Abstract
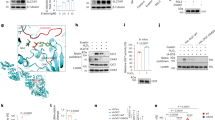
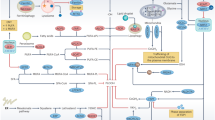
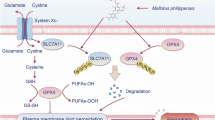
Gastric adenocarcinoma (STAD), a leading cause of cancer mortality, faces major therapeutic challenges due to intrinsic and acquired chemoresistance. Chemoresistance is intricately linked to ferroptosis.Elucidating the mechanisms of chemotherapy resistance in STAD represents a critical unmet need to improve patient survival. This study identifies ODC1 as a crucial driver of 5-Fu resistance and suppressor of ferroptosis in STAD. Multi-dataset analysis revealed significant ODC1 overexpression in STAD tissues, correlating with advanced stage and poor survival. Functionally, ODC1 depletion inhibited proliferation, migration, invasion, and tumor growth in vitro and in vivo, while its overexpression exacerbated malignant phenotypes. Critically, ODC1 was upregulated in 5-Fu-resistant cell models, and its knockdown restored chemosensitivity by triggering ferroptosis—an iron-dependent cell death characterized by lipid peroxidation, glutathione depletion, and malondialdehyde accumulation. Mechanistically, ODC1 interacts with transcription factor YBX1 through its PLPDE_III_ODC domain. This complex binds the promoter of SLC7A11, enhancing its transcription. YBX1 silencing phenocopied ODC1 knockdown, increasing ferroptosis susceptibility; conversely, SLC7A11 overexpression or GPX4 activation (via ML334) reversed ferroptosis induced by ODC1/YBX1 inhibition. Significantly, Erastin—a SLC7A11 inhibitor—overcame YBX1-mediated resistance, synergizing with 5-Fu to induce ferroptosis and suppress tumor growth. Collectively, we unveil the ODC1-YBX1-SLC7A11-ferroptosis axis as a central mechanism of chemoresistance in STAD. Targeting this axis—via ODC1 inhibition or ferroptosis induction—represents a novel therapeutic strategy to reverse treatment resistance in gastric adenocarcinoma.
Similar content being viewed by others
Data availability
The datasets generated and/or analysed during the current study are not publicly available due to patient privacy concerns but are available from the corresponding author on reasonable request.The transcriptome sequencing data have been uploaded to a public database.https://ngdc.cncb.ac.cn/gsa-human/browse/HRA016361.
References
Smyth EC, Nilsson M, Grabsch HI, van Grieken NC, Lordick F. Gastric cancer. Lancet. 2020;396:635–48.
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209–49.
Sreeram A, Stroobant EE, Laszkowska M, Guilford P, Shimada S, Nishimura M, et al. Disappearing Signet Ring Cell Adenocarcinoma in Gastric Cancer Patients. Ann Surg Oncol. 2024;31:9030–8.
Ajani JA, D'amico TA, Bentrem DJ, Corvera CU, Das P, Enzinger PC, et al. Gastric Cancer, Version 2.2025, NCCN Clinical Practice Guidelines In Oncology. J Natl Compr Canc Netw. 2025;23:169–91.
Sharma S, Carey N, McConnell D, Lowery M, O'Sullivan J, McCullagh L, et al. Systematic Review of Economic Evaluations of Systemic Treatments for Advanced and Metastatic Gastric Cancer. Pharmacoeconomics. 2024;42:1091–110.
Chen Z, Song Z, Den S, Zhang W, Han M, Lan T, et al. Application of Immune Checkpoint Inhibitors in Cancer. MedComm. 2025;6:e70176.
Al-Batran SE, Homann N, Pauligk C, Goetze TO, Meiler J, Kasper S, et al. Perioperative chemotherapy with fluorouracil plus leucovorin, oxaliplatin, and docetaxel versus fluorouracil or capecitabine plus cisplatin and epirubicin for locally advanced, resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4): a randomised, phase 2/3 trial. Lancet. 2019;393:1948–57.
Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376:687–97.
Hofer SJ, Daskalaki I, Abdellatif M, Stelzl U, Sedej S, Tavernarakis N, et al. A surge in endogenous spermidine is essential for rapamycin-induced autophagy and longevity. Autophagy. 2024;20:2824–6.
Zhu G, Xie Y, Wang J, Wang M, Qian Y, Sun Q, et al. Multifunctional copper-phenolic nanopills achieve comprehensive polyamines depletion to provoke enhanced pyroptosis and cuproptosis for cancer immunotherapy. Adv Mater. 2024;36:2409066.
Wang T, Liu L, Fang J, Jin H, Natarajan S, Sheppard H, et al. Conditional activation of c-MYC in distinct catecholaminergic cells drives development of neuroblastoma or somatostatinoma. Cancer Res. 2025;85:424–41.
