncol. 2011;137:1309–16. https://doi.org/10.1007/s00432-011-1001-5.
Chen L, Cai J, Huang Y, Tan X, Guo Q, Lin X, et al. Identification of cofilin-1 as a novel mediator for the metastatic potentials and chemoresistance of the prostate cancer cells. Eur J Pharm. 2020;880:173100. https://doi.org/10.1016/j.ejphar.2020.173100.
Becker M, De Bastiani MA, Müller CB, Markoski MM, Castro MA, Klamt F. High cofilin-1 levels correlate with cisplatin resistance in lung adenocarcinomas. Tumour Biol. 2014;35:1233–8. https://doi.org/10.1007/s13277-013-1164-6.
Sousa-Squiavinato ACM, Morgado-Diaz JA. A glimpse into cofilin-1 role in cancer therapy: A potential target to improve clinical outcomes? Biochim Biophys Acta Rev Cancer. 2024;1879:189087. https://doi.org/10.1016/j.bbcan.2024.189087.
Jiang C, Xuan Z, Zhao F, Zhang MQ. TRED: a transcriptional regulatory element database, new entries and other development. Nucleic Acids Res. 2007;35:D137–140. https://doi.org/10.1093/nar/gkl1041.
Lebel R, McDuff FO, Lavigne P, Grandbois M. Direct visualization of the binding of c-Myc/Max heterodimeric b-HLH-LZ to E-box sequences on the hTERT promoter. Biochemistry. 2007;46:10279–86. https://doi.org/10.1021/bi700076m.
Tsuneoka M, Nakano F, Ohgusu H, Mekada E. c-myc activates RCC1 gene expression through E-box elements. Oncogene. 1997;14:2301–11. https://doi.org/10.1038/sj.onc.1201067.
Conacci-Sorrell M, Ngouenet C, Eisenman RN. Myc-nick: a cytoplasmic cleavage product of Myc that promotes alpha-tubulin acetylation and cell differentiation. Cell. 2010;142:480–93. https://doi.org/10.1016/j.cell.2010.06.037.
Coppe JP, Desprez PY, Krtolica A, Campisi J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol. 2010;5:99–118. https://doi.org/10.1146/annurev-pathol-121808-102144.
Kumari R, Jat P. Mechanisms of Cellular Senescence: Cell Cycle Arrest and Senescence Associated Secretory Phenotype. Front Cell Dev Biol. 2021;9:645593. https://doi.org/10.3389/fcell.2021.645593.
Chen JW, Dhahbi J. Lung adenocarcinoma and lung squamous cell carcinoma cancer classification, biomarker identification, and gene expression analysis using overlapping feature selection methods. Sci Rep. 2021;11:13323. https://doi.org/10.1038/s41598-021-92725-8.
White MC, Holman DM, Boehm JE, Peipins LA, Grossman M, Henley SJ. Age and cancer risk: a potentially modifiable relationship. Am J Prev Med. 2014;46:S7–15. https://doi.org/10.1016/j.amepre.2013.10.029.
Xiao S, Qin D, Hou X, Tian L, Yu Y, Zhang R, et al. Cellular senescence: a double-edged sword in cancer therapy. Front Oncol. 2023;13:1189015. https://doi.org/10.3389/fonc.2023.1189015.
Milanovic M, Fan D, Belenki D, Däbritz J, Zhao Z, Yu Y, et al. Senescence-associated reprogramming promotes cancer stemness. Nature. 2018;553:96–100. https://doi.org/10.1038/nature25167.
Laberge RM, Sun Y, Orjalo AV, Patil CK, Freund A, Zhou L, et al. Author Correction: MTOR regulates the pro-tumorigenic senescence-associated secretory phenotype by promoting IL1A translation. Nat Cell Biol. 2021;23:564–5. https://doi.org/10.1038/s41556-021-00655-4.
Orjalo AV, Bhaumik D, Gengler BK, Scott GK, Campisi J. Cell surface-bound IL-1alpha is an upstream regulator of the senescence-associated IL-6/IL-8 cytokine network. Proc Natl Acad Sci USA. 2009;106:17031–6. https://doi.org/10.1073/pnas.0905299106.
Yu Y, Schleich K, Yue B, Ji S, Lohneis P, Kemper K, et al. Targeting the Senescence-Overriding Cooperative Activity of Structurally Unrelated H3K9 Demethylases in Melanoma. Cancer Cell. 2018;33:322–36.e328. https://doi.org/10.1016/j.ccell.2018.01.002.
Zampetidis CP, Galanos P, Angelopoulou A, Zhu Y, Polyzou A, Karamitros T, et al. A recurrent chromosomal inversion suffices for driving escape from oncogene-induced senescence via subTAD reorganization. Mol Cell. 2021;81:4907–23.e4908. https://doi.org/10.1016/j.molcel.2021.10.017.
Martínez-Zamudio RI, Stefa A, Nabuco Leva Ferreira Freitas JA, Vasilopoulos T, Simpson M, Doré G, et al. Escape from oncogene-induced senescence is controlled by POU2F2 and memorized by chromatin scars. Cell Genom. 2023;3:100293. https://doi.org/10.1016/j.xgen.2023.100293.
Hayflick L. The Limited in Vitro Lifetime of Human Diploid Cell Strains. Exp Cell Res. 1965;37:614–36. https://doi.org/10.1016/0014-4827(65)90211-9.