Zhang XP, Zou WB, Li ZQ, Yu ZT, Yu SB, Lin ZY, et al. The heterogeneity of cellular metabolism in the tumour microenvironment of hepatocellular carcinoma with portal vein tumour thrombus. Cell Prolif. 2025;58:e13738.
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149:1060–72.
Stockwell BR, Friedmann Angeli JP, Bayir H, Bush AI, Conrad M, Dixon SJ, et al. Ferroptosis: a regulated cell death nexus linking metabolism, redox biology, and disease. Cell. 2017;171:273–85.
Galluzzi L, Vitale I, Aaronson SA, Abrams JM, Adam D, Agostinis P, et al. Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018;25:486–541.
Lei G, Zhuang L, Gan B. Targeting ferroptosis as a vulnerability in cancer. Nat Rev Cancer. 2022;22:381–96.
Mao C, Liu X, Zhang Y, Lei G, Yan Y, Lee H, et al. DHODH-mediated ferroptosis defence is a targetable vulnerability in cancer. Nature. 2021;593:586–90.
Lei G, Zhang Y, Koppula P, Liu X, Zhang J, Lin SH, et al. The role of ferroptosis in ionizing radiation-induced cell death and tumor suppression. Cell Res. 2020;30:146–62.
Bi G, Liang J, Zhao M, Zhang H, Jin X, Lu T, et al. miR-6077 promotes cisplatin/pemetrexed resistance in lung adenocarcinoma via CDKN1A/cell cycle arrest and KEAP1/ferroptosis pathways. Mol Ther Nucleic Acids. 2022;28:366–86.
Wang W, Green M, Choi JE, Gijón M, Kennedy PD, Johnson JK, et al. CD8(+) T cells regulate tumour ferroptosis during cancer immunotherapy. Nature. 2019;569:270–4.
Bi G, Liang J, Bian Y, Shan G, Huang Y, Lu T, et al. Polyamine-mediated ferroptosis amplification acts as a targetable vulnerability in cancer. Nat Commun. 2024;15:2461.
Koppula P, Lei G, Zhang Y, Yan Y, Mao C, Kondiparthi L, et al. A targetable CoQ-FSP1 axis drives ferroptosis- and radiation-resistance in KEAP1 inactive lung cancers. Nat Commun. 2022;13:2206.
Dar HH, Tyurina YY, Mikulska-Ruminska K, Shrivastava I, Ting HC, Tyurin VA, et al. Pseudomonas aeruginosa utilizes host polyunsaturated phosphatidylethanolamines to trigger theft-ferroptosis in bronchial epithelium. J Clin Invest. 2018;128:4639–53.
Wu Y, Yu C, Luo M, Cen C, Qiu J, Zhang S, et al. Ferroptosis in Cancer Treatment: Another Way to Rome. Front Oncol. 2020;10:571127.
Zhang C, Liu X, Jin S, Chen Y, Guo R. Ferroptosis in cancer therapy: a novel approach to reversing drug resistance. Mol Cancer. 2022;21:47.
Janjigian YY, Kawazoe A, Bai Y, Xu J, Lonardi S, Metges JP, et al. Pembrolizumab plus trastuzumab and chemotherapy for HER2-positive gastric or gastro-oesophageal junction adenocarcinoma: interim analyses from the phase 3 KEYNOTE-811 randomised placebo-controlled trial. Lancet. 2023;402:2197–208.
Shah MA, Schwartz GK. Cell cycle-mediated drug resistance: an emerging concept in cancer therapy. Clin Cancer Res. 2001;7:2168–81.
Liang D, Feng Y, Zandkarimi F, Wang H, Zhang Z, Kim J, et al. Ferroptosis surveillance independent of GPX4 and differentially regulated by sex hormones. Cell. 2023;186:2748–64.e22.
Koppula P, Zhuang L, Gan B. Cystine transporter SLC7A11/xCT in cancer: ferroptosis, nutrient dependency, and cancer therapy. Protein Cell. 2021;12:599–620.
Tuo QZ, Liu Y, Xiang Z, Yan HF, Zou T, Shu Y, et al. Thrombin induces ACSL4-dependent ferroptosis during cerebral ischemia/reperfusion. Signal Transduct Target Ther. 2022;7:59.
Chen L, Ma N, Liu D, Li Y, Ci X, Wei Z. Tiliroside induces ferroptosis and suppresses tumor growth by synergistically targeting AKR1B1 and modulating iron metabolism in ovarian cancer cells. Eur J Pharmacol. 2025;997:177591.
Schurch NJ, Schofield P, Gierliński M, Cole C, Sherstnev A, Singh V, et al. How many biological replicates are needed in an RNA-seq experiment and which differential expression tool should you use? Rna. 2016;22:839–51.
Wang S, Osgood AO, Chatterjee A. Uncovering post-translational modification-associated protein-protein interactions. Curr Opin Struct Biol. 2022;74:102352.
Lang X, Green MD, Wang W, Yu J, Choi JE, Jiang L, et al. Radiotherapy and Immunotherapy Promote Tumoral Lipid Oxidation and Ferroptosis via Synergistic Repression of SLC7A11. Cancer Discov. 2019;9:1673–85.
Lu H, Tong W, Jiang M, Liu H, Meng C, Wang K, et al. Mitochondria-Targeted Multifunctional Nanoprodrugs by Inhibiting Metabolic Reprogramming for Combating Cisplatin-Resistant Lung Cancer. ACS Nano. 2024;18:21156–70.
Lyabin DN, Eliseeva IA, Ovchinnikov LP. YB-1 protein: functions and regulation. Wiley Interdiscip Rev RNA. 2014;5:95–110.
Jayavelu AK, Schnöder TM, Perner F, Herzog C, Meiler A, Krishnamoorthy G, et al. Splicing factor YBX1 mediates persistence of JAK2-mutated neoplasms. Nature. 2020;588:157–63.
Mordovkina D, Lyabin DN, Smolin EA, Sogorina EM, Ovchinnikov LP, Eliseeva I Y-Box Binding Proteins in mRNP Assembly, Translation, and Stability Control. Biomolecules, 2020, 10.
Hu J, Liu L, Gong Y, Zhang L, Gan X, Luo X, et al. Linc02527 promoted autophagy in Intrahepatic cholestasis of pregnancy. Cell Death Dis. 2018;9:979.
Cui Y, Li F, Xie Q, Zhao S, Guo T, Guo P, et al. YBX1 mediates autophagy by targeting p110β and decreasing the sensitivity to cisplatin in NSCLC. Cell Death Dis. 2020;11:476.
Shi X, Xiang S, Cao J, Zhu H, Yang B, He Q, et al. Kelch-like proteins: physiological functions and relationships with diseases. Pharmacol Res. 2019;148:104404.
Silva-Islas CA, Maldonado PD. Canonical and non-canonical mechanisms of Nrf2 activation. Pharmacol Res. 2018;134:92–9.
Hu X, Ma Z, Xu B, Li S, Yao Z, Liang B, et al. Glutamine metabolic microenvironment drives M2 macrophage polarization to mediate trastuzumab resistance in HER2-positive gastric cancer. Cancer Commun (Lond). 2023;43:909–37.
Yasuda T, Wang YA. Gastric cancer immunosuppressive microenvironment heterogeneity: implications for therapy development. Trends Cancer. 2024;10:627–42.
Ouyang S, Li H, Lou L, Huang Q, Zhang Z, Mo J, et al. Inhibition of STAT3-ferroptosis negative regulatory axis suppresses tumor growth and alleviates chemoresistance in gastric cancer. Redox Biol. 2022;52:102317.
Gu J, Huang C, Hu X, Xia J, Shao W, Lin D. Nuclear magnetic resonance-based tissue metabolomic analysis clarifies molecular mechanisms of gastric carcinogenesis. Cancer Sci. 2020;111:3195–209.
Dong Y, Kang H, Peng R, Liu Z, Liao F, Hu SA, et al. A clinical-stage Nrf2 activator suppresses osteoclast differentiation via the iron-ornithine axis. Cell Metab. 2024;36:1679–95.e6.
Khan A, Gamble LD, Upton DH, Ung C, Yu D, Ehteda A, et al. Dual targeting of polyamine synthesis and uptake in diffuse intrinsic pontine gliomas. Nat Commun. 2021;12:971.
Miller AK, Tavera G, Dominguez RL, Camargo MC, Waterboer T, Wilson KT, et al. Ornithine decarboxylase (ODC1) gene variant (rs2302615) is associated with gastric cancer independently of Helicobacter pylori CagA serostatus. Oncogene. 2021;40:5963–9.
Friedmann Angeli JP, Schneider M, Proneth B, Tyurina YY, Tyurin VA, Hammond VJ, et al. Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat Cell Biol. 2014;16:1180–91.
Jin R, Li H, Nan S, Wang H. FOXA1 co-activates circODC1 and ODC1 in HPV-positive cervical cancer cell growth. Syst Biol Reprod Med. 2024;70:113–23.
Holbert CE, Cullen MT, Casero RA Jr, Stewart TM. Polyamines in cancer: integrating organismal metabolism and antitumour immunity. Nat Rev Cancer. 2022;22:467–80.