Wallis R, Milligan D, Hughes B, Mizen H, López-Domínguez JA, Eduputa U, et al. Senescence-associated morphological profiles (SAMPs): an image-based phenotypic profiling method for evaluating the inter and intra model heterogeneity of senescence. Aging (Albany NY). 2022;14:4220–46. https://doi.org/10.18632/aging.204072.
Wang E, Gundersen D. Increased organization of cytoskeleton accompanying the aging of human fibroblasts in vitro. Exp Cell Res. 1984;154:191–202. https://doi.org/10.1016/0014-4827(84)90679-7.
Kwak IH, Kim HS, Choi OR, Ryu MS, Lim IK. Nuclear accumulation of globular actin as a cellular senescence marker. Cancer Res. 2004;64:572–80. https://doi.org/10.1158/0008-5472.can-03-1856.
Prasad AV, Mohan N, Chandrasekar B, Meltz ML. Induction of transcription of “immediate early genes” by low-dose ionizing radiation. Radiat Res. 1995;143:263–72.
Leu JD, Wang CY, Lo CC, Lin MY, Chang CY, Hung WC, et al. Involvement of c-Myc in low dose radiation-induced senescence enhanced migration and invasion of unirradiated cancer cells. Aging (Albany NY). 2021;13:22208–31. https://doi.org/10.18632/aging.203527.
Nuszkiewicz J, Wozniak A, Szewczyk-Golec K. Ionizing radiation as a source of oxidative stress-the protective role of melatonin and vitamin D. Int J Mol Sci. 2020;21. https://doi.org/10.3390/ijms21165804.
Wang F, Wu D, Fu H, He F, Xu C, Zhou J, et al. Cofilin 1 promotes bladder cancer and is regulated by TCF7L2. Oncotarget. 2017;8:92043–54. https://doi.org/10.18632/oncotarget.20664.
Corrigendum to Hypoxia-induced cofilin 1 promotes hepatocellular carcinoma progression by regulating the PLD1/AKT pathway. Clin Transl Med. 2022;12, e1110. https://doi.org/10.1002/ctm2.1110.
Yao B, Li Y, Chen T, Niu Y, Wang Y, Yang Y, et al. Hypoxia-induced cofilin 1 promotes hepatocellular carcinoma progression by regulating the PLD1/AKT pathway. Clin Transl Med. 2021;11:366. https://doi.org/10.1002/ctm2.366.
Ciribilli Y, Singh P, Inga A, Borlak J. c-Myc targeted regulators of cell metabolism in a transgenic mouse model of papillary lung adenocarcinoma. Oncotarget. 2016;7:65514–39. https://doi.org/10.18632/oncotarget.11804.
Wiley CD. Bubble bubble, senescent cells are a cauldron of tumor trouble. Cancer Res. 2020;80:3193–4. https://doi.org/10.1158/0008-5472.CAN-20-1811.
Lujambio A, Akkari L, Simon J, Grace D, Tschaharganeh DF, Bolden JE, et al. Non-cell-autonomous tumor suppression by p53. Cell. 2013;153:449–60. https://doi.org/10.1016/j.cell.2013.03.020.
Hu L, Li H, Zi M, Li W, Liu J, Yang Y, et al. Why senescent cells are resistant to apoptosis: an insight for senolytic development. Front Cell Dev Biol. 2022;10:822816. https://doi.org/10.3389/fcell.2022.822816.
Gorgoulis VG, Halazonetis TD. Oncogene-induced senescence: the bright and dark side of the response. Curr Opin Cell Biol. 2010;22:816–27. https://doi.org/10.1016/j.ceb.2010.07.013.
ArulJothi KN, Kumaran K, Senthil S, Nidhu AB, Munaff N, Janitri VB, et al. Implications of reactive oxygen species in lung cancer and exploiting it for therapeutic interventions. Med Oncol. 2022;40:43. https://doi.org/10.1007/s12032-022-01900-y.
Thai AA, Solomon BJ, Sequist LV, Gainor JF, Heist RS. Lung cancer. Lancet. 2021;398:535–54. https://doi.org/10.1016/S0140-6736(21)00312-3.
Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature. 2018;553:446–54.
Polzar B, Nowak E, Goody RS, Mannherz HG. The complex of actin and deoxyribonuclease I as a model system to study the interactions of nucleotides, cations and cytochalasin D with monomeric actin. Eur J Biochem. 1989;182:267–75. https://doi.org/10.1111/j.1432-1033.1989.tb14826.x.
Acknowledgements
These works were supported by a grant of National Science and Technology Council of Taiwan (NSTC 111-2314-B-A49-037-MY3, NSTC 114-2314-B-A49 -065 -MY3), and a grant of Taipei City Hospital, RenAi Branch (TPCH-112-12), and grants from the Ministry of Education, Higher Education SPROUT Project for Cancer and Immunology Research Center (112W31101 and 113W031101). MRC-5 cells were kindly provided by Dr. Chien-Hsien Chen from Dept. of Bioengineering, Tatung University. We thank Mr. Guan Zeng for his assistance on raw data management. We thank Ms. Ke-Xin Huang, Ms. Chia-Chien Lo, and Ms. Bo-Han Huang for their technical supports.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chou, YT., Leu, JD., Yang, WY. et al. c-Myc transactivates CFL1 to induce senescence-like phenotype and potentiate the bystander effects for the migration and proliferation in lung cancer cells. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-03065-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-03065-3