Bello-Fernandez C, Packham G, Cleveland JL. The ornithine decarboxylase gene is a transcriptional target of c-Myc. Proc Natl Acad Sci USA. 1993;90:7804–8.
Nilsson JA, Keller UB, Baudino TA, Yang C, Norton S, Old JA, et al. Targeting ornithine decarboxylase in Myc-induced lymphomagenesis prevents tumor formation. Cancer Cell. 2005;7:433–44.
Ye Z, Zeng Z, Shen Y, Yang Q, Chen D, Chen Z, et al. ODC1 promotes proliferation and mobility via the AKT/GSK3β/β-catenin pathway and modulation of acidotic microenvironment in human hepatocellular carcinoma. Onco Targets Ther. 2019;12:4081–92.
Li J, Cao F, Yin HL, Huang ZJ, Lin ZT, Mao N, et al. Ferroptosis: past, present and future. Cell Death Dis. 2020;11:88.
Tang D, Chen X, Kang R, Kroemer G. Ferroptosis: molecular mechanisms and health implications. Cell Res. 2021;31:107–25.
Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22:266–82.
Yang YC, Zhu Y, Sun SJ, Zhao CJ, Bai Y, Wang J, et al. ROS regulation in gliomas: implications for treatment strategies. Front Immunol. 2023;14:1259797.
Zhang Q, Li Q, Xu T, Jiang H, Xu LG. miR-491-5p suppresses cell growth and invasion by targeting Notch3 in nasopharyngeal carcinoma. Oncol Rep. 2016;35:3541–7.
Lim J, Delaidelli A, Minaker SW, Zhang HF, Colovic M, Yang H, et al. Cystine/glutamate antiporter xCT (SLC7A11) facilitates oncogenic RAS transformation by preserving intracellular redox balance. Proc Natl Acad Sci USA. 2019;116:9433–42.
Koppula P, Zhang Y, Zhuang L, Gan B. Amino acid transporter SLC7A11/xCT at the crossroads of regulating redox homeostasis and nutrient dependency of cancer. Cancer Commun (Lond). 2018;38:12.
Zhang Z, Wang ZX, Chen YX, Wu HX, Yin L, Zhao Q, et al. Integrated analysis of single-cell and bulk RNA sequencing data reveals a pan-cancer stemness signature predicting immunotherapy response. Genome Med. 2022;14:45.
Dong S, Liang S, Cheng Z, Zhang X, Luo L, Li L, et al. ROS/PI3K/Akt and Wnt/β-catenin signalings activate HIF-1α-induced metabolic reprogramming to impart 5-fluorouracil resistance in colorectal cancer. J Exp Clin Cancer Res. 2022;41:15.
Marx C, Sonnemann J, Maddocks O, Marx-Blümel L, Beyer M, Hoelzer D, et al. Global metabolic alterations in colorectal cancer cells during irinotecan-induced DNA replication stress. Cancer Metab. 2022;10:10.
Longley DB, Harkin DP, Johnston PG. 5-fluorouracil: mechanisms of action and clinical strategies. Nat Rev Cancer. 2003;3:330–8.
Fu D, Wang C, Yu L, Yu R. Induction of ferroptosis by ATF3 elevation alleviates cisplatin resistance in gastric cancer by restraining Nrf2/Keap1/xCT signaling. Cell Mol Biol Lett. 2021;26:26.
Liu J, Kang R, Tang D. Signaling pathways and defense mechanisms of ferroptosis. Febs j. 2022;289:7038–50.
Acknowledgements
Not applicable.
Funding
This study was financially supported by National Natural Science Foundation of China (NO.82373014).
Author information
Authors and Affiliations
Contributions
Conceptualization: Ruiqi Li, Jiajie Zhou, Chenkai Zhang; Shantanu Baral, Data production, analysis and investigation: Ruiqi Li, Fanyu Zhao, Yifan Cheng, Zijie Xu, Shuai Zhao, Jie Wang, Yayan Fu, Longhe Sun, Zhen Tian, Chenkai Zhang, Xuetong Jiang, Mengli Zi; Writing, Review and Edit: Ruiqi Li, Jianyue Ding,Dengyang Fang, Ben Li, Qiannan Sun; Supervision: Ruiqi Li. Funding acquisition: Daorong Wang. The author(s) read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests
Patient consent statement and ethics approval statement
This study was approved by the Ethics Committee of Northern Jiangsu People’s Hospital, and written informed consent was obtained from all participants. All animal procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of Yangzhou University (Approval No. 202303827).
Consent for publication
Not applicable.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, R., Baral, S., Zhao, F. et al. Targeting the ODC1-YBX1 axis reverses gastric cancer chemoresistance via transcriptional control of SLC7A11-mediated ferroptosis. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-03067-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-03067-